
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Kyung-Min Kim | + 2678 word(s) | 2678 | 2022-03-11 06:47:14 | | | |
| 2 | Catherine Yang | + 73 word(s) | 2751 | 2022-03-21 03:47:13 | | |
Video Upload Options
Brown planthopper (BPH, Nilaparvata lugens Stal.) is the most damaging rice pest affecting stable rice yields worldwide. Currently, methods for controlling BPH include breeding a BPH‐resistant cultivar and using synthetic pesticides. Nevertheless, the continuous cultivation of resistant cultivars allows for the emergence of various resistant races, and the use of synthetic pesticides can induce environmental pollution as well as the emergence of unpredictable new pest species. As plants cannot migrate to other locations on their own to combat various stresses, the production of secondary metabolites allows plants to protect themselves from stress and tolerate their reproduction. Pesticides using natural products are currently being developed to prevent environmental pollution and ecosystem disturbance caused by synthetic pesticides. In this study, after BPH infection in rice, chrysoeriol7 (C7), a secondary metabolite that induces resistance against BPH, was assessed. After C7 treatment and BPH infection, relative expression levels of the flavonoid‐related genes were elevated, suggesting that in plants subjected to BPH, compounds related to flavonoids, among the secondary metabolites, play an important role in inducing resistance. The plant‐derived natural compound chrysoeriol7 can potentially thus be used to develop environmentally friendly pesticides. The suggested control of BPH can be effectively used to alleviate concerns regarding environmental pollution and to construct a relatively safe rice breeding environment.
1. Introduction
2. Assessment of C7 Efficacy against BPH
2.1. Analysis of Phenotypes in Rice after Infection with BPH
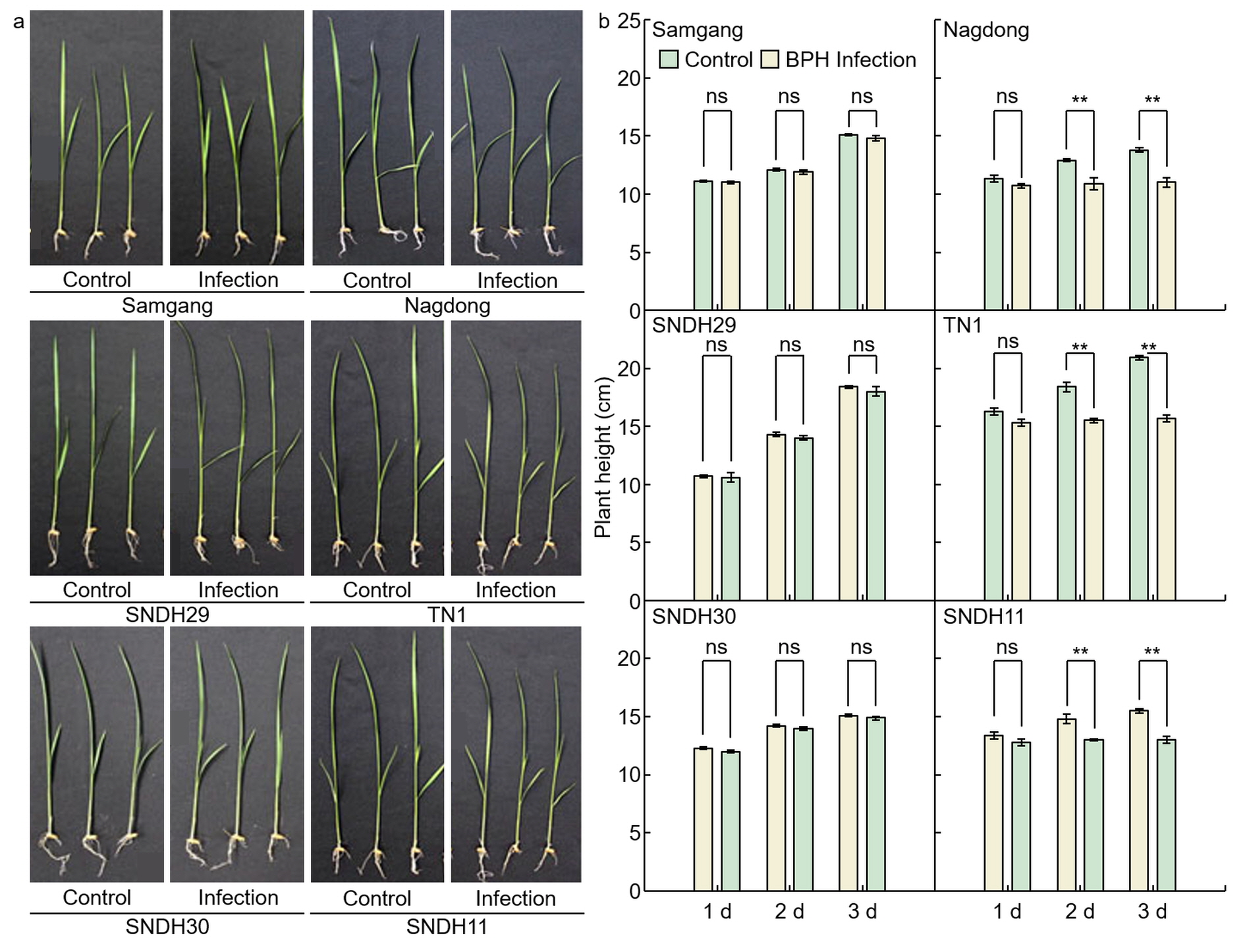
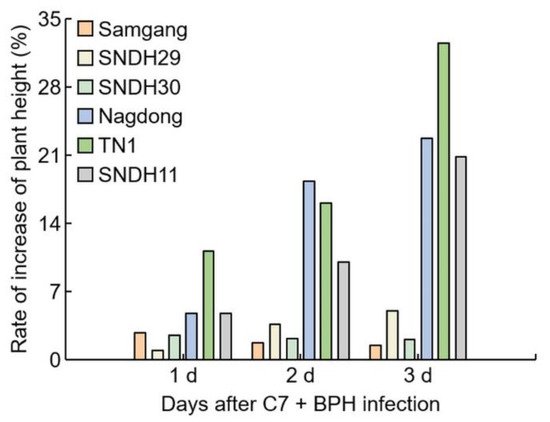
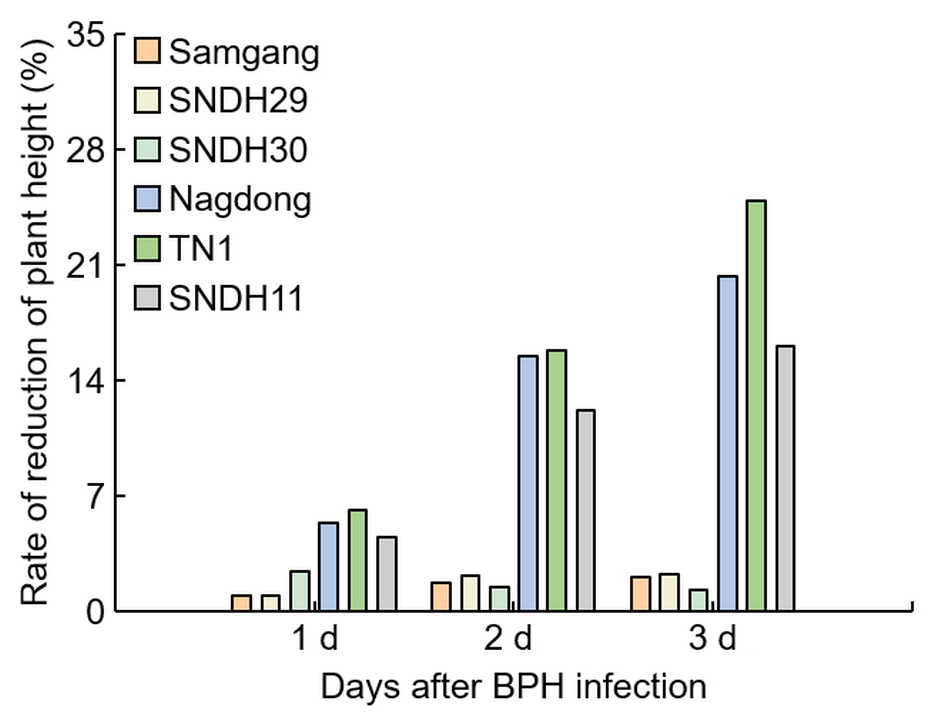 Figure 2. Analysis of the reduction rate of plant growth for 3 days after BPH infection. 1 d: One day after BPH infection; 2 d: two days after BPH infection; 3 d: three days after BPH infection. Bars are shown as the value of the rate of reduction in plant height (n = 5).
Figure 2. Analysis of the reduction rate of plant growth for 3 days after BPH infection. 1 d: One day after BPH infection; 2 d: two days after BPH infection; 3 d: three days after BPH infection. Bars are shown as the value of the rate of reduction in plant height (n = 5).2.2. Analysis of Concentration of C7 after BPH Infection
To identify that C7 is involved in BPH resistance, the concentration of C7 after BPH infection was investigated in Samgang, a cultivar resistant to BPH, and Nagdong, a cultivar susceptible to BPH (Figure 3). In Samgang, the C7 concentration of the BPH‐infected population was 15.30 ± 1.01, the uninfected population was 12.32 ± 0.85, and the C7 concentration rate of increase (%) was 24.24 ± 1.79 (Table 1). In Nagdong, the C7 concentration of the BPH‐infected population was 12.48 ± 0.96, the uninfected population was 11.54 ± 1.21, and the C7 concentration rate of increase (%) was 8.4 1± 6.41. The BPH‐infected population had a greater C7 concentration than the uninfected population in Samgang, and Samgang synthesized more C7 than Nagdong (p < 0.05).
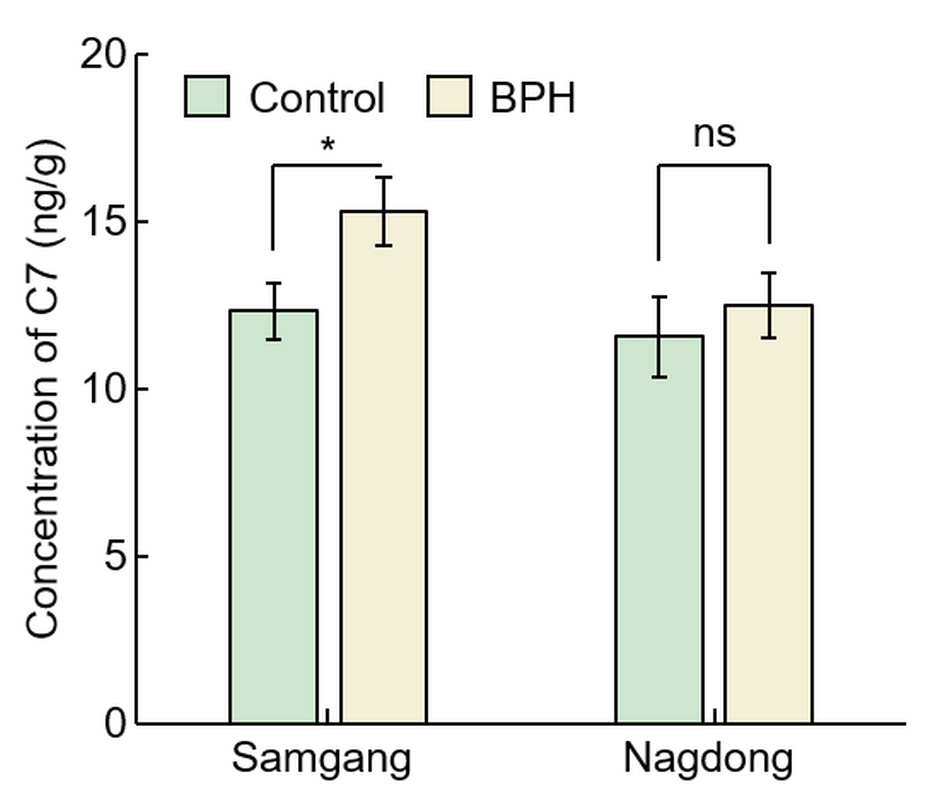 Figure 3. Analysis of concentration of C7 after BPH infection in Samgang and Nagdong. Control: BPH non‐infection. BPH: BPH infection. Samgang: BPH‐resistant cultivar. Nagdong: BPH‐susceptible cultivar. Data are shown as the mean ± SD (n = 5). * indicates a significant difference at p < 0.05. ns indicates not significant.
Figure 3. Analysis of concentration of C7 after BPH infection in Samgang and Nagdong. Control: BPH non‐infection. BPH: BPH infection. Samgang: BPH‐resistant cultivar. Nagdong: BPH‐susceptible cultivar. Data are shown as the mean ± SD (n = 5). * indicates a significant difference at p < 0.05. ns indicates not significant.2.3. Assessment of C7 Efficacy Against BPH
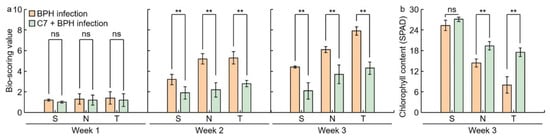
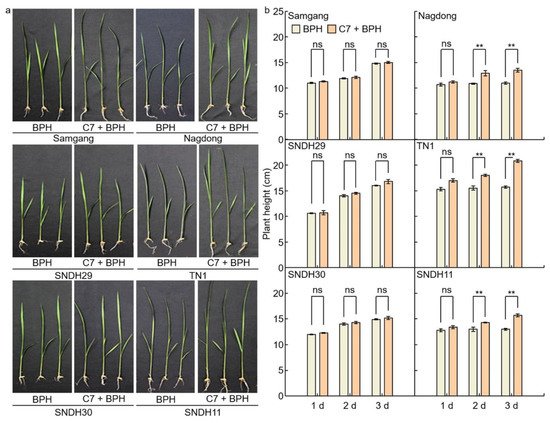
2.4. Flavonoid and Plant Resistant Gene Expression Levels Analysis in BPH‐infected Rice
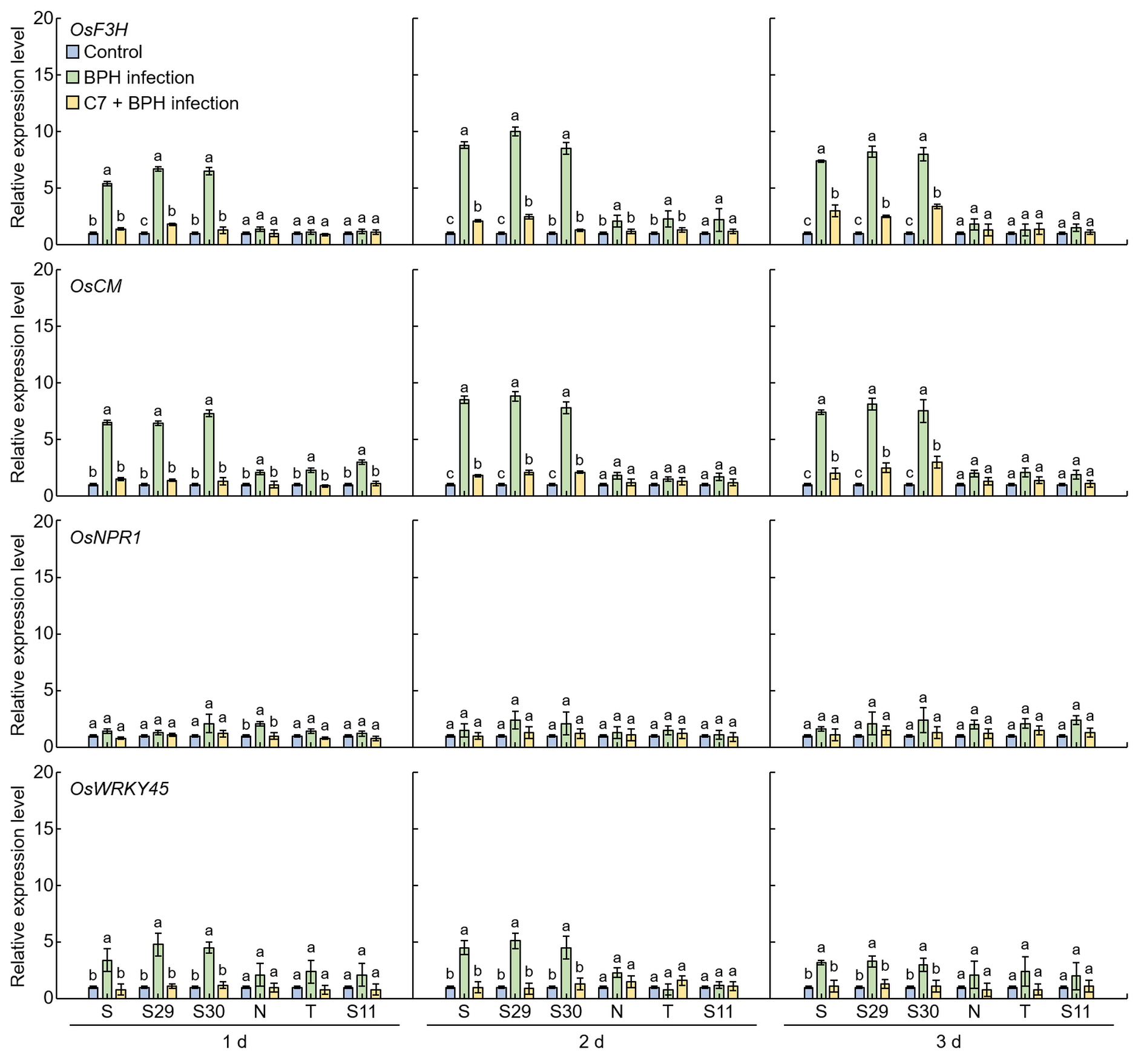
Figure 7. Expression levels of the genes involved in plant defense in the leaves of rice. 1 d: One day after BPH infection; 2 d: two days after BPH infection; 3 d: three days after BPH infection. S: Samgang; S29: SNDH29; S30: SNDH30; N: Nagdong; T: TN1; S11: SNDH11. Bars represent means ± SD (n = 5). Mean denoted by the same letter are not significantly different (p < 0.05) as evaluated by Duncan’s multiple range test (DMRT).
3. Researches and Findings
References
- Taub, D.A.; Wei, J.T. The economics of benign prostatic hyperplasia and lower urinary tract symptoms in the United States. Curr. Urol. Rep. 2006, 7, 272–281.
- Xue, J.; Zhou, X.; Zhang, C.X.; Yu, L.L.; Fan, H.W.; Wang, Z.; Xu, H.J.; Xi, Y.; Zhu, Z.R.; Zhou, W.W.; et al. Genomes of the rice pest brown planthopper and its endosymbionts reveal complex complementary contributions for host adaptation. Genome Biol. 2014, 15, 521.
- Ling, Y.; Weilin, Z. Genetic and biochemical mechanisms of rice resistance to planthopper. Plant Cell Rep. 2016, 35, 1559–1572.
- Sun, Z.; Shi, J.H.; Liu, H.; Yin, L.T.; Abdelnabby, H.; Wang, M.Q. Phytopathogenic infection alters rice–pest–parasitoid tri-trophic interactions. Pest Manag. Sci. 2021, 77, 4530–4538.
- Jing, S.; Zhao, Y.; Du, B.; Chen, R.; Zhu, L.; He, G. Genomics of interaction between the brown planthopper and rice. Curr. Opin. Insect Sci. 2017, 19, 82–87.
- Roy, D.; Chakraborty, G. Bio-efficacy of novel chemicals and tribal pesticide-based integrated modules against brown planthopper in rice. Int. J. Trop. Insect Sci. 2021, 1–11.
- Zhang, F.; Zhu, L.; He, G. Differential gene expression in response to brown planthopper feeding in rice. J. Plant Physiol. 2004, 161, 53–62.
- Wu, S.F.; Zeng, B.; Zheng, C.; Mu, X.C.; Zhang, Y.; Hu, J.; Zhang, S.; Gao, C.F.; Shen, J.L. The evolution of insecticide resistance in the brown planthopper (Nilaparvata lugens Stål) of China in the period 2012–2016. Sci. Rep. 2018, 8, 4586.
- Tschoeke, P.H.; Oliveira, E.E.; Dalcin, M.S.; Silveira-Tschoeke, M.C.A.C.; Sarmento, R.A.; Santos, G.R. Botanical and synthetic pesticides alter the flower visitation rates of pollinator bees in Neotropical melon fields. Environ. Pollut. 2019, 251, 591–599.
- Mesnage, R.; Séralini, G.-E. Editorial: Toxicity of Pesticides on Health and Environment. Front. Public Health 2018, 6, 1–2.
- Dubey, N.K.; Shukla, R.; Kumar, A.; Singh, P.; Prakash, B. Prospects of botanical pesticides in sustainable agriculture. Curr. Sci. 2010, 98, 479–480.
- Amoabeng, B.W.; Johnson, A.C.; Gurr, G.M. Natural enemy enhancement and botanical insecticide source: A review of dual use companion plants. Appl. Entomol. Zool. 2019, 54, 1–19.
- Kedia, A.; Prakash, B.; Mishra, P.K.; Singh, P.; Dubey, N.K. Botanicals as eco friendly biorational alternatives of synthetic pesticides against Callosobruchus spp. (Coleoptera: Bruchidae)—A review. J. Food Sci. Technol. 2015, 52, 1239–1257.
- Mfarrej, M.F.B.; Rara, F.M. Competitive, Sustainable Natural Pesticides. Acta Ecol. Sin. 2019, 39, 145–151.
- Tomas, F.; Abbott, J.M.; Steinberg, C.; Balk, M.; Williams, S.L.; Stachowicz, J.J. Plant genotype and nitrogen loading influence seagrass productivity, biochemistry, and plant-herbivore interactions. Ecology 2011, 92, 1807–1817.
- Bezemer, T.M.; Wagenaar, R.; Van Dam, N.M.; Wäckers, F.L. Interactions between above- and belowground insect herbivores as mediated by the plant defense system. Oikos 2003, 101, 555–562.
- Rampe, H.L.; Tulung, M.; Pelealu, J.; Runtunuwu, S.D. The Antibiotic and Antixenotic Resistance of Some Peanut (Arachis hypogea L.) Varieties after the Organic Fertilizer Application. Int. J. Res. Eng. Sci. 2015, 3, 40–44.
- Aznar-Fernández, T.; Rubiales, D. Identification and characterisation of antixenosis and antibiosis to pea aphid (Acyrthosiphon pisum) in Pisum spp. germplasm. Ann. Appl. Biol. 2018, 172, 268–281.
- Carr, J.P.; Murphy, A.M.; Tungadi, T.; Yoon, J.Y. Plant defense signals: Players and pawns in plant-virus-vector interactions. Plant Sci. 2019, 279, 87–95.
- Bueno, A.F.; Panizzi, A.R.; Hunt, T.E.; Dourado, P.M.; Pitta, R.M.; Gonçalves, J. Challenges for Adoption of Integrated Pest Management (IPM): The Soybean Example. Neotrop. Entomol. 2021, 50, 5–20.
- Lin, D.; Xu, Y.; Wu, H.; Liu, X.; Zhang, L.; Wang, J.; Rao, Q. Plant defense responses induced by two herbivores and consequences for whitefly Bemisia tabaci. Front. Physiol. 2019, 10, 1–9.
- Emamverdian, A.; Ding, Y.; Mokhberdoran, F.; Xie, Y. Heavy metal stress and some mechanisms of plant defense response. Sci. World J. 2015, 25, 27–54.
- Dalin, P.; Ågren, J.; Björkman, C.; Huttunen, P.; Kärkkäinen, K. Leaf trichome formation and plant resistance to herbivory. In Induced Plant Resistance to Herbivory; Springer: Dordrecht, The Netherlands, 2008; pp. 89–105.
- Wang, Y.; Cao, L.; Zhang, Y.; Cao, C.; Liu, F.; Huang, F.; Qiu, Y.; Li, R.; Lou, X. Map-based cloning and characterization of BPH29, a B3 domain-containing recessive gene conferring brown planthopper resistance in rice. J. Exp. Bot. 2015, 66, 6035–6045.
- Saxena, S.C.; Salvi, P.; Kamble, N.U.; Joshi, P.K.; Majee, M.; Arora, S. Ectopic overexpression of cytosolic ascorbate peroxidase gene (Apx1) improves salinity stress tolerance in Brassica juncea by strengthening antioxidative defense mechanism. Acta Physiol. Plant. 2020, 42, 45.
- Xing, Z.; Liu, Y.; Cai, W.; Huang, X.; Wu, S.; Lei, Z. Efficiency of trichome-based plant defense in phaseolus vulgaris depends on insect behavior, plant ontogeny, and structure. Front. Plant Sci. 2017, 8, 1–8.
- Darvill, A.G.; Albersheim, P. Phytoalexins and their Elicitors-A Defense against Microbial Infection in Plants. Annu. Rev. Plant Physiol. 1984, 35, 243–275.
- Saddique, M.; Kamran, M.; Shahbaz, M. Differential Responses of Plants to Biotic Stress and the Role of Metabolites; Elsevier Inc.: Amsterdam, The Netherlands, 2018; ISBN 9780128126905.
- Zhang, X.; Abrahan, C.; Colquhoun, T.A.; Liu, C.J. A proteolytic regulator controlling chalcone synthase stability and flavonoid biosynthesis in Arabidopsis. Plant Cell 2017, 29, 1157–1174.
- Onkokesung, N.; Reichelt, M.; Van Doorn, A.; Schuurink, R.C.; Van Loon, J.J.A.; Dicke, M. Modulation of flavonoid metabolites in Arabidopsis thaliana through overexpression of the MYB75 transcription factor: Role of kaempferol-3,7- dirhamnoside in resistance to the specialist insect herbivore Pieris brassicae. J. Exp. Bot. 2014, 65, 2203–2217.
- Simmonds, M.S.J. Flavonoid-insect interactions: Recent advances in our knowledge. Phytochemistry 2003, 64, 21–30.
- Simmonds, M.S.J. Importance of flavonoids in insect-plant interactions: Feeding and oviposition. Phytochemistry 2001, 56, 245–252.
- Aboshi, T.; Ishiguri, S.; Shiono, Y.; Murayama, T. Flavonoid glycosides in Malabar spinach Basella alba inhibit the growth of Spodoptera litura larvae. Biosci. Biotechnol. Biochem. 2018, 82, 9–14.
- Du, S.S.; Zhang, H.M.; Bai, C.Q.; Wang, C.F.; Liu, Q.Z.; Liu, Z.L.; Wang, Y.Y.; Deng, Z.W. Nematocidal flavone-C-glycosides against the root-knot nematode (Meloidogyne incognita) from Arisaema erubescens tubers. Molecules 2011, 16, 5079–5086.
- Zhang, Z.; Cui, B.; Yan, S.; Li, Y.; Xiao, H.; Li, Y.; Zhang, Y. Evaluation of tricin, a stylet probing stimulant of brown planthopper, in infested and non-infested rice plants. J. Appl. Entomol. 2017, 141, 393–401.
- Kong, C.H.; Xu, X.H.; Zhang, M.; Zhang, S.Z. Allelochemical tricin in rice hull and its aurone isomer against rice seedling rot disease. Pest Manag. Sci. 2010, 66, 1018–1024.
- Bing, L.; Hongxia, D.; Maoxin, Z.; Di, X.; Jingshu, W. Potential resistance of tricin in rice against brown planthopper Nilaparvata lugens (Stål). Acta Ecol. Sin. 2007, 27, 1300–1306.
- Piasecka, A.; Jedrzejczak-Rey, N.; Bednarek, P. Secondary metabolites in plant innate immunity: Conserved function of divergent chemicals. New Phytol. 2015, 206, 948–964.
- Xiao, Y.; Wang, Q.; Erb, M.; Turlings, T.C.J.; Ge, L.; Hu, L.; Li, J.; Han, X.; Zhang, T.; Lu, J.; et al. Specific herbivore-induced volatiles defend plants and determine insect community composition in the field. Ecol. Lett. 2012, 15, 1130–1139.
- Jang, Y.; Park, J.; Kim, K. Antimicrobial Activity of Chrysoeriol 7 and Chochlioquinone 9, White-Backed Planthopper-Resistant Compounds, Against Rice Pathogenic Strains. Biology 2020, 9, 382.
- Wang, H.; Ye, S.; Mou, T. Molecular Breeding of Rice Restorer Lines and Hybrids for Brown Planthopper (BPH) Resistance Using the Bph14 and Bph15 Genes. Rice 2016, 9, 53.
- Fan, F.; Li, N.; Chen, Y.; Liu, X.; Sun, H.; Wang, J.; He, G.; Zhu, Y.; Li, S. Development of elite BPH-resistant wide-spectrum restorer lines for three and two line hybrid rice. Front. Plant Sci. 2017, 8, 1–9.
- Zhou, S.; Chen, M.; Zhang, Y.; Gao, Q.; Noman, A.; Wang, Q.; Li, H.; Chen, L.; Zhou, P.; Lu, J.; et al. Osmkk3, a stress-responsive protein kinase, positively regulates rice resistance to nilaparvata lugens via phytohormone dynamics. Int. J. Mol. Sci. 2019, 20, 3023.
- Verma, N.; Shukla, S. Impact of various factors responsible for fluctuation in plant secondary metabolites. J. Appl. Res. Med. Aromat. Plants 2015, 2, 105–113.
- Mishra, B.; Priyadarsini, K.I.; Kumar, M.S.; Unnikrishnan, M.K.; Mohan, H. Effect of O-glycosilation on the antioxidant activity and free radical reactions of a plant flavonoid, chrysoeriol. Bioorg. Med. Chem. 2003, 11, 2677–2685.
- Plazonić, A.; Bucar, F.; Maleŝ, Ẑeljan; Mornar, A.; Nigović, B.; Kujundẑić, N. Identification and quantification of flavonoids and phenolic acids in burr parsley (Caucalis platycarpos L.), using high-performance liquid chromatography with diode array detection and electrospray ionization mass spectrometry. Molecules 2009, 14, 2466–2490.
- Kelebek, H.; Canbas, A.; Jourdes, M.; Teissedre, P.L. Characterization of colored and colorless phenolic compounds in Öküzgözü wines from Denizli and Elazig regions using HPLC-DAD-MS. Ind. Crops Prod. 2010, 31, 499–508.
- Gross, B.L.; Skare, K.J.; Olsen, K.M. Novel Phr1 mutations and the evolution of phenol reaction variation in US weedy rice (Oryza sativa). New Phytol. 2009, 184, 842–850.
- Lou, Y.G.; Ma, B.; Cheng, J.A. Attraction of the parasitoid Anagrus nilaparvatae to rice volatiles induced by the rice brown planthopper Nilaparvata lugens. J. Chem. Ecol. 2005, 31, 2357–2372.
- Ashraf, U.; Kanu, A.S.; Mo, Z.; Hussain, S.; Anjum, S.A.; Khan, I.; Abbas, R.N.; Tang, X. Lead toxicity in rice: Effects, mechanisms, and mitigation strategies—A mini review. Environ. Sci. Pollut. Res. 2015, 22, 18318–18332.
- Xu, T.; Zhou, Q.; Xia, Q.; Zhang, W.; Zhang, G.; Gu, D. Effects of herbivore-induced rice volatiles on the host selection behavior of brown planthopper, Nilaparvata lugens. Chin. Sci. Bull. 2002, 47, 1355–1360.
- Shah, A.; Smith, D.L. Flavonoids in agriculture: Chemistry and roles in, biotic and abiotic stress responses, and microbial associations. Agronomy 2020, 10, 1209.
- Jan, R.; Khan, M.A.; Asaf, S.; Lee, I.J.; Kim, K.M. Overexpression of OsF3H modulates WBPH stress by alteration of phenylpropanoid pathway at a transcriptomic and metabolomic level in Oryza sativa. Sci. Rep. 2020, 10.
- Park, J.R.; Yun, S.; Jan, R.; Kim, K.M. Screening and identification of brown planthopper resistance genes OsCM9 in rice. Agronomy 2020, 10, 1865.
- Shimono, M.; Koga, H.; Akagi, A.; Hayashi, N.; Goto, S.; Sawada, M.; Kurihara, T.; Matsushita, A.; Sugano, S.; Jiang, C.J.; et al. Rice WRKY45 plays important roles in fungal and bacterial disease resistance. Mol. Plant Pathol. 2012, 13, 83–94.
- Yuan, Y.; Zhong, S.; Li, Q.; Zhu, Z.; Lou, Y.; Wang, L.; Wang, J.; Wang, M.; Li, Q.; Yang, D.; et al. Functional analysis of rice NPR1-like genes reveals that OsNPR1/NH1 is the rice orthologue conferring disease resistance with enhanced herbivore susceptibility. Plant Biotechnol. J. 2007, 5, 313–324.
- Hassan, S.; Mathesius, U. The role of flavonoids in root-rhizosphere signalling: Opportunities and challenges for improving plant-microbe interactions. J. Exp. Bot. 2012, 63, 3429–3444.





