| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Diaconeasa Zorita | + 2673 word(s) | 2673 | 2020-09-16 05:48:29 | | | |
| 2 | Bruce Ren | + 1 word(s) | 2674 | 2020-09-23 02:55:24 | | |
Video Upload Options
Anthocyanins are water-soluble vacuolar pigments that occur ubiquitously in the plant kingdom, and they are widely distributed in fruits and vegetables as glycosides, having different sugars, such as glucose, rhamnose, xylose or arabinose, attached to an aglycon nucleus. Till now have been shown to have antioxidant properties in vitro and in vivo.
This work aim to provide an up-to-date overview regarding anthocyanins as functional molecules and their chemopreventive effects on melanoma in vitro and in vivo as well as a comprehensive description of major sources of anthocyanins.
Until today, numerous studies evaluated the topic of anthocyanins and various types of cancer, regarding the anthocyanins’ preventative and inhibitory effects, underlying molecular mechanisms, and such. However, there is no targeted review available regarding the anticarcinogenic effects of dietary anthocyanins on skin cancers. If diagnosed at the early stages, the survival rate of skin cancer is quite high. Nevertheless, the metastatic form has a short prognosis. In fact, the incidence of melanoma skin cancer, the type with high mortality, has increased exponentially over the last 30 years, causing the majority of skin cancer deaths. Malignant melanoma is considered a highly destructive type of skin cancer due to its particular capacity to grow and spread faster than any other type of cancers. Plants, in general, have been used in disease treatment for a long time, and medicinal plants are commonly a part of anticancer drugs on the market.
1. Introduction
Today, it is a generally accepted concept that including fruits and vegetables in our daily diet is beneficial for the prevention of a vast array of human diseases. Accordingly, numerous studies based on the hypothesis of the therapeutic potential of fruits and vegetables, as well as several medicinal plants, have been conducted. Following a diet that consists of fruits and vegetables has been associated with a lower frequency of diagnosed patients suffering from several pathologies such as obesity, cardiovascular and neurological diseases, diabetes, Alzheimer’s disease, and finally, yet importantly, cancer, due to their high content in phytochemicals, such as polyphenols [1][2][3]. Phytochemicals are secondary plant metabolites, known to stimulate plant reproduction as well to protect against pathogens and phytochemistry, including a systematic study of phytochemicals which has been of significant interest to researchers in the last decades [4].
Incontrovertible evidence of various health benefits arising from incorporating anthocyanins in our diet was presented by many researchers. The worldwide interest of the research community in respect to biochemical and biological aspects of anthocyanins has considerably increased not only due to their wide therapeutic potential but also due to their anticarcinogenic effects. Currently, the potential health benefits of these extraordinary molecules are strongly related to their potent antioxidant activity. Recent studies involving anthocyanin-based extracts carried out in vitro and in vivo, along with the epidemiological studies, have granted these pigments potential anticarcinogenic properties.
The most abundant compounds with antioxidant potential in the human daily diet are represented by polyphenols. There are over 15,000 polyphenols identified in nature, widely distributed not only among fruits and vegetables but also in different types of grains, oils, alcoholic and non-alcoholic beverages [5]. They may protect against an array of chronic diseases or even various cancers, as exhibited in some pre-clinical, clinical, and epidemiologic studies [1]. However, the nature and the underlying mechanisms of the most protective effects of polyphenols are still under investigation [6]. Dietary polyphenols have gained the attention of researchers worldwide owing to their antioxidant potential. They are able to protect the human organism from free-radical-induced damage to DNA and defend against the harmful UV radiation or pathogen aggressiveness.
The most common polyphenols in plants that have proved potential health benefits are the well-known class of flavonoids. Furthermore, anthocyanins are natural-occurring plant pigments which are included in this class. Nearly 700 different anthocyanins have been discovered in nature [7], and they are considered the vastest and probably the most remarkable category of water-soluble pigments occurring in plants [8]. These natural plant pigments grant the color palette of flowers, leaves, fruits, and some vegetables. The red, blue, magenta, purple, and orange colors are the result of the conjugates bonds in anthocyanins structure, which are able to absorb the light at wavelengths around 500 nm [1]. The wide range of hues which an anthocyanin solution may present is not only the consequence of the pH variations. In this regard, the various structural patterns of anthocyanins play a key role as well the proportions of these diverse forms, particularly the flavylium cation under red appearance, the quinonoidal bases under violet appearance, the colorless solution or sulfite adducts, and the chalcones, which appear as yellow [9]. However, the phenomenon called “copigmentation” is the one that can offer a more elaborate explanation for the diversity of anthocyanin-derived colors observed in nature. To date, anthocyanins have had multiple applications, not only in different industrial branches, such as the textile industry, where they are used as natural colorants, but in the health sector as well, where they serve as bioactive components in the form of nutraceuticals [10].
Recently, several studies have focused on the anthocyanins’ potential, especially their anticarcinogenic potential; and numerous publications have shown the positive effects of this family of flavonoids in cancer therapy. However, to the best of our knowledge, there have been only two review papers published on the anticancer effect of anthocyanins. One of these papers overviewed the improvements on the anticancer activities of anthocyanins and the associated molecular mechanism, without targeting a specific type of cancer [11]. The other review paper was written on the preventative and inhibitory effects of anthocyanins on a specific kind of cancer: colorectal cancer [12]. On the other hand, a review paper on natural polyphenols as anticancer agents for skin cancer was recently published [1]. However, this study reviewed the polyphenols, focusing not on anthocyanins but other phenolics such as ellagitannins, quercetin, and resveratrol in purified forms (excluding total extracts and fractions). Their chemopreventive effects against skin cancer metastasis were illustrated by reviewing 34 in vivo mechanistic studies. Still, there is no comprehensive review of the anticarcinogenic effects of dietary anthocyanins on skin cancers available in the literature.
One of the leading causes of deaths among patients diagnosed with cancer is represented by skin cancer. This type of cancer is the most encountered type of cancer in the United States [13]. Depending on the cell type, skin cancers are divided into two types: cutaneous melanoma and non-melanoma. Although cutaneous melanoma, also known as malignant melanoma, is not very common compared to other skin cancers (prevalence rate of about 4%), it is the most deadly malignancy (responsible for the 80% of mortality of skin cancer), and the number of young patients diagnosed with melanoma increases faster than the number of patients diagnosed with any other types of cancer [1]. Malignant melanoma starts in melanocytes at the basal layer of the epidermis, which are the cells specialized for producing the brown pigment called melanin. Melanocytes are part of the basal cell layer of the epidermis, but they can be found in the eye as well [14]. On the other hand, non-melanoma skin cancer originates from keratinocytes of epidermis, and this cancer is also grouped into two categories: basal cell carcinoma (BCC) and squamous cell carcinoma (SCC). As opposed to the low prevalence and high mortality of malignant melanoma, BCC is the most prevalent type of skin cancer with a low mortality rate due to its low probability to metastasize [1].
One of the most critical environmental factors for melanoma development is the UV light exposure, and the risk considerably increases in lightly pigmented skin compared to darkly pigmented skin, and as people become aged [15]. To date, several in vitro studies conducted on melanoma cell lines have demonstrated that anthocyanins have the ability to induce apoptosis [16], block the cell cycle at G0/G1 phase [17], increase the oxidative stress [18], decrease the cell viability [19], cell proliferation [20] and tumor progression [21]. Moreover, the polyphenol concentrated extract from acerola was used to examine the skin-lightening effect on brownish guinea pigs, which had been subjected to controlled UVB irradiation. The results showed that an oral administration of the applied treatment effectively lightened the pigmented skin of guinea pigs. This effect might have mainly been because of the suppression of melanin biosynthesis by inhibiting the tyrosinase activity in melanocytes [22].
In this review, we aim to provide an up-to-date overview regarding anthocyanins as functional molecules and their chemopreventive effects on melanoma in vitro and in vivo as well as a comprehensive description of major sources of anthocyanins.
2. Anthocyanins’ Chemistry
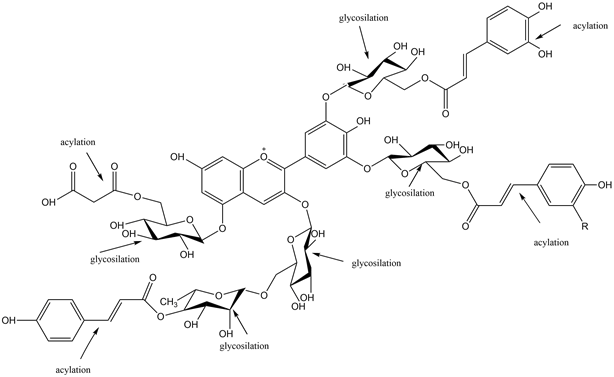
Anthocyanins are water-soluble vacuolar pigments that occur ubiquitously in the plant kingdom, and they are widely distributed in fruits and vegetables as glycosides, having different sugars, such as glucose, rhamnose, xylose or arabinose, attached to an aglycon nucleus [23]. Basically, anthocyanins are glycosylated or acylglycosylated forms of polyhydroxy or polymethroxy molecules, derivates of 2-phenylbenzopyrylium cation [24] (Figure 1).
Figure 1. Typically glycosylation and acylation of anthocyanins (adapted from [25][26]).
They present a characteristic chemical structure, having a carbon chain (C6—C3—C6) where an intermediate heterocyclic ring divides the two aromatic rings. Anthocyanin molecules may contain variations in the number of hydroxyl groups or different degrees of methylation [27]. The rich structural diversity of anthocyanins is also sustained by their nature, number, and position of sugar molecules bound to aromatic rings, as well as their nature and number of aliphatic or aromatic acids attached to sugars [28]. The sugar components of anthocyanins are usually conjugated to the anthocyanidin skeleton via the C3-hydroxyl group in ring C [28].
Anthocyanins are unstable molecules, and they are prone to degradation. There are several factors which may influence the color stability of anthocyanins, such as temperature, pH, enzymes activity, light, oxygen, chemical structure, anthocyanin concentration, and also the presence of complex compounds such as other flavonoids, phenolic acids, copigments, sugars and metal ions [29]. The stability of the colored structural forms of the anthocyanins and their vibrant colors is the result of copigmentation [30]. The copigmentation occurs due to molecular associations between pigments and other usually uncolored organic molecules present in the solutions that are often reported as cofactors [31]. Unlike other flavonoids, the anthocyanins carry a positive charge in acidic solution [28]. In acidic conditions, anthocyanins appear as red but turn to blue when the pH increases [29]. They all present the basic flavylium cationic structure at low pH values, and they can be distinguished from one another by the occurrence of different substituents in ring B [32].
The most common anthocyanin aglycones, also called anthocyanidins, found in plants include delphinidin (De), cyanidin (Cy), petunidin (Pt), peonidin (Pn), pelargonidin (Pg), and malvidin (Mv) [33]. Cyanidin has the highest proportion in tissues of plants (50%), followed by pelargonidin (12%), peonidin (12%), delphinidin (12%), petunidin (7%), and malvidin (7%). Nevertheless, these aglycones by themselves are very stable and convert to anthocyanins via glycosylation [29]. These structural variations among anthocyanins and anthocyanidins may influence their anticancer efficiency [20] Although a previous study demonstrated that anthocyanidins caused more potent growth inhibition of human hepatoma cell lines than anthocyanins [34], another study reported that both anthocyanins and their aglycones selectively inhibited the tumor growth [35]. Other biological effects influenced by the structural differences between anthocyanins and their aglycones are related to their antioxidant activities and bioavailability.
Although there has been an abundant number of studies available regarding the protective effects of dietary anthocyanins against health problems such as myocardial infarction, cardiovascular diseases, and cancer-related mortality, the exact mechanism is still complicated and not fully elucidated yet. Accordingly, a good grasp of how anthocyanins proceed in the human body from their consumption to their absorption, distribution, metabolism, and excretion is also crucial. In order to show their effect, anthocyanins need to be present in blood circulation and tissues upon their consumption through the diet. This is also known as “bioavailability,” indicating that a part of the dietary anthocyanins digested reaches the general circulation and specific locates and becomes available to exert their functions. Earlier, anthocyanins were considered to have deficient levels of bioavailability, such as between 0.26 and 1.8% in animal studies [36][37][38][39][40]. Nevertheless, a relevant study tracking the isotopically labeled cyanidin-3-glucoside in humans has further clarified the anthocyanin metabolism in the human body, reporting a remarkable higher recovery level of anthocyanins (12.4%) [41]. The authors stated that this unexpectedly higher bioavailability of the specific anthocyanin could be due to newly identified metabolites.
Once ingested, anthocyanins travel through the gastrointestinal tract, including stomach, small intestine, large intestine (fecal elimination), portal vein, liver, general circulation, organs, and tissues as well as being discarded through urine. As opposed to other flavonoids, previous studies have revealed that the glycosides follow a unique pattern and can be present in the human circulation after a few minutes of anthocyanin glycosides intake (reaching 100nM within 0.5–2 h and disappearing from the bloodstream in less than 6 h), indicating the intact anthocyanin absorption from the stomach [41][42][43]. Animal studies also validated the effective absorption of anthocyanins from the stomach [44][45][46]. However, the small intestine is the major part where anthocyanin absorption occurs. Various factors, including the type of aglycone, sugar moiety, or acylated groups and the presence of other flavonoids, may alter the anthocyanin absorption rate, and the extent [47][48]. Still, monitoring the intact anthocyanins may not be the best approach to evaluate the level of anthocyanin absorptions. Several studies evaluating the bioavailability of the anthocyanins have reported that along with the anthocyanins, their conjugates, and other related metabolites such as a variety of phenolic acids, might be present in the plasma [49]. In one human study, 35 anthocyanin metabolites were determined after labeled cyanidin-3-glucoside intake, with 17 being in the bloodstream, 31 in urine, and 28 in feces [49]. Indeed, the concentrations of phenolic acid metabolites detected in the bloodstream were remarkably higher than those of their parent anthocyanins. Thus, the health benefits of orally administered dietary anthocyanins could be due to these higher rates of metabolites [42]. On the other hand, some anthocyanins can reach the colon in substantial amounts. These anthocyanins go through decomposition there in the large intestine by the present microbiota [42]. A previous study demonstrated the hydrolyzation of the anthocyanin glycosides in intestinal microflora within 20 min to 2 h [50]. Furthermore, some studies conducted in rats and pigs in the literature also showed that anthocyanins could reach the liver, eye, and brain tissue [51].
3. Anthocyanins, as Part of the Daily Human Diet
A key factor for maintaining human homeostasis is food intake, but more importantly, the nature of the food, which constitutes our diet [52]. As mentioned earlier, evidence regarding the potential health benefits provided by the consumption of fruits and vegetables was reported by many researchers. In these studies, the biggest challenge was to identify the specific compounds responsible for the health benefits. Then, the new fields of research turned their attention towards flavonoid compounds [53], mainly on anthocyanins. In the flavonoid-rich foods ingested, anthocyanins represent a large subclass [54]. Almost all berry types are abundant in anthocyanins, as well as many other dark-colored fruits and vegetables (Figure 2) [55][56][57][58], fruit-derived products such as juices, wines, and jams. Tables 1–3 summarize major flowers, berries, and vegetables as sources of anthocyanins and their quantitative occurrence, respectively.
Figure 2. Food rich in the source of anthocyanins.
In 2007–2008, the National Health and Nutrition Examination Survey reported the estimated dietary intake of anthocyanins to be ~11.6 ± 1.1 mg/d for individuals aged ≥20 years. This number is significant compared with other flavonoids ingested, such as genistein, quercetin, and apigenin, whose daily intake was estimated at only 20–25 mg [59]. However, anthocyanin intake largely depends on dietary habits. For instance, only one serving of berries can significantly increase daily anthocyanin intake. Additionally, the daily intake of anthocyanins can be estimated from 500 mg to 1 g if flavonoid supplements are included in the diet [60].
Another important subject to discuss, considering that anthocyanins are found in a significant amount in edible foods, is their toxicity. So far, no adverse effects of anthocyanins associated with their normal dietary intake have been reported [61]. Moreover, evaluations concerning anthocyanin toxicity suggest that adverse effects may occur only if anthocyanins are ingested at extremely high levels [62]. Certainly, further complex toxicological assessments are necessary in order to have complete data regarding side-effects of different concentrations of anthocyanins. To date, there is no specific level of anthocyanin intake to be recommended for optimal health benefits; therefore, this could be a new field of research, considering the consumer demands for natural health-promoting ingredients growing to a greater extent [62].
References
- Zhou, Y.; Zheng, J.; Li, Y.; Xu, D.-P.; Li, S.; Chen, Y.-M.; Li, H.-B. Natural polyphenols for prevention and treatment of cancer. Nutrients 2016, 8, 515.
- Lage, N.N.; Layosa, M.A.A.; Arbizu, S.; Chew, B.P.; Pedrosa, M.L.; Mertens-Talcott, S.; Talcott, S.; Noratto, G.D. Dark sweet cherry (prunus avium) phenolics enriched in anthocyanins exhibit enhanced activity against the most aggressive breast cancer subtypes without toxicity to normal breast cells. J. Funct. Foods 2020, 64, 12.
- Koch, W. Dietary polyphenols-important non-nutrients in the prevention of chronic noncommunicable diseases. A systematic review. Nutrients 2019, 11, 1039.
- Isah, T. Stress and defense responses in plant secondary metabolites production. Biol Res. 2019, 52, 39–39.
- Lila, M.A. Anthocyanins and human health: An in vitro investigative approach. J. Biomed. Biotechnol. 2004, 2004, 306–313.
- Scalbert, A.; Johnson, I.T.; Saltmarsh, M. Polyphenols: Antioxidants and beyond. Am. J. Clin. Nutr. 2005, 81, 215S-217S.
- Wallace, T.C.; Giusti, M.M. Anthocyanins. Adv. Nutr. 2015, 6, 620–622.
- Takeoka, G.R.; Dao, L.; Harden, L.; Pantoja, A.; Kuhl, J.C. Antioxidant activity, phenolic and anthocyanin contents of various rhubarb (rheum spp.) varieties. Food Sci. Technol. 2013, 48, 172–178.
- Bimpilas, A.; Panagopoulou, M.; Tsimogiannis, D.; Oreopoulou, V. Anthocyanin copigmentation and color of wine: The effect of naturally obtained hydroxycinnamic acids as cofactors. Food Chem 2016, 197, 39–46.
- Khoo, H.E.; Azlan, A. Anthocyanidins and anthocyanins: Colored pigments as food, pharmaceutical ingredients, and the potential health benefits. Food Nutr. Res. 2017, 61, 1361779.
- Lin, B.W.; Gong, C.C.; Song, H.F.; Cui, Y.Y. Effects of anthocyanins on the prevention and treatment of cancer. Br. J. Pharmacol. 2017, 174, 1226–1243.
- Zhao, X.; Feng, P.; He, W.; Du, X.; Chen, C.; Suo, L.; Liang, M.; Zhang, N.; Na, A.; Zhang, Y. The prevention and inhibition effect of anthocyanins on colorectal cancer. Curr. Pharm. Des. 2019, 25, 4919–4927.
- Rager, E.L.; Bridgeford, E.P.; Ollila, D.W. Cutaneous melanoma: Update on prevention, screening, diagnosis, and treatment. Am. Fam. Physician 2005, 72, 269–276.
- Hurst, E.A.; Harbour, J.W.; Cornelius, L.A. Ocular melanoma: A review and the relationship to cutaneous melanoma. Arch. Dermatol. 2003, 139, 1067–1073.
- Wiecker, T.S.; Luther, H.; Buettner, P.; Bauer, J.; Garbe, C. Moderate sun exposure and nevus counts in parents are associated with development of melanocytic nevi in childhood: A risk factor study in 1,812 kindergarten children. Cancer 2003, 97, 628–638.
- Bunea, A.; Rugina, D.; Sconta, Z.; Pop, R.M.; Pintea, A.; Socaciu, C.; Tabaran, F.; Grootaert, C.; Struijs, K.; VanCamp, J. Anthocyanin determination in blueberry extracts from various cultivars and their antiproliferative and apoptotic properties in b16-f10 metastatic murine melanoma cells. Phytochemistry 2013, 95, 436–444.
- Wang, E.; Liu, Y.; Xu, C.; Liu, J. Antiproliferative and proapoptotic activities of anthocyanin and anthocyanidin extracts from blueberry fruits on b16-f10 melanoma cells. Food Nutr. Res. 2017, 61, 1325308.
- Diaconeasa, Z.; Ayvaz, H.; Rugina, D.; Leopold, L.; Stanila, A.; Socaciu, C.; Tabaran, F.; Luput, L.; Mada, D.C.; Pintea, A.; et al. Melanoma inhibition by anthocyanins is associated with the reduction of oxidative stress biomarkers and changes in mitochondrial membrane potential. Plant Foods Hum. Nutr. 2017, 72, 404–410.
- Benedec, D.; Oniga, I.; Cuibus, F.; Sevastre, B.; Stiufiuc, G.; Duma, M.; Hanganu, D.; Iacovita, C.; Stiufiuc, R.; Lucaciu, C.M. Origanum vulgare mediated green synthesis of biocompatible gold nanoparticles simultaneously possessing plasmonic, antioxidant and antimicrobial properties. Int. J. Nanomed. 2018, 13, 1041–1058.
- Rugină, D.; Hanganu, D.; Diaconeasa, Z.; Tăbăran, F.; Coman, C.; Leopold, L.; Bunea, A.; Pintea, A. Antiproliferative and apoptotic potential of cyanidin-based anthocyanins on melanoma cells. Int. J. Mol. Sci. 2017, 18, 0949.
- Lu, J.N.; Panchanathan, R.; Lee, W.S.; Kim, H.J.; Kim, D.H.; Choi, Y.H.; Kim, G.S.; Shin, S.C.; Hong, S.C. Anthocyanins from the fruit of vitis coignetiae pulliat inhibit tnf-augmented cancer proliferation, migration, and invasion in a549 cells. Asian Pac. J. Cancer Prev. Apjcp 2017, 18, 2919–2923.
- Hanamura, T.; Uchida, E.; Aoki, H. Skin-lightening effect of a polyphenol extract from acerola (Malpighia emarginata DC.) fruit on uv-induced pigmentation. Biosci. Biotechnol. Biochem. 2008, 72, 3211–3218.
- Mazza, G.; Francis, F.J. Anthocyanins in grapes and grape products. Crit. Rev. Food Sci. Nutr. 1995, 35, 341–371.
- Felgines, C.; Talavera, S.; Texier, O.; Gil-Izquierdo, A.; Lamaison, J.L.; Remesy, C. Blackberry anthocyanins are mainly recovered from urine as methylated and glucuronidated conjugates in humans. J. Agric. Food Chem. 2005, 53, 7721–7727.
- Andersen, O.M.; Markham, K.R. The Anthocyanins. In Flavonoids Chemistry, Biochemistry and Applications; Andersen, O.M., Markham, K.R., eds.; CRC Press Taylor & Francis Group: Boca Raton, FL, USA, 2006; 471–551.
- Hsu, Y.H.; Tagami, T.; Matsunaga, K.; Okuyama, M.; Suzuki, T.; Noda, N.; Suzuki, M.; Shimura, H. Functional characterization of udp-rhamnose-dependent rhamnosyltransferase involved in anthocyanin modification, a key enzyme determining blue coloration in lobelia erinus. Plant J. 2017, 89, 325–337.
- Vicente Dragano, N.R.; Castro Marques, A.Y. Chapter 11—Native Fruits, Anthocyanins in Nutraceuticals, and the Insulin Receptor/Insulin Receptor Substrate-1/Akt/Forkhead Box Protein Pathway. In Molecular Nutrition and Diabetes; Mauricio, D., Ed.; Academic Press: San Diego, CA, USA, 2016; pp 131–145.
- Wang, L.-S.S.; Gary, D. Anthocyanins and their role in cancer prevention. Cancer Lett. 2008, 269, 281–290.
- Khoo, H.E.; Azlan, A.; Tang, S.T.; Lim, S.M. Anthocyanidins and anthocyanins: Colored pigments as food, pharmaceutical ingredients, and the potential health benefits. Food Nutr. Res. 2017, 61, 1361779–1361779.
- Trouillas, P.; Sancho-García, J.C.; De Freitas, V.; Gierschner, J.; Otyepka, M.; Dangles, O. Stabilizing and modulating color by copigmentation: Insights from theory and experiment. Chem. Rev. 2016, 116, 4937–4982.
- Boulton, R. The copigmentation of anthocyanins and its role in the color of red wine: A critical review. Am. J. Enol. Vitic. 2001, 52, 67–87.
- Huang, H.-P.; Shih, Y.-W.; Chang, Y.-C.; Hung, C.-N.; Wang, C.-J. Chemoinhibitory effect of mulberry anthocyanins on melanoma metastasis involved in the ras/pi3k pathway. J. Agric. Food Chem. 2008, 56, 9286–9293.
- Tsuda, T. Dietary anthocyanin-rich plants: Biochemical basis and recent progress in health benefits studies. Mol. Nutr. Food Res. 2012, 56, 159–170.
- Yeh, C.T.; Yen, G.C. Induction of apoptosis by the anthocyanidins through regulation of bcl-2 gene and activation of c-jun n-terminal kinase cascade in hepatoma cells. J. Agric. Food Chem. 2005, 53, 1740–1749.
- Katsube, N.; Iwashita, K.; Tsushida, T.; Yamaki, K.; Kobori, M. Induction of apoptosis in cancer cells by bilberry (vaccinium myrtillus) and the anthocyanins. J. Agric. Food Chem 2003, 51, 68–75.
- Borges, G.; Roowi, S.; Rouanet, J.M.; Duthie, G.G.; Lean, M.E.; Crozier, A. The bioavailability of raspberry anthocyanins and ellagitannins in rats. Mol. Nutr. Food Res. 2007, 51, 714–725.
- Felgines, C.; Texier, O.; Besson, C.; Fraisse, D.; Lamaison, J.L.; Rémésy, C. Blackberry anthocyanins are slightly bioavailable in rats. J. Nutr. 2002, 132, 1249–1253.
- Felgines, C.; Talavéra, S.; Gonthier, M.P.; Texier, O.; Scalbert, A.; Lamaison, J.L.; Rémésy, C. Strawberry anthocyanins are recovered in urine as glucuro- and sulfoconjugates in humans. J. Nutr. 2003, 133, 1296–1301.
- Ichiyanagi, T.; Shida, Y.; Rahman, M.M.; Hatano, Y.; Konishi, T. Bioavailability and tissue distribution of anthocyanins in bilberry (vaccinium myrtillus l.) extract in rats. J. Agric. Food Chem. 2006, 54, 6578–6587.
- Marczylo, T.H.; Cooke, D.; Brown, K.; Steward, W.P.; Gescher, A.J. Pharmacokinetics and metabolism of the putative cancer chemopreventive agent cyanidin-3-glucoside in mice. Cancer Chemother. Pharmacol. 2009, 64, 1261–1268.
- Matsumoto, H.; Ichiyanagi, T.; Iida, H.; Ito, K.; Tsuda, T.; Hirayama, M.; Konishi, T. Ingested delphinidin-3-rutinoside is primarily excreted to urine as the intact form and to bile as the methylated form in rats. J. Agric. Food Chem. 2006, 54, 578–582.
- Fang, J. Bioavailability of anthocyanins. Drug Metab. Rev. 2014, 46, 508–520.
- Czank, C.; Cassidy, A.; Zhang, Q.; Morrison, D.J.; Preston, T.; Kroon, P.A.; Botting, N.P.; Kay, C.D. Human metabolism and elimination of the anthocyanin, cyanidin-3-glucoside: A (13)c-tracer study. Am. J. Clin. Nutr. 2013, 97, 995–1003.
- Passamonti, S.; Vrhovsek, U.; Vanzo, A.; Mattivi, F. The stomach as a site for anthocyanins absorption from food. Febs Lett. 2003, 544, 210–213.
- Passamonti, S.; Vrhovsek, U.; Vanzo, A.; Mattivi, F. Fast access of some grape pigments to the brain. J. Agric. Food Chem. 2005, 53, 7029–7034.
- Vanzo, A.; Terdoslavich, M.; Brandoni, A.; Torres, A.M.; Vrhovsek, U.; Passamonti, S. Uptake of grape anthocyanins into the rat kidney and the involvement of bilitranslocase. Mol. Nutr. Food Res. 2008, 52, 1106–1116.
- Tian, Q.; Giusti, M.M.; Stoner, G.D.; Schwartz, S.J. Urinary excretion of black raspberry (rubus occidentalis) anthocyanins and their metabolites. J. Agric. Food Chem. 2006, 54, 1467–1472.
- Wu, X.; Pittman, H.E.III.; McKay, S.; Prior, R.L. Aglycones and sugar moieties alter anthocyanin absorption and metabolism after berry consumption in weanling pigs. J. Nutr. 2005, 135, 2417–2424.
- de Ferrars, R.M.; Czank, C.; Zhang, Q.; Botting, N.P.; Kroon, P.A.; Cassidy, A.; Kay, C.D. The pharmacokinetics of anthocyanins and their metabolites in humans. Br. J. Pharmacol. 2014, 171, 3268–3282.
- Keppler, K.; Humpf, H.U. Metabolism of anthocyanins and their phenolic degradation products by the intestinal microflora. Bioorganic Med. Chem. 2005, 13, 5195–5205.
- Talavéra, S.; Felgines, C.; Texier, O.; Besson, C.; Gil-Izquierdo, A.; Lamaison, J.L.; Rémésy, C. Anthocyanin metabolism in rats and their distribution to digestive area, kidney, and brain. J. Aagricultural Food Chem. 2005, 53, 3902–3908.
- Forni, C.; Braglia, R.; Mulinacci, N.; Urbani, A.; Ronci, M.; Gismondi, A.; Tabolacci, C.; Provenzano, B.; Lentini, A.; Beninati, S. Antineoplastic activity of strawberry (fragaria × ananassa duch.) crude extracts on b16-f10 melanoma cells. Mol. Biosyst. 2014, 10, 1255–1263.
- Konczak, I.; Zhang, W. Anthocyanins—More than nature’s colours. J. Biomed. Biotechnol. 2004, 2004, 239–240.
- Hogan, S.; Chung, H.; Zhang, L.; Li, J.; Lee, Y.; Dai, Y.; Zhou, K. Antiproliferative and antioxidant properties of anthocyanin-rich extract from açai. Food Chem. 2010, 118, 208–214.
- Jaakola, L. New insights into the regulation of anthocyanin biosynthesis in fruits. Trends Plant Sci. 2013, 18, 477–483.
- Fang, J. Classification of fruits based on anthocyanin types and relevance to their health effects. Nutrition 2015, 31, 1301–1306.
- Escribano-Bailón, M.T.; Santos-Buelga, C.; Rivas-Gonzalo, J.C. Anthocyanins in cereals. J. Chromatogr. A 2004, 1054, 129–141.
- Kruger, M.J.; Davies, N.; Myburgh, K.H.; Lecour, S. Proanthocyanidins, anthocyanins and cardiovascular diseases. Food Res. Int. 2014, 59, 41–52.
- Hertog, M.G.; Feskens, E.J.; Hollman, P.C.; Katan, M.B.; Kromhout, D. Dietary antioxidant flavonoids and risk of coronary heart disease: The zutphen elderly study. Lancet 1993, 342, 1007–1011.
- Skibola, C.F.; Smith, M.T. Potential health impacts of excessive flavonoid intake. Free Radic Biol Med. 2000, 29, 375–383.
- Markakis, P. Anthocyanins as Food Colors. Academic Press: Cambridge, MA, USA, 1982.
- Burton-Freeman, B.; Sandhu, A.; Edirisinghe, I. Chapter 35—Anthocyanins. In Nutraceuticals; Gupta, R.C., Ed. Academic Press: Boston, MA, USA, 2016; pp 489–500.