Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Laura Kresty | + 1632 word(s) | 1632 | 2022-02-25 09:33:52 | | | |
| 2 | Beatrix Zheng | Meta information modification | 1632 | 2022-03-04 02:26:19 | | | | |
| 3 | Beatrix Zheng | Meta information modification | 1632 | 2022-03-04 07:22:26 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Kresty, L. Cranberry Polyphenols in Esophageal Cancer Inhibition. Encyclopedia. Available online: https://encyclopedia.pub/entry/20182 (accessed on 07 February 2026).
Kresty L. Cranberry Polyphenols in Esophageal Cancer Inhibition. Encyclopedia. Available at: https://encyclopedia.pub/entry/20182. Accessed February 07, 2026.
Kresty, Laura. "Cranberry Polyphenols in Esophageal Cancer Inhibition" Encyclopedia, https://encyclopedia.pub/entry/20182 (accessed February 07, 2026).
Kresty, L. (2022, March 03). Cranberry Polyphenols in Esophageal Cancer Inhibition. In Encyclopedia. https://encyclopedia.pub/entry/20182
Kresty, Laura. "Cranberry Polyphenols in Esophageal Cancer Inhibition." Encyclopedia. Web. 03 March, 2022.
Copy Citation
Esophageal adenocarcinoma (EAC) is a cancer characterized by rapidly rising incidence and poor survival, resulting in the need for new prevention and treatment options. Cranberry extracts including proanthocyanidins (C-PAC) and anthocyanins, flavonoids and glycosides (AFG) mitigate key networks dysregulated in EAC progression including NOTCH, NRF2, immune, EMT and TP53 signaling.
Barrett’s esophagus
esophageal adenocarcinoma
cranberry polyphenols
anthocyanins
flavonoids
glycosides
reverse phase protein array
proanthocyanidins
1. Introduction
In recent years, rates of esophageal adenocarcinoma (EAC) and the only known precursor lesion, Barrett’s esophagus (BE), have increased sharply in the United States and the Westernized world. Mechanisms by which BE progresses to EAC are still being unraveled, but repeated exposure of the esophagus to bile at an acidic pH can result in gastroesophageal reflux disease (GERD), the primary risk factor for EAC. Subsequent inflammation, loss of barrier integrity and molecular alterations resulting from GERD contribute to EAC progression [1][2]. TP53 mutation is recognized as the strongest known driver for EAC progression, documented to occur in over 70% of EAC cases [3][4]. Overall, mutational burden for EAC is considered high, but mutational events beyond TP53 tend to be spread across a large number of genes (i.e., ARID1A, CDKN2A, SMAD4, CTNNB1, EGFR, ERBB2, MET) at relatively low frequency, as recently reviewed [5]. EAC disproportionality occurs in males and is obesity linked [6]. Efficacious prevention and treatment strategies are urgently needed considering that EAC is the seventh leading cause of cancer mortality among Caucasian males in the United States and one with a 5-year survival rate consistently below 20% [7].
2. Current Insights
Considering the poor 5-year survival statistics and rising incidence of EAC, there is a critical need for improving targeted efforts for prevention and treatment of this deadly malignancy. It has been previously shown that cranberry extracts have cancer inhibitory properties in numerous in vitro studies utilizing an array of diverse cancer cell lines [8][9][10][11][12]. Additionally, a few in vivo studies, largely centered around carcinogen-induced and xenograft models, have supported cranberries’ cancer inhibitory potential [8][13][14][15]. Few clinical studies targeting cancer or premalignancy have been conducted with cranberry or cranberry derivatives to date; however, the limited research does support antibacterial activity, favorable effects on the microbiome, and one study reported a 22.5% drop in PSA levels following a 30 day intervention [16][17][18][19][20][21].
RPPA results support that C-PAC and AFG treatment have rather pleiotropic effects on diverse cellular processes, ultimately resulting in EAC cell death. C-PAC and AFG significantly modulated the signal transduction process network NOTCH signaling. Increased NOTCH signaling is commonly observed in cancer and chemoresistance [22], with polyphenols including quercetin and resveratrol reportedly reducing NOTCH intracellular domain protein levels [23][24]. Interestingly, polyphenols including picatannol, apigenin, chrysin and genistein upregulate NOTCH signaling to elicit anti-cancer effects via inhibition of cellular proliferation and migration [25][26][27]. NOTCH signaling is increased in progression from BE to EAC [28][29][30][31] and decreased NOTCH signaling inhibits EAC xenografts [32]. The ability of cranberry polyphenols to downregulate the intracellular domains of NOTCH1 and NOTCH2 is promising for targeting NOTCH-linked progression of BE to EAC (Figure 1). Kunze et al. recently showed that NOTCH2 and NOTCH3 were upregulated with progression to EAC in human tissues and expression of the intracellular domain of NOTCH2 led to increased dysplasia and decreased survival rates in the L2-ILB mouse model of BE [31]. The data show that the NOTCH signaling pathway is significantly modulated by C-PAC and AFG and that the canonical NOTCH target and transcriptional repressor HES1 [33] are downregulated by C-PAC and AFG in JHAD1 cells and by AFG in OE19 cells. C-PAC and AFG impact cell cycle processes in both JHAD1 and OE19 cells, with G1-S and G2-M highlighted in the process networks. The results for Cyclin B1 are consistent with the researchers' previous findings reporting that C-PAC induces cell cycle arrest at G2-M, elicits an S-Phase delay and largely induces caspase-independent cell death, which is also consistent with the enrichment analysis herein [34].
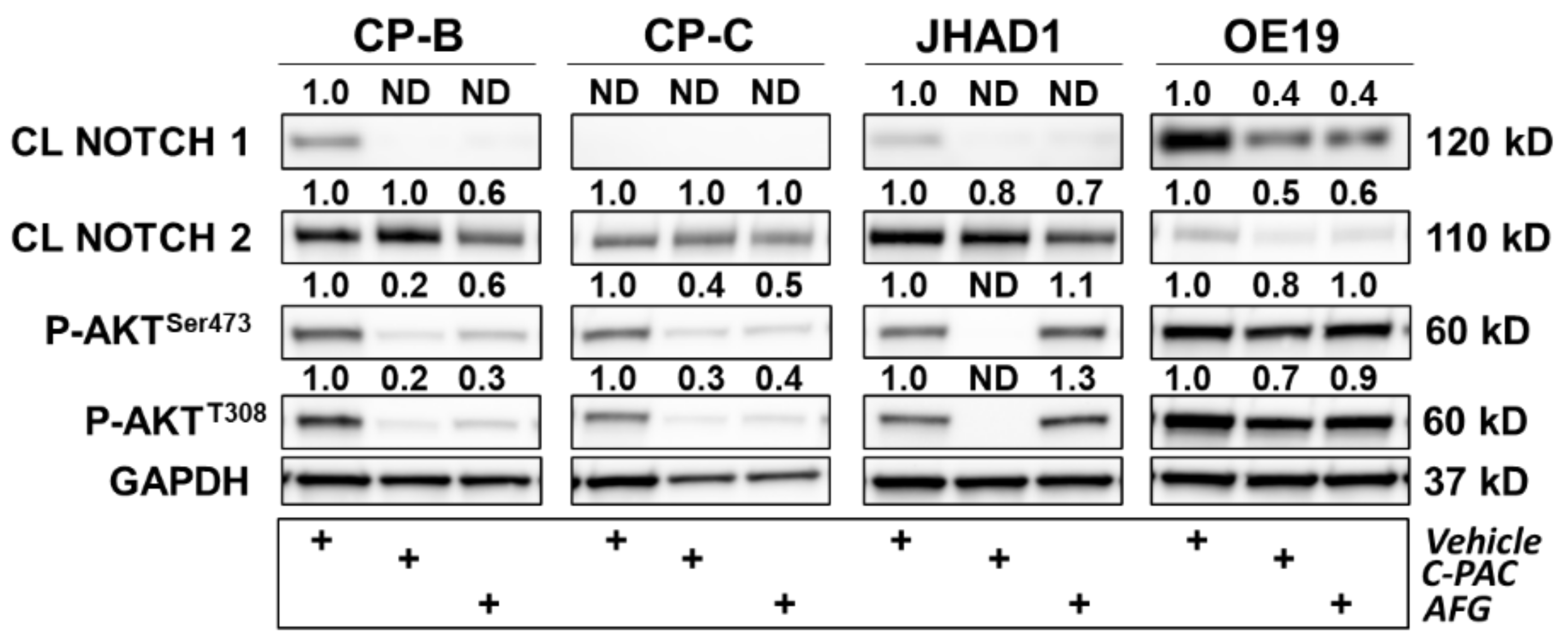
Figure 1. Polyphenols modulate several proteins involved in inflammation, DNA damage and cell cycle in premalignant and EAC cell lines. CP-B, CP-C, JHAD1 and OE19 cells were treated with C-PAC (75 µg/mL), AFG (400 µg/mL) or vehicle and lysates isolated at 24 h following treatment. Western blotting was performed using commercially available antibodies to proteins of interest. Expression values were normalized to GAPDH and fold change from vehicle was calculated using Imagelab. Bands with no detectable expression are denoted as ND. CL denotes a cleaved protein. The plus (+) sign denotes treatment group.
C-PAC and AFG similarly modulate inflammation and immune responses. The immunosuppressive tumor microenvironment in EAC, not only results in immunotherapy being relatively ineffective but also allows tumors to evade cell death and continue to aggressively grow [35][36]. Immune cells that surround BE and EAC, including myeloid-derived suppressor cells, T-regulatory cells and Th17 cells, secrete proinflammatory cytokines including IL-6, IL-19, TNFα and TGF-β, resulting in a tumor permissive environment that promotes tumor cell survival, proliferation and metastasis [37][38][39]. In alignment with the anti-inflammatory capacity of cranberry polyphenols [40], C-PAC and AFG modulated several anti-inflammatory process networks including those linked to IL-2, IL-6, IL-10 signaling and neutrophil activation as well as immune response pathway maps related to interferon, IL-2, IL-3, IL-4, IL6 and BAFF signaling. One additional pathway map to highlight from the RPPA analysis is that of oxidative stress due to reactive oxygen species (ROS)-induced cellular signaling in JHAD1 cells. These results are consistent with data the researchers previously published [41] showing that C-PAC induces ROS in both JHAD1 and OE19 cells leading to cell death, but more so in JHAD1 cells, which aligns with oxidative stress being the most modulated pathway by C-PAC in JHAD1 cells. Additionally, oxidative stress due to the ROS-induced cellular signaling pathway map is the 13th-most significantly modulated in OE19 cells, further highlighting its central role in cell death induction in both cell lines. Protein level results following C-PAC, and to a lesser extent AGF treatment, also align with oxidative-linked changes as evidenced by increased levels of the DNA damage marker phospho-H2AXSer139 in OE19 and CP-B cell lines. OE19 is a phenotypically aggressive EAC cell line and, although CP-B is a BE derived cell line, the patient from which the line was developed progressed to EAC, supporting the notion that this BE line was rewired prior to pathologic progression to EAC. This concept aligns with recent research by Fitzgerald et al. which identified TP53 mutational events among EAC progressors prior to the onset of high-risk pathology [42]. These results support that both C-PAC and AFG possess cancer inhibitory properties, but the precise molecules modulated may reflect cell line diversity and patient heterogeneity.
Given that TP53 is the best-known and most frequent mutation involved in EAC development [5][43][44], the ability of cranberry polyphenols to target mutant P53 and key signal transduction pathways implicated in EAC progression is promising and warrants further investigation in early phase clinical trials. These results show that cranberry polyphenols reduce P53 levels by 10–40%, presumably reflecting reduced levels of the mutated form of the gene. This phenomenon is encouraging to note in BE-derived cell lines which may reflect sensitivity of premalignant esophageal cells to TP53 reprogramming by polyphenols. In alignment with these results, others have shown that polyphenols including curcumin, resveratrol, epigallocatechin-3-gallate, as well as other black and green tea derivatives, alter TP53 signaling pathways, leading to cell death in a variety of cancer cell lines [8].
Matrix metalloproteases, including MMP-9, are linked to progression in many cancers and are responsible for the remodeling of the extracellular matrix, resulting in tumor invasion and metastasis [45]. Increased levels of MMP-9 are observed in tissues from patients with dysplastic BE and EAC, but not those with esophagitis or metaplastic BE [46][47]. Furthermore, MMP-9 expression is inversely correlated with protein levels of the phase II detoxification glutathione-s-transferase pi (GSTP1), with increased levels of GSTP1 in normal patients and those with esophagitis [47]. The researchers recently showed that C-PAC increases levels of the phase 2 detoxification enzyme glutathione-s-transferase theta 2 (GSTT2) and protects against acidified bile acid induced cell death in patient-derived primary normal esophageal cells [48]. Thus, the ability of cranberry constituents to favorably impact esophageal cells, both primary and immortalized cell cultures, across a range of histopathology lends support for the investigation of cranberry constituents in cohorts at increased risk for progression to EAC, such as BE patients. Consistent with decreased MMP-9 levels, C-PAC and AFG decreased expression of L1CAM in JHAD1 cells, a neuronal adhesion protein linked to metastasis and chemoresistance, and is overexpressed in many solid tumor cancers including esophageal squamous cell carcinoma and gastric cancer [49][50][51]. AFG modulated similar process networks as C-PAC in JHAD1 cells including cell cycle kinetics, but also different examples including downregulation of epithelial to mesenchymal transition (EMT) and proinflammatory signaling cascades involving IL-2 and IL-4. Both C-PAC and AFG modulated ERBB family signaling in JHAD1 and OE19 cells. ERBB family members including ERBB1/EGFR and ERBB2/HER2, are receptor tyrosine kinases that are often aberrantly activated leading to cancer cell migration, EMT, antitumor immunity and cancer cell survival [52][53]. Upregulation of both ERBB1/EGFR and ERBB2/HER2 is observed in progression from BE to EAC and increased surface expression has been used for in vivo detection using fluorescent peptides targeting these two proteins [54][55][56][57][58].
The ability of C-PAC and AFG to decrease NRF2 in BE and EAC cell lines is intriguing as the NRF2/KEAP1 pathway is upregulated in BE and EAC based on recent COSMIC database analysis [59]. Polyphenols including luteolin, apigenin and chrysin inhibit NRF2 in multiple cancer cell lines resulting in inhibition of multidrug-resistant drug transporters, increased sensitivity to chemotherapeutic drugs and ROS-induced cell death [60]. Finally, there is preliminary data suggesting that brusatol, a natural compound and NRF2 inhibitor [61], selectively kills EAC cells versus normal or premalignant metaplastic BE cells and increases sensitivity of EAC cells to the chemotherapeutic agent cisplatin via increased ROS [62]. Therefore, the ability of C-PAC and AFG to downregulate NRF2 protein levels in BE and EAC cells is promising.
References
- Ustaoglu, A.; Nguyen, A.; Spechler, S.; Sifrim, D.; Souza, R.; Woodland, P. Mucosal pathogenesis in gastro-esophageal reflux disease. Neurogastroenterol. Motil. 2020, 32, e14022.
- Napier, K.J.; Scheerer, M.; Misra, S. Esophageal cancer: A Review of epidemiology, pathogenesis, staging workup and treatment modalities. World J. Gastrointest. Oncol. 2014, 6, 112–120.
- Dulak, A.M.; Stojanov, P.; Peng, S.; Lawrence, M.S.; Fox, C.; Stewart, C.; Bandla, S.; Imamura, Y.; Schumacher, S.E.; Shefler, E.; et al. Exome and whole-genome sequencing of esophageal adenocarcinoma identifies recurrent driver events and mutational complexity. Nat. Genet. 2013, 45, 478–486.
- Grady, W.M.; Yu, M.; Markowitz, S.D.; Chak, A. Barrett’s Esophagus and Esophageal Adenocarcinoma Biomarkers. Cancer Epidemiol. Biomark. Prev. 2020, 29, 2486–2494.
- Contino, G.; Vaughan, T.L.; Whiteman, D.; Fitzgerald, R.C. The Evolving Genomic Landscape of Barrett’s Esophagus and Esophageal Adenocarcinoma. Gastroenterology 2017, 153, 657–673.e651.
- Domper Arnal, M.J.; Ferrandez Arenas, A.; Lanas Arbeloa, A. Esophageal cancer: Risk factors, screening and endoscopic treatment in Western and Eastern countries. World J. Gastroenterol. 2015, 21, 7933–7943.
- American Cancer Society. Cancer Facts & Figures 2021; American Cancer Society: Atlanta, GA, USA, 2021.
- Weh, K.M.; Clarke, J.; Kresty, L.A. Cranberries and Cancer: An Update of Preclinical Studies Evaluating the Cancer Inhibitory Potential of Cranberry and Cranberry Derived Constituents. Antioxidants 2016, 5, 27.
- Jozkowiak, M.; Skupin-Mrugalska, P.; Nowicki, A.; Borys-Wojcik, S.; Wierzchowski, M.; Kaczmarek, M.; Ramlau, P.; Jodynis-Liebert, J.; Piotrowska-Kempisty, H. The Effect of 4-hydroxy-3,4,5-trimetoxystilbene, the Metabolite of Resveratrol Analogue DMU-212, on Growth, Cell Cycle and Apoptosis in DLD-1 and LOVO Colon Cancer Cell Lines. Nutrients 2020, 12, 1327.
- Mansouri, R.A.; Percival, S.S. Cranberry extract initiates intrinsic apoptosis in HL-60 cells by increasing BAD activity through inhibition of AKT phosphorylation. BMC Complement. Med. Ther. 2020, 20, 71.
- Khairnar, M.R.; Wadgave, U.; Jadhav, H.; Naik, R. Anticancer activity of chlorhexidine and cranberry extract: An in-vitro study. J. Exp. Ther. Oncol. 2018, 12, 201–205.
- Prasain, J.K.; Rajbhandari, R.; Keeton, A.B.; Piazza, G.A.; Barnes, S. Metabolism and growth inhibitory activity of cranberry derived flavonoids in bladder cancer cells. Food Funct. 2016, 7, 4012–4019.
- Wu, X.; Song, M.; Cai, X.; Neto, C.; Tata, A.; Han, Y.; Wang, Q.; Tang, Z.; Xiao, H. Chemopreventive Effects of Whole Cranberry (Vaccinium macrocarpon) on Colitis-Associated Colon Tumorigenesis. Mol. Nutr. Food Res. 2018, 62, e1800942.
- Wu, X.; Xue, L.; Tata, A.; Song, M.; Neto, C.C.; Xiao, H. Bioactive Components of Polyphenol-Rich and Non-Polyphenol-Rich Cranberry Fruit Extracts and Their Chemopreventive Effects on Colitis-Associated Colon Cancer. J. Agric. Food Chem. 2020, 68, 6845–6853.
- Jin, D.; Liu, T.; Dong, W.; Zhang, Y.; Wang, S.; Xie, R.; Wang, B.; Cao, H. Dietary feeding of freeze-dried whole cranberry inhibits intestinal tumor development in Apcmin/+ mice. Oncotarget 2017, 8, 97787–97800.
- Straub, T.J.; Chou, W.C.; Manson, A.L.; Schreiber, H.L.; Walker, B.J.; Desjardins, C.A.; Chapman, S.B.; Kaspar, K.L.; Kahsai, O.J.; Traylor, E.; et al. Limited effects of long-term daily cranberry consumption on the gut microbiome in a placebo-controlled study of women with recurrent urinary tract infections. BMC Microbiol. 2021, 21, 53.
- Li, Z.X.; Ma, J.L.; Guo, Y.; Liu, W.D.; Li, M.; Zhang, L.F.; Zhang, Y.; Zhou, T.; Zhang, J.Y.; Gao, H.E.; et al. Suppression of Helicobacter pylori infection by daily cranberry intake: A double-blind, randomized, placebo-controlled trial. J. Gastroenterol. Hepatol. 2021, 36, 927–935.
- Maki, K.C.; Kaspar, K.L.; Khoo, C.; Derrig, L.H.; Schild, A.L.; Gupta, K. Consumption of a cranberry juice beverage lowered the number of clinical urinary tract infection episodes in women with a recent history of urinary tract infection. Am. J. Clin. Nutr. 2016, 103, 1434–1442.
- Tao, W.; Zhang, Y.; Shen, X.; Cao, Y.; Shi, J.; Ye, X.; Chen, S. Rethinking the Mechanism of the Health Benefits of Proanthocyanidins: Absorption, Metabolism, and Interaction with Gut Microbiota. Compr. Rev. Food Sci. Food Saf. 2019, 18, 971–985.
- Bekiares, N.; Krueger, C.G.; Meudt, J.J.; Shanmuganayagam, D.; Reed, J.D. Effect of Sweetened Dried Cranberry Consumption on Urinary Proteome and Fecal Microbiome in Healthy Human Subjects. OMICS 2018, 22, 145–153.
- Student, V.; Vidlar, A.; Bouchal, J.; Vrbkova, J.; Kolar, Z.; Kral, M.; Kosina, P.; Vostalova, J. Cranberry intervention in patients with prostate cancer prior to radical prostatectomy. Clinical, pathological and laboratory findings. Biomed. Pap. Med. Fac. Univ. Palacky Olomouc. Czech Repub. 2016, 160, 559–565.
- Mungamuri, S.K.; Yang, X.; Thor, A.D.; Somasundaram, K. Survival signaling by Notch1: Mammalian target of rapamycin (mTOR)-dependent inhibition of p53. Cancer Res. 2006, 66, 4715–4724.
- Cecchinato, V.; Chiaramonte, R.; Nizzardo, M.; Cristofaro, B.; Basile, A.; Sherbet, G.V.; Comi, P. Resveratrol-induced apoptosis in human T-cell acute lymphoblastic leukaemia MOLT-4 cells. Biochem. Pharmacol. 2007, 74, 1568–1574.
- Kawahara, T.; Kawaguchi-Ihara, N.; Okuhashi, Y.; Itoh, M.; Nara, N.; Tohda, S. Cyclopamine and quercetin suppress the growth of leukemia and lymphoma cells. Anticancer Res. 2009, 29, 4629–4632.
- Pinchot, S.N.; Jaskula-Sztul, R.; Ning, L.; Peters, N.R.; Cook, M.R.; Kunnimalaiyaan, M.; Chen, H. Identification and validation of Notch pathway activating compounds through a novel high-throughput screening method. Cancer 2011, 117, 1386–1398.
- LaFoya, B.; Munroe, J.A.; Albig, A.R. A comparison of resveratrol and other polyphenolic compounds on Notch activation and endothelial cell activity. PLoS ONE 2019, 14, e0210607.
- Yu, X.M.; Jaskula-Sztul, R.; Ahmed, K.; Harrison, A.D.; Kunnimalaiyaan, M.; Chen, H. Resveratrol induces differentiation markers expression in anaplastic thyroid carcinoma via activation of Notch1 signaling and suppresses cell growth. Mol. Cancer Ther. 2013, 12, 1276–1287.
- Wang, Z.; Chen, J.; Capobianco, A.J. The Notch signaling pathway in esophageal adenocarcinoma. Cell Mol. Biol. 2015, 61, 24–32.
- Kunze, B.; Wein, F.; Fang, H.Y.; Anand, A.; Baumeister, T.; Strangmann, J.; Gerland, S.; Ingermann, J.; Munch, N.S.; Wiethaler, M.; et al. Notch Signaling Mediates Differentiation in Barrett’s Esophagus and Promotes Progression to Adenocarcinoma. Gastroenterology 2020, 159, 575–590.
- Tamagawa, Y.; Ishimura, N.; Uno, G.; Yuki, T.; Kazumori, H.; Ishihara, S.; Amano, Y.; Kinoshita, Y. Notch signaling pathway and Cdx2 expression in the development of Barrett’s esophagus. Lab. Investig. 2012, 92, 896–909.
- Kunze, B.; Middelhoff, M.; Maurer, H.C.; Agibalova, T.; Anand, A.; Buhrer, A.M.; Fang, H.Y.; Baumeister, T.; Steiger, K.; Strangmann, J.; et al. Notch signaling drives development of Barrett’s metaplasia from Dclk1-positive epithelial tuft cells in the murine gastric mucosa. Sci. Rep. 2021, 11, 4509.
- Wang, Z.; Da Silva, T.G.; Jin, K.; Han, X.; Ranganathan, P.; Zhu, X.; Sanchez-Mejias, A.; Bai, F.; Li, B.; Fei, D.L.; et al. Notch signaling drives stemness and tumorigenicity of esophageal adenocarcinoma. Cancer Res. 2014, 74, 6364–6374.
- Monahan, P.; Rybak, S.; Raetzman, L.T. The notch target gene HES1 regulates cell cycle inhibitor expression in the developing pituitary. Endocrinology 2009, 150, 4386–4394.
- Kresty, L.A.; Weh, K.M.; Zeyzus-Johns, B.; Perez, L.N.; Howell, A.B. Cranberry proanthocyanidins inhibit esophageal adenocarcinoma in vitro and in vivo through pleiotropic cell death induction and PI3K/AKT/mTOR inactivation. Oncotarget 2015, 6, 33438–33455.
- Bhat, A.A.; Nisar, S.; Maacha, S.; Carneiro-Lobo, T.C.; Akhtar, S.; Siveen, K.S.; Wani, N.A.; Rizwan, A.; Bagga, P.; Singh, M.; et al. Cytokine-chemokine network driven metastasis in esophageal cancer; promising avenue for targeted therapy. Mol. Cancer 2021, 20, 2.
- Lagisetty, K.H.; McEwen, D.P.; Nancarrow, D.J.; Schiebel, J.G.; Ferrer-Torres, D.; Ray, D.; Frankel, T.L.; Lin, J.; Chang, A.C.; Kresty, L.A.; et al. Immune determinants of Barrett’s progression to esophageal adenocarcinoma. JCI Insight 2021, 6, e143888.
- Milano, F.; Jorritsma, T.; Rygiel, A.M.; Bergman, J.J.; Sondermeijer, C.; Ten Brinke, A.; vanHam, S.M.; Krishnadath, K.K. Expression pattern of immune suppressive cytokines and growth factors in oesophageal adenocarcinoma reveal a tumour immune escape-promoting microenvironment. Scand. J. Immunol. 2008, 68, 616–623.
- Blum, A.E.; Venkitachalam, S.; Ravillah, D.; Chelluboyina, A.K.; Kieber-Emmons, A.M.; Ravi, L.; Kresak, A.; Chandar, A.K.; Markowitz, S.D.; Canto, M.I.; et al. Systems Biology Analyses Show Hyperactivation of Transforming Growth Factor-beta and JNK Signaling Pathways in Esophageal Cancer. Gastroenterology 2019, 156, 1761–1774.
- Ebbing, E.A.; van der Zalm, A.P.; Steins, A.; Creemers, A.; Hermsen, S.; Rentenaar, R.; Klein, M.; Waasdorp, C.; Hooijer, G.K.J.; Meijer, S.L.; et al. Stromal-derived interleukin 6 drives epithelial-to-mesenchymal transition and therapy resistance in esophageal adenocarcinoma. Proc. Natl. Acad. Sci. USA 2019, 116, 2237–2242.
- Caldas, A.P.S.; Coelho, O.G.L.; Bressan, J. Cranberry antioxidant power on oxidative stress, inflammation and mitochondrial damage. Int. J. Food Prop. 2018, 21, 582–592.
- Weh, K.M.; Aiyer, H.S.; Howell, A.B.; Kresty, L.A. Cranberry proanthocyanidins modulate reactive oxygen species in Barrett’s and esophageal adenocarcinoma cell lines. J. Berry Res. 2016, 6, 125–136.
- Frankell, A.M.; Jammula, S.; Li, X.; Contino, G.; Killcoyne, S.; Abbas, S.; Perner, J.; Bower, L.; Devonshire, G.; Ococks, E.; et al. The landscape of selection in 551 esophageal adenocarcinomas defines genomic biomarkers for the clinic. Nat. Genet. 2019, 51, 506–516.
- Stachler, M.D.; Camarda, N.D.; Deitrick, C.; Kim, A.; Agoston, A.T.; Odze, R.D.; Hornick, J.L.; Nag, A.; Thorner, A.R.; Ducar, M.; et al. Detection of Mutations in Barrett’s Esophagus before Progression to High-Grade Dysplasia or Adenocarcinoma. Gastroenterology 2018, 155, 156–167.
- Ross-Innes, C.S.; Becq, J.; Warren, A.; Cheetham, R.K.; Northen, H.; O’Donovan, M.; Malhotra, S.; di Pietro, M.; Ivakhno, S.; He, M.; et al. Whole-genome sequencing provides new insights into the clonal architecture of Barrett’s esophagus and esophageal adenocarcinoma. Nat. Genet. 2015, 47, 1038–1046.
- Huang, H. Matrix Metalloproteinase-9 (MMP-9) as a Cancer Biomarker and MMP-9 Biosensors: Recent Advances. Sensors 2018, 18, 3249.
- Lu, X.; Duan, L.; Xie, H.; Lu, X.; Lu, D.; Lu, D.; Jiang, N.; Chen, Y. Evaluation of MMP-9 and MMP-2 and their suppressor TIMP-1 and TIMP-2 in adenocarcinoma of esophagogastric junction. Oncol. Targets Ther. 2016, 9, 4343–4349.
- Herszenyi, L.; Hritz, I.; Pregun, I.; Sipos, F.; Juhasz, M.; Molnar, B.; Tulassay, Z. Alterations of glutathione S-transferase and matrix metalloproteinase-9 expressions are early events in esophageal carcinogenesis. World J. Gastroenterol. 2007, 13, 676–682.
- Weh, K.M.; Turgeon, D.K.; Rubenstein, J.H.; Clarke, J.L.; Howell, A.B.; Chang, A.C.; Kresty, L.A. Proanthocyanidins mitigate bile acid-induced changes in GSTT2 levels in a panel of racially diverse patient-derived primary esophageal cell cultures. Mol. Carcinog. 2021, 61, 281–287.
- Guo, J.C.; Xie, Y.M.; Ran, L.Q.; Cao, H.H.; Sun, C.; Wu, J.Y.; Wu, Z.Y.; Liao, L.D.; Zhao, W.J.; Fang, W.K.; et al. L1CAM drives oncogenicity in esophageal squamous cell carcinoma by stimulation of ezrin transcription. J. Mol. Med. 2017, 95, 1355–1368.
- Chen, D.L.; Zeng, Z.L.; Yang, J.; Ren, C.; Wang, D.S.; Wu, W.J.; Xu, R.H. L1cam promotes tumor progression and metastasis and is an independent unfavorable prognostic factor in gastric cancer. J. Hematol. Oncol. 2013, 6, 43.
- Ganesh, K.; Basnet, H.; Kaygusuz, Y.; Laughney, A.M.; He, L.; Sharma, R.; O’Rourke, K.P.; Reuter, V.P.; Huang, Y.H.; Turkekul, M.; et al. L1CAM defines the regenerative origin of metastasis-initiating cells in colorectal cancer. Nat. Cancer 2020, 1, 28–45.
- Jacobi, N.; Seeboeck, R.; Hofmann, E.; Eger, A. ErbB Family Signalling: A Paradigm for Oncogene Addiction and Personalized Oncology. Cancers 2017, 9, 33.
- Kumagai, S.; Koyama, S.; Nishikawa, H. Antitumour immunity regulated by aberrant ERBB family signalling. Nat. Rev. Cancer 2021, 21, 181–197.
- Chen, J.; Zhou, J.; Gao, Z.; Li, X.; Wang, F.; Duan, X.; Li, G.; Joshi, B.P.; Kuick, R.; Appelman, H.D.; et al. Multiplexed Targeting of Barrett’s Neoplasia with a Heterobivalent Ligand: Imaging Study on Mouse Xenograft in Vivo and Human Specimens ex Vivo. J. Med. Chem. 2018, 61, 5323–5331.
- Joshi, B.P.; Zhou, J.; Pant, A.; Duan, X.; Zhou, Q.; Kuick, R.; Owens, S.R.; Appelman, H.; Wang, T.D. Design and Synthesis of Near-Infrared Peptide for in Vivo Molecular Imaging of HER2. Bioconjug. Chem. 2016, 27, 481–494.
- Kashyap, M.K.; Abdel-Rahman, O. Expression, regulation and targeting of receptor tyrosine kinases in esophageal squamous cell carcinoma. Mol. Cancer 2018, 17, 54.
- Caspa Gokulan, R.; Garcia-Buitrago, M.T.; Zaika, A.I. From genetics to signaling pathways: Molecular pathogenesis of esophageal adenocarcinoma. Biochim. Biophys. Acta Rev. Cancer 2019, 1872, 37–48.
- Chiba, T. What are the real roles of different erbB proteins in Barrett’s Esophagus. Digestion 2004, 70, 93–94.
- Peng, D.; Zaika, A.; Que, J.; El-Rifai, W. The antioxidant response in Barrett’s tumorigenesis: A double-edged sword. Redox. Biol. 2021, 41, 101894.
- Suraweera, T.L.; Rupasinghe, H.P.V.; Dellaire, G.; Xu, Z. Regulation of Nrf2/ARE Pathway by Dietary Flavonoids: A Friend or Foe for Cancer Management? Antioxidants 2020, 9, 973.
- Tao, S.; Rojo de la Vega, M.; Chapman, E.; Ooi, A.; Zhang, D.D. The effects of NRF2 modulation on the initiation and progression of chemically and genetically induced lung cancer. Mol. Carcinog. 2018, 57, 182–192.
- Peng, D.; Lu, H.; Hu, T.; Sriramajayam, K.; El-Rifai, W. Abstract 1938: Targeting constitutively overexpressed NRF2 in esophageal adenocarcinoma. Cancer Res. 2020, 80, 1938.
More
Information
Subjects:
Oncology
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
680
Revisions:
3 times
(View History)
Update Date:
04 Mar 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No




