Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Michelangelo Bisconti | -- | 4768 | 2022-04-04 09:11:38 | | | |
| 2 | Vivi Li | -51 word(s) | 4717 | 2022-04-06 05:49:16 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Bisconti, M.; , . Skeletal Transformations and the Origin of Baleen Whales. Encyclopedia. Available online: https://encyclopedia.pub/entry/21343 (accessed on 17 May 2026).
Bisconti M, . Skeletal Transformations and the Origin of Baleen Whales. Encyclopedia. Available at: https://encyclopedia.pub/entry/21343. Accessed May 17, 2026.
Bisconti, Michelangelo, . "Skeletal Transformations and the Origin of Baleen Whales" Encyclopedia, https://encyclopedia.pub/entry/21343 (accessed May 17, 2026).
Bisconti, M., & , . (2022, April 04). Skeletal Transformations and the Origin of Baleen Whales. In Encyclopedia. https://encyclopedia.pub/entry/21343
Bisconti, Michelangelo and . "Skeletal Transformations and the Origin of Baleen Whales." Encyclopedia. Web. 04 April, 2022.
Copy Citation
Mysticeti or baleen whales are a group of cetaceans including a few extant species in only six genera. Despite their scarce diversity, baleen whales represent key species in the trophic webs of the oceans as they are able to sequester enormous quantities of carbon and to disseminate nutrients that support large planktonic blooms. The study of their evolution is thus crucial to understand how and when the ecological characteristics of today’s oceans came to be.
character distribution
evolution
fossil record
Mysticeti
paleontology
pattern
phylogeny
1. Introduction
Mysticeti or baleen whales are a group of cetaceans including a few extant species in only six genera [1]. Despite their scarce diversity, baleen whales represent key species in the trophic webs of the oceans as they are able to sequester enormous quantities of carbon and to disseminate nutrients that support large planktonic blooms [2][3][4]. The study of their evolution is thus crucial to understand how and when the ecological characteristics of today’s oceans came to be.
It was thought for long time that the origin of baleen whales was related to the origin of those morphological characters that are related to their typical way of feeding: baleen-assisted filter feeding. Different mysticete families use the baleen plates in peculiar ways depending upon their own feeding style [5][6]. However, recent studies have shown that the earliest-diverging mysticetes had skull characters that were remarkably different from those of the extant species, possessed functional dentitions and did not filter feed. The actual origin of the baleen is still not fully understood as the fossil record provides only a few clues on this process, and a cogent debate has developed in the past two decades in the scientific literature on this topic (e.g., [7][8][9][10]).
A number of new paleontological discoveries illuminated some aspects of the earliest phases of the evolution of mysticete whales. Late Eocene and Oligocene fossils have helped researchers to understand part of the transformation process that happened at the origin stage of Mysticeti, suggesting that the assembly of the mysticete bauplan was realized through a step-wise process of character transformation (e.g., [7][11]). However, an extraordinarily limited amount of work has been published dealing with the evolution of characters that are not closely related to feeding in this group. For example, there have been only a few attempts to analyze the origin and evolution of the mysticete forelimb and vertebral column (e.g., [12][13][14]).
2. Rostrum
In the earliest-diverging mysticetes, i.e., Mammalodontidae, the rostrum is short and the neurocranium represents a higher portion of the skull length (Figure 1). However, differing from archaeocetes, in Mammalodon and Janjucetus, the dorsal infraorbital foramen is represented by a cluster of foramina located in the posterior portion of the maxilla. The dorsal infraorbital foramen provides the maxillary ramus of the trigeminal nerve a path through the maxilla from the endocranial cavity where it is represented by a robust, bilateral nerve trunk in both the Odontoceti and Mysticeti ([15][16] and literature therein). In the Mammalodontidae, the frontal-rostrum articulation is reduced to the ascending processes of the maxilla and premaxilla, the latter being transversely and anteroposteriorly reduced. The posterolateral border of the maxilla produces a short and narrow lateral process and a flattened infraorbital process developed ventrally to the supraorbital process of the frontal. Anteriorly to the nasal, the narial fossa is prolonged anteriorly and forms a mesorostral groove. Most of the rostral characters of balaenomorph mysticetes were already present in the earliest-diverging mysticetes known up to now, the Mammalodontidae, in which the rostrum lacks the strong elongation observed in the later-diverging branches (e.g., Aetiocetidae, Eomysticetidae and so on).
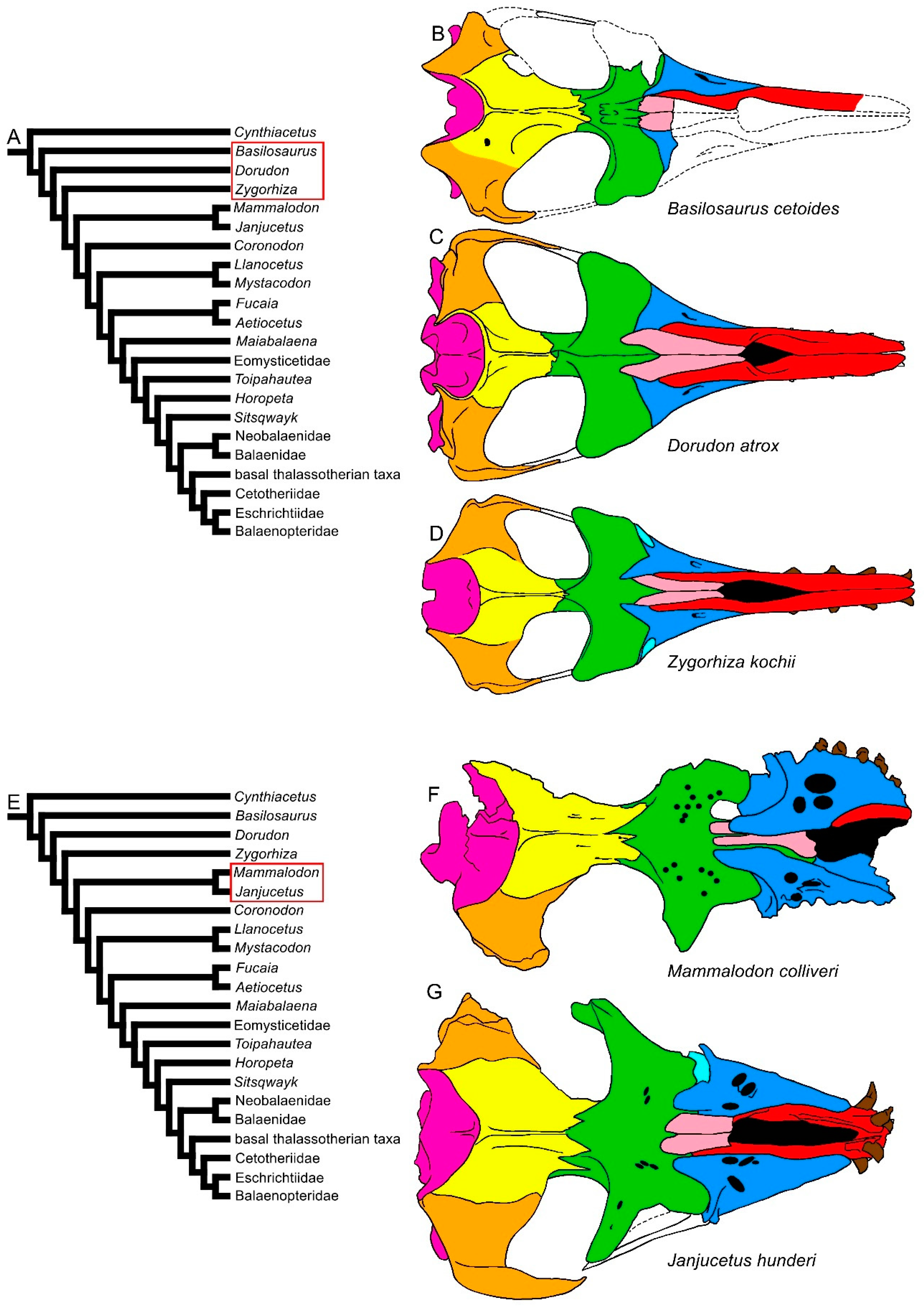
Figure 1. Diversity of the skull in dorsal view in basilosaurine and dorudontine archaeocetes and in mysticetes. (A) Phylogenetic relationships among archaeocetes and mysticetes showing the archaeocete branching (red ellipsis). (B) Skull of Basilosaurus cetoides. (C) Skull of Dorudon atrox. (D) Skull of Zygorhiza kochii. (E) Phylogenetic relationships among archeocetes and mysticetes showing the ramus leading to Mammalodontidae (red ellipsis). (F) Skull of Mammalodon colliveri. (G) Skull of Janjucetus hunderi. Not to scale. The skulls are represented as if they shared the same condylobasal length.
The subsequent branches are represented by the Oligocene Coronodon havensteini, Llanocetus denticrenatus and Mystacodon selenensis (Figure 2). In these taxa, the rostrum is longer compared to that of Mammalodontidae. Mystacodon shows an archaeocete-like rostrum in that the mesorostral groove is short and the maxilla is transversely shortened, giving the rostrum a narrow and pointed shape that resembles that observed in Dorudon, Cynthiacetus, Basilosaurus and Zygorhiza (compare Figure 1 and Figure 2A–D). In both Coronodon and Mystacodon, the rostrum represents about half of the whole skull length. In Coronodon, moreover, the lateral borders of the maxilla are thin as in more recent mysticetes.
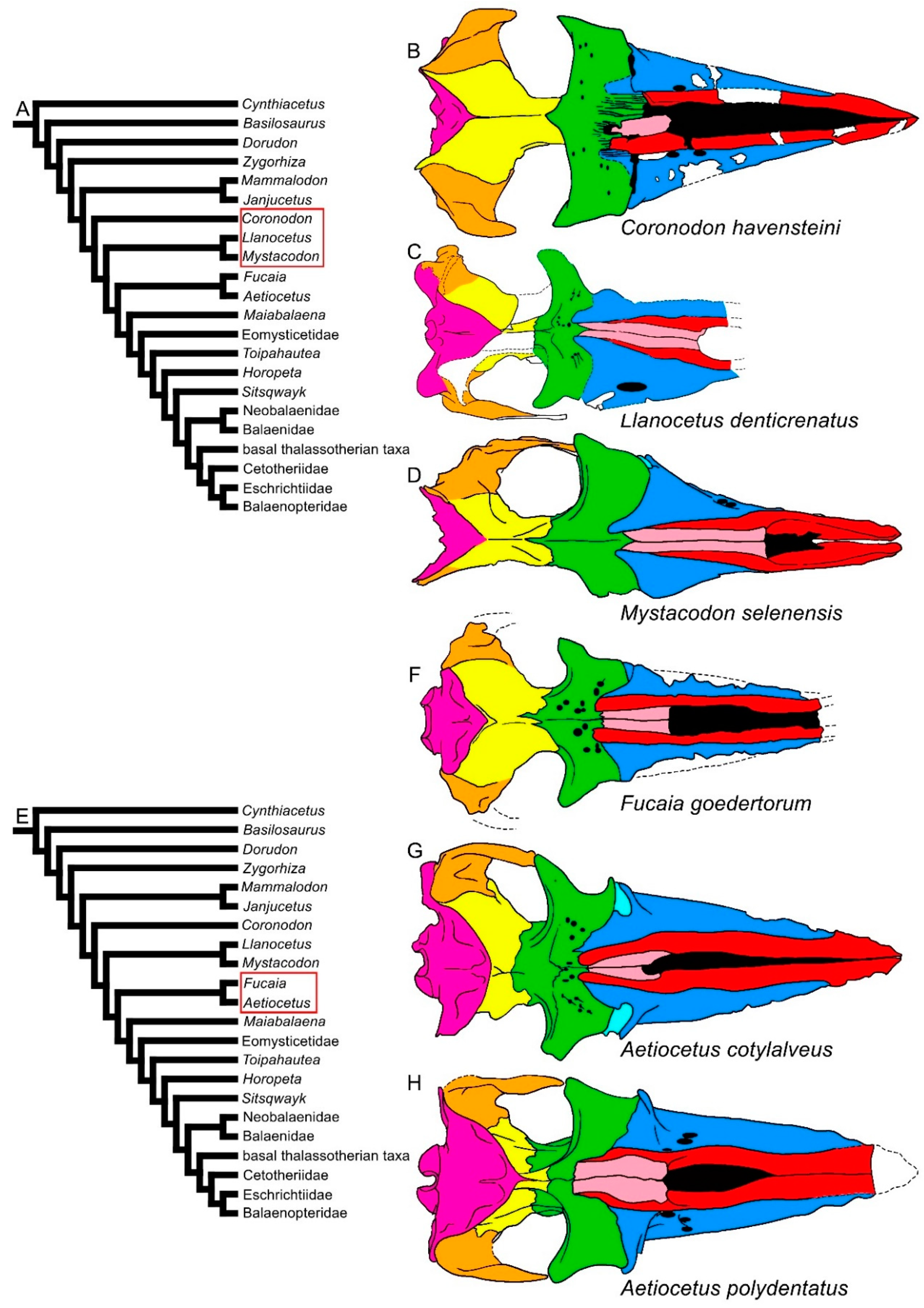
Figure 2. Diversity of mysticete skulls in dorsal view. (A) Phylogenetic relationships among mysticetes showing the branching order of Coronodon, Llanocetus and Mystacodon (red ellipsis). (B) Skull of Coronodon havensteini. (C) Skull of Llanocetus denticrenatus. (D) Skull of Mystacodon selenensis. (E) Phylogenetic relationships among mysticetes showing the branching order of Fucaia and Aetiocetus (red ellipsis). (F) Skull of Fucaia goedertorum. (G) Skull of Aetiocetus cotylalveus. (H) Skull of Aetiocetus polydentatus. Not to scale. The skulls are represented as if they shared the same condylobasal length.
In the clade formed by Fucaia and Aetiocetus (corresponding to the Aetiocetidae family), the rostrum is transversely expanded, and its external outline is externally convex, resembling that of most Chaeomysticeti (Figure 2E–H). The elongation of the rostrum remarkably increases in the Eomysticetidae (Figure 3C–E), while it continues to represent about the half of the skull length in Maiabalaena and Sitsqwayk (Figure 3A,F). In the Eomysticetidae, the rostrum represents about 60% of the whole skull length, shows a border that is thin and externally convex, and a broadly expanded maxilla. The nasal is elongated and reaches c. 30% of the rostrum length. Interestingly, the nasal bone diminishes in proportional length independently in different clades. For example, in Mystacodon, Maiabalaena and Sitsqwayk, the nasal length is about 50% of the rostrum length, but in Aetiocetidae the nasal length is about 20% of such a length. Nasal reduction occurred independently in different mysticete clades.
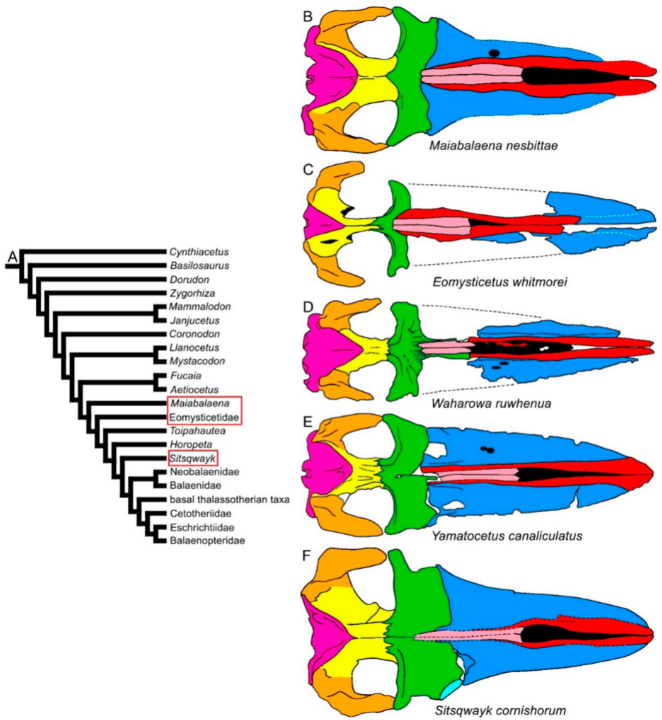
Figure 3. Diversity of mysticete skulls in dorsal view: stem- and early Chaeomysticeti. (A) Phylogenetic relationships among mysticetes showing the branching order of Maiabalaena, Eomysticetidae and Sitsqwayk(red ellipses). (B) Skull of Maiabalaena nesbittae. (C) Skull of Eomysticetus whitmorei. (D) Skull of Waharowa ruwhenua. (E) Skull of Yamatocetus canaliculatus. (F) Skull of Sitsqwayk cornishorum. Not to scale. The skulls are represented as if they shared the same condylobasal length.
Within the Chaeomysticeti, the diversity of the rostrum increases in terms of transverse expansion and articulation patterns with the frontal. In Balaenoidea, two distinct patterns are observed: one is that of the Neobalaenidae, in which the rostrum is transversely expanded and greatly arched dorsoventrally [17]; the ascending process of the maxilla is reduced to a squared posteromedial projection. The other is that of the Balaenidae, in which the ascending process of the maxilla may be wide and posteriorly pointed or squared and short, and the maxilla is transversely narrowed, showing lateral borders parallel to the longitudinal axis of the skull (Figure 4). The balaenid rostra are strongly arched along the dorsoventral axis like that of the Neobalaenidae. Compared to the mysticetes in which the rostrum is mostly horizontal, the space between the ventral surface of the rostrum and the floor of the mouth cavity is massively enlarged in the Balaenoidea, and this allows the allocation of a large and muscular tongue and up to 4 m-long baleen plates [6]. In both the Balaenidae and Neobalaenidae, the squamosal is elongated dorsoventrally, and the craniomandibular joint is comparatively more ventral than in the other baleen-bearing mysticetes [18][19]. Both these families mainly filter feed upon calanoid copepods and share a number of feeding-related morphological characters (e.g., reduced coronoid process of the dentary, presence of mylohyoidal groove in the dentary and elongated baleen).
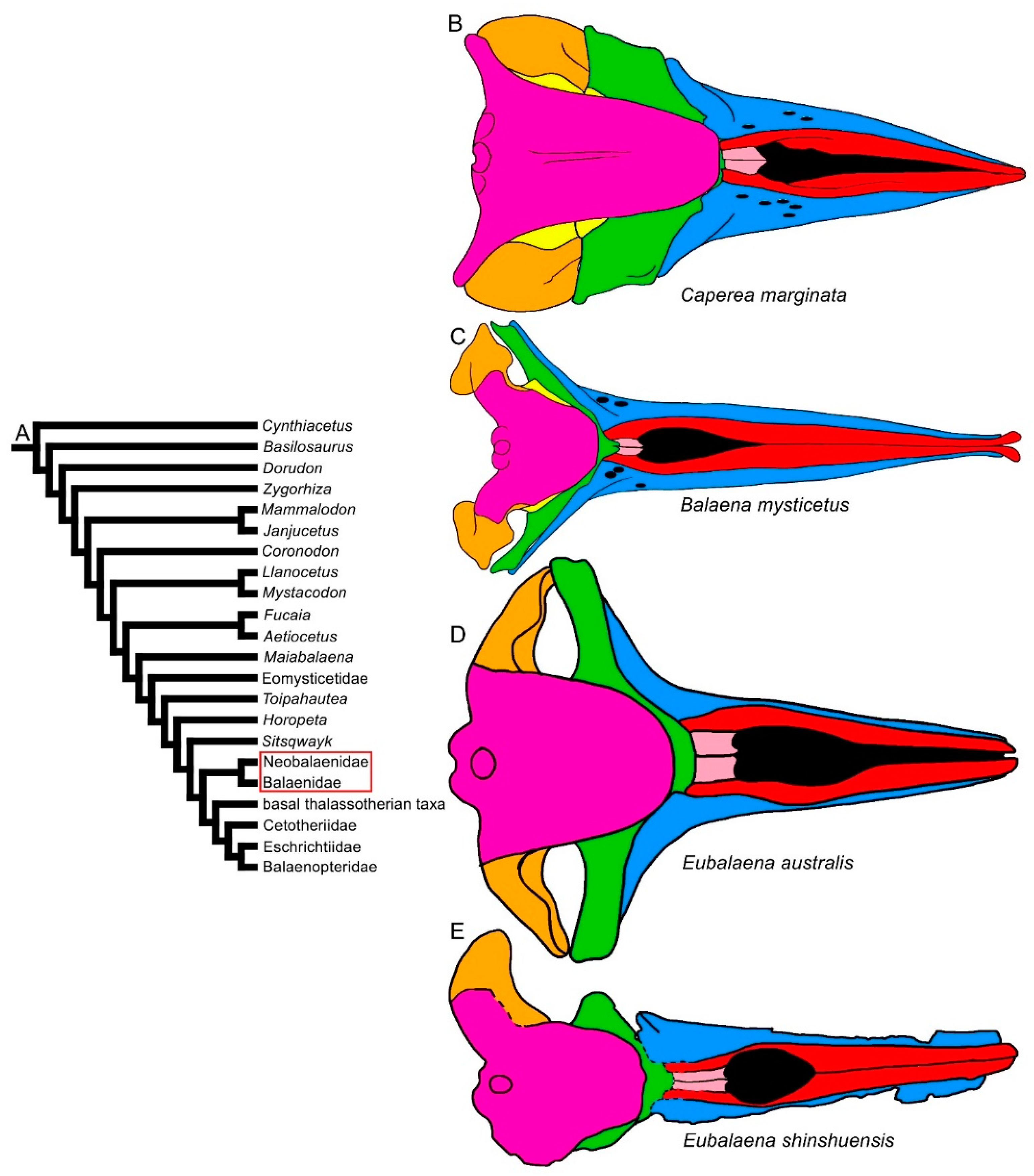
Figure 4. Diversity of balaenomorph mysticete skulls in dorsal view: Balaenoidea. (A) Phylogenetic relationships among mysticetes showing the branching order of Balaenoidea. (B) Skull of Caperea marginata. (C) Skull of Balaena mysticetus. (D) Skull of Eubalaena australis. (E) Skull of Eubalaena shinshuensis. Not to scale. The skulls are represented as if they shared the same condylobasal length.
The superfamily Thalassotherii is characterized by four families in which the rostrum is mainly horizontal and flat. The gray whales, Eschrichtiidae, are the only group within this superfamily in which the rostrum is arched in at least one species (i.e., Eschrichtius robustus). Two different patterns of articulation between rostrum and frontal are observed: (1) in the basal thalassotherian taxa, the ascending process of the maxilla is short, wide and posteriorly pointed resembling that of Eubalaena glacialis (Figure 5); and (2) in the Cetotheriidae and Balaenopteroidea, the ascending process of the maxilla is much more elongated and narrow (Figure 6 and Figure 7). These two patterns are related to the different shapes of the parietal-frontal suture that is anteriorly convex in dorsal view in the basal thalassotherian taxa and anteriorly concave in most Cetotheriidae and Balaenopteroidea. The length of the ascending process of the maxilla is especially increased in a number of cetotheriid (e.g., Piscobalaena nana, Herentalia nigra and Tranatocetus maregermanicum) and balaenopterid taxa (e.g., all the balaenopterid genera with the exception of ‘Balaenoptera’ ryaniand ‘Balaenoptera’ cortesii var. portisi), so that the parietal interdigitates with it [20][21][22][23][24][25].
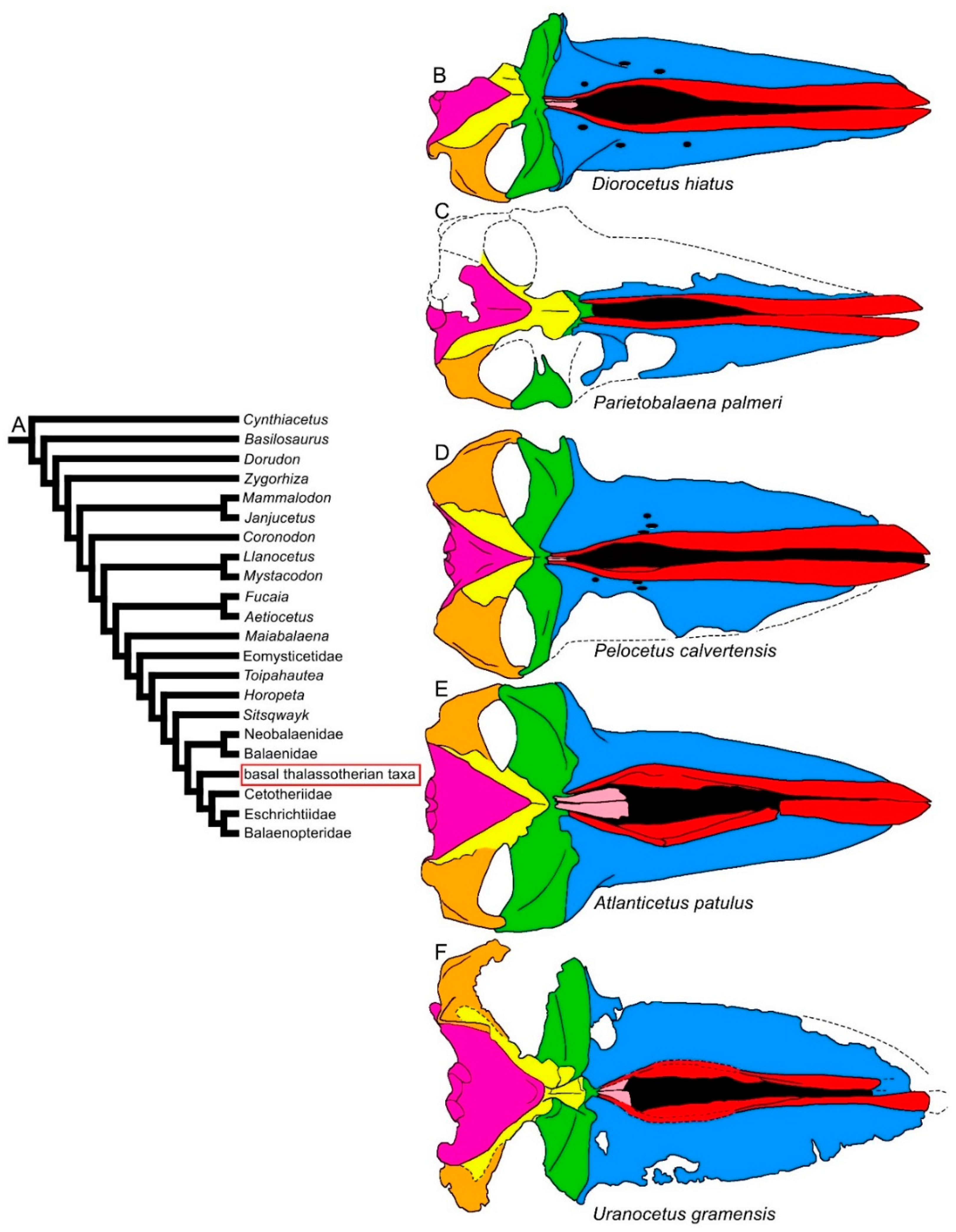
Figure 5. Diversity of balaenomorph mysticete skulls in dorsal view: basal thalassotherian taxa. (A) Phylogenetic relationships among mysticetes showing the branching order of the basal thalassotherian clade. (B) Skull of Diorocetus hiatus. (C) Skull of Parietobalaena palmeri (United States National Museum, Smithsonian Institution No. 10677). (D) Skull of Pelocetus calvertensis. (E) Skull of Atlanticetus patulus. (F) Skull of Uranocetus gramensis. Not to scale. The skulls are represented as if they shared the same condylobasal length.
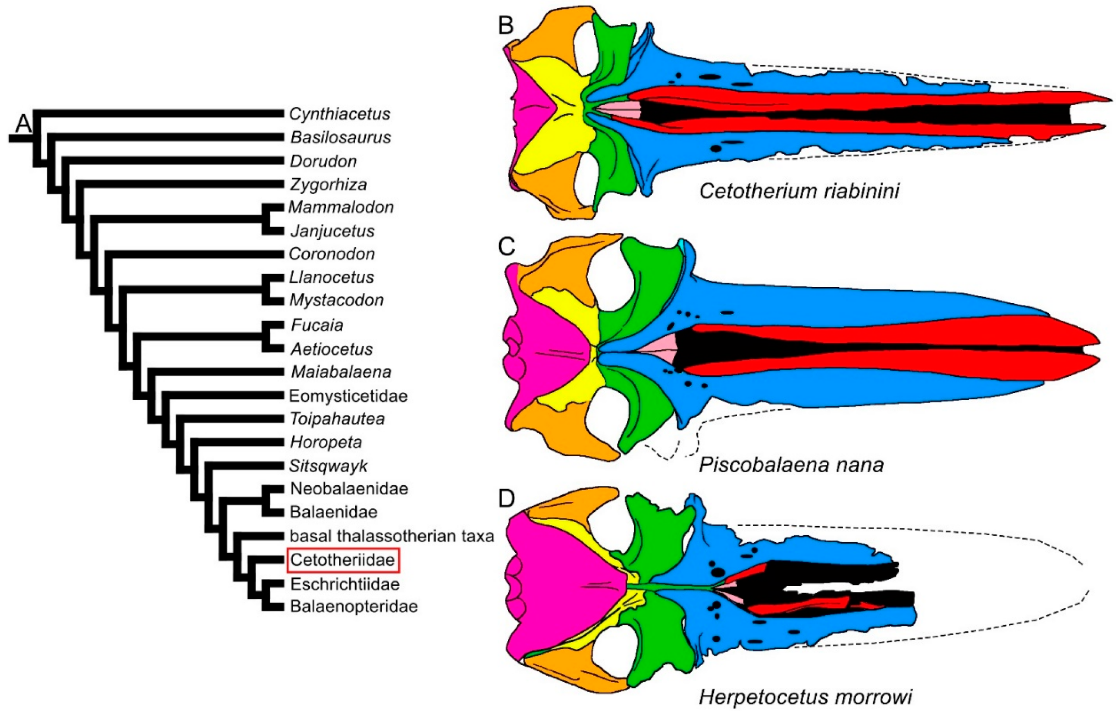
Figure 6. Diversity of balaenomorph mysticete skulls in dorsal view: Cetotheriidae. (A) Phylogenetic relationships among mysticetes showing the position of Cetotheriidae. (B) Skull of Cetotherium riabinini. (C) Skull of Piscobalaena nana. (D) Skull of Herpetocetus morrowi. Not to scale. The skulls are represented as if they shared the same condylobasal length.
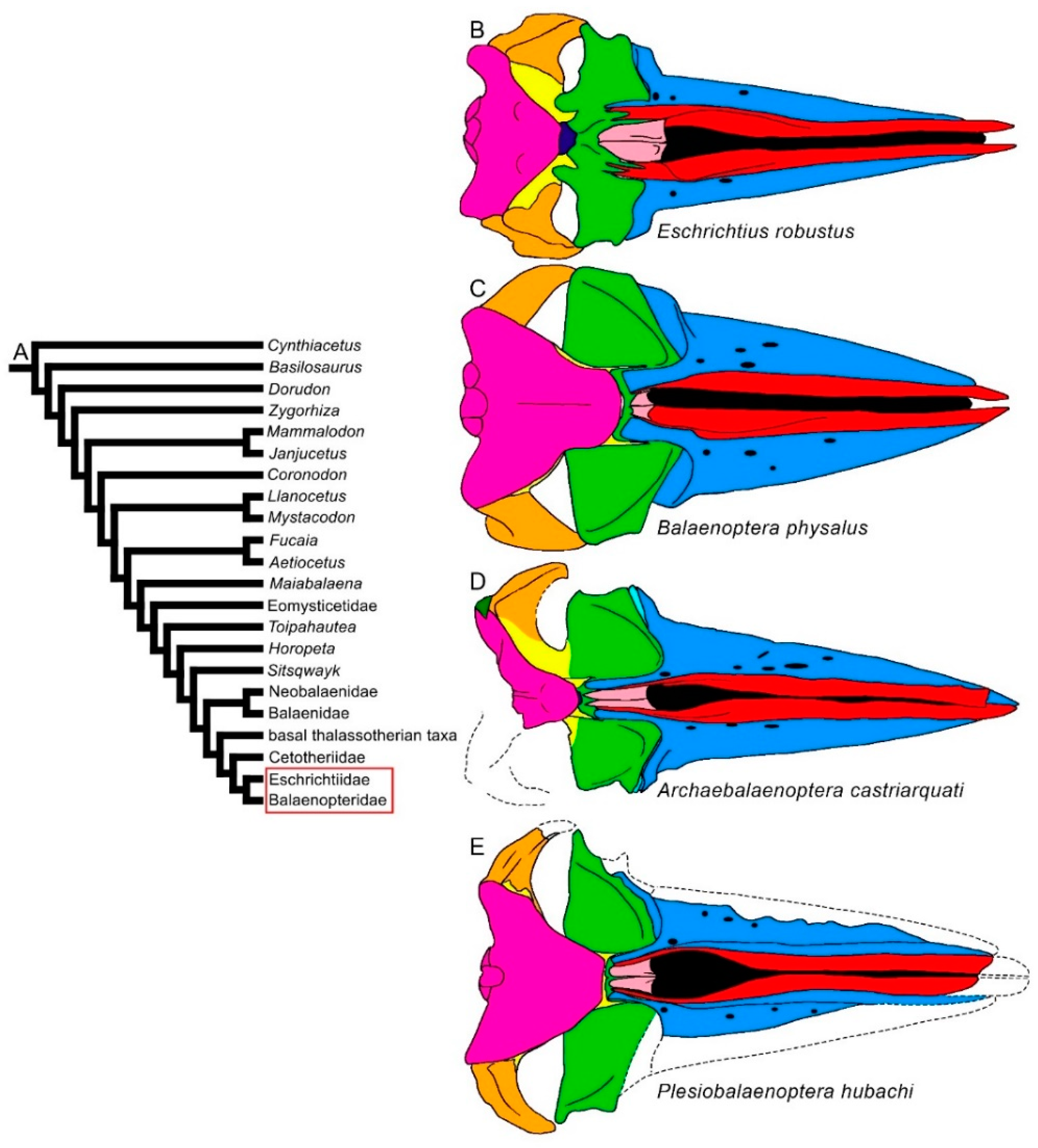
Figure 7. Diversity of balaenomorph mysticete skulls in dorsal view: Balaenopteroidea. (A) Phylogenetic relationships among mysticetes showing the position of Balaenopteroidea (Eschrichtiidae and Balaenopteridae). (B) Skull of Eschrichtius robustus. (C) Skull of Balaenoptera physalus. (D) Archebalaenoptera castriarquati. (E) Plesiobalaenoptera hubachi. Not to scale. The skulls are represented as if they shared the same condylobasal length.
The transverse width of the rostrum may be highly diverse within the Thalassotherii. In basal thalassotherian taxa, it is possible to recognize two groups (based on the phylogenetic results of [26][27][28][29]): one in which the rostrum width is particularly expanded (including Atlanticetus patulus, Pelocetus calvertensis, Isanacetus laticephalus and Uranocetus gramensis) and another in which the rostrum is comparatively narrower (including Diorocetus hiatus, Parietobalaena palmeri, P. campiniana and P. yamaokai) (see differences shown in Figure 5). A wide rostrum is related to an increased gape, suggesting that feeding adaptations were finely modulated within this monophyletic group. The same pattern is observed within the Balaenopteridae, in which broad-nosed and sharp-nosed species are observed within the same genera. Examples are the sharp-nosed Balaenoptera acutorostrata and Balaenoptera physalus and the broad-nosed Balaenoptera musculus; additional examples are the sharp-nosed Archaebalaenoptera castriarquati and the broad-nosed Archaebalaenoptera eusebioi [30][31]. The differences that can be observed in the rostrum of cetotheriids (compare, for example, the narrow rostrum of Cetotherium riabinini and the broader rostrum of Piscobalaena nana and Herpetocetus morrowi in Figure 6) may be related to similar functional differences, suggesting that different feeding adaptations were present in this extinct family. Additional work is still needed, however, to determine the fine differences in the rostra of these taxa.
Long nasal bones are present in early-diverging balaenopterids, such as Archaebalaenoptera castriarquati and Parabalaenoptera baulinensis [18][32], although the nasal bones are strongly reduced in both length and width in all the other Balaenomorpha. In the Cetotheriidae and Balaenopteroidea, most of the taxa show the nasofrontal suture located well within the interorbital region of the frontal, a character not observed in the Balaenoidea and basal thalassotherian taxa. In Aetiocetidae, the same trend is observed as the nasofrontal suture is located at the anterior border of the interorbital region of the frontal in Fucaia and Aetiocetus cotylalveus but it is located within the interorbital region of the frontal in Aetiocetus polydentatus.
In conclusion, the examination of the morphological diversity of the rostrum in the fossil record of baleen whales shows that the earliest mysticetes were equipped with most of the morphological characters observed in later-diverging clades (articulation between rostrum and frontal provided by the ascending process of the maxilla and premaxilla, presence of mesorostral groove, lateral process of the maxilla, infraorbital process of the maxilla and multiple dorsal maxillary foramina). The rostrum-length-to-skull-length ratio increased in early mysticetes but only in the Eomysticetidae is the rostrum markedly longer than the neurocranium, a character present in all Balaenomorpha probably related to a peramorphic process as discussed by [33][34][35]. Rostrum diversity expands to Balaenomorpha, where different patterns of articulation between rostrum and frontal and different maxillary and premaxillary morphologies are observed, possibly related to different feeding strategies [5].
3. Vertex
According to ([36], p. 155), “The vertex has assumed a new role in the cetacean skull as a result of the posterior movement of the external nares. The vertex in terrestrial mammals was a craniometric point, defined as the most dorsal point on the skull, and did not have a definite function. In cetaceans […] it serves as a nexus for many groups of muscles. The vertex serves as a foundation on which the muscles that control the movements of the nasal passage and associated diverticula rely. The vertex is primarily centered on the nasal bones. In groups in which the vertex is more highly developed, […] the vertex consists of the nasals and surrounding portions of the maxillae, premaxillae, and frontals.”
Comparative illustrations of the mysticete vertex have been provided by [31] for the balaenopterid genus Archaebalaenoptera, [37] for Cetotheriidae, [17] for the neobalaenidCaperea and Miocaperea[38] and for some species of extant Balaenoptera.
Early-diverging mysticetes (i.e., Mammalodontidae, Aetiocetidae, Llanocetus, Mystacodon, Maiabalaena and Coronodon) and early chaeomysticetes (i.e., Eomysticetidae and Sitsqwayk) show archaeocete-like morphological patterns of the vertex in that the nasofrontal suture is located at the anterior border of the interorbital region of the frontal (with the exception of Aetiocetus polydentatus, in which the nasofrontal suture is located well within the interorbital region of the frontal); the anterior border of the nasal is located at a considerable distance from the frontal, reaching a point located between one-third and half of the length of the maxilla; the ascending process of the maxilla is present, narrow and triangular in outline, forming an evident angle with the posterolateral border of the maxilla; the frontal is flat and wide along both the anteroposterior and the transverse axes; the frontal-parietal (coronal) suture is developed transversely; and the postorbital constriction is transversely narrow and long.
This pattern undergoes numerous changes in the Balaenomorpha in all the structures mentioned above. The nasofrontal suture is retained at the anterior border of the interorbital region of the frontal in the Balaenoidea and basal thalassotherian taxa but it is located more posteriorly within the interorbital region of the frontal in the Cetotheriidae and Balaenopteroidea. The posterior development of the nasofrontal suture is paralleled by an anterior shift of the anterior border of the parietal, which in Balaenopteroidea and some cetotheriid taxa is located more anteriorly than the posterior-most point of the maxilla. This configuration results in an anteriorly concave coronal suture and in a massive reduction in the size of the interorbital region of the frontal, which may be completely superimposed by the posteromedial elements of the rostrum (ascending process of maxilla, ascending process of the premaxilla and nasal). This morphological pattern is traditionally known as ‘telescoping’ [24][39] and was recently reviewed by [40], who analyzed the possible developmental mechanisms underlying suture formation in telescoped cetacean skulls.
In the Balaenoidea and basal thalassotherian taxa, the coronal suture is anteriorly convex. In the Balaenoidea, it is completely superimposed by a massive anterior projection of the supraoccipital that reaches the posterior portion of the interorbital region of the frontal and, consequently, a shortening of the interorbital region of the frontal is due to the anterior thrust of the supraoccipital that excludes the parietal from being observed in dorsal view (Figure 4). Moreover, the ascending process of the maxilla is reduced in length. The balaenoid pattern is also observed in the earliest known balaenoid species, Morenocetus parvus, which shows a less advanced degree of bone overlap between the rostrum and frontal so that the interorbital region of the frontal is anteroposteriorly longer in comparative terms than in other, later balaenoid taxa. Despite the longer interorbital region of the frontal, the ascending process of the maxilla is short, as suggested by the extent of the articular groove between maxilla and frontal, even though there would be enough space to allocate a longer ascending process of the maxilla. This pattern is in contrast with the hypothesis that the length of the interorbital region of the frontal is related to the available space in the interorbital region of the frontal suggested by [41] and indicates that different developmental mechanisms may be responsible for the patterning of these two anatomical structures.
In basal thalassotherian taxa, the coronal suture is anteriorly triangular, and the parietal is superimposed on the posterior portion of the interorbital region of the frontal. In this way, the interorbital region is consistently shortened along the anteroposterior axis being constricted between the anterior border of the parietal posteriorly and by the wide, triangular and short ascending processes of the maxilla anteriorly (Figure 5).
In conclusion, in early mysticetes, the vertex closely resembles that of the basilosaurine and dorudontine archaeocetes. The articulation between the rostrum and the frontal is the major difference since in early mysticetes it is realized exclusively by the ascending process of maxilla and premaxilla, and the posterolateral border of the maxilla and the anterior border of the frontal are spaced. At the same time, it was concluded that the evolution of the rostrum, including the vertex structure, underwent a remarkable diversification in the Balaenomorpha with the evolution of two main styles, as follows: (1) anteriorly convex coronal suture, short and wide ascending process of the maxilla and premaxilla in the Balaenoidea and basal thalassotherian taxa; and (2) anteriorly concave coronal suture, long and narrow ascending process of the maxilla in Cetotheriidae and Balaenopteroidea. Fine differences are then observed within these two main patterns: (i) development of the superimposition of the supraoccipital on the posterior portion of the interorbital region of the frontal in the Balaenoidea but not in basal thalassotherian taxa, and (ii) the ascending processes of the maxilla meet along the longitudinal axis of the skull in the Cetotheriidae but not in the Balaenopteroidea.
4. Temporal Fossa
In basilosaurine and dorudontine archaeocetes, the temporal fossa has an anteroposteriorly elongate, oval shape (Figure 1). The anterior portion of the temporal crest for the attachment of the temporalis muscle is located at the posterodorsal edge of the flat supraorbital process of the frontal and continues along a sagittal crest located on the top of the skull up to the anterior edge of the supraoccipital. The temporal crest projects posterolaterally to a posterior apex named the nuchal crest. Finally, the temporal crest projects anterolaterally from the nuchal crest along the dorsal edge of the squamosal where it may form a prominent supramastoid crest. In the archaeocetes illustrated in Figure 1, the nuchal crest is triangular and protrudes posteriorly, reaching a point that is located more posteriorly than the occipital condyle. This pattern is retained in some of the earliest-diverging mysticetes, such asCoronodon, Llanocetus and Mystacodon, and in some balaenomorph mysticetes, such as the basal thalassotherians Pelocetus, Atlanticetus and Uranocetus (Figure 5).
The position of the orbitotemporal crest at the posterodorsal edge of the supraorbital process of the frontal is retained in all the toothed mysticetes (Mammalodontidae, Aetiocetidae, Coronodon, Llanocetus and Mystacodon) and eomysticetes. Among the Balaenomorpha, only the Cetotheriidae retains this character. In all the other balaenomorph baleen whales, the orbitotemporal crest shows different shapes. In the Balaenoidea and basal thalassotherian taxa, the orbitotemporal crest is developed from an anteromedial point to a point close to the postorbital process, diagonally crossing the supraorbital process of the frontal (Figure 5E). In the Balaenopteroidea, the orbitotemporal crest forms a wide posterior concavity because most of its development occurs close to the anterior border of the supraorbital process of the frontal.
The temporal crest (which runs parallel to the lateral borders of the supraoccipital) overhangs the temporal fossa and prevents the posterior portion of the parietal and the anterior part of the squamosal from being exposed in dorsal view in the Balaenoidea and late Balaenopteridae. As observed in the advanced archaeocetes, toothed mysticetes, early chaeomysticetes, basal thalassotherian taxa and cetotheriids, the plesiomorphic condition consists of a temporal crest that does not overhang the temporal fossa and the posterior parietal so that the anterior part of the squamosal can be observed in dorsal view. Even the earliest known balaenoid species, Morenocetus parvus, shows the plesiomorphic state, in which the temporal crest does not overhang the temporal fossa [42]. In the Balaenopteridae, early-diverging species (e.g., Archaebalaenoptera castriarquati, ‘Balaenoptera’ ryani) may show the plesiomorphic condition but, in general, the temporal crest does overhang the temporal fossa like in extant and fossil balaenoids (with the exclusion of M. parvus).
In the Balaenomorpha, the anteroposterior length of the temporal fossa is visibly diminished, paralleling the shortening of the intertemporal constriction. The geometry of the temporalis muscle and the shape of the ensemble formed by the orbitotemporal, temporal, nuchal and supramastoid crests show morphological patterns that are largely different from those observed in nonbalaenomorph mysticetes. Again, the origin of the Balaenomorpha marked the beginning of a diversification process in mysticete morphology.
5. Occipital Region
In ([40], p. 1062), Roston and Roth define the term telescoping in reference to skulls that have “(i) extensive bone overlap and (ii) extreme proximity of anterior and posterior cranial elements that is observed in modern cetaceans”. In mysticetes, the second feature is attained by a posterior elongation of the posteromedial elements of the rostrum together with an anterior placement of the anterior border of the supraoccipital. In the earliest-diverging mysticetes, such as the Mammalodontidae, Mystacodon, Coronodon, Fucaia and some aetiocetids (including Aetiocetus cotylalveus), the anterior apex of the supraoccipital is located on a transverse line crossing the skull posteriorly to the posterior border of the temporal fossa. The advanced aetiocetid Aetiocetus polydentatus, however, shows a more anterior placement of the anterior apex of the supraoccipital, which is located anteriorly to the posterior border of the temporal fossa. This pattern is observed in eomysticetids, Maiabalaena and Sitsqwaykand is retained by balaenomorph mysticetes. In all the nonbalaenomorph mysticetes, the anterior apex of the supraoccipital is located more posteriorly than the anterior end of the zygomatic process of the squamosal. This pattern is not retained in balaenomorph mysticetes, in which the supraoccipital reaches a point that is more anterior than the anterior end of the zygomatic process of the squamosal. The cetotheriids are an exception in that they are the only balaenomorph family in which the anterior apex of the supraoccipital is posterior to the anterior end of the zygomatic process of the squamosal [24][39]. The origin of the Balaenomorpha marks a major shift in supraoccipital development with a supraoccipital that develops more anteriorly, superimposing onto the cranial vault and reducing the anteroposterior extent of the parietal exposure in dorsal view. In the case of the Balaenoidea, the supraoccipital completely excludes the parietal from dorsal exposure. In the Balaenopteroidea, a complex system of sutures occurs at the supraoccipital–parietal interface; the eschrichtiids show the presence of a bony block interposed between the anterior end of the supraoccipital and the interorbital region of the frontal that was interpreted as an interparietal [43], which is not separated from the supraoccipital itself. This structure forms a platform anterior to the anterior border of the supraoccipital and replaces the sagittal crest observed in the archaeocetes, early-diverging mysticetes and basal thalassotherian taxa with a couple of externally concave attachment sites for the temporalis muscle connected to the orbitotemporal crest anteriorly and to the temporal crest posteriorly. This same morphological pattern is observed in the Cetotheriidae. In the Balaenopteridae, the intertemporal constriction is transversely wider, and the interparietal, when present, is anteroposteriorly reduced to a thin sheet of bone interposed between the anterior border of the supraoccipital and the interorbital region of the frontal (see [38] for an overview).
The anterior border of the supraoccipital is wide and round in archaeocetes, and the supraoccipital is mainly developed along the dorsoventral axis. A rounded outline is also observed in the Mammalodontidae (Figure 1), Fucaia and Aetiocetus cotylalveus, but a triangular outline is present inLlanocetus, Mystacodon, Coronodon and Aetiocetus polydentatus (Figure 2). The triangular shape is shared with later chaeomysticetes, including the Eomysticetidae, basal thalassotherian taxa, early balaenopterids and some cetotheriids. The triangular shape of the supraoccipital is thus an evolutionary novelty that originated in the period between the origin of the Mysticeti and the origin of the Balaenomorpha.
Another evolutionary novelty that appeared independently in different balaenomorph clades is the transverse expansion of the intertemporal constriction, which can be related to the increase in the transverse width of the anterior border of the supraoccipital in the advanced balaenopterids and in Eubalaena. This character was discussed in [16] in relation to the anterior expansion of the forebrain of the extant Balaenopteridae. Such an expansion is observed in later balaenopterids (genera Balaenoptera, Megaptera, Norrisanima and Diunatans) and in the Balaenoidea.
Several accounts illustrated the posterior view of archaeocete (e.g., [44][45]) and mysticete (e.g., [11][28][46][29][47][17][48][49] skulls. Based on these accounts, it is possible to observe that the exoccipital protrudes laterally and forms an approximately right angle with the supraoccipital in archaeocetes. In the mysticetes, the exoccipital protrusion is less marked as the posterior width of the supraoccipital is wider compared to that of archaeocetes, and the angle is obtuse. In many cases (e.g., balaenids) the angle cannot be observed because the lateral border of the supraoccipital and the borders of the exoccipital are continuous.
In more general terms, in dorsal view, the archaeocete occipital region shows a transverse constriction at the level of the nuchal crest (Figure 1A–C); this constriction was already lost in the earliest-diverging mysticetes (Mammalodontidae) because of the larger distance between the posterior apices of the nuchal crest (Figure 1D,E).
Finally, a change in supraoccipital orientation occurred at the Mysticeti node: in the archaeocetes, the supraoccipital is mostly developed along the dorsoventral axis, but in the mysticetes it projects anteriorly and superimposes on the parietal. The anterior shift of the supraoccipital is one of the key points related to the concept of telescoping in the cetacean skull [24][39][40].
6. Earbones
Extensive descriptions and comparisons of the mysticete earbones have been published in the last decade, including, for example, those of [50][51][52][53]. Periotic and cochlear morphologies have been intensely studied by CT scans and 3D renderings showing resemblances between the mysticetes and later archaeocetes [53]. Tomographic analyses of the mammalodontid and aetiocetid cochleae revealed that the earliest-diverging mysticetes had cochlear morphologies similar to those of the archaeocetes rather than those of the chaeomysticetes [52]. This observation suggests that both the advanced archaeocetes and the earliest-diverging mysticetes could share the characteristic of low-frequency hearing with the Chaeomysticeti [52].
This suggestion is reinforced by bullar morphology. In Figure 8, schematic representations of the tympanic bullae of the archaeocetes and mysticetes are plotted around the mysticete phylogeny adopted in this entry. As shown, the archaeocetes and early-diverging mysticetes share the absence of the anterior lobe and the presence of an extended median furrow. In general, the bulla of archaic, nonbalaenomorph mysticetes is morphologically similar to that of the basilosaurine and dorudontine archaeocetes [34]. In a group of basal thalassotherian taxa (including the genera Diorocetus and Parietobalaena) and in the cetotheriids, the tympanic bulla retains the lack of the anterior lobe; the anterior lobe occurs in a diverse group of basal thalassotherian taxa (including the genera Atlanticetus and Uranocetus), in the Balaenoidea and in the Balaenopteroidea. Observing the distribution of this character in the phylogeny of Figure 8, it appears that the anterior lobe evolved independently in these three lineages. Further work is necessary to understand the function of the anterior lobe of the tympanic bulla because it is still unclear what function this structure may have in the hearing physiology of the mysticetes.
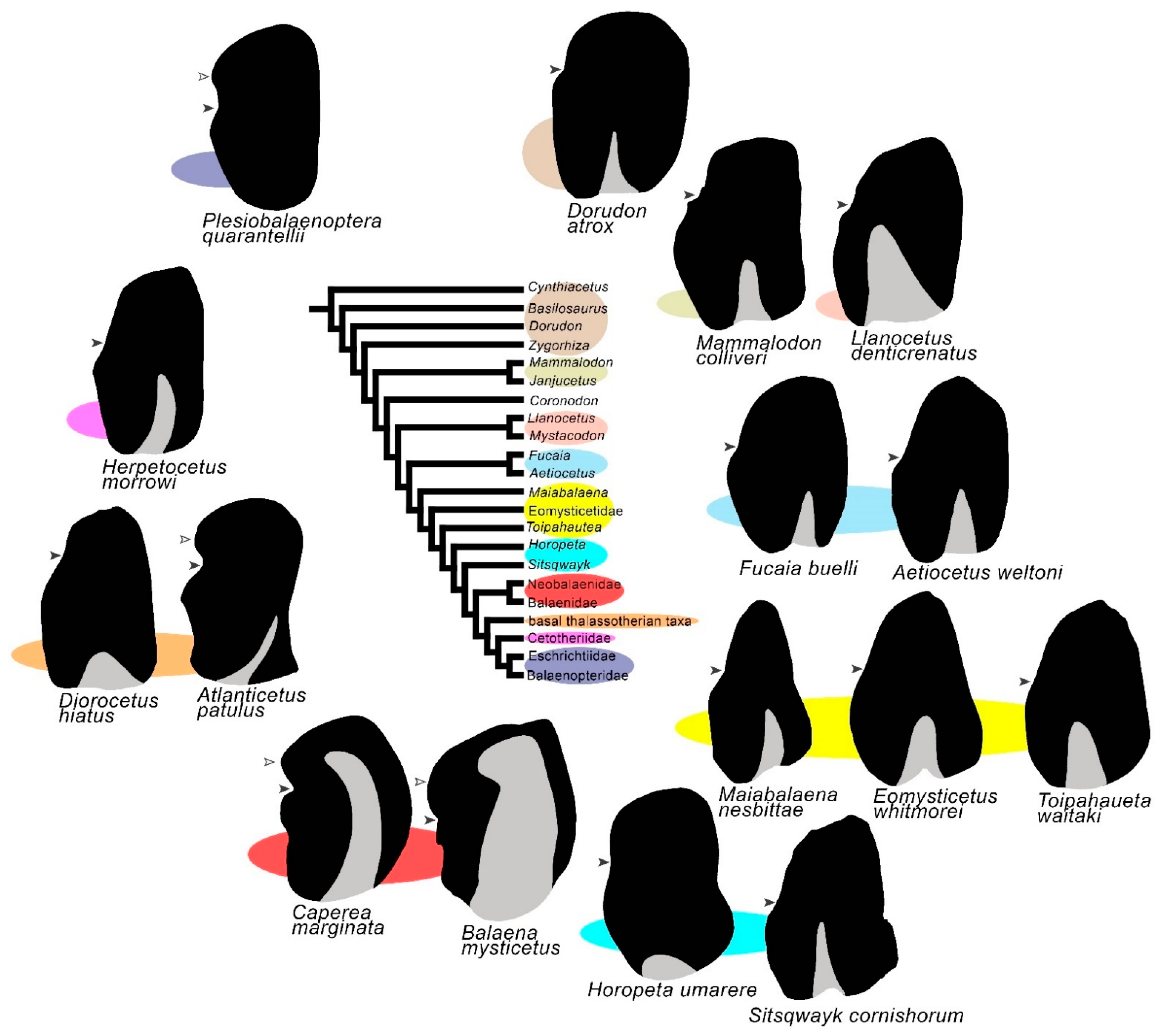
Figure 8. Schematic representations of tympanic bullae of advanced archaeocetes and mysticetes in relation to mysticete phylogeny. The black arrowhead indicates the position of the lateral furrow; the gray triangle indicates the position and extent of the median furrow; the white arrowhead indicates the anterior lobe in Balaenopteridae, Balaenoidea and in a group of basal thalassotherian taxa (here represented by Atlanticetus patulus). Note the similar extent of the median furrow and the anterior widening of the tympanic bulla in neobalaenids and balaenids. Color ellipses link bullae to the corresponding taxa in the cladogram.
In conclusion, the earliest mysticetes share a number of characters of both the tympanic bulla and the cochlea, with the later archaeocetes suggesting that they shared similar hearing characteristics. Evident structural changes occurred in the tympanic bulla after the divergence of the main balaenomorph clades with the recurrent evolution of the anterior lobe in the tympanic bulla.
References
- Bannister, J.D. Baleen whales. In Encyclopedia of Marine Mammals; Perrin, W.F., Würsig, B., Thewissen, J.G.M., Eds.; Academic Press: New York, NY, USA, 2002; pp. 62–72.
- Savoca, M.S.; Czapanskiy, M.F.; Kahane-Rapport, S.R.; Gough, W.T.; Fahlbusch, J.A.; Bierlich, K.C.; Segre, P.S.; Di Clemente, J.; Penry, G.S.; Wiley, D.N.; et al. Baleen whale prey consumption based on high-resolution foraging measurements. Nature 2021, 599, 85–90.
- Pershing, A.J.; Christensen, L.B.; Record, N.R.; Sherwood, G.D.; Stetson, P.B. The impact of whaling on the oceancarbon cycle: Why bigger was better. PLoS ONE 2010, 5, e12444.
- Pershing, A.J.; Stamieszkin, K. The North Atlantic ecosystem, from plankton to whales. Annu. Rev. Mar. Sci. 2020, 12, 339–359.
- Sanderson, L.R.; Wassersug, R. Convergent and alternative designs for vertebrate suspension feeding. In The Skull. Functional and Evolutionary Mechanisms; Hanken, J., Hall, B.K., Eds.; University of Chicago Press: Chicago, IL, USA, 1993; Volume 3, pp. 37–112.
- Werth, A.J. Feeding in marine mammals. In Feeding: Form, Function and Evolution in Tetrapod Vertebrates; Schwenk, K., Ed.; Academic Press: New York, NY, USA, 2000; pp. 475–514.
- Deméré, T.A.; McGowen, M.R.; Berta, A.; Gatesy, J. Morphological and molecular evidence for a stepwise evolutionary transition from teeth to baleen in mysticete whales. Syst. Biol. 2008, 57, 15–37.
- Ekdale, E.G.; Deméré, T.A. Neurovascular evidence for a co-occurrence of teeth and baleen in an Oligocene mysticete and the transition to filter-feeding in baleen whales. Zool. J. Linn. Soc. 2021, 194, 395–415.
- Marx, F.D.; Hocking, D.P.; Park, T.; Ziegler, T.; Evans, A.R.; Fitzgerald, E.M.G. Suction feeding preceded filtering in baleen whale evolution. Mem. Mus. Vic. 2016, 75, 71–82.
- Geisler, J.H.; Boessenecker, R.W.; Brown, M.; Beatty, B.L. The origin of filter feding in whales. Curr. Biol. 2017, 27, 2036–2042.e2.
- Fitzgerald, E.M.G. The morphology and systematic of Mammalodon colliveri (Cetacea: Mysticeti), a toothed mysticete from the Oligocene of Australia. Zool. J. Linn. Soc. 2010, 158, 367–476.
- Boessenecker, R.W.; Churchill, M.; Buchholtz, E.A.; Beatty, B.L.; Geisler, J.H. Convergent Evolution of Swimming Adaptations in Modern Whales Revealed by a Large Macrophagous Dolphin from the Oligocene of South Carolina. Curr. Biol. 2020, 30, 3267–3273.e2.
- Cooper, L.N.; Berta, A.; Dawson, S.D.; Reidenberg, J.S. Evolution of hyperphalangy and digit reduction in the cetacean manus. Anat. Rec. 2007, 290, 654–672.
- Benke, H. Investigations on the osteology and the functional morphology of the flipper of whales and dolphins (Cetacea). Investig. Cetacea 1993, 24, 9–252.
- Bisconti, M.; Damarco, P.; Tartarelli, G.; Pavia, M.; Carnevale, G. A natural endocast of an early Miocene odontocete and its implications in cetacean brain evolution. J. Comp. Neurol. 2021, 529, 1198–1227.
- Bisconti, M.; Daniello, R.; Damarco, P.; Tartarelli, G.; Pavia, M.; Carnevale, G. High encephalization in a fossil rorqual illuminates baleen whale brain evolution. Br. Behav. Evol. 2021, 96, 78–90.
- Bisconti, M. Comparative osteology and phylogenetic relationships of Miocaperea pulchra, the first fossilpygmy right whale genus and species (Cetacea, Mysticeti, Neobalaenidae). Zool. J. Linn. Soc. 2012, 166, 876–911.
- Bisconti, M. Evolutionary history of Balaenidae. Cranium 2003, 20, 9–50.
- McLeod, S.A.; Whitmore, F.C., Jr.; Barnes, L.G. Evolutionary relationships and classification. In The Bowhead Ehale; Burns, J.J., Montague, J.J., Cowles, C.J., Eds.; Society for Marine Mammalogy: Yarmouth Port, MA, USA, 1993; Volume 2, pp. 45–70.
- Marx, F.G.; Post, K.; Bosselaers, M.; Munsterman, D. A large Late Miocene cetotheriid (Cetacea, Mysticeti) from the Netherlands clarifies the status of Tranatocetidae. PeerJ 2019, 7, e6426.
- Bisconti, M. Anatomy of a new cetotheriid genus and species from the Miocene of Herentals, Belgium, and the phylogenetic and paleobiogeographic relationships of Cetotheriidae s.s. (Mammalia, Cetacea, Mysticeti). J. Syst. Palaeontol. 2015, 13, 377–395.
- Bouetel, V.; de Muizon, C. The anatomy and relationships of Piscobalaena nana (Cetacea, Mysticeti), a Cetotheriidae s.s. from the early Pliocene of Peru. Geodiversitas 2006, 28, 319–395.
- Hanna, G.D.; McLellan, M. A new species of whale from the type locality of the Monterey Group. Proc. Calif. Acad. Sci. 1924, 13, 237–241.
- Kellogg, R. The history of whales—Their adaptation to life in the water. Q. Rev. Biol. 1928, 3, 29–76.
- Portis, A. Catalogo descrittivo dei Talassoterii rinvenuti nei terreni terziari del Piemonte e della Liguria. Mem. R. Acc. Sci. Torino 1884, 37, 247–365.
- Bisconti, M.; Pellegrino, L.; Carnevale, G. Evolution of gigantism in right and bowhead whales (Cetacea: Mysticeti: Balaenidae). Biol. J. Linn. Soc. 2021, 134, 498–524.
- Bisconti, M.; Damarco, P.; Mao, S.; Pavia, M.; Carnevale, G. The earliest baleen whale from the Mediterranean: Large-scale implications of an early Miocene thalassotherian mysticete from Piedmont, Italy. Pap. Palaeontol. 2021, 7, 1147–1166.
- Bisconti, M.; Bosselaers, M. A new balaenopterid species from the southern North Sea Basin informs about phylogeny and taxonomy of Burtinopsis and Protororqualus (Cetacea, Mysticeti, Balaenopteridae). PeerJ 2020, 8, e9570.
- Bisconti, M.; Damarco, P.; Pavia, M.; Sorce, B.; Carnevale, G. Marzanoptera tersillae, a new balaenopterid genus and species from the Pliocene of Piedmont, north-west Italy. Zool. J. Linn. Soc. 2021, 192, 1253–1292.
- Bisconti, M. A new basal balaenopterid from the Early Pliocene of northern Italy. Palaeontology 2007, 50, 1103–1122.
- Bisconti, M.; Ochoa, D.; Urbina, M.; Salas-Gismondi, R. Archaebalaenoptera eusebioi, a new rorqual from the late Miocene of Peru (Cetacea, Mysticeti, Balaenopteridae) and its impact in reconstructing body size evolution, ecomorphology and paleobiogeography of Balaenopteridae. J. Syst. Palaeontol. 2022, in press.
- Zeigler, C.V.; Chan, G.L.; Barnes, L.G. A new Late Miocene balaenopterid whale (Cetacea: Mysticeti), Parabalaenoptera baulinensis, (new genus and species) from the Santa Cruz Mudstone, Point Reyes Peninsula, California. Proc. Calif. Acad. Sci. USA 1997, 50, 115–138.
- Boessenecker, R.W.; Fordyce, R.E. A new eomysticetid from the Oligocene Kokoamu Greensand of New Zealand and a review of the Eomysticetidae (Mammalia, Cetacea). J. Syst. Palaeontol. 2016, 15, 429–469.
- Boessenecker, R.W.; Fordyce, R.E. A new genus and species of eomysticetid (Cetacea: Mysticeti) and a reinterpretation of “Mauicetus” lophocephalus Marples, 1956: Transitional baleen whales from the upper Oligocene of New Zealand. Zool. J. Linn. Soc. 2015, 175, 607–660.
- Boessenecker, R.W.; Fordyce, R.E. Anatomy, feeding ecology, and ontogeny of a transitional baleen whale: A new genus and species of Eomysticetidae (Mammalia: Cetacea) from the Oligocene of New Zealand. PeerJ 2015, 3, e1129.
- Mead, J.G.; Fordyce, R.E. The therian skull: A lexicon with emphasis on the odontocetes. Smiths. Contrib. Zool. 2009, 627, 1–216.
- Gol’Din, P.; Steeman, M.E. From problem taxa to problem solver: A new Miocene family, Tranatocetidae, brings perspective on baleen whale evolution. PLoS ONE 2015, 10, e0135500.
- Wada, S.; Oishi, M.; Yamada, T.K. A newly discovered species of living baleen whale. Nature 2003, 426, 278–281.
- Miller, G.S. The telescoping of the cetacean skull. Smiths. Misc. Coll. 1923, 76, 1–70.
- Roston, R.A.; Roth, V.L. Cetacean Skull Telescoping Brings Evolution of Cranial Sutures into Focus. Anat. Rec. 2019, 302, 1055–1073.
- Marx, F.G.; Fordyce, R.E. A link no longer missing: New evidence for the cetotheriid affinities of Caperea. PLoS ONE 2016, 11, e0164059.
- Buono, M.R.; Fernández, M.S.; Cozzuol, M.A.; Cuitiño, J.I.; Fitzgerald, E.M.G. The early Miocene balaenid Morenocetus parvus from Patagonia (Argentina) and the evolution of right whales. PeerJ 2018, 5, e4148.
- Tsai, C.-H.; Fordyce, R.E. Disparate heterochronic processes in baleen whale evolution. Evol. Biol. 2014, 41, 299–307.
- Uhen, M.D. Form, function, and anatomy of Dorudon atrox (Mammalia, Cetacea): An archaeocete from the middle to late Eocene of Egypt. Univ. Michigan P. Palaeont. 2004, 34, 1–238.
- Kellogg, R. A review of the Archaeoceti. Carnegie Inst. Wash. Publ. 1936, 482, 1–455.
- Kellogg, R. Fossil marine mammals from the Miocene Calvert Formation of Maryland and Virginia. US Natl. Mus. Bull. 1968, 247, 103–197.
- Bisconti, M.; Bosselaers, M. Fragilicetus velponi: A new mysticete genus and species and its implications for the origin of Balaenopteridae (Mammalia, Cetacea, Mysticeti). Zool. J. Linn. Soc. 2016, 177, 450–474.
- Gol’Din, P.; Startsev, D. Brandtocetus, a new genus of baleen whales (Cetacea, Cetotheriidae) from the late Miocene of Crimea, Ukraine. J. Vert. Paleontol. 2014, 34, 419–433.
- Kellogg, R. A new whalebone whale from the Miocene Calvert Formation. US Natl. Mus. Bull. 1965, 247, 1–45.
- Ekdale, E.G.; Berta, A.; Deméré, T.A. The comparative osteology of the petrotympanic complex (ear region) of extant baleen whales (Cetacea: Mysticeti). PLoS ONE 2011, 6, e21311.
- Ritsche, I.S.; Fahlke, J.M.; Wieder, F.; Hilger, A.; Manke, I.; Hampe, O. Relationships of cochlear coiling shape and hearing frequencies in cetaceans, and the occurrence of infrasonic hearing in Miocene Mysticeti. Foss. Rec. 2018, 21, 33–45.
- Park, T.; Evans, A.R.; Gallagher, S.J.; Fitzgerald, E.M.G. Low-frequency hearing preceded the evolution of giant body size and filter feeding in baleen whales. Proc. R. Soc. B 2017, 284, 20162528.
- Ekdale, E.G. Morphological Variation Among the Inner Ears of Extinct and Extant Baleen Whales (Cetacea: Mysticeti). J. Morphol. 2016, 277, 1599–1615.
More
Information
Subjects:
Paleontology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.0K
Revisions:
2 times
(View History)
Update Date:
06 Apr 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





