Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Manuela Giraldo Acosta | + 1236 word(s) | 1236 | 2022-02-28 07:29:05 | | | |
| 2 | Amina Yu | + 65 word(s) | 1301 | 2022-03-09 03:24:03 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Acosta, M. Melatonin in Brassicaceae. Encyclopedia. Available online: https://encyclopedia.pub/entry/20342 (accessed on 25 May 2026).
Acosta M. Melatonin in Brassicaceae. Encyclopedia. Available at: https://encyclopedia.pub/entry/20342. Accessed May 25, 2026.
Acosta, Manuela. "Melatonin in Brassicaceae" Encyclopedia, https://encyclopedia.pub/entry/20342 (accessed May 25, 2026).
Acosta, M. (2022, March 08). Melatonin in Brassicaceae. In Encyclopedia. https://encyclopedia.pub/entry/20342
Acosta, Manuela. "Melatonin in Brassicaceae." Encyclopedia. Web. 08 March, 2022.
Copy Citation
The family Brassicaceae, also known as Cruciferae, includes approximately 372 genera and 4060 species. It is distributed throughout the world, and its distribution pattern suggests that this plant family originated and was diversified from the eastern Mediterranean. Brassicaceae is a large family of plants that have a wide range of applications, including human consumption in vegetables, seed oils and condiments, livestock in fodder and others.Arabidopsis thaliana, which is well known in the research, belongs to this family.
broccoli
melatonin
postharvest
glucosinolates
vegetables
1. Brassicaceae Plants and Melatonin Studies
The family Brassicaceae, also known as Cruciferae, includes approximately 372 genera and 4060 species. It is distributed throughout the world, and its distribution pattern suggests that this plant family originated and was diversified from the eastern Mediterranean. Brassicaceae is a large family of plants that have a wide range of applications, including human consumption in vegetables, seed oils and condiments, livestock in fodder and others. Arabidopsis thaliana, which is well known in the research, belongs to this family. Among the vegetables that stand out for their high consumption are broccolis, cauliflowers, cabbages, collard greens, turnips and radishes; all of these derive from cultivars of Brassica rapa L. and Brassica oleracea L., which present a multitude of varieties and/or subspecies that provide an enormous culinary richness in the different regions where they are grown. Brassica napus L. var oleracea is cultivated worldwide for rapeseed oil. The three types of mustard: yellow (Sinapsis alba L.), black (Brassica nigra L.) and brown or Indian (Brassica juncea L.) are also widely cultivated and appreciated in gastronomy [1][2].
Multiple studies of melatonin with A. thaliana can be consulted. In general, melatonin has a vegetative-growth-promoting effect, activating the biosynthesis of plant hormones, and balancing redox homeostasis, especially in situations of abiotic stress. A clear effect of Brassicaceae is the activation of anthocyanin biosynthesis, together with a foliar senescence-retarding effect. Therefore, melatonin treatments in plants, either foliar or root, result in plants of larger size, biomass and color and those with greater tolerance to stress—all very interesting aspects to be applied in the crops of the different Brassicaceae.
2. Postharvest Application of Melatonin in Brassicaceae
One of the most interesting aspects on melatonin is its ability as a biomodulator of ripening and senescence. There are many interesting results in the postharvest treatments of fruits such as apple, pear, tomato, cucumber, grape, plum, peach, apricot, cherry, strawberry, pomegranate, banana and so on, and in flowers such as carnation, anthurium, devil’s trumpet and peony. Some recent reviews on this topic, in which multiple data are analyzed and a scheme of action of melatonin on postharvest conservation is proposed, can be consulted [3][4][5][6][7].
Melatonin treatment in broccoli florets shows interesting results for the postharvest conservation of this vegetable frequently consumed worldwide. Visual and quality characteristics are clearly improved by treatments around 100 μM melatonin. Short treatments by immersion or spray induce a lower loss of chlorophylls and carotenoids, and therefore, an improvement in the chromatic spectrum of broccoli florets, as can be seen by a greater degree of hue angle (greener, bluer, less yellow). This anti-senescence effect was first described in 2009 in barley leaves [8], and has subsequently been verified in a multitude of plant species, both edible and wildtype, where an inhibitory effect of melatonin on activating senescence transcription factors was demonstrated [9][10][11][4][12][13]. In addition, broccoli florets preserve freshness better, with less hydric and texture loss. In general, the maintenance and cold transport of broccoli florets is essential for an acceptable shelf life. Temperatures around 4 °C are ideal for reaching a maximum duration of about 20–22 days. Melatonin treatments clearly enhance visual and quality characteristics, improving color, texture and shine, and are able to extend its shelf life between 5–8 days with maximum quality. Other parameters such as soluble solids, total acid content, astringency, bitterness and vitamin C content are improved with melatonin treatments.
Regarding the compounds of nutraceutical interest in broccoli florets, melatonin treatments increase the contents of total phenols and flavonoids, and some specific ones such as rutin and quercetin. Melatonin induces the expression of diverse transcripts of phenolic metabolism. In white and red cabbage seedlings, melatonin upregulates phenolic-related genes such as PAL, cinnamic acid 4-hydroxylase (C4H), CHS, CHI, F3H, DFR and UFGT, among others [14]. Moreover, in fruits such as berries and kiwifruit, melatonin induces several flavonoid biosynthesis genes [15][16]. Melatonin has also been found to cause a general activation of the primary and secondary metabolism that leads to high levels of carbohydrates, lipids and amino acids, and a better energy level and redox status [17][4].
A characteristic of Brassicaceae, though not exclusive to it, is its relevant level in glucosinolates, which are a group of secondary metabolites. Evolutionarily, glucosinolates originated twice, so that they are found in two unrelated lines of plants: in the Brassicales order (mainly Brassicaceae, Capparaceae and Caricaceae families) and in the Putranjivaceae family [18]. These compounds contain N- and/or S- in their chemical structure, formulated as β-thioglucoside-N-hydroxysulfates. More than 120 glucosinolate compounds have been identified, and they can be classified as aliphatic glucosinolates (originating from methionine, valine, leucine or isoleucine), indole glucosinolates (originating from tryptophan) and aromatic glucosinolates (from phenylalanine or tyrosine) [19]. These secondary metabolites are relevant in the defense system of plants due their fungicide, bactericide, nematicide and allelopathic properties [20]. Moreover, Brassicaceae’s powerful odor and taste (“mustard oil bomb”) seems to repel herbivores, defending the plant from excessive consumption [21]. Likewise, the potent cancer chemoprotective and/or anti-oncogenic activity of glucosinolates and isothiocyanates (their hydrolysis products) has been described, promoting the cultivation and human consumption of glucosinolate-containing plant species as healthy foods [19][22][23].
In an experiment on broccoli florets without cooling, the contents of total aliphatic and indolic glucosinolates declined by more than 50% after 3 days of storage. However, the decrease was alleviated by melatonin treatment [24]. In broccoli florets stored at 20 °C [24] and also at 4 °C [25], melatonin induced total glucosinolate content, including the biosynthesis of several aliphatic glucosinolates such as GER and GRA, and decreased others such as GNA and PRO; moreover, indolic glucosinolates such as GBS, NGBS and 4-methoxyglucobrassicin (4MGBS) were increased. Sulforaphane (SFR), an isothiocyanate product, was additionally increased (Figure 1). Melatonin upregulated the expression of several genes related to glucosinolate biosynthesis such as the transcription factors MYB28 and MYB34, and several transcripts of glucosinolate biosynthesis enzymes such as GS-Elong, UGT74B1, ST5b, FMOGS-OX1 and TGG1, and CYP83A1, CYP79F1 and CYP79B2, but AOP2 and ESP were downregulated by melatonin. The upregulation by melatonin of MYO/TGGs induced glucosinolate hydrolysis, reflecting the accumulated levels of sulforaphane in stored broccoli (Figure 1) [24][25]. In Chinese cabbage, a similar glucosinolate biosynthesis activation by melatonin was observed. An interesting study on the fungicide activity of glucosinolate-rich extracts against Sclerotinia sclerotiorum (stem rot disease) and its relationship with the melatonin-inducing capacity of glucosinolate biosynthesis was demonstrated [26]. An overall view is provided in the proposed model of Figure 1, but although it was unknown that during the storage of broccoli glucosinolates are synthesized [27], and that melatonin is able to over-activate the biosynthesis of these compounds, it is important to point out that the conditions of temperature and light/dark determine the balance between glucosinolate biosynthesis and hydrolysis up to isothiocyanates, which considerably alter the flavors of the product by the time it reaches the consumer.
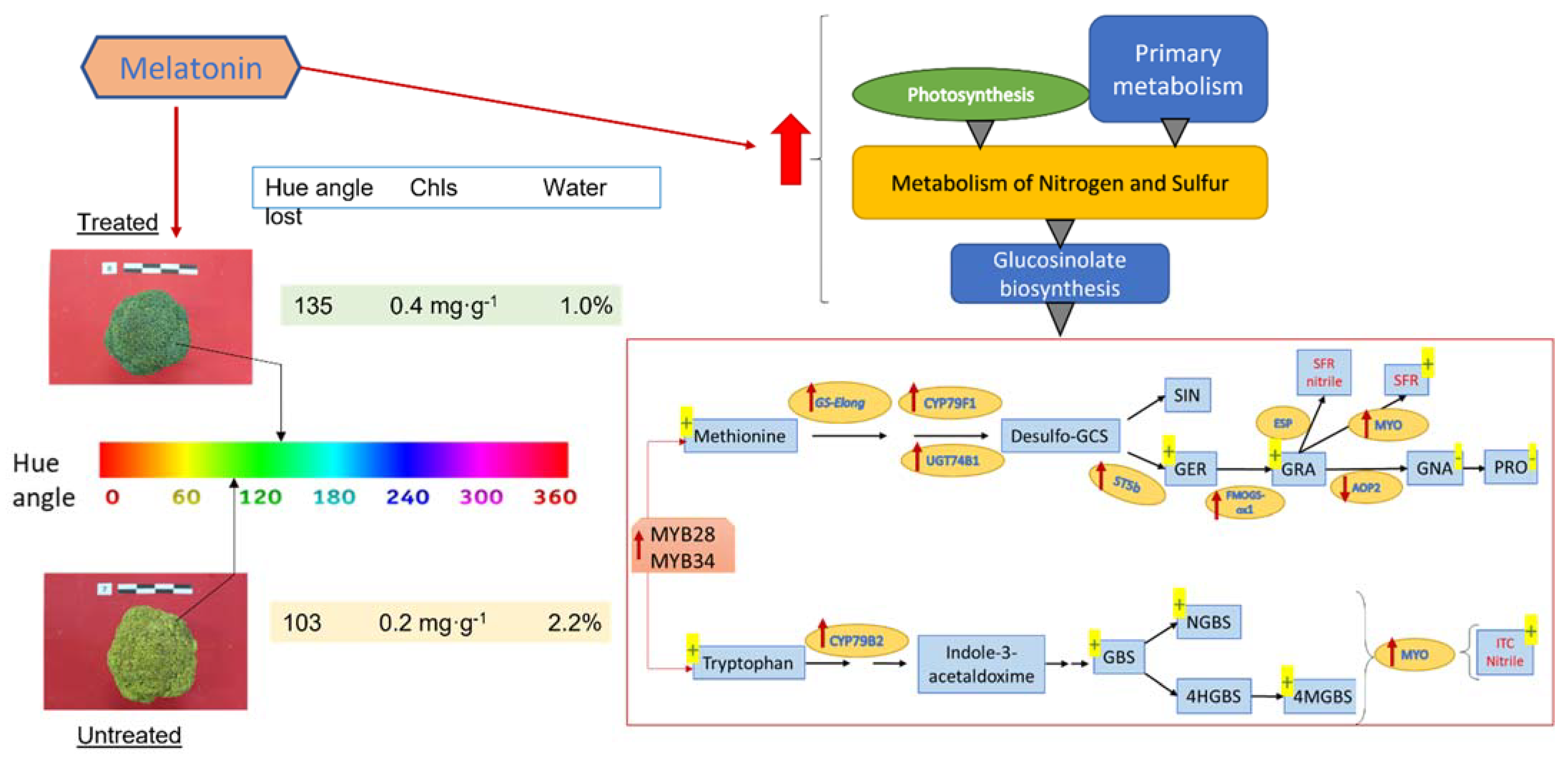
Figure 1. Representative model of melatonin’s effects on broccoli florets postharvest. Red arrows mean up-/downregulation, and yellow +/− means higher/lower levels with respect to untreated broccoli heads.
Abbreviations
| 4MGBS | 4-methoxyglucobrassicin |
| AOP2 | 2-oxoglutarate-dependent dioxygenase |
| C4H | cinnamic acid 4-hydroxylase |
| CHI | chalcone isomerase |
| CHS | chalcone synthase |
| CYP79B1,2 | cytochome P450 |
| CYP79F1 | dihomomethionine N-hydroxylase |
| CYP83A1 | cytochome P450 |
| DFR | dihydroflavonol 4-reductase |
| EC | electroconductivity |
| ESP | epithiospecifier protein |
| F3H: | flavanone 3-hydroxylase |
| FMOGS-OX1 | flavin monooxygenase |
| GBS | glucobrassicin |
| GER | glucoerucin |
| GNA | gluconapin |
| GRA | glucoraphanin |
| GS-Elong | glucosinolate biosynthesis enzyme |
| MYO | myrosinase |
| NGBS | neoglucobrassicin |
| PAL | phenylalanine ammonia-lyase |
| SIN | sinigrin |
| ST5b | sulfotransferase |
| TGG1 | myrosinase |
| UFGT, UDP-glucose | flavonoid 3-O-glucosyltransferase |
| UGT74B1 | UDP-glycosyltransferase |
References
- Al-Shehbaz, I.A. A Generic and Tribal Synopsis of the Brassicaceae (Cruciferae). TAXON 2012, 61, 931–954.
- Kapusta-Duch, J.; Kopeć, A.; Piatkowska, E.; Borczak, B.; Leszczyńska, T. The Beneficial Effects of Brassica Vegetables on Human Health. Rocz. Państwowego Zakładu Hig. 2012, 63, 389–395.
- Arnao, M.B.; Hernández-Ruiz, J. Melatonin in Flowering, Fruit Set and Fruit Ripening. Plant Reprod. 2020, 33, 77–87.
- Arnao, M.B.; Cano, A.; Hernández-Ruiz, J. Phytomelatonin: An Unexpected Molecule with Amazing Performances in Plants. J. Exp. Bot. 2022, erac009.
- Jayarajan, S.; Sharma, R.R. Melatonin: A Blooming Biomolecule for Postharvest Management of Perishable Fruits and Vegetables. Trends Food Sci. Technol. 2021, 116, 318–328.
- Wang, S.Y.; Shi, X.C.; Wang, R.; Wang, H.L.; Liu, F.; Laborda, P. Melatonin in Fruit Production and Postharvest Preservation: A Review. Food Chem. 2020, 320, 126642.
- Aghdam, M.S.; Mukherjee, S.; Flores, F.B.; Arnao, M.B.; Luo, Z.; Corpas, F.J. Functions of Melatonin During Postharvest of Horticultural Crops. Plant Cell Physiol. 2021, pcab175.
- Arnao, M.B.; Hernández-Ruiz, J. Growth Activity, Rooting Capacity, and Tropism: Three Auxinic Precepts Fulfilled by Melatonin. Acta Physiol. Plant 2017, 39, 127.
- Arnao, M.B.; Hernández-Ruiz, J. Functions of Melatonin in Plants: A Review. J. Pineal Res. 2015, 59, 133–150.
- Arnao, M.B.; Hernández-Ruiz, J. Melatonin: A New Plant Hormone and/or a Plant Master Regulator? Trends Plant Sci. 2019, 24, 38–48.
- Arnao, M.B.; Hernández-Ruiz, J. Regulatory Role of Melatonin in the Redox Network of Plants and Plant Hormone Relationship in Stress. In Hormones and Plant Response; Gupta, D.K., Corpas, F.J., Eds.; Springer International Publishing: Cham, Switzerland, 2021; pp. 235–272. ISBN 978-3-030-77477-6.
- Arnao, M.B.; Hernández-Ruiz, J. Protective Effect of Melatonin against Chlorophyll Degradation during the Senescence of Barley Leaves. J. Pineal Res. 2009, 46, 58–63.
- Zhao, Y.Q.; Zhang, Z.W.; Chen, Y.E.; Ding, C.B.; Yuan, S.; Reiter, R.J.; Yuan, M. Melatonin: A Potential Agent in Delaying Leaf Senescence. Crit. Rev. Plant Sci. 2021, 40, 1–22.
- Vadnie, C.A.; McClung, C.A. Circadian Rhythm Disturbances in Mood Disorders: Insights into the Role of the Suprachiasmatic Nucleus. Neural Plast. 2017, 2017, 1504507.
- Sharif, R.; Xie, C.; Zhang, H.; Arnao, M.B.; Ali, M.; Ali, Q.; Muhammad, I.; Shalmani, A.; Nawaz, M.; Chen, P.; et al. Melatonin and Its Effects on Plant Systems. Molecules 2018, 23, 2352.
- Moustafa-Farag, M.; Almoneafy, A.; Mahmoud, A.; Elkelish, A.; Arnao, M.B.; Li, L.; Ai, S. Melatonin and Its Protective Role against Biotic Stress Impacts on Plants. Biomolecules 2020, 10, 54.
- Arnao, M.B.; Hernández-Ruiz, J.; Cano, A.; Reiter, R.J. Melatonin and Carbohydrate Metabolism in Plant Cells. Plants 2021, 10, 1917.
- Rodman, J.E.; Soltis, P.S.; Soltis, D.E.; Sytsma, K.J.; Karol, K.G. Parallel Evolution of Glucosinolate Biosynthesis Inferred from Congruent Nuclear and Plastid Gene Phylogenies. Am. J. Bot. 1998, 85, 997–1006.
- Fahey, J.; Zalcmann, A.; Talalay, P. The Chemical Diversity and Distribution of Glucosinolates and Isothiocyanates among Plants. Phytochemistry 2001, 56, 5–51.
- Selmar, D. Biosynthesis of Cyanogenic Glycosides, Glucosinolates and Non-Protein Amino Acids. In Annual Plant Reviews: Biochemistry of Plant Secondary Metabolism; Wink, M., Ed.; Blackwell Pub.: New York, NY, USA, 2010; Volume 2, pp. 92–181. ISBN 978-1-4443-2050-3.
- Halkier, B.A.; Gershenzon, J. Biology and Biochemistry of Glucosinolates. Annu. Rev. Plant Biol. 2006, 57, 303–333.
- Herr, I.; Büchler, M.W. Dietary Constituents of Broccoli and Other Cruciferous Vegetables: Implications for Prevention and Therapy of Cancer. Cancer Treat. Rev. 2010, 36, 377–383.
- Mandrich, L.; Caputo, E. Brassicaceae-Derived Anticancer Agents: Towards a Green Approach to Beat Cancer. Nutrients 2020, 12, 868.
- Miao, H.; Zeng, W.; Zhao, M.; Wang, J.; Wang, Q. Effect of Melatonin Treatment on Visual Quality and Health-Promoting Properties of Broccoli Florets under Room Temperature. Food Chem. 2020, 319, 126498.
- Wei, L.; Liu, C.; Zheng, H.; Zheng, L. Melatonin Treatment Affects the Glucoraphanin-Sulforaphane System in Postharvest Fresh-Cut Broccoli (Brassica oleracea L.). Food Chem. 2020, 307, 125562.
- Teng, Z.; Yu, Y.; Zhu, Z.; Hong, S.B.; Yang, B.; Zang, Y. Melatonin Elevated Sclerotinia Sclerotiorum Resistance via Modulation of ATP and Glucosinolate Biosynthesis in Brassica Rapa Ssp. Pekinensis. J. Proteom. 2021, 243, 104264.
- Ku, K.M.; Choi, J.H.; Kim, H.S.; Kushad, M.M.; Jeffery, E.H.; Juvik, J.A. Methyl Jasmonate and 1-Methylcyclopropene Treatment Effects on Quinone Reductase Inducing Activity and Post-Harvest Quality of Broccoli. PLoS ONE 2013, 8, e77127.
More
Information
Subjects:
Agriculture, Dairy & Animal Science
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
680
Revisions:
2 times
(View History)
Update Date:
09 Mar 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





