
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Christian Chapa | + 1639 word(s) | 1639 | 2022-02-25 10:07:36 | | | |
| 2 | Lindsay Dong | + 77 word(s) | 1716 | 2022-03-01 03:17:28 | | | | |
| 3 | Lindsay Dong | + 77 word(s) | 1716 | 2022-03-01 03:19:50 | | |
Video Upload Options
Pancreatic cancer has one of the highest mortality rates among cancers, and a combination of nab-paclitaxel with gemcitabine remains the cornerstone of first-line therapy. Nab-paclitaxel with gemcitabine in combination with other therapeutic agents can be new treatment strategies in pancreatic cancer. Seven therapeutic agents (ibrutinib, necuparanib, tarextumab, apatorsen, cisplatin, enzalutamide, and momelotinib) are found.
1. Introduction
2. Pancreatic Cancer Treatment by Nab-Paclitaxel with Gemcitabine Combination
Table 1 shows the diversity of therapeutic agents under investigation in clinical trials. Considering the diversity in the chemical structures of the therapeutic agents enlisted, molecular biology studies will likely be needed to relate cellular or molecular events with the responses of patients to the triple regimen.
| Ref | Therapeutic Agent | Structure | Description |
|---|---|---|---|
| [47] | Ibrutinib | 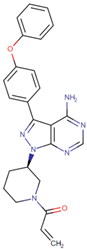 |
Ibrutinib is a Bruton’s tyrosine kinase inhibitor that forms a covalent bond with a cysteine residue (Cys 481). Ibrutinib is used to treat chronic lymphocytic leukemia, mantle cell lymphoma, and Waldenstrom‘s macroglobulinemia, leading to inhibition of BTK activity [48][49]. ClinicalTrials.gov identifier: NCT024366. Phase III RESOLVE study. Ibrutinib plus nab-paclitaxel/gemcitabine did not improve OS or PFS for patients with metastatic PDAC. |
| [50] | Necuparanib | — | Necuparanib (a heparin mimetic) acts as a multitargeting therapeutic, altering multiple signaling pathways simultaneously by binding and sequestering different proteins [51][52]. ClinicalTrials.gov identifier: NCT01621243. A randomized phase II trial. Necuparanib plus nab-paclitaxel/gemcitabine did not improve OS. |
| [53] | Tarextumab | — | Monoclonal antibodies (mAb, anti-Notch2/3, OMP-59R5) are fully human monoclonal antibodies that target the Notch2 and Notch3 receptors. They have been used in trials studying the treatment of solid tumors, stage IV pancreatic cancer, and stage IV small cell lung cancer [54][55]. ClinicalTrials.gov identifier: NCT01647828. A randomized phase II trial. Tarextumab plus nab-paclitaxel/gemcitabine did not improve OS, PFS, or ORR in first-line metastatic PDAC |
| [56] | Apatorsen |  |
Apatorsen is a second-generation antisense drug in preclinical experiments that inhibits the production of heat shock protein 27 (Hsp27), a cell survival protein found at elevated levels in many human cancers, including prostate, lung, breast, ovarian, bladder, renal, pancreatic, multiple myeloma, and liver cancer [57][58]. ClinicalTrials.gov identifier: NCT01844817. A randomized, double-blinded, phase II trial. The RAINIER trial. Addition of apatorsen to nab-paclitaxel/gemcitabine regimen did not improve survival or other clinically relevant endpoints in patients with metastatic pancreatic cancer. |
| [59] | Cisplatin | 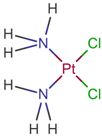 |
Cisplatin is a platinum-based chemotherapy agent used to treat various sarcomas, carcinomas, lymphomas, and germ cell tumors. Cisplatin exerts its anticancer activities by generating DNA lesions through interactions with purine bases, leading to the activation of various signal transduction pathways leading to apoptosis [60][61][62]. ClinicalTrials.gov identifier: NCT01893801. A nonrandomized phase 1b/2 pilot clinical trial. The addition of cisplatin to nab-paclitaxel/gemcitabine resulted in a high response rate and evolving OS. |
| [63] | Enzalutamide | 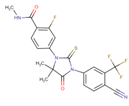 |
Enzalutamide is a rationally designed, targeted androgen-receptor inhibitor used to treat castration-resistant prostate cancer. Enzalutamide acts both by inhibiting the translocation of the androgen receptor into the nucleus and by reducing the transcriptional activity of this receptor [64][65]. ClinicalTrials.gov identifier: NCT02138383. A phase I trial. Enzalutamide plus nab-paclitaxel/gemcitabine was safely administered with no unexpected toxicities and resulted in consistent reductions in CA 19–9 (biological marker) levels. |
| [66] | Momelotinib | 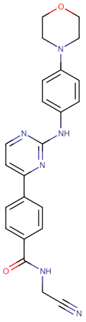 |
Momelotinib is a benzamide that acts as an ATP-competitive JAK1/JAK2 inhibitor. Momelotinib has been used in trials studying the treatment of polycythemia vera, primary myelofibrosis, post-polycythemia vera, essential thrombocythemia, and primary myelofibrosis (PMF), among others [67][68]. ClinicalTrials.gov identifier: NCT02101021. Phase 1 dose-escalation study. Momelotinib plus nab-paclitaxel/gemcitabine was safe and well tolerated, with no OS or PFS benefits. |
The molecular effects of paclitaxel, gemcitabine, and cisplatin are well characterized in cancer cells [69][70][71][72][73][74][75][76][60]. However, a successful triple regimen whose cellular events are known with certainty will bring about a new paradigm for the treatment of pancreatic cancer. Clinical trials of combination therapies that are effective and safe should be complemented by molecular studies to understand the pathways for their biological activities.
The MPACT, a randomized phase III study, reported that NP/G had an OS of 8.5 months, a PFS of 5.5 months, a CR of less than 1%, and an RP of 23% in 431 patients, resulting in greater efficacy than gemcitabine monotherapy. The AE of third grade or higher were as follows: neutropenia (38%), fatigue (17%), and neuropathy (17%). Febrile neutropenia was also present in 3% of the patients [77], whereas in [47][53][56], placebo in combination with the NP/G regimen exceeded the formulation of the main regimen with the additional drug in ORR, OS, and PFS, which means that adding ibrunitib (Bruton’s tyrosine kinase inhibitor), tarextumab (IgG2 antibody against Notch2 and Notch3 receptors), or apatorsen (antisense oligonucleotide targeting heat shock protein 27 messenger RNA) was not more effective than the standard therapy of NP/G. While the formulation with necuparanib (heparin mimetic) was slightly superior to the placebo formulation, there was no significant improvement in OS and PFS, resulting in the same ORR as the NP/G standard therapy.
3. Conclusions
Abbreviations and Acronyms
| 5-FU | Fluorouracil |
| AE | Adverse events |
| CNTs | concentrative nucleoside transporters |
| CTCAE | Common Terminology Criteria for Adverse Events |
| CR | Complete response |
| ENTs | Equilibrative nucleoside transporters |
| FOLFIRINOX | Chemotherapy regimen containing fluorouracil, folinic acid, irinotecan, and oxaliplatin |
| MMRI | Molecular magnetic resonance imaging |
| NP/G | Nab-paclitaxel plus gemcitabine |
| ORR | Objective response rate |
| OS | Overall survival |
| PDAC | Pancreatic ductal adenocarcinoma |
| PFS | Progression-free survival |
| TNBC | Triple-negative breast cancer |
| TNM | Tumor/Node/Metastasis staging system from the American Joint Committee on Cancer |
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249.
- Rawla, P.; Sunkara, T.; Gaduputi, V. Epidemiology of Pancreatic Cancer: Global Trends, Etiology and Risk Factors. World J. Oncol. 2019, 10, 10–27.
- Hand, F.; Conlon, K.C. Pancreatic Cancer. Surgery 2019, 37, 319–326.
- Tuveson, D.A.; Neoptolemos, J.P. Understanding Metastasis in Pancreatic Cancer: A Call for New Clinical Approaches. Cell 2012, 148, 21–23.
- Lippi, G.; Mattiuzzi, C. The Global Burden of Pancreatic Cancer. Arch. Med. Sci. 2020, 16, 820–824.
- Tempero, M.A.; Malafa, M.P.; Al-Hawary, M.; Behrman, S.W.; Benson, A.B.; Cardin, D.B.; Chiorean, E.G.; Chung, V.; Czito, B.; del Chiaro, M.; et al. Pancreatic Adenocarcinoma, Version 2.2021, NCCN Clinical Practice Guidelines in Oncology. J. Natl. Compr. Cancer Netw. 2021, 19, 439–457.
- Soweid, A.M. The Borderline Resectable and Locally Advanced Pancreatic Ductal Adenocarcinoma: Definition. Endosc. Ultrasound 2017, 6, 76.
- Roalsø, M.; Aunan, J.R.; Søreide, K. Refined TNM-Staging for Pancreatic Adenocarcinoma—Real Progress or Much Ado about Nothing? Eur. J. Surg. Oncol. 2020, 46, 1554–1557.
- Liu, C.; Cheng, H.; Jin, K.; Guo, M.; Lu, Y.; Wang, Z.; Yang, C.; Long, J.; Ni, Q.; Yu, X.; et al. Application of the Eighth Edition of the American Joint Committee on Cancer Staging for Pancreatic Adenocarcinoma. Pancreas 2018, 47, 742–747.
- Allen, P.J.; Kuk, D.; del Castillo, C.F.; Basturk, O.; Wolfgang, C.L.; Cameron, J.L.; Lillemoe, K.D.; Ferrone, C.R.; Morales-Oyarvide, V.; He, J.; et al. Multi-Institutional Validation Study of the American Joint Commission on Cancer (8th Edition) Changes for T and N Staging in Patients with Pancreatic Adenocarcinoma. Ann. Surg. 2017, 265, 185.
- Mostafa, M.E.; Erbarut-Seven, I.; Pehlivanoglu, B.; Adsay, V. Pathologic Classification of “Pancreatic Cancers”: Current Concepts and Challenges. Chin. Clin. Oncol. 2017, 6, 59.
- Puckett, Y.; Garfield, K. Pancreatic Cancer; StatPearls: Treasure Island, FL, USA, 2022.
- McGuigan, A.; Kelly, P.; Turkington, R.C.; Jones, C.; Coleman, H.G.; McCain, R.S. Pancreatic Cancer: A Review of Clinical Diagnosis, Epidemiology, Treatment and Outcomes. World J. Gastroenterol. 2018, 24, 4846–4861.
- Qiao, P.L.; Gargesha, M.; Liu, Y.; Laney, V.E.A.; Hall, R.C.; Vaidya, A.M.; Gilmore, H.; Gawelek, K.; Scott, B.B.; Roy, D.; et al. Magnetic Resonance Molecular Imaging of Extradomain B Fibronectin Enables Detection of Pancreatic Ductal Adenocarcinoma Metastasis. Magn. Reson. Imaging 2022, 86, 37–45.
- Yan, T.-B.; Huang, J.-Q.; Huang, S.-Y.; Ahir, B.K.; Li, L.-M.; Mo, Z.-N.; Zhong, J.-H. Advances in the Detection of Pancreatic Cancer Through Liquid Biopsy. Front. Oncol. 2021, 11, 801173.
- Goulart, M.R.; Watt, J.; Siddiqui, I.; Lawlor, R.T.; Imrali, A.; Hughes, C.; Saad, A.; ChinAleong, J.; Hurt, C.; Cox, C.; et al. Pentraxin 3 Is a Stromally-Derived Biomarker for Detection of Pancreatic Ductal Adenocarcinoma. NPJ Precis. Oncol. 2021, 5, 1–10.
- Hipperson, L.; Hadden, W.J.; Nahm, C.B.; Gill, A.J.; Samra, J.S.; Dona, A.; Mittal, A.; Sahni, S. Urinary Metabolite Prognostic Biomarker Panel for Pancreatic Ductal Adenocarcinomas. Biochim. Biophys. Acta Gen. Subj. 2021, 1865, 129966.
- Mollinedo, F.; Gajate, C. Direct Endoplasmic Reticulum Targeting by the Selective Alkylphospholipid Analog and Antitumor Ether Lipid Edelfosine as a Therapeutic Approach in Pancreatic Cancer. Cancers 2021, 13, 4173.
- Qi, D.; Song, X.; Xue, C.; Yao, W.; Shen, P.; Yu, H.; Zhang, Z. AKT1/FOXP3 Axis-Mediated Expression of CerS6 Promotes P53 Mutant Pancreatic Tumorigenesis. Cancer Lett. 2021, 522, 105–118.
- Yuan, L.; Zhao, J.; Zhao, S.; Dong, T.; Dong, R.; Liu, D.; Ma, E.; Li, Y. ASPER-29 Suppresses the Metastasis of Pancreatic Cancer Cells by Dual Inhibition of Cathepsin-L and Cathepsin-S. Chem.-Biol. Interact. 2022, 353, 109811.
- Roacho-Pérez, J.A.; Garza-Treviño, E.N.; Delgado-Gonzalez, P.; G-Buentello, Z.; Delgado-Gallegos, J.L.; Chapa-Gonzalez, C.; Sánchez-Domínguez, M.; Sánchez-Domínguez, C.N.; Islas, J.F. Target Nanoparticles against Pancreatic Cancer: Fewer Side Effects in Therapy. Life 2021, 11, 1187.
- Demirtürk, N.; Bilensoy, E. Nanocarriers Targeting the Diseases of the Pancreas. Eur. J. Pharm. Biopharm. 2022, 170, 10–23.
- Brachi, G.; Bussolino, F.; Ciardelli, G.; Mattu, C. Nanomedicine for Imaging and Therapy of Pancreatic Adenocarcinoma. Front. Bioeng. Biotechnol. 2019, 7, 307.
- Mousa, D.S.; El-Far, A.H.; Saddiq, A.A.; Sudha, T.; Mousa, S.A. Nanoformulated Bioactive Compounds Derived from Different Natural Products Combat Pancreatic Cancer Cell Proliferation. Int. J. Nanomed. 2020, 15, 2259–2268.
- Redruello, P.; Perazzoli, G.; Cepero, A.; Quiñonero, F.; Mesas, C.; Doello, K.; Láinez-Ramos-Bossini, A.; Rivera-Izquierdo, M.; Melguizo, C.; Prados, J. Nanomedicine in Pancreatic Cancer: A New Hope for Treatment. Curr. Drug Targets 2020, 21, 1580–1592.
- Zhang, T.; Jiang, Z.; Chen, L.; Pan, C.; Sun, S.; Liu, C.; Li, Z.; Ren, W.; Wu, A.; Huang, P. PCN-Fe(III)-PTX Nanoparticles for MRI Guided High Efficiency Chemo-Photodynamic Therapy in Pancreatic Cancer through Alleviating Tumor Hypoxia. Nano Res. 2020, 13, 273–281.
- Ren, S.; Song, L.; Tian, Y.; Zhu, L.; Guo, K.; Zhang, H.; Wang, Z. Emodin-Conjugated PEGylation of Fe3O4 Nanoparticles for FI/MRI Dual-Modal Imaging and Therapy in Pancreatic Cancer. Int. J. Nanomed. 2021, 16, 7463–7478.
- Lei, F.; Xi, X.; Rachagani, S.; Seshacharyulu, P.; Talmon, G.A.; Ponnusamy, M.P.; Batra, S.K.; Bronich, T.K. Nanoscale Platform for Delivery of Active IRINOX to Combat Pancreatic Cancer. J. Control. Release 2021, 330, 1229–1243.
- Ou, A.; Zhao, X.; Lu, Z. The Potential Roles of P53 Signaling Reactivation in Pancreatic Cancer Therapy. Biochim. Biophys. Acta (BBA) Rev. Cancer 2022, 1877, 188662.
- Polireddy, K.; Chen, Q. Cancer of the Pancreas: Molecular Pathways and Current Advancement in Treatment. J. Cancer 2016, 7, 1497–1514.
- Digiacomo, G.; Volta, F.; Garajova, I.; Balsano, R.; Cavazzoni, A. Biological Hallmarks and New Therapeutic Approaches for the Treatment of PDAC. Life 2021, 11, 843.
- Koga, F.; Kawaguchi, Y.; Shimokawa, M.; Murayama, K.; Nakashita, S.; Oza, N.; Ureshino, N.; Takahashi, H.; Ueda, Y.; Nakazawa, J.; et al. Gemcitabine plus Nab-Paclitaxel in Older Patients with Metastatic Pancreatic Cancer: A Post-Hoc Analysis of the Real-World Data of a Multicenter Study (the NAPOLEON Study). J. Geriatr. Oncol. 2022, 13, 82–87.
- Vogl, U.M.; Andalibi, H.; Klaus, A.; Vormittag, L.; Schima, W.; Heinrich, B.; Kafka, A.; Winkler, T.; Öhler, L. Nab-Paclitaxel and Gemcitabine or FOLFIRINOX as First-Line Treatment in Patients with Unresectable Adenocarcinoma of the Pancreas: Does Sequence Matter? 11 Medical and Health Sciences 1112 Oncology and Carcinogenesis. BMC Cancer 2019, 19, 28.
- Saito, T.; Ishido, K.; Kudo, D.; Kimura, N.; Wakiya, T.; Nakayama, Y.; Hakamada, K. Combination Therapy with Gemcitabine and Nab-Paclitaxel for Locally Advanced Unresectable Pancreatic Cancer. Mol. Clin. Oncol. 2017, 6, 963–967.
- Bukhari, N.; Abdalla, K.; Ibnshamsa, F.; Alselwi, W.; Al-Shakir, S.; Alqahtani, M. Exceptional Response to Second-Line Gemcitabine/Nab-Paclitaxel Chemotherapy in Patients with Metastatic Pancreatic Adenocarcinoma. Cureus 2021, 13, e18756.
- Cao, D.; Song, Q.; Li, J.; Jiang, Y.; Wang, Z.; Lu, S. Opportunities and Challenges in Targeted Therapy and Immunotherapy for Pancreatic Cancer. Expert Rev. Mol. Med. 2021, 23, e21.
- Lambert, A.; Gavoille, C.; Conroy, T. Current Status on the Place of FOLFIRINOX in Metastatic Pancreatic Cancer and Future Directions. Ther. Adv. Gastroenterol. 2017, 10, 631–645.
- Moris, D.; Karachaliou, G.-S.; Lazarou, V.; Giannis, D.; Astras, G.; Petrou, A. Initial Experience with Neoadjuvant FOLFIRINOX as First Line Therapy for Locally Advanced Pancreatic Cancer. J. Balk. Union Oncol. 2020, 25, 2525–2527.
- Loveday, B.P.T.; Lipton, L.; Thomson, B.N. Pancreatic Cancer: An Update on Diagnosis and Management. Aust. J. Gen. Pract. 2019, 48, 826–831.
- Chiorean, E.G.; Coveler, A.L. Pancreatic Cancer: Optimizing Treatment Options, New, and Emerging Targeted Therapies. Drug Des. Dev. Ther. 2015, 9, 3529–3545.
- Muranaka, T.; Kuwatani, M.; Komatsu, Y.; Sawada, K.; Nakatsumi, H.; Kawamoto, Y.; Yuki, S.; Kubota, Y.; Kubo, K.; Kawahata, S.; et al. Comparison of Efficacy and Toxicity of FOLFIRINOX and Gemcitabine with Nab-Paclitaxel in Unresectable Pancreatic Cancer. J. Gastrointest. Oncol. 2017, 8, 566–571.
- Blomstrand, H.; Scheibling, U.; Bratthäll, C.; Green, H.; Elander, N.O. Real World Evidence on Gemcitabine and Nab-Paclitaxel Combination Chemotherapy in Advanced Pancreatic Cancer. BMC Cancer 2019, 19, 40.
- Khot, V.M.; Salunkhe, A.B.; Pricl, S.; Bauer, J.; Thorat, N.D.; Townley, H. Nanomedicine-Driven Molecular Targeting, Drug Delivery, and Therapeutic Approaches to Cancer Chemoresistance. Drug Discov. Today 2021, 26, 724–739.
- Giordano, G.; Pancione, M.; Olivieri, N.; Parcesepe, P.; Velocci, M.; di Raimo, T.; Coppola, L.; Toffoli, G.; D’Andrea, M.R. Nano Albumin Bound-Paclitaxel in Pancreatic Cancer: Current Evidences and Future Directions. World J. Gastroenterol. 2017, 23, 5875–5886.
- Yang, C.P.H.; Horwitz, S.B. Taxol®: The First Microtubule Stabilizing Agent. Int. J. Mol. Sci. 2017, 18, 1733.
- Cretella, D.; Fumarola, C.; Bonelli, M.; Alfieri, R.; la Monica, S.; Digiacomo, G.; Cavazzoni, A.; Galetti, M.; Generali, D.; Petronini, P.G. Pre-Treatment with the CDK4/6 Inhibitor Palbociclib Improves the Efficacy of Paclitaxel in TNBC Cells. Sci. Rep. 2019, 9, 1–11.
- Tempero, M.; Oh, D.Y.; Tabernero, J.; Reni, M.; van Cutsem, E.; Hendifar, A.; Waldschmidt, D.T.; Starling, N.; Bachet, J.B.; Chang, H.M.; et al. Ibrutinib in Combination with Nab-Paclitaxel and Gemcitabine for First-Line Treatment of Patients with Metastatic Pancreatic Adenocarcinoma: Phase III RESOLVE Study. Ann. Oncol. 2021, 32, 600–608.
- Ibrutinib: Uses, Interactions, Mechanism of Action. DrugBank Online. Available online: https://go.drugbank.com/drugs/DB09053 (accessed on 16 January 2022).
- Hughes, D.L. Patent Review of Manufacturing Routes to Recently Approved Oncology Drugs: Ibrutinib, Cobimetinib, and Alectinib. Org. Process Res. Dev. 2016, 20, 1855–1869.
- O’Reilly, E.M.; Barone, D.; Mahalingam, D.; Bekaii-Saab, T.; Shao, S.H.; Wolf, J.; Rosano, M.; Krause, S.; Richards, D.A.; Yu, K.H.; et al. Randomised Phase II Trial of Gemcitabine and Nab-Paclitaxel with Necuparanib or Placebo in Untreated Metastatic Pancreas Ductal Adenocarcinoma. Eur. J. Cancer 2020, 132, 112–121.
- Necuparanib: Uses, Interactions, Mechanism of Action. DrugBank Online. Available online: https://go.drugbank.com/drugs/DB16251 (accessed on 16 January 2022).
- MacDonald, A.; Priess, M.; Curran, J.; Guess, J.; Farutin, V.; Oosterom, I.; Chu, C.L.; Cochran, E.; Zhang, L.; Getchell, K.; et al. Necuparanib, A Multitargeting Heparan Sulfate Mimetic, Targets Tumor and Stromal Compartments in Pancreatic Cancer. Mol. Cancer Ther. 2019, 18, 245–256.
- Hu, Z.I.; Bendell, J.C.; Bullock, A.; LoConte, N.K.; Hatoum, H.; Ritch, P.; Hool, H.; Leach, J.W.; Sanchez, J.; Sohal, D.P.S.; et al. A Randomized Phase II Trial of Nab-Paclitaxel and Gemcitabine with Tarextumab or Placebo in Patients with Untreated Metastatic Pancreatic Cancer. Cancer Med. 2019, 8, 5148–5157.
- Tarextumab: Uses, Interactions, Mechanism of Action. DrugBank Online. Available online: https://go.drugbank.com/drugs/DB12104 (accessed on 16 January 2022).
- Shah, J.; O’Young, G.; Wei, J.; Fischer, M.; Yen, W.-C.; Cancilla, B.; Kapoun, A.; Lewicki, J.; Cain, J.; Hoey, T. Tarextumab (Anti-NOTCH2/3) Reverses NOTCH2 and NOTCH3-Dependent Tumorigenicity and Metastases in Small Cell Lung Cancer. Cancer Res. 2015, 75, 2323.
- Ko, A.H.; Murphy, P.B.; Peyton, J.D.; Shipley, D.L.; Al-Hazzouri, A.; Rodriguez, F.A.; Womack, M.S.; Xiong, H.Q.; Waterhouse, D.M.; Tempero, M.A.; et al. A Randomized, Double-Blinded, Phase II Trial of Gemcitabine and Nab-Paclitaxel Plus Apatorsen or Placebo in Patients with Metastatic Pancreatic Cancer: The RAINIER Trial. Oncologist 2017, 22, 1427-e129.
- Apatorsen: Uses, Interactions, Mechanism of Action. DrugBank Online. Available online: https://go.drugbank.com/drugs/DB06094 (accessed on 16 January 2022).
- Crooke, S.T.; Baker, B.F.; Crooke, R.M.; Liang, X. Hai Antisense Technology: An Overview and Prospectus. Nat. Rev. Drug Discov. 2021, 20, 427–453.
- Jameson, G.S.; Borazanci, E.; Babiker, H.M.; Poplin, E.; Niewiarowska, A.A.; Gordon, M.S.; Barrett, M.T.; Rosenthal, A.; Stoll-D’Astice, A.; Crowley, J.; et al. Response Rate Following Albumin-Bound Paclitaxel Plus Gemcitabine Plus Cisplatin Treatment Among Patients with Advanced Pancreatic Cancer: A Phase 1b/2 Pilot Clinical Trial. JAMA Oncol. 2020, 6, 125–132.
- Reedijk, J.; Lohman, P.H.M. Cisplatin: Synthesis, Antitumour Activity and Mechanism of Action. Pharm. Weekbl. Sci. Ed. 1985, 7, 7–173.
- Cisplatin: Uses, Interactions, Mechanism of Action. DrugBank Online. Available online: https://go.drugbank.com/drugs/DB00515 (accessed on 16 January 2022).
- Ghosh, S. Cisplatin: The First Metal Based Anticancer Drug. Bioorg. Chem. 2019, 88, 102925.
- Mahipal, A.; Tella, S.H.; Kommalapati, A.; Goyal, G.; Soares, H.; Neuger, A.; Copolla, D.; Kim, J.; Kim, R. Phase 1 Trial of Enzalutamide in Combination with Gemcitabine and Nab-Paclitaxel for the Treatment of Advanced Pancreatic Cancer. Investig. New Drugs 2019, 37, 473–481.
- Enzalutamide: Uses, Interactions, Mechanism of Action. DrugBank Online. Available online: https://go.drugbank.com/drugs/DB08899 (accessed on 16 January 2022).
- Erdogan, B.; Kostek, O.; Bekirhacioglu, M. Enzalutamide in Prostate Cancer, A Review on Enzalutamide and Cancer. Eurasian J. Med. Oncol. 2018, 2, 121–129.
- Ng, K.; Hendifar, A.; Starodub, A.; Chaves, J.; Yang, Y.; Koh, B.; Barbie, D.; Hahn, W.C.; Fuchs, C.S. Phase 1 Dose-Escalation Study of Momelotinib, a Janus Kinase 1/2 Inhibitor, Combined with Gemcitabine and Nab-Paclitaxel in Patients with Previously Untreated Metastatic Pancreatic Ductal Adenocarcinoma. Investig. New Drugs 2019, 37, 159–165.
- Momelotinib: Uses, Interactions, Mechanism of Action. DrugBank Online. Available online: https://go.drugbank.com/drugs/DB11763 (accessed on 16 January 2022).
- Azhar, M.; Kincaid, Z.; Kesarwani, M.; Ahmed, A.; Wunderlich, M.; Latif, T.; Starczynowski, D.; Azam, M. Momelotinib Is a Highly Potent Inhibitor of FLT3-Mutant AML. Blood Adv. 2021, 6, 1186–1192.
- Weaver, B.A. How Taxol/Paclitaxel Kills Cancer Cells. Mol. Biol. Cell 2014, 25, 2677–2681.
- Yardley, D.A. Nab-Paclitaxel Mechanisms of Action and Delivery. J. Control. Release 2013, 170, 365–372.
- Veltkamp, S.A.; Jansen, R.S.; Callies, S.; Pluim, D.; Visseren-Grul, C.M.; Rosing, H.; Kloeker-Rhoades, S.; Andre, V.A.M.; Beijnen, J.H.; Slapak, C.A.; et al. Oral Administration of Gemcitabine in Patients with Refractory Tumors: A Clinical and Pharmacologic Study. Clin. Cancer Res. 2008, 14, 3477–3486.
- Espey, M.G.; Chen, P.; Chalmers, B.; Drisko, J.; Sun, A.Y.; Levine, M.; Chen, Q. Pharmacologic Ascorbate Synergizes with Gemcitabine in Preclinical Models of Pancreatic Cancer. Free Radic. Biol. Med. 2011, 50, 1610–1619.
- Barton-Burke, M. Gemcitabine: A Pharmacologic and Clinical Overview. Cancer Nurs. 1999, 22, 176–183.
- Dilruba, S.; Kalayda, G.V. Platinum-Based Drugs: Past, Present and Future. Cancer Chemother. Pharmacol. 2016, 77, 1103–1124.
- Hato, S.V.; Khong, A.; de Vries, I.J.M.; Lesterhuis, W.J. Molecular Pathways: The Immunogenic Effects of Platinum-Based Chemotherapeutics. Clin. Cancer Res. 2014, 20, 2831–2837.
- Galluzzi, L.; Vitale, I.; Michels, J.; Brenner, C.; Szabadkai, G.; Harel-Bellan, A.; Castedo, M.; Kroemer, G. Systems Biology of Cisplatin Resistance: Past, Present and Future. Cell Death Dis. 2014, 5, e1257.
- Von Hoff, D.D.; Ervin, T.; Arena, F.P.; Chiorean, E.G.; Infante, J.; Moore, M.; Seay, T.; Tjulandin, S.A.; Ma, W.W.; Saleh, M.N.; et al. Increased Survival in Pancreatic Cancer with Nab-Paclitaxel plus Gemcitabine. N. Engl. J. Med. 2013, 369, 1691–1703.





