
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Shulamit Katzav | + 2680 word(s) | 2680 | 2022-03-10 14:03:27 | | | |
| 2 | Beatrix Zheng | + 17 word(s) | 2697 | 2022-03-23 10:14:15 | | | | |
| 3 | Beatrix Zheng | -1 word(s) | 2696 | 2022-03-25 06:58:59 | | |
Video Upload Options
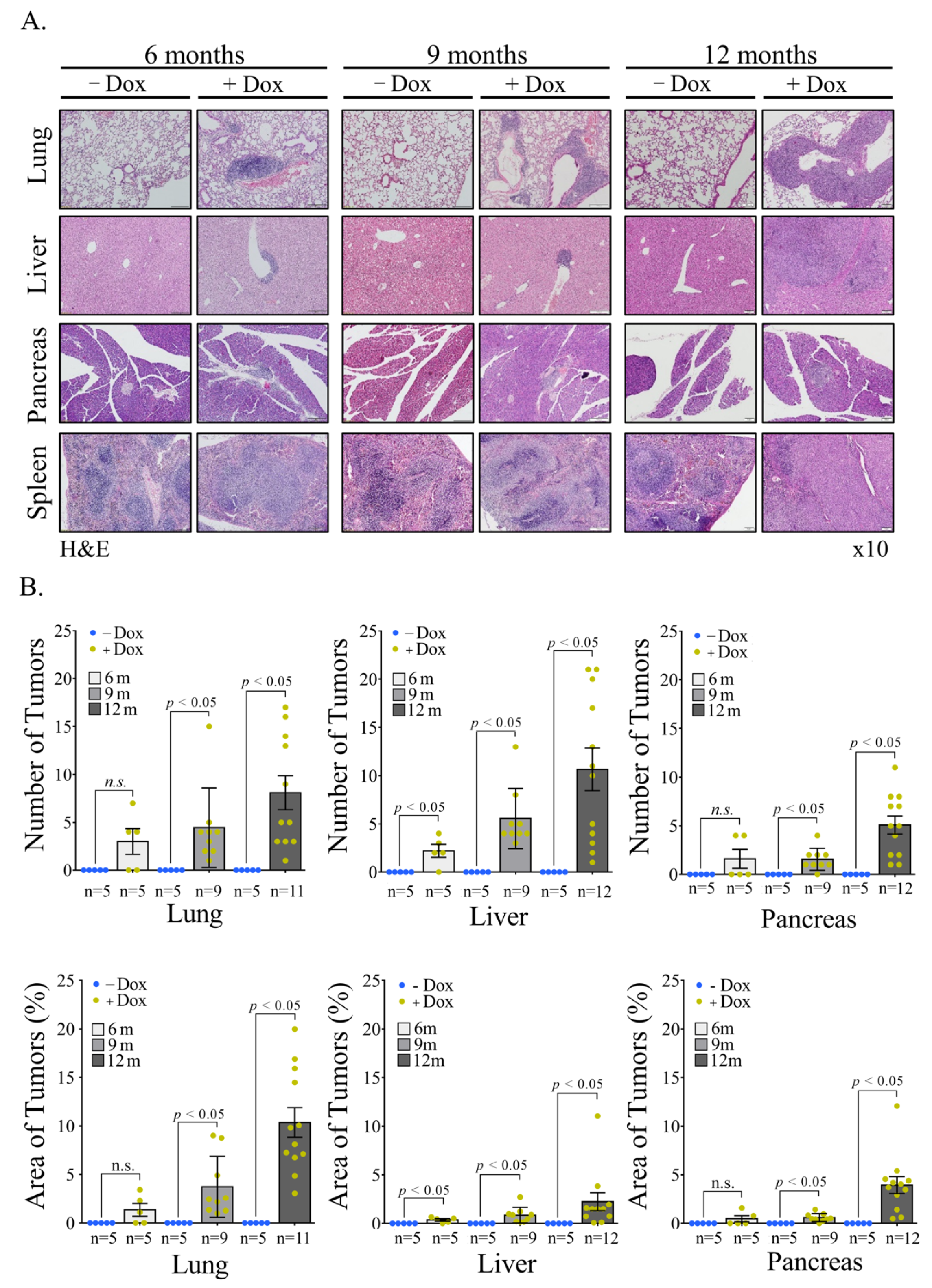
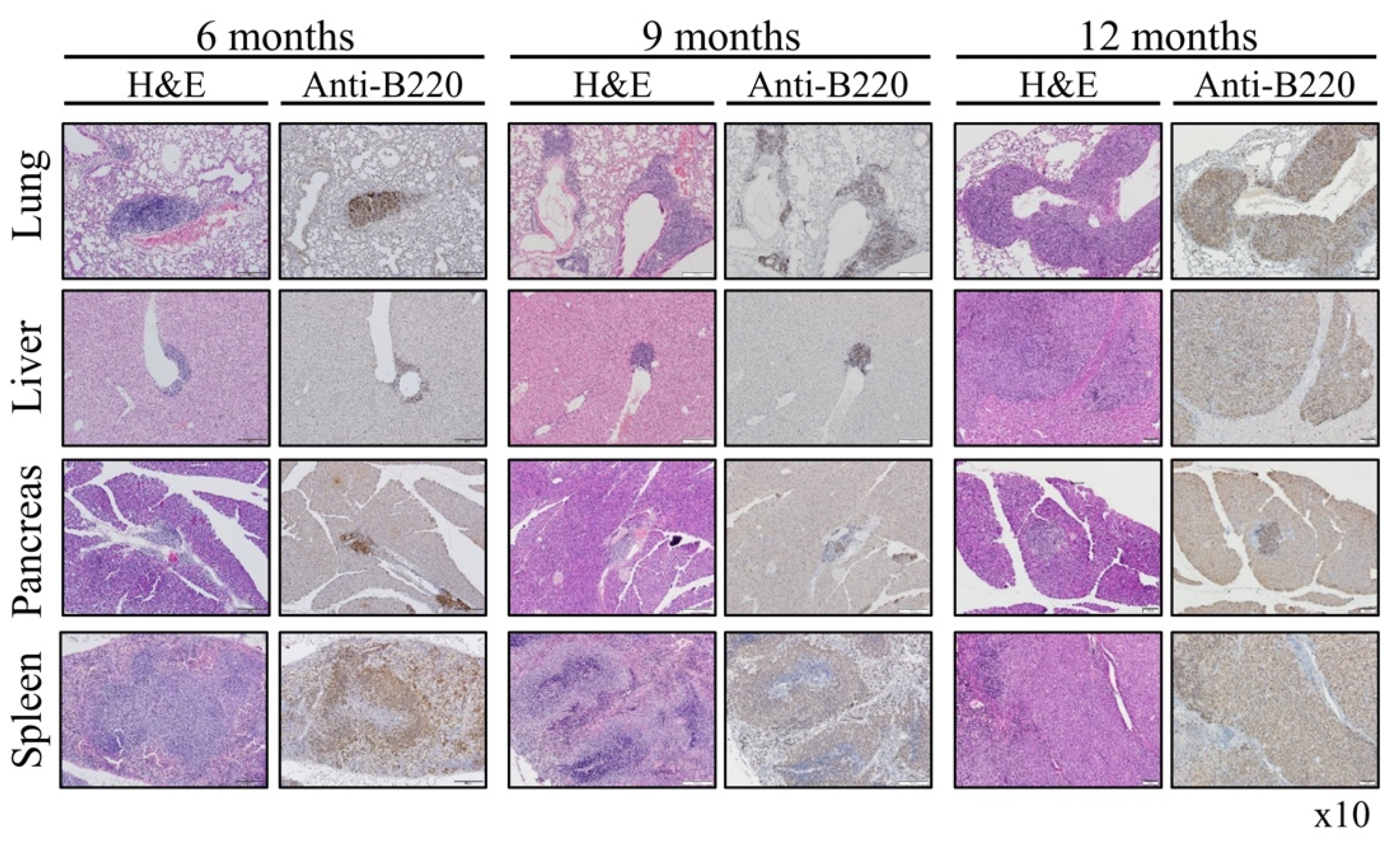
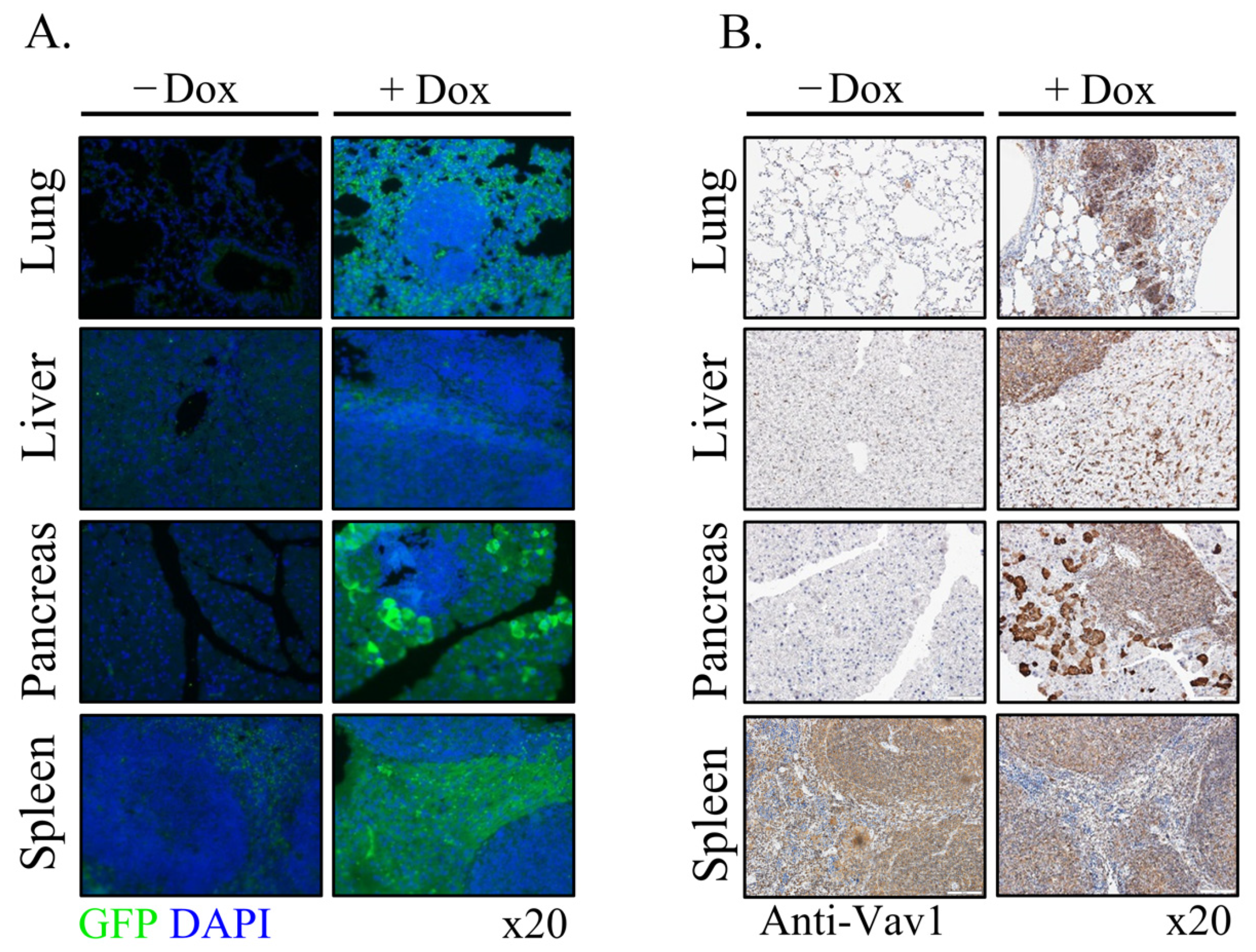
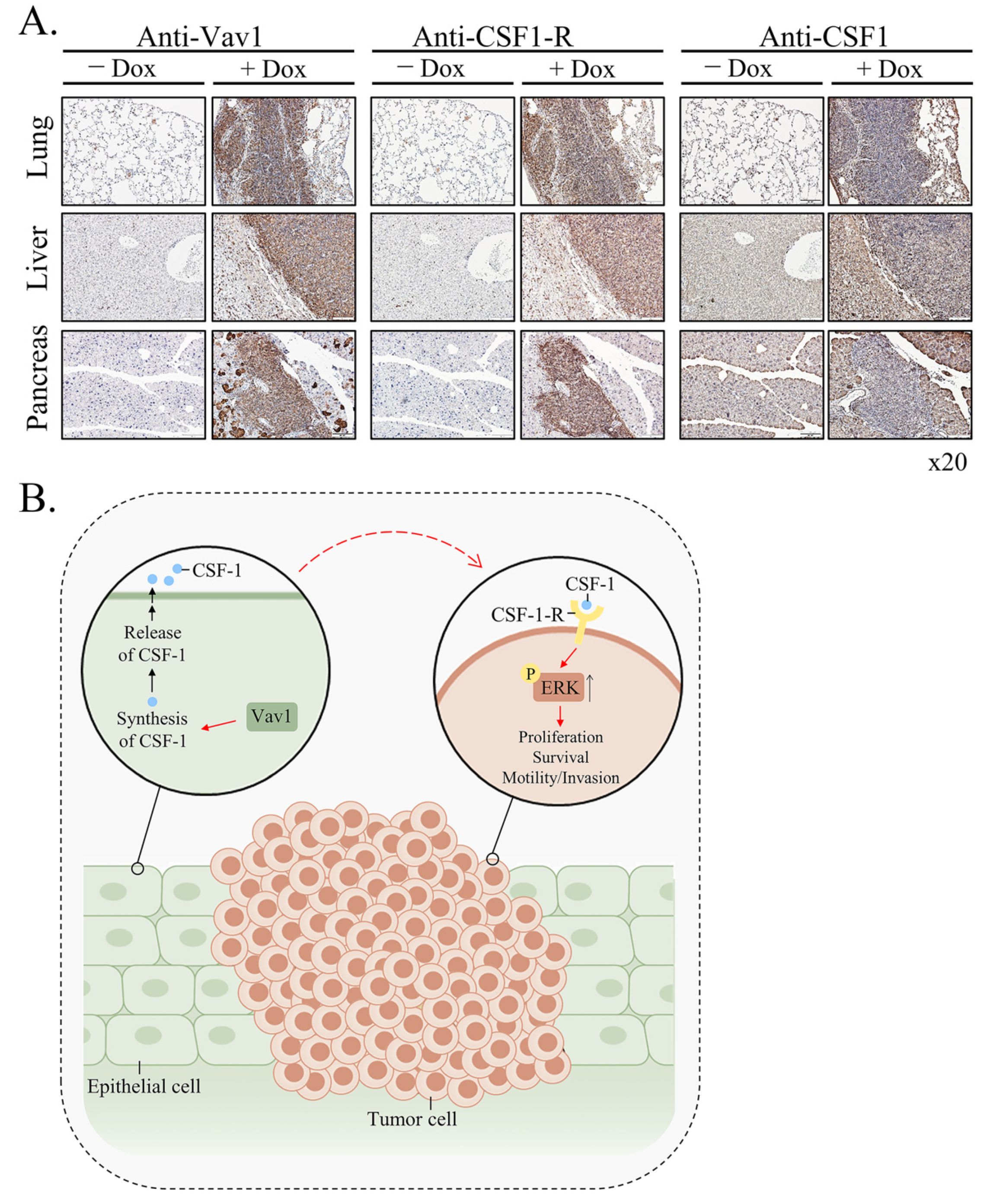
Vav1 is normally and exclusively expressed in the hematopoietic system where it functions as a specific GDP/GTP nucleotide exchange factor (GEF), firmly regulated by tyrosine phosphorylation. Mutations and overexpression of Vav1 in hematopoietic malignancies, and in human cancers of various histologic origins, are well documented. The research results suggest that overexpressing Vav1 in epithelial tissues induced chronic inflammatory reactions eventually leading to B-cell lymphomas development. The development of the lymphomas was accompanied by an increase in ERK phosphorylation, elevation of CSF- in the epithelial tissue, and an increase in CSF1-R expression in the lymphomas. These findings provide a novel mechanism by which Vav1 contributes to tumor propagation.
1. Introduction
2. Current Insights
References
- Katzav, S.; Martin-Zanca, D.; Barbacid, M. Vav, a novel human oncogene derived from a locus ubiquitously expressed in hematopoietic cells. EMBO J. 1989, 8, 2283–2290.
- Katzav, S. Flesh and blood: The story of Vav1, a gene that signals in hematopoietic cells but can be transforming in human malignancies. Cancer Lett. 2007, 255, 241–254.
- Katzav, S. Vav1: A hematopoietic signal transduction molecule involved in human malignancies. Int. J. Biochem. Cell Biol. 2009, 41, 1245–1248.
- Tybulewicz, V.L.J. Vav-family proteins in T-cell signalling. Curr. Opin. Immunol. 2005, 17, 267–274.
- Crespo, P.; Schuebel, K.E.; Ostrom, A.A.; Gutkind, J.S.; Bustelo, X.R. Phosphotyrosine-dependent activation of Rac-1 GDP/GTP exchange by the vav proto-oncogene product. Nature 1997, 385, 169–172.
- Kataoka, K.; Nagata, Y.; Kitanaka, A.; Shiraishi, Y.; Shimamura, T.; Yasunaga, J.-I.; Totoki, Y.; Chiba, K.; Sato-Otsubo, A.; Nagae, G.; et al. Integrated molecular analysis of adult T cell leukemia/lymphoma. Nat. Genet. 2015, 47, 1304–1315.
- Abate, F.; da Silva-Almeida, A.C.; Zairis, S.; Robles-Valero, J.; Couronne, L.; Khiabanian, H.; Quinn, S.A.; Kim, M.-Y.; Laginestra, M.A.; Kim, C.; et al. Activating mutations and translocations in the guanine exchange factor VAV1 in peripheral T-cell lymphomas. Proc. Natl. Acad. Sci. USA 2017, 114, 764–769.
- Prieto-Sánchez, R.M.; Hernández, J.A.; García, J.L.; Gutiérrez, N.C.; San Miguel, J.; Bustelo, X.R.; Hernández, J.M. Overexpression of the VAV proto-oncogene product is associated with B-cell chronic lymphocytic leukaemia displaying loss on 13q. Br. J. Haematol. 2006, 133, 642–645.
- Oberley, M.J.; Wang, D.-S.; Yang, D.T. Vav1 in hematologic neoplasms, a mini review. Am. J. Blood Res. 2012, 2, 1–8.
- Hornstein, I.; Pikarsky, E.; Groysman, M.; Amir, G.; Peylan-Ramu, N.; Katzav, S. The haematopoietic specific signal transducer Vav1 is expressed in a subset of human neuroblastomas. J. Pathol. 2003, 199, 526–533.
- Lazer, G.; Idelchuk, Y.; Schapira, V.; Pikarsky, E.; Katzav, S. The haematopoietic specific signal transducer Vav1 is aberrantly expressed in lung cancer and plays a role in tumourigenesis. J. Pathol. 2009, 219, 25–34.
- Fernandez-Zapico, M.E.; Gonzalez-Paz, N.C.; Weiss, E.; Savoy, D.N.; Molina, J.R.; Fonseca, R.; Smyrk, T.C.; Chari, S.T.; Urrutia, R.; Billadeau, D.D. Ectopic expression of VAV1 reveals an unexpected role in pancreatic cancer tumorigenesis. Cancer Cell 2005, 7, 39–49.
- Sebban, S.; Farago, M.; Gashai, D.; Ilan, L.; Pikarsky, E.; Ben-Porath, I.; Katzav, S. Vav1 fine tunes p53 control of apoptosis versus proliferation in breast cancer. PLoS ONE 2013, 8, e54321.
- Grassilli, S.; Brugnoli, F.; Lattanzio, R.; Rossi, C.; Perracchio, L.; Mottolese, M.; Marchisio, M.; Palomba, M.; Nika, E.; Natali, P.G.; et al. High nuclear level of Vav1 is a positive prognostic factor in early invasive breast tumors: A role in modulating genes related to the efficiency of metastatic process. Oncotarget 2014, 5, 4320–4336.
- Wakahashi, S.; Sudo, T.; Oka, N.; Ueno, S.; Yamaguchi, S.; Fujiwara, K.; Ohbayashi, C.; Nishimura, R. VAV1 represses E-cadherin expression through the transactivation of Snail and Slug: A potential mechanism for aberrant epithelial to mesenchymal transition in human epithelial ovarian cancer. Transl. Res. 2013, 162, 181–190.
- Kniazev, I.P.; Cheburkin, I.V.; Spikermann, K.; Peter, S.; Jenster, G.; Bangma, K.H.; Karelin, M.I.; Shkol’nik, M.I.; Urbanskiĭ, A.I.; Evtushenko, V.I.; et al. Gene expression profiles of protein kinases and phosphatases obtained by hybridization with cDNA arrays: Molecular portrait of human prostate carcinoma. Mol. Biol. 2003, 37, 97–111.
- Zhu, X.; Jin, H.; Xia, Z.; Wu, X.; Yang, M.; Zhang, H.; Shang, X.; Cheng, R.; Zhan, Z.; Yu, Z. Vav1 expression is increased in esophageal squamous cell carcinoma and indicates poor prognosis. Biochem. Biophys. Res. Commun. 2017, 486, 571–576.
- Lindsey, J.C.; Kawauchi, D.; Schwalbe, E.C.; Solecki, D.J.; Selby, M.P.; McKinnon, P.J.; Olson, J.M.; Hayden, J.T.; Grundy, R.G.; Ellison, D.W.; et al. Cross-species epigenetics identifies a critical role for VAV1 in SHH subgroup medulloblastoma maintenance. Oncogene 2015, 34, 4746–4757.
- Barnes, C.J.; Vadlamudi, R.K.; Kumar, R. Novel estrogen receptor coregulators and signaling molecules in human diseases. Cell. Mol. Life Sci. 2004, 61, 281–291.
- Hornstein, I.; Alcover, A.; Katzav, S. Vav proteins, masters of the world of cytoskeleton organization. Cell. Signal. 2004, 16, 1–11.
- Lazer, G.; Katzav, S. Guanine nucleotide exchange factors for RhoGTPases: Good therapeutic targets for cancer therapy? Cell. Signal. 2011, 23, 969–979.
- Salaymeh, Y.; Farago, M.; Sebban, S.; Shalom, B.; Pikarsky, E.; Katzav, S. Vav1 and mutant K-Ras synergize in the early development of pancreatic ductal adenocarcinoma in mice. Life Sci. Alliance 2020, 3, e202000661.
- Fukumoto, K.; Sakata-Yanagimoto, M.; Fujisawa, M.; Sakamoto, T.; Miyoshi, H.; Suehara, Y.; Nguyen, T.B.; Suma, S.; Yanagimoto, S.; Shiraishi, Y.; et al. VAV1 mutations contribute to development of T-cell neoplasms in mice. Blood 2020, 136, 3018–3032.
- Soriano, P.; Friedrich, G.; Lawinger, P. Promoter interactions in retrovirus vectors introduced into fibroblasts and embryonic stem cells. J. Virol. 1991, 65, 2314–2319.
- Hollmann, A.; Aloyz, R.; Baker, K.; Dirnhofer, S.; Owens, T.; Sladek, R.; Tzankov, A. Vav-1 expression correlates with NFκB activation and CD40-mediated cell death in diffuse large B-cell lymphoma cell lines. Hematol. Oncol. 2010, 28, 142–150.
- Yin, J.; Wan, Y.; Li, S.; Du, M.; Zhang, C.; Zhou, X.; Cao, Y. The distinct role of guanine nucleotide exchange factor Vav1 in Bcl-2 transcription and apoptosis inhibition in Jurkat leukemia T cells. Acta Pharmacol. Sin. 2011, 32, 99–107.
- Bertagnolo, V.; Grassilli, S.; Bavelloni, A.; Brugnoli, F.; Piazzi, M.; Candiano, G.; Petretto, A.; Benedusi, M.; Capitani, S. Vav1 modulates protein expression during ATRA-induced maturation of APL-derived promyelocytes: A proteomic-based analysis. J. Proteome Res. 2008, 7, 3729–3736.
- Bertagnolo, V.; Brugnoli, F.; Grassilli, S.; Nika, E.; Capitani, S. Vav1 in differentiation of tumoral promyelocytes. Cell. Signal. 2012, 24, 612–620.
- Huang, P.H.; Lu, P.J.; Ding, L.Y.; Chu, P.C.; Hsu, W.Y.; Chen, C.S.; Tsao, C.C.; Chen, B.H.; Lee, C.T.; Shan, Y.S.; et al. TGFβ promotes mesenchymal phenotype of pancreatic cancer cells, in part, through epigenetic activation of VAV1. Oncogene 2017, 36, 2202–2214.
- Ilan, L.; Katzav, S. Human Vav1 expression in hematopoietic and cancer cell lines is regulated by c-Myb and by CpG methylation. PLoS ONE 2012, 7, e29939.
- Zugaza, J.L.; López-Lago, M.A.; Caloca, M.J.; Dosil, M.; Movilla, N.; Bustelo, X.R. Structural determinants for the biological activity of Vav proteins. J. Biol. Chem. 2002, 277, 45377–45392.
- Shalom, B.; Farago, M.; Pikarsky, E.; Katzav, S. Vav1 mutations identified in human cancers give rise to different oncogenic phenotypes. Oncogenesis 2018, 7, 80.
- Strathdee, D.; Ibbotson, H.; Grant, S.G.N. Expression of transgenes targeted to the Gt(ROSA)26Sor locus is orientation dependent. PLoS ONE 2006, 1, e4.
- Feng, Y.-Q.; Warin, R.; Li, T.; Olivier, E.; Besse, A.; Lobell, A.; Fu, H.; Lin, C.M.; Aladjem, M.I.; Bouhassira, E.E. The human beta-globin locus control region can silence as well as activate gene expression. Mol. Cell. Biol. 2005, 25, 3864–3874.
- Mullangi, S.; Lekkala, M.R. Mucosa-associated lymphoma tissue. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022.
- Alvarez-Lesmes, J.; Chapman, J.R.; Poveda, J.C. Pitfalls in gastrointestinal tract haematopoietic lesions. Pathology 2021, 54, 177–183.
- Mnatsakanian, A.; Heil, J.R.; Sharma, S. Anatomy, head and neck, adenoids. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022.
- Farooq, U.; Chakinala, R.C. MALToma. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022.
- Genco, I.S.; Gur, H.; Hajiyeva, S. Lymphoma of the breast: A clinicopathologic analysis of 51 cases with a specific emphasis on patients with a history of breast carcinoma. Breast J. 2021, 27, 456–460.
- Borie, R.; Wislez, M.; Antoine, M.; Copie-Bergman, C.; Thieblemont, C.; Cadranel, J. Pulmonary mucosa-associated lymphoid tissue lymphoma revisited. Eur. Respir. J. 2016, 48, 1252.
- Rivière, E.; Pascaud, J.; Tchitchek, N.; Boudaoud, S.; Paoletti, A.; Ly, B.; Dupré, A.; Chen, H.; Thai, A.; Allaire, N.; et al. Salivary gland epithelial cells from patients with Sjögren’s syndrome induce B-lymphocyte survival and activation. Ann. Rheum. Dis. 2020, 79, 1468–1477.
- Ahmed, S.; Shahid, R.K.; Sison, C.P.; Fuchs, A.; Mehrotra, B. Orbital lymphomas: A clinicopathologic study of a rare disease. Am. J. Med. Sci. 2006, 331, 79–83.
- Kogame, T.; Kabashima, K.; Egawa, G. Putative Immunological Functions of Inducible Skin-Associated Lymphoid Tissue in the Context of Mucosa-Associated Lymphoid Tissue. Front. Immunol. 2021, 12, 733484.
- Verstappen, G.M.; Pringle, S.; Bootsma, H.; Kroese, F.G.M. Epithelial-immune cell interplay in primary Sjögren syndrome salivary gland pathogenesis. Nat. Rev. Rheumatol. 2021, 17, 333–348.
- Jones, B.E.; Maerz, M.D.; Buckner, J.H. IL-6: A cytokine at the crossroads of autoimmunity. Curr. Opin. Immunol. 2018, 55, 9–14.
- Batten, M.; Groom, J.; Cachero, T.G.; Qian, F.; Schneider, P.; Tschopp, J.; Browning, J.L.; Mackay, F. BAFF mediates survival of peripheral immature B lymphocytes. J. Exp. Med. 2000, 192, 1453–1466.
- Mackay, F.; Schneider, P. Cracking the BAFF code. Nat. Rev. Immunol. 2009, 9, 491–502.
- Kyriakidis, N.C.; Kapsogeorgou, E.K.; Gourzi, V.C.; Konsta, O.D.; Baltatzis, G.E.; Tzioufas, A.G. Toll-like receptor 3 stimulation promotes Ro52/TRIM21 synthesis and nuclear redistribution in salivary gland epithelial cells, partially via type I interferon pathway. Clin. Exp. Immunol. 2014, 178, 548–560.
- Ittah, M.; Miceli-Richard, C.; Gottenberg, E.; Lavie, F.; Lazure, T.; Ba, N.; Sellam, J.; Lepajolec, C.; Mariette, X. B cell-activating factor of the tumor necrosis factor family (BAFF) is expressed under stimulation by interferon in salivary gland epithelial cells in primary Sjögren’s syndrome. Arthritis Res. Ther. 2006, 8, R51.
- Gottenberg, J.-E.; Cagnard, N.; Lucchesi, C.; Letourneur, F.; Mistou, S.; Lazure, T.; Jacques, S.; Ba, N.; Ittah, M.; Lepajolec, C.; et al. Activation of IFN pathways and plasmacytoid dendritic cell recruitment in target organs of primary Sjögren’s syndrome. Proc. Natl. Acad. Sci. USA 2006, 103, 2770–2775.
- Kapsogeorgou, E.K.; Manoussakis, M.N. Salivary gland epithelial cells (SGEC): Carriers of exquisite B7-2 (CD86) costimulatory molecules. J. Autoimmun. 2010, 35, 188–191.
- Kim, N. Chemoprevention of gastric cancer by Helicobacter pylori eradication and its underlying mechanism. J. Gastroenterol. Hepatol. 2019, 34, 1287–1295.
- Sinkovics, J.G. Molecular biology of oncogenic inflammatory processes. I. Non-oncogenic and oncogenic pathogens, intrinsic inflammatory reactions without pathogens, and microRNA/DNA interactions (Review). Int. J. Oncol. 2012, 40, 305–349.
- Wilsbacher, J.L.; Moores, S.L.; Brugge, J.S. An active form of Vav1 induces migration of mammary epithelial cells by stimulating secretion of an epidermal growth factor receptor ligand. Cell Commun. Signal. 2006, 4, 5.
- Schapira, V.; Lazer, G.; Katzav, S. Osteopontin is an oncogenic Vav1- but not wild-type Vav1-responsive gene: Implications for fibroblast transformation. Cancer Res. 2006, 66, 6183–6191.
- Sebban, S.; Farago, M.; Rabinovich, S.; Lazer, G.; Idelchuck, Y.; Ilan, L.; Pikarsky, E.; Katzav, S. Vav1 promotes lung cancer growth by instigating tumor-microenvironment cross-talk via growth factor secretion. Oncotarget 2014, 5, 9214–9226.
- Lin, E.Y.; Nguyen, A.V.; Russell, R.G.; Pollard, J.W. Colony-stimulating factor 1 promotes progression of mammary tumors to malignancy. J. Exp. Med. 2001, 193, 727–740.
- Goswami, S.; Sahai, E.; Wyckoff, J.B.; Cammer, M.; Cox, D.; Pixley, F.J.; Stanley, E.R.; Segall, J.E.; Condeelis, J.S. Macrophages promote the invasion of breast carcinoma cells via a colony-stimulating factor-1/epidermal growth factor paracrine loop. Cancer Res. 2005, 65, 5278–5283.
- Stanley, E.R.; Berg, K.L.; Einstein, D.B.; Lee, P.S.; Pixley, F.J.; Wang, Y.; Yeung, Y.G. Biology and action of colony--stimulating factor-1. Mol. Reprod. Dev. 1997, 46, 4–10.
- Achkova, D.; Maher, J. Role of the colony-stimulating factor (CSF)/CSF-1 receptor axis in cancer. Biochem. Soc. Trans. 2016, 44, 333–341.
- Sun, X.; Ingman, W.V. Cytokine networks that mediate epithelial cell-macrophage crosstalk in the mammary gland: Implications for development and cancer. J. Mammary Gland Biol. Neoplasia 2014, 19, 191–201.
- Guilbert, L.J.; Stanley, E.R. Specific interaction of murine colony-stimulating factor with mononuclear phagocytic cells. J. Cell Biol. 1980, 85, 153–159.
- Mo, H.; Hao, Y.; Lv, Y.; Chen, Z.; Shen, J.; Zhou, S.; Yin, M. Overexpression of macrophage-colony stimulating factor-1 receptor as a prognostic factor for survival in cancer: A systematic review and meta-analysis. Medicine 2021, 100, e25218.
- Dos Anjos Cassado, A. F4/80 as a major macrophage marker: The case of the peritoneum and spleen. Results Probl. Cell Differ. 2017, 62, 161–179.
- Martín-Moreno, A.M.; Roncador, G.; Maestre, L.; Mata, E.; Jiménez, S.; Martínez-Torrecuadrada, J.L.; Reyes-García, A.I.; Rubio, C.; Tomás, J.F.; Estévez, M.; et al. CSF1R protein expression in reactive lymphoid tissues and lymphoma: Its relevance in classical hodgkin lymphoma. PLoS ONE 2015, 10, e0125203.
- Ullrich, K.; Wurster, K.D.; Lamprecht, B.; Köchert, K.; Engert, A.; Dörken, B.; Janz, M.; Mathas, S. BAY 43-9006/Sorafenib blocks CSF1R activity and induces apoptosis in various classical Hodgkin lymphoma cell lines. Br. J. Haematol. 2011, 155, 398–402.
- Lamprecht, B.; Walter, K.; Kreher, S.; Kumar, R.; Hummel, M.; Lenze, D.; Köchert, K.; Bouhlel, M.A.; Richter, J.; Soler, E.; et al. Derepression of an endogenous long terminal repeat activates the CSF1R proto-oncogene in human lymphoma. Nat. Med. 2010, 16, 571–579.
- Murga-Zamalloa, C.; Rolland, D.C.M.; Polk, A.; Wolfe, A.; Dewar, H.; Chowdhury, P.; Onder, O.; Dewar, R.; Brown, N.A.; Bailey, N.G.; et al. Colony-Stimulating Factor 1 Receptor (CSF1R) Activates AKT/mTOR Signaling and Promotes T-Cell Lymphoma Viability. Clin. Cancer Res. 2020, 26, 690–703.
- Krause, G.; Hassenrück, F.; Hallek, M. Relevant Cytokines in the B Cell Lymphoma Micro-Environment. Cancers 2020, 12, 2525.





