
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Masahiro Terasawa | -- | 2308 | 2022-04-08 12:02:34 | | | |
| 2 | Peter Tang | -19 word(s) | 2289 | 2022-04-08 16:45:13 | | |
Video Upload Options
Monostroma nitidum is a green single-cell layered algae that grows on the southwest coast of Japan. It is often used for salad ingredients, boiled tsukudani, soups, etc., due to its health benefits. M. nitidum is composed of many cell aggregates, and the various substances that fill the intercellular space are dietary fibers, vitamins, and minerals. Rhamnan sulfate (RS), a sulfated polysaccharide, is main the component of the fiber extracted from M. nitidum. Recently, some biological properties of RS have been demonstrated by in vitro and in vivo studies that probably protect human subjects from viruses and ameliorate vascular dysfunction caused by metabolic disorders, especially lifestyle-related diseases.
1. Introduction
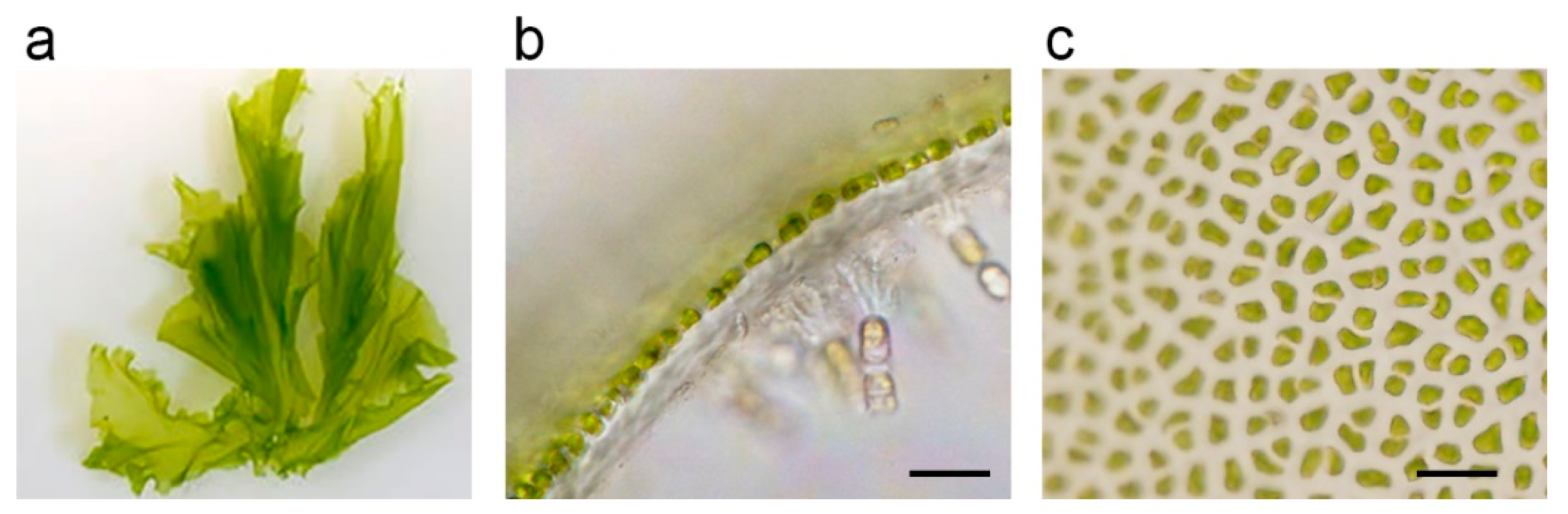
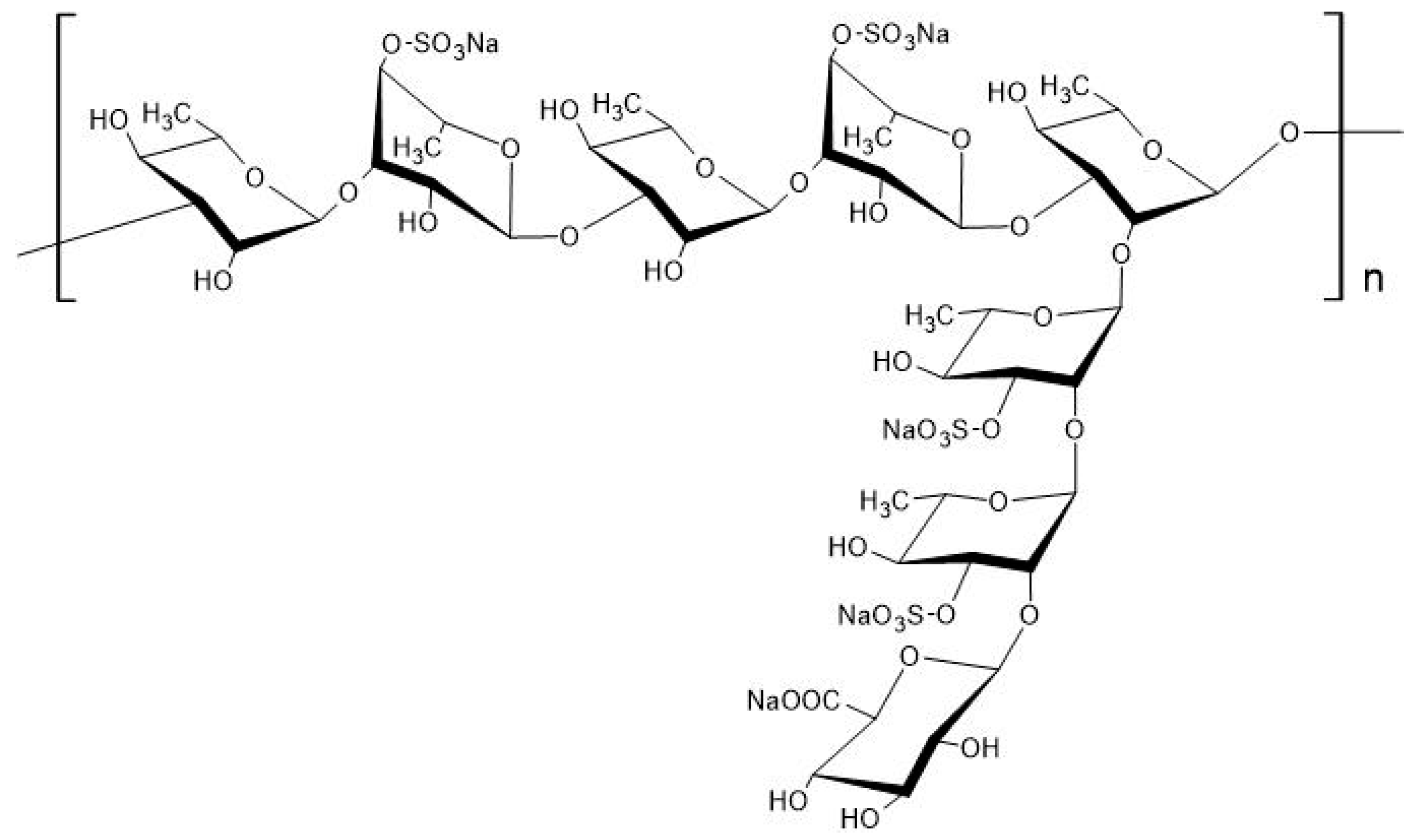
2. Biochemical Characteristics of Rhamnan Sulfate

3. Biological Activities of RS
|
Biological Activity of RS |
References |
|---|---|
|
Anti-hyaluronidase |
in vitro study [23] |
|
Antitumor |
in vitro study [22] |
|
Anti-obesity |
in vivo study [25] |
|
Anti-thrombosis |
in vitro study [14][15][16][17][18][19][20][21], in vivo study [16][17] |
|
Antivirus |
|
|
Anti-hypercholesterolemia |
|
|
Anti-hyperglycemia |
3.1. Antiviral Effect of RS
3.2. Anti-Obesity and Anti-Hypercholesterolemic Effects of RS
3.3. Anti-Glycemia Effect of RS
3.4. Antithrombotic Effects of RS
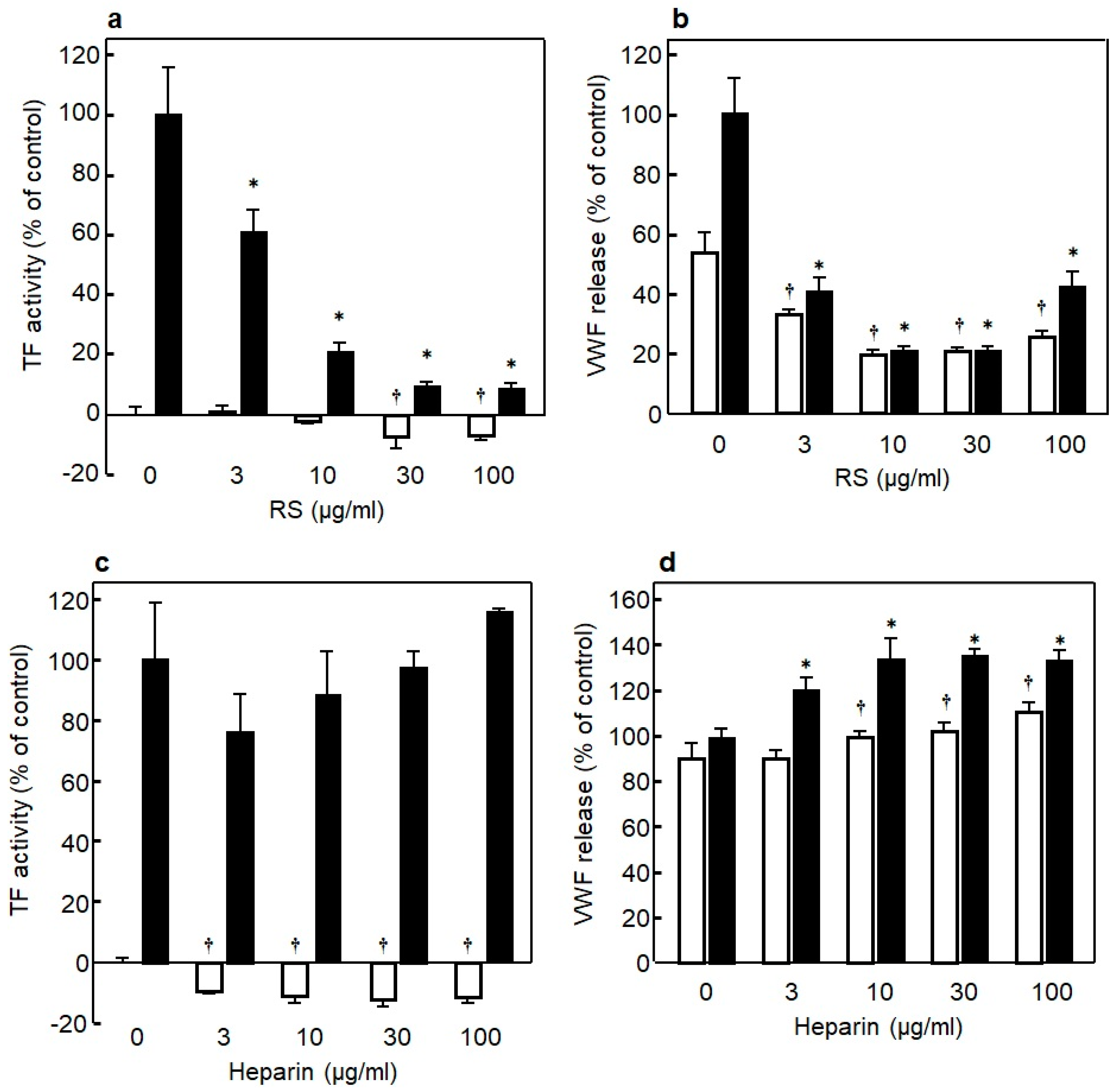
3.5. Anti-Inflammation Effect of RS on Endothelial Cells
References
- Gitzendanner, M.A.; Soltis, P.S.; Wong, G.K.; Ruhfel, B.R.; Soltis, D.E. Plastid phylogenomic analysis of green plants: A billion years of evolutionary history. Am. J. Bot. 2018, 105, 291–301.
- Sanchez-Baracaldo, P.; Raven, J.A.; Pisani, D.; Knoll, A.H. Early photosynthetic eukaryotes inhabited low-salinity habitats. Proc. Natl. Acad. Sci. USA 2017, 114, E7737–E7745.
- Marine Organisms Data. Available online: https://www.jodc.go.jp/data/biology.html (accessed on 19 March 2020).
- Patel, S. Therapeutic importance of sulfated polysaccharides from seaweeds: Updating the recent findings. 3 Biotech. 2012, 2, 171–185.
- Lee, J.B.; Yamagaki, T.; Maeda, M.; Nakanishi, H. Rhamnan sulfate from cell walls of Monostroma latissimum. Phytochemistry 1998, 48, 921–925.
- Lee, J.B.; Koizumi, S.; Hayashi, K.; Hayashi, T. Structure of rhamnan sulfate from the green alga Monostroma nitidum and its anti-herpetic effect. Carbohydr. Polym. 2010, 81, 572–577.
- Tako, M.; Yamashiro, Y.; Teruya, T.; Uechi, S. Structure-function relationship of rhamnan sulfate isolated from commercially cultured edible green seaweed, Monostroma nitidum. Am. J. Appl. Chem. 2017, 5, 38–44.
- Nakamura, M.; Yamashiro, Y.; Konishi, T.; Hanasiro, I.; Tako, M. Structural characterization of rhamnan sulfate isolated from commercially cultured Monostroma nitidum (Hitoegusa). J. Jpn. Soc. Food Sci. 2011, 58, 245–251.
- Lee, J.B.; Hayashi, K.; Hayashi, T.; Sankawa, U.; Maeda, M. Antiviral activities against HSV-1, HCMV, and HIV-1 of rhamnan sulfate from Monostroma latissimum. Planta Med. 1999, 65, 439–441.
- Lee, J.B.; Hayashi, K.; Maeda, M.; Hayashi, T. Antiherpetic activities of sulfated polysaccharides from green algae. Planta Med. 2004, 70, 813–817.
- Wang, S.; Wang, W.; Hou, L.; Qin, L.; He, M.; Li, W.; Mao, W. A sulfated glucuronorhamnan from the green seaweed Monostroma nitidum: Characteristics of its structure and antiviral activity. Carbohydr. Polym. 2020, 227, 115280.
- Wang, S.; Wang, W.; Hao, C.; Yunjia, Y.; Qin, L.; He, M.; Mao, W. Antiviral activity against enterovirus 71 of sulfated rhamnan isolated from the green alga Monostroma latissimum. Carbohydr. Polym. 2018, 200, 43–53.
- Terasawa, M.; Hayashi, K.; Lee, J.B.; Nishiura, K.; Matsuda, K.; Hayashi, T.; Kawahara, T. Anti-influenza A virus activity of rhamnan sulfate from green algae Monostroma nitidum in mice with normal and compromised immunity. (unpublished but already submitted).
- Okamoto, T.; Akita, N.; Terasawa, M.; Hayashi, T.; Suzuki, K. Rhamnan sulfate extracted from Monostroma nitidum attenuates blood coagulation and inflammation of vascular endothelial cells. J. Nat. Med. 2019, 73, 614–619.
- Liu, X.; Hao, J.; He, X.; Wang, S.; Cao, S.; Qin, L.; Mao, W. A rhamnan-type sulfated polysaccharide with novel structure from Monostroma angicava Kjellm (Chlorophyta) and its bioactivity. Carbohydr. Polym. 2017, 173, 732–748.
- Liu, X.; Du, P.; Liu, X.; Cao, S.; Qin, L.; He, M.; He, X.; Mao, W. Anticoagulant properties of a green algal rhamnan-type sulfated polysaccharide and its low-molecular-weight fragments prepared by mild acid degradation. Mar. Drugs 2018, 16, 445.
- Liu, X.; Wang, S.; Cao, S.; He, X.; Qin, L.; He, M.; Yang, Y.; Hao, J.; Mao, W. Structural characteristics and anticoagulant property in vitro and in vivo of a seaweed sulfated rhamnan. Mar. Drugs 2018, 16, 243.
- Li, H.; Mao, W.; Hou, Y.; Gao, Y.; Qi, X.; Zhao, C.; Chen, Y.; Chen, Y.; Li, N.; Wang, C. Preparation, structure and anticoagulant activity of a low molecular weight fraction produced by mild acid hydrolysis of sulfated rhamnan from Monostroma latissimum. Bioresour. Technol. 2012, 114, 414–418.
- Li, N.; Liu, X.; He, X.; Wang, S.; Cao, S.; Xia, Z.; Xian, H.; Qin, L.; Mao, W. Structure and anticoagulant property of a sulfated polysaccharide isolated from the green seaweed Monostroma angicava. Carbohydr. Polym. 2017, 159, 195–206.
- Harada, N.; Maeda, M. Chemical structure of antithrombin-active rhamnan sulfate from Monostrom nitidum. Biosci. Biotechnol. Biochem. 1998, 62, 1647–1652.
- Li, H.; Mao, W.; Zhang, X.; Qi, X.; Chen, Y.; Chen, Y.; Xu, J.; Zhao, C.; Hou, Y.; Yang, Y.; et al. Structural characterization of an anticoagulant-active sulfated polysaccharide isolated from green alga Monostroma latissimum. Carbohydr. Polym. 2011, 85, 394–400.
- Ropellato, J.; Carvalho, M.M.; Ferreira, L.G.; Noseda, M.D.; Zuconelli, C.R.; Goncalves, A.G.; Ducatti, D.R.; Kenski, J.C.; Nasato, P.L.; Winnischofer, S.M.; et al. Sulfated heterorhamnans from the green seaweed Gayralia oxysperma: Partial depolymerization, chemical structure and antitumor activity. Carbohydr. Polym. 2015, 117, 476–485.
- Yamamoto, Y.; Ozono, M.; Oishi, T.; Oshima, K.; Mitsuiki, S.; Kakihara, H.; Mukae, K. Hyaluronidase-inhibitory activity of rhamnan sulfate obtained from cultivated Monostroma nitidum (Hitoegusa). J. Jpn. Soc. Food Sci. 2016, 63, 545–549.
- Nishikawa, M.; Mitsui, M.; Umeda, K.; Kitaoka, Y.; Takahashi, Y.; Tanaka, S. Effect of sulfated polysaccharides extracted from sea alga (Monostroma latissium and Monostroma nitidum) on serum cholesterol in subjects with borderline or mild hypercholesterolemia. J. New Rem. Clin. 2006, 55, 1763–1770.
- Zang, L.; Shimada, Y.; Tanaka, T.; Nishimura, N. Rhamnan sulphate from Monostroma nitidum attenuates hepatic steatosis by suppressing lipogenesis in a diet-induced obesity zebrafish model. J. Funct. Foods 2015, 17, 364–370.
- Kamimura, Y.; Hashiguchi, K.; Nagata, Y.; Saka, T.; Yoshida, M.; Makino, Y.; Amano, H. Inhibitory effects of edible green algae Monostroma nitidum on glycemic responses. J. Jpn. Soc. Food Sci. 2010, 57, 441–445.
- Panlasigui, L.N.; Baello, O.Q.; Dimatangal, J.M.; Dumelod, B.D. Blood cholesterol and lipid-lowering effects of carrageenan on human volunteers. Asia Pac. J. Clin. Nutr. 2003, 12, 209–214.
- Fuller, S.; Beck, E.; Salman, H.; Tapsell, L. New horizons for the study of dietary fiber and health: A Review. Plant Foods Hum. Nutr. 2016, 71, 1–12.
- Yu, K.; Ke, M.Y.; Li, W.H.; Zhang, S.Q.; Fang, X.C. The impact of soluble dietary fibre on gastric emptying, postprandial blood glucose and insulin in patients with type 2 diabetes. Asia Pac. J. Clin. Nutr. 2014, 23, 210–218.
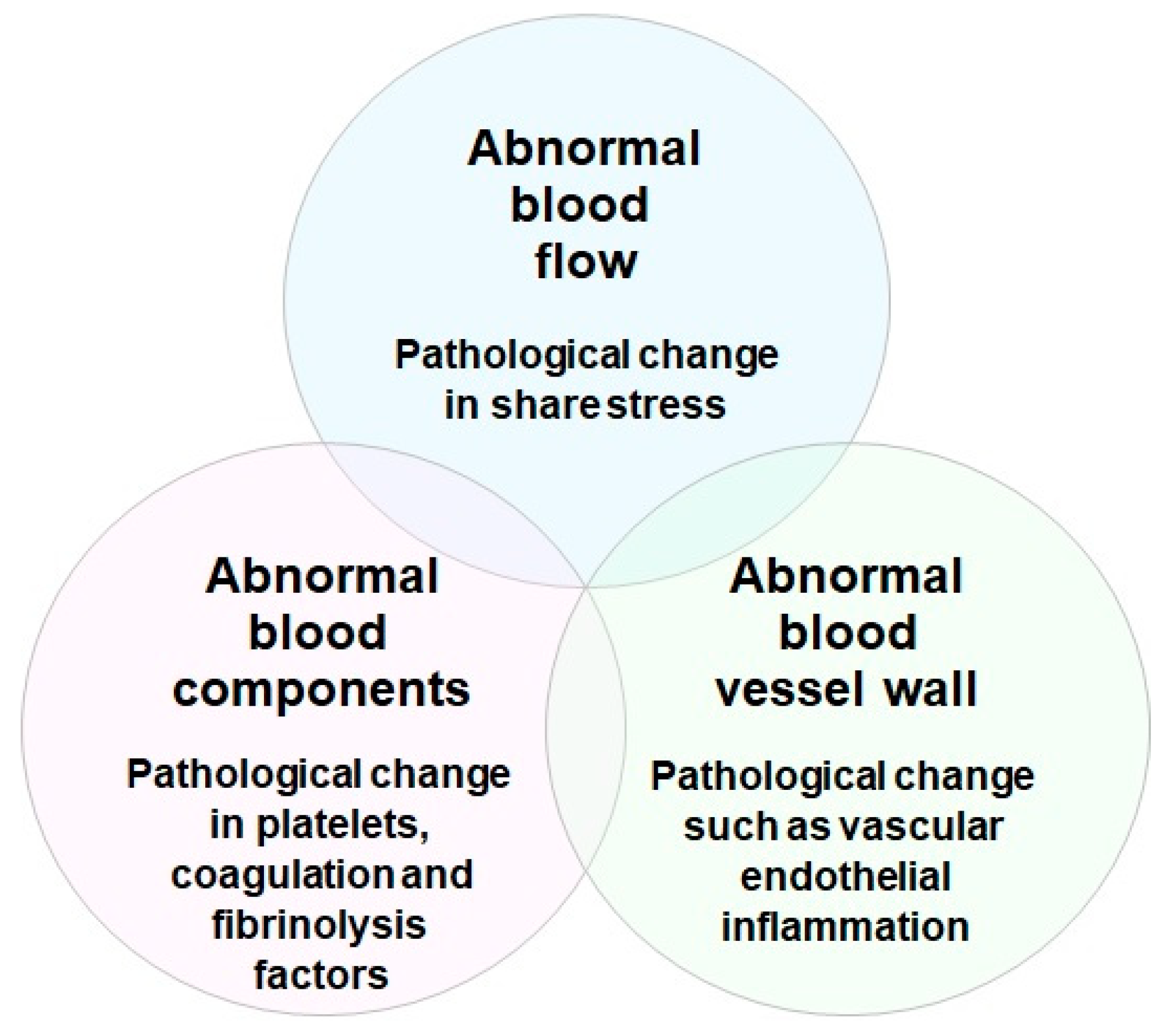
- Kumar, D.R.; Hanlin, E.; Glurich, I.; Mazza, J.J.; Yale, S.H. Virchow’s contribution to the understanding of thrombosis and cellular biology. Clin. Med. Res. 2010, 8, 168–172.
- Bagot, C.N.; Arya, R. Virchow and his triad: A question of attribution. Br. J. Haematol. 2008, 143, 180–190.
- Watson, T.; Shantsila, E.; Lip, G.Y. Mechanisms of thrombogenesis in atrial fibrillation: Virchow’s triad revisited. Lancet 2009, 373, 155–166.
- Hosseinzadegan, H.; Tafti, D.K. Prediction of Thrombus Growth: Effect of Stenosis and Reynolds Number. Cardiovasc. Eng. Technol. 2017, 8, 164–181.





