
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Eloah Rabello Suarez | + 2808 word(s) | 2808 | 2022-03-15 06:40:27 | | | |
| 2 | Peter Tang | Meta information modification | 2808 | 2022-03-17 02:02:19 | | |
Video Upload Options
Carbonic anhydrases are metalloenzymes that reversibly catalyze the hydration of carbon dioxide, generating bicarbonate ions and protons. Several tumors, such as clear cell renal cell carcinoma (ccRCC), glioblastoma, triple-negative breast cancer, ovarian cancer, colorectal, and others overexpress carbonic anhydrase isoform IX (CAIX). The CAIX enzyme is constitutively overexpressed in the vast majority of clear cell renal cell carcinoma (ccRCC) and can also be induced in hypoxic microenvironments, a major hallmark of most solid tumors. CAIX expression is restricted to a few sites in healthy tissues, positioning this molecule as a strategic target for cancer immunotherapy.
1. Introduction
2. Anti-CAIX Monoclonal Antibodies: Preclinical and Clinical Efficacy
2.1. Murine G250 IgG1 mAb—Isolated and Associated with Radionuclides
|
Author |
Antibody Type |
Tumor Type |
Dosage |
Response |
|---|---|---|---|---|
|
Surfus et al. (1996) [17] |
cG250 |
RCC and breast carcinoma cell lines |
cG250: 0.5 µg/mL, IL2: 100 U/mL |
ADCC with PBMCs (effector to target rate 100:1) after 4 h RCC—SK-RC-13: cG250 48%; cG250 + IL2 50%; SK-RC-30: cG250 25%; cG250 + IL2 65%; Breast cancer—BT-20: cG250 38%; cG250 + IL2 28% |
|
Liu et al. (2002) [18] |
cG250 |
RCC and chronic myelogenous leukemia |
cG250: 1 µg/mL, IL2 10 IU/mL; IFNγ, IFN-2a, IFN-2b 1000 IU/mL |
ADCC with PBMC (effector to target rate 25:1) after 2 days RCC—SK-RC-52: cG250 + IL2 42%; cG250 + IFN-? 33%; cG250 + IFN-?-2a or cG250 + IFN-α-2b 25%; SK-RC-09: cG250 + IL2 28%; cG250 + IFN-?; cG250 + IFN-?-2a, and cG250 + IFN-α-2b < 10%; Leukemia—K562: cG250 + IL2 60%; cG250 + IFN-? 30%; cG250 + IFN-?-2a or cG250 + INF-α-2b 43% |
|
Brouwers et al. (2004) [19] |
131I-cG250, 90Y-SCN-Bz- DTPA-cG250, 177Lu-SCN-Bz-DTPA-cG250, or 186Re-MAG3 cG250 |
RCC |
30 µg 131I-cG250, 30 µg 90Y-SCN-Bz-DTPA-cG250, 60 µg 177Lu-SCN-Bz-DTPA-cG250, or 35 µg 186Re-MAG3-cG250; Variable doses of radioisotopes |
Best median survival (SK-RC-52 cells) 177Lu-SCN-Bz-DTPA cG250: 294 days; 90Y-SCN-Bz-DTPA cG250: 241 days; 186Re-MAG3-cG250: 211 days; 131I-cG250: 164 days; Control groups < 150 days |
|
Bauer et al. (2009) [20] |
cG250-TNF and cG250 |
RCC |
100 µg of cG250 or cG250-TNF 300 ng every 3 days |
In vivo tumor size after 78 days (SK-RC-52 cells) cG250-TNF + IFNγ: 60% decrease; cG250-TNF: 50% decrease; cG250 + IFNγ: no difference in tumor size compared to negative control |
|
Zatovicova et al. (2010) [21] |
VII/20 |
Colorectal carcinoma |
100 μg twice a week |
In vivo tumor weight/volume reduction (HT-29 cells) 60%/73% treatment initiated after 10 days of tumor implantation; 88%/93% treatment initiated in the same day of tumor implantation |
|
Oosterwijk-Wakka et al. (2011) [22] |
125I-cG250 + sorafenib, sunitinib, or vandetanib |
RCC |
125I-cG250 185 kBq/5 μg 35 mg/kg of sunitinib, 50 mg/kg of sorafenib, 50 mg/kg of vandetanib |
In vivo tumor volume (NU-12 cells) decrease for continuous treatment (14 days) Vandetanib: 57%, sunitinib: 49%, and sorafenib: 37%, all compared to 125I-cG250 alone |
|
Petrul et al. (2012) [23] |
BAY 79-4620 |
Colorectal cancer, gastric carcinoma, and NSCLC-PDX |
Variable |
In vivo tumor regression (3 doses of every 4 days) Colorectal cancer (dose 10 mg/kg): HT-29: 100%, Colo205: 85%; Gastric carcinoma (dose 60 mg/kg): NCI-N87: 87%, MKN-45: 90%, SNU-16: 75%; NSCLC-PDX: complete regression in 2/5, partial regression in 3/5 |
|
Muselaers et al. (2014) [15] |
111In-DOTA-mG250 and 177Lu-DOTA-mG250 |
RCC |
13 MBq 177Lu-DOTA-mG250, 13 MBq nonspecific 177Lu-DOTA-MOPC21, 20 MBq 111In-DOTA-mG250 |
Median survival (SK-RC-52 cells) 177Lu-DOTA-mG250: 139 days; 177Lu-DOTA-MOPC21: 49 days; 111In-DOTA-mG250: 53 days; Control: 49–53 days |
|
Zatovicova et al. (2014) [16] |
mG250 |
Colorectal carcinoma |
100 µg/dose |
In vivo tumor weight/volume reduction (HT-29 cells) Treatment initiated after 10 days of tumor implantation: 55%/73%; Treatment initiated at the same day of tumor implantation: 90%/93% |
|
Chang et al. (2015) [24] |
In vitro: G10, G36, G37, G39, and G119; In vivo: only G37 and G119 were tested |
RCC |
ADCC in vitro: 5 µg/mL, In vivo: 10 mg/kg |
ADCC in SK-RC-09 cells: 25:1 effector to target cells: 25% for G36 and G119; 15–20% for G10, G37, and G39; 50:1 effector to target cells: 45% for G10, G36, G37, and G119; 30% for G39 In vivo tumor weight (Day 29)/volume (Day 28) reduction (SK-RC-59 CAIX+ cells): 85%/75% for G37, G119, mG37, and mG119 |
|
Oosterwijk-Wakka et al. (2015) [25] |
111In-cG250 and Sunitinib |
RCC |
0.4 MBq/5 µg 111In-cG250 three days after administration of 40–50 mg/kg of sunitinib for 13 days |
In vivo tumor growth reduction 20 days after the beginning of the treatment with sunitinib NU-12: 60%; SK-RC-52: not statistically significant compared to control |
|
Yamaguchi et al. (2015) [26] |
chKM4927 and chKM4927_N297D |
RCC |
10 mg/kg i.p. twice a week for three weeks |
In vivo tumor volume (VMRC-RCW cells) reduction after 32 days chKM4927 and chKM4927_N297D: 60% compared to negative control |
|
Lin et al. (2017) [27] |
Anti-CAIX functionalized liposomes with TPL |
Lung cancer cells |
0.15 mg/kg once every 3–4 days for 8 times via pulmonary delivery |
Median survival time (A549 cells) CAIX-TPL-Lips: 90 days (statistically significant compared to saline control); Nontargeted TPL-lips: 71 days (not statistically significant compared to saline control); Control group: 45 days |
|
De Luca et al. (2019) [28] |
IL2-Anti-CAIX(XE114)-TNFmut and IL2-Anti-CAIX(F8)-TNFmut |
Colon Carcinoma |
30 µg i.v. four times every 24 h |
Tumor volume reduction (CT26-CAIX cells) after 18 days IL2-F8-TNFmut: 58%; mIL2-F8-mTNFmut: 72%; IL2-XE114-TNFmut: 63%; mIL2-XE114-mTNFmut: 50% |
|
Author |
Phase |
Treatment |
Clinical Response |
Adverse Effects (≥3 Grade) |
|---|---|---|---|---|
|
Divgi et al. (1998) [14] |
I/II |
mG250 (10 mg single i.v. infusion) combined with 131I (30, 45, 60, 75, and 90 mCi/m2) |
1/33 CR; 17/33 SD—2 months after treatment |
19/33 grade 3 (thrombocytopenia, hematotoxicity, hepatoxicity); 3/33 grade 4 (thrombocytopenia and hematotoxicity); 33/33 HAMA |
|
Steffens et al. (1999) [29] |
I |
cG250 (5 mg single i.v. infusion) combined with 131l (222–2775 MBq/m2) |
6/12 PD; 1/12 SD—lasting 3–6 months; 1/12 PR—9 months or longer |
1/12 grade 3 (leukocytopenia); 2/12 grade 4 (thrombocytopenia and leukocytopenia); 1/12 HACA |
|
Bleumer et al. (2004) [30] |
II |
cG250 (25 mg/m2 weekly i.v. infusion for 12 weeks) |
10/36 SD, 17/36 PD—week 16; 8/36 SD—week 24; 1/36 CR, 1/36 PR—week 38–44 |
* 33/36 grade 3 (pain, pulmonary, cardiovascular, constitutional symptoms, neurological, bone marrow, genitourinary, hemorrhage, hepatic, metabolic/laboratory); 5/36 grade 4 (pulmonary, hemorrhage) |
|
Bleumer et al. (2006) [31] |
III |
cG250 (20 mg by i.v. infusion for 11 weeks) combined with IL2 (1.8–5.4 MIU daily for 12 consecutive weeks) |
1/35 PR, 11/35 SD, 23/35 PD—week 16; 1/35 PR, 7/35 SD, 4/35 PD—week 22 |
17/35 grade 3 (constitutional symptoms, pain, pulmonary, blood/bone marrow, hepatic); 2/35 grade 4 (renal/genitourinary and metabolic/laboratory); 2/36 HACA |
|
Davis et al. (2007) [32] |
Pilot |
cG250 (10 mg/m2/week, first and fifth doses trace-labeled with 131I) and 1.25 × 106 IU/m2/day IL2 for six weeks |
2/9 SD, 7/9 PD—after six-week cycle 1; 1/9 SD, 1/9 PD—after six-week cycle 2 |
* 3/9 grade 3 or 4 (dyspnea and anemia) |
|
Davis et al. (2007) [33] |
I |
cG250 (5, 10, 25, or 50 mg/m2 i.v. for 6 weeks) combined with 131l (200–350 MBq/m2) weeks 1 and 5 |
1/13 CR, 8/13 SD, 3/13 PD—first six-weeks cycle; 1/13 CR, 6/13 SD, 2/13 PD—second six-weeks cycle |
* 1/13 grade 3 (bone pain), 1/13 HACA |
|
Siebels et al. (2010) [34] |
I/II |
cG250 (20 mg i.v. infusion; week 2–12) combined with LD-IFNα (3 MIU s.c. 3 times/week; weeks 1–12) |
2/26 PR, 14/26 SD—week 16; 1/26 CR, 9/26 SD—24 weeks or longer |
11/26 grade 3 (constitutional symptoms, pain, pulmonary, musculoskeletal, cardiovascular, secondary malignancy, lymphatics); 1/26 grade 4 (gastrointestinal) |
|
Stillebroer et al. (2013) [35] |
I |
cG250 (10 mg i.v. infusion—three consecutive) combined with 131ln (1110–2405 MBq/m2) |
17/23 SD—during the 3 months 1/23 PR—lasted 9 months |
3/23 grade 4 (myelotoxicity); 4/23 HACA |
|
Muselaers et al. (2016) [36] |
II |
cG250 (10 mg i.v. infusion) combined with 111In (185 MBq/m2); 177Lu (2405 MBq/m2) 9–10 days after infusion; 177Lu (1805 MBq/m2) weeks 12–14 |
1/14 PR, 8/14 SD, 5/9 PD—after cycle 1; 1/14 PR, 4/14 SD, 1/14 PD—after cycle 2 |
12/14 grade 3–4 (thrombocytopenia); 9/14 grade 3–4 (leukocytopenia); 2/14 grade 3 (fatigue and anorexia); 4/14 grade 4 (neutropenia) |
|
Chamie et al. (2017) [37] |
III |
cG250 (50 mg i.v.; week 1; 20 mg i.v. weeks 2–24) |
NR |
72/864 grade 3 or 4—type not mentioned |
PD: progressive disease, SD: stable disease, PR: partial response, CR: complete response, MTD: maximum tolerated dose, ND: not detected, NE: not evaluable, NR: no response, HAMA: human antimouse antibodies, HACA: human anti-chimeric antibodies. * All grade 3 and 4 toxicities were not related to the study medication. Doses highlighted in bold are related to clinical responses reported.
2.2. Humanized Chimeric Monoclonal Antibody IgG1 G250 (cG250)—Isolated or Associated with Cytokines
2.3. Chimeric Monoclonal Antibody G250 (cG250) Conjugate with Radionuclides
2.4. cG250 and Other Associations
2.5. Other Antibodies
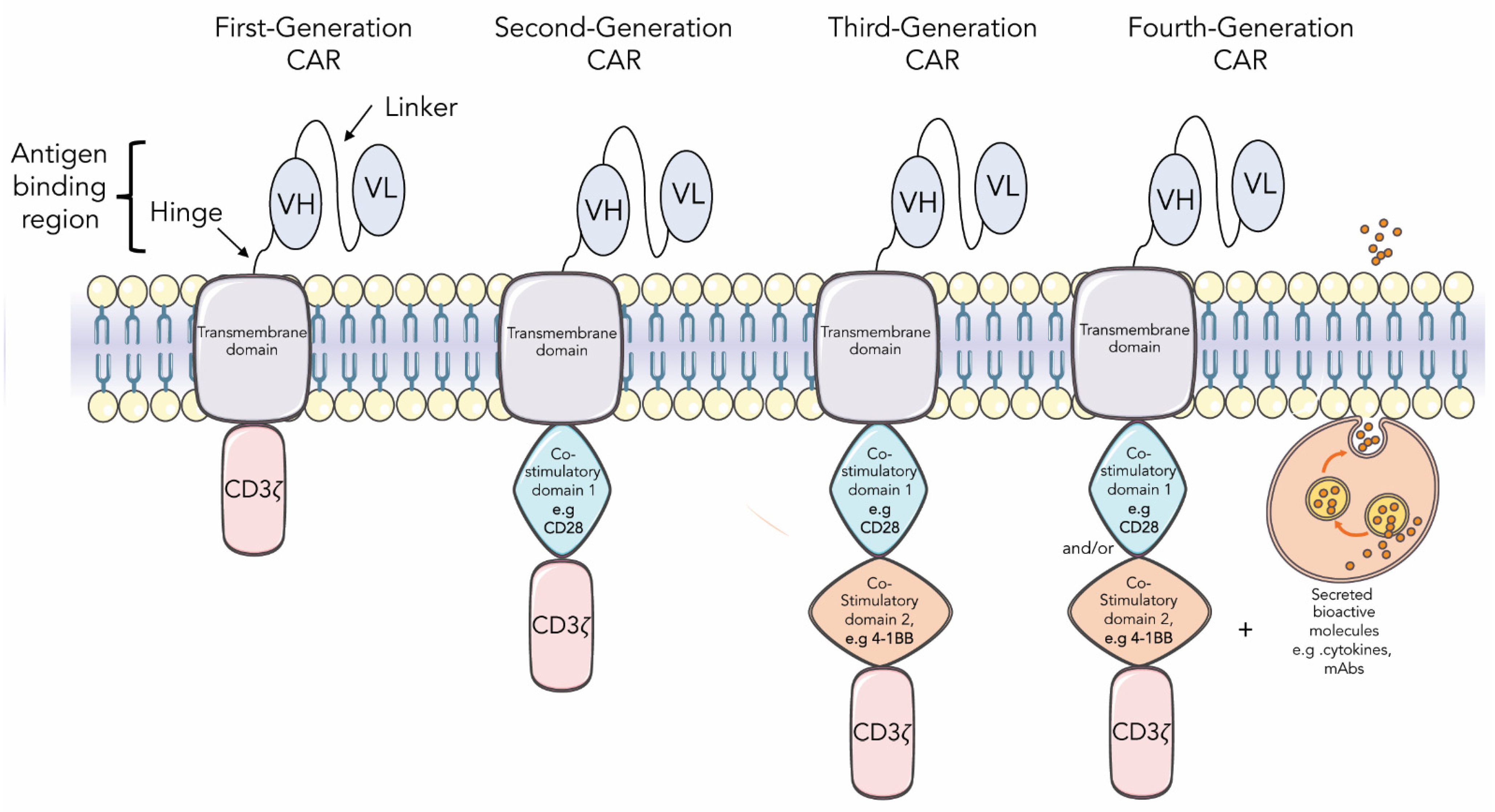
2.6. Fusion Proteins
3. Current Insights
References
- Mahon, B.P.; Pinard, M.A.; McKenna, R. Targeting carbonic anhydrase IX activity and expression. Molecules 2015, 20, 2323–2348.
- Genega, E.M.; Ghebremichael, M.; Najarian, R.; Fu, Y.; Wang, Y.; Argani, P.; Grisanzio, C.; Signoretti, S. Carbonic anhydrase IX expression in renal neoplasms: Correlation with tumor type and grade. Am. J. Clin. Pathol. 2010, 134, 873–879.
- Parks, S.K.; Chiche, J.; Pouyssegur, J. pH control mechanisms of tumor survival and growth. J. Cell. Physiol. 2011, 226, 299–308.
- Maxwell, P.H.; Wiesener, M.S.; Chang, G.-W.; Clifford, S.C.; Vaux, E.C.; Cockman, M.E.; Wykoff, C.C.; Pugh, C.W.; Maher, E.R.; Ratcliffe, P.J. The tumour suppressor protein VHL targets hypoxia-inducible factors for oxygen-dependent proteolysis. Nature 1999, 399, 271–275.
- Lau, J.; Lin, K.-S.; Bénard, F. Past, Present, and Future: Development of Theranostic Agents Targeting Carbonic Anhydrase IX. Theranostics 2017, 7, 4322–4339.
- Takacova, M.; Barathova, M.; Zatovicova, M.; Golias, T.; Kajanova, I.; Jelenska, L.; Sedlakova, O.; Svastova, E.; Kopacek, J.; Pastorekova, S. Carbonic Anhydrase IX-Mouse versus Human. Int. J. Mol. Sci. 2019, 21, 246.
- Ramachandran, K.; Bindhu, M.R.; Jojo, A.; Pooleri, G.K.; Thomas, A. Role of CAIX Expression in Conventional Renal Cell Carcinomas as a Diagnostic Marker and its Prognostic Importance. Indian J. Surg. Oncol. 2021, 12, 79–84.
- Luong-Player, A.; Liu, H.; Wang, H.L.; Lin, F. Immunohistochemical reevaluation of carbonic anhydrase IX (CA IX) expression in tumors and normal tissues. Am. J. Clin. Pathol. 2014, 141, 219–225.
- Singh, A.K.; McGuirk, J.P. CAR T cells: Continuation in a revolution of immunotherapy. Lancet Oncol. 2020, 21, e168–e178.
- Cui, J.; Zhang, Q.; Song, Q.; Wang, H.; Dmitriev, P.; Sun, M.Y.; Cao, X.; Wang, Y.; Guo, L.; Indig, I.H.; et al. Targeting hypoxia downstream signaling protein, CAIX, for CAR T-cell therapy against glioblastoma. Neuro-Oncology 2019, 21, 1436–1446.
- Oosterwdk, E.; Ruiter, D.J.; Hoedemaeker, P.J.; Pauwels, E.K.J.; Jonas, U.; Zwartendijk, I.; Warnaar, S.O. Monoclonal antibody G 250 recognizes a determinant present in renal-cell carcinoma and absent from normal kidney. Int. J. Cancer 1986, 38, 489–494.
- van Dijk, J.; Oosterwijk, E.; van Kroonenburgh, M.J.; Jonas, U.; Fleuren, G.J.; Pauwels, E.K.; Warnaar, S.O. Perfusion of tumor-bearing kidneys as a model for scintigraphic screening of monoclonal antibodies. J. Nucl. Med. 1988, 29, 1078–1082.
- Oosterwijk, E.; Bander, N.H.; Divgi, C.R.; Welt, S.; Wakka, J.C.; Finn, R.D.; Carswell, E.A.; Larson, S.M.; Warnaar, S.O.; Fleuren, G.J.; et al. Antibody localization in human renal cell carcinoma: A phase I study of monoclonal antibody G250. J. Clin. Oncol. 1993, 11, 738–750.
- Divgi, C.R.; Bander, N.H.; Scott, A.M.; O’Donoghue, J.A.; Sgouros, G.; Welt, S.; Finn, R.D.; Morrissey, F.; Capitelli, P.; Williams, J.M.; et al. Phase I/II radioimmunotherapy trial with iodine-131-labeled monoclonal antibody G250 in metastatic renal cell carcinoma. Clin. Cancer Res. 1998, 4, 2729–2739.
- Muselaers, C.H.J.; Oosterwijk, E.; Bos, D.L.; Oyen, W.J.G.; Mulders, P.F.A.; Boerman, O.C. Optimizing lutetium 177-anti-carbonic anhydrase IX radioimmunotherapy in an intraperitoneal clear cell renal cell carcinoma xenograft model. Mol. Imaging 2014, 13, 1–7.
- Zatovicova, M.; Jelenska, L.; Hulikova, A.; Ditte, P.; Ditte, Z.; Csaderova, L.; Svastova, E.; Schmalix, W.; Boettger, V.; Bevan, P.; et al. Monoclonal antibody G250 targeting CA IX: Binding specificity, internalization and therapeutic effects in a non-renal cancer model. Int. J. Oncol. 2014, 45, 2455–2467.
- Surfus, J.E.; Hank, J.A.; Oosterwijk, E.; Welt, S.; Lindstrom, M.J.; Albertini, M.R.; Schiller, J.H.; Sondel, P.M. Anti-renal-cell carcinoma chimeric antibody G250 facilitates antibody-dependent cellular cytotoxicity with in vitro and in vivo interleukin-2-activated effectors. J. Immunother. Emphas. Tumor Immunol. 1996, 19, 184–191.
- Liu, Z.; Smyth, F.E.; Renner, C.; Lee, F.-T.; Oosterwijk, E.; Scott, A.M. Anti-renal cell carcinoma chimeric antibody G250: Cytokine enhancement of in vitro antibody-dependent cellular cytotoxicity. Cancer Immunol. Immunother. 2002, 51, 171–177.
- Brouwers, A.H.; van Eerd, J.E.M.; Frielink, C.; Oosterwijk, E.; Oyen, W.J.G.; Corstens, F.H.M.; Boerman, O.C. Optimization of Radioimmunotherapy of Renal Cell Carcinoma: Labeling of Monoclonal Antibody cG250 with 131I, 90Y, 177Lu, or 186Re. J. Nucl. Med. 2004, 45, 327–337.
- Bauer, S.; Oosterwijk-Wakka, J.C.; Adrian, N.; Oosterwijk, E.; Fischer, E.; Wüest, T.; Stenner, F.; Perani, A.; Cohen, L.; Knuth, A.; et al. Targeted therapy of renal cell carcinoma: Synergistic activity of cG250-TNF and IFNg. Int. J. Cancer 2009, 125, 115–123.
- Zatovicova, M.; Jelenska, L.; Hulikova, A.; Csaderova, L.; Ditte, Z.; Ditte, P.; Goliasova, T.; Pastorek, J.; Pastorekova, S. Carbonic Anhydrase IX as an Anticancer Therapy Target: Preclinical Evaluation of Internalizing Monoclonal Antibody Directed to Catalytic Domain. Curr. Pharm. Des. 2010, 16, 3255–3263.
- Oosterwijk-Wakka, J.C.; Kats-Ugurlu, G.; Leenders, W.P.J.; Kiemeney, L.A.L.M.; Old, L.J.; Mulders, P.F.A.; Oosterwijk, E. Effect of tyrosine kinase inhibitor treatment of renal cell carcinoma on the accumulation of carbonic anhydrase IX-specific chimeric monoclonal antibody cG250: Effect of TKIs on the accumulation of caix-specific cG250 in RCC. BJU Int. 2011, 107, 118–125.
- Petrul, H.M.; Schatz, C.A.; Kopitz, C.C.; Adnane, L.; McCabe, T.J.; Trail, P.; Ha, S.; Chang, Y.S.; Voznesensky, A.; Ranges, G.; et al. Therapeutic mechanism and efficacy of the antibody-drug conjugate BAY 79-4620 targeting human carbonic anhydrase 9. Mol. Cancer Ther. 2012, 11, 340–349.
- Chang, D.-K.; Moniz, R.J.; Xu, Z.; Sun, J.; Signoretti, S.; Zhu, Q.; Marasco, W.A. Human anti-CAIX antibodies mediate immune cell inhibition of renal cell carcinoma in vitro and in a humanized mouse model in vivo. Mol. Cancer 2015, 14, 119.
- Oosterwijk-Wakka, J.C.; de Weijert, M.C.A.; Franssen, G.M.; Leenders, W.P.J.; van der Laak, J.A.W.M.; Boerman, O.C.; Mulders, P.F.A.; Oosterwijk, E. Successful Combination of Sunitinib and Girentuximab in Two Renal Cell Carcinoma Animal Models: A Rationale for Combination Treatment of Patients with Advanced RCC. Neoplasia 2015, 17, 215–224.
- Yamaguchi, A.; Usami, K.; Shimabe, M.; Hasegawa, K.; Asada, M.; Motoki, K.; Tahara, T.; Masuda, K. The novel CA IX inhibition antibody chKM4927 shows anti-tumor efficacy in vivo. Anticancer Res. 2015, 35, 1997–2004.
- Lin, C.; Wong, B.C.K.; Chen, H.; Bian, Z.; Zhang, G.; Zhang, X.; Kashif Riaz, M.; Tyagi, D.; Lin, G.; Zhang, Y.; et al. Pulmonary delivery of triptolide-loaded liposomes decorated with anti-carbonic anhydrase IX antibody for lung cancer therapy. Sci. Rep. 2017, 7, 1097.
- De Luca, R.; Gouyou, B.; Ongaro, T.; Villa, A.; Ziffels, B.; Sannino, A.; Buttinoni, G.; Galeazzi, S.; Mazzacuva, M.; Neri, D. A Novel Fully-Human Potency-Matched Dual Cytokine-Antibody Fusion Protein Targets Carbonic Anhydrase IX in Renal Cell Carcinomas. Front. Oncol. 2019, 9, 1228.
- Steffens, M.G.; Boerman, O.C.; de Mulder, P.H.; Oyen, W.J.; Buijs, W.C.; Witjes, J.A.; van den Broek, W.J.; Oosterwijk-Wakka, J.C.; Debruyne, F.M.; Corstens, F.H.; et al. Phase I radioimmunotherapy of metastatic renal cell carcinoma with 131I-labeled chimeric monoclonal antibody G250. Clin. Cancer Res. 1999, 5, 3268s–3274s.
- Bleumer, I.; Knuth, A.; Oosterwijk, E.; Hofmann, R.; Varga, Z.; Lamers, C.; Kruit, W.; Melchior, S.; Mala, C.; Ullrich, S.; et al. A phase II trial of chimeric monoclonal antibody G250 for advanced renal cell carcinoma patients. Br. J. Cancer 2004, 90, 985–990.
- Bleumer, I.; Oosterwijk, E.; Oosterwijk-Wakka, J.C.; Völler, M.C.W.; Melchior, S.; Warnaar, S.O.; Mala, C.; Beck, J.; Mulders, P.F.A. A Clinical Trial with Chimeric Monoclonal Antibody WX-G250 and Low Dose Interleukin-2 Pulsing Scheme for Advanced Renal Cell Carcinoma. J. Urol. 2006, 175, 57–62.
- Davis, I.D.; Liu, Z.; Saunders, W.; Lee, F.-T.; Spirkoska, V.; Hopkins, W.; Smyth, F.E.; Chong, G.; Papenfuss, A.T.; Chappell, B.; et al. A pilot study of monoclonal antibody cG250 and low dose subcutaneous IL-2 in patients with advanced renal cell carcinoma. Cancer Immun. 2007, 7, 14.
- Davis, I.D.; Wiseman, G.A.; Lee, F.-T.; Gansen, D.N.; Hopkins, W.; Papenfuss, A.T.; Liu, Z.; Moynihan, T.J.; Croghan, G.A.; Adjei, A.A.; et al. A phase I multiple dose, dose escalation study of cG250 monoclonal antibody in patients with advanced renal cell carcinoma. Cancer Immun. 2007, 7, 13.
- Siebels, M.; Rohrmann, K.; Oberneder, R.; Stahler, M.; Haseke, N.; Beck, J.; Hofmann, R.; Kindler, M.; Kloepfer, P.; Stief, C. A clinical phase I/II trial with the monoclonal antibody cG250 (RENCAREX®) and interferon-alpha-2a in metastatic renal cell carcinoma patients. World J. Urol. 2010, 29, 121–126.
- Stillebroer, A.B.; Boerman, O.C.; Desar, I.M.E.; Boers-Sonderen, M.J.; van Herpen, C.M.L.; Langenhuijsen, J.F.; Smith-Jones, P.M.; Oosterwijk, E.; Oyen, W.J.G.; Mulders, P.F.A. Phase 1 radioimmunotherapy study with lutetium 177-labeled anti-carbonic anhydrase IX monoclonal antibody girentuximab in patients with advanced renal cell carcinoma. Eur. Urol. 2013, 64, 478–485.
- Muselaers, C.H.J.; Boers-Sonderen, M.J.; van Oostenbrugge, T.J.; Boerman, O.C.; Desar, I.M.E.; Stillebroer, A.B.; Mulder, S.F.; van Herpen, C.M.L.; Langenhuijsen, J.F.; Oosterwijk, E.; et al. Phase 2 Study of Lutetium 177-Labeled Anti-Carbonic Anhydrase IX Monoclonal Antibody Girentuximab in Patients with Advanced Renal Cell Carcinoma. Eur. Urol. 2016, 69, 767–770.
- Chamie, K.; Donin, N.M.; Klöpfer, P.; Bevan, P.; Fall, B.; Wilhelm, O.; Störkel, S.; Said, J.; Gambla, M.; Hawkins, R.E.; et al. Adjuvant Weekly Girentuximab Following Nephrectomy for High-Risk Renal Cell Carcinoma: The ARISER Randomized Clinical Trial. JAMA Oncol. 2017, 3, 913–920.
- Muselaers, C.H.; Stillebroer, A.B.; Desar, I.M.; Boers-Sonderen, M.J.; van Herpen, C.M.; de Weijert, M.C.; Langenhuijsen, J.F.; Oosterwijk, E.; Leenders, W.P.; Boerman, O.C.; et al. Tyrosine kinase inhibitor sorafenib decreases 111In-girentuximab uptake in patients with clear cell renal cell carcinoma. J. Nucl. Med. 2014, 55, 242–247.
- Arjaans, M.; Oude Munnink, T.H.; Oosting, S.F.; Terwisscha van Scheltinga, A.G.; Gietema, J.A.; Garbacik, E.T.; Timmer-Bosscha, H.; Lub-de Hooge, M.N.; Schroder, C.P.; de Vries, E.G. Bevacizumab-induced normalization of blood vessels in tumors hampers antibody uptake. Cancer Res. 2013, 73, 3347–3355.
- Ahlskog, J.K.; Schliemann, C.; Mårlind, J.; Qureshi, U.; Ammar, A.; Pedley, R.B.; Neri, D. Human monoclonal antibodies targeting carbonic anhydrase IX for the molecular imaging of hypoxic regions in solid tumours. Br. J. Cancer 2009, 101, 645–657.
- Xu, C.; Lo, A.; Yammanuru, A.; Tallarico, A.S.C.; Brady, K.; Murakami, A.; Barteneva, N.; Zhu, Q.; Marasco, W.A. Unique Biological Properties of Catalytic Domain Directed Human Anti-CAIX Antibodies Discovered through Phage-Display Technology. PLoS ONE 2010, 5, e9625.
- Rafiq, S.; Hackett, C.S.; Brentjens, R.J. Engineering strategies to overcome the current roadblocks in CAR T cell therapy. Nat. Rev. Clin. Oncol. 2019, 17, 147–167.





