Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Frantz Rom Poulsen | + 1182 word(s) | 1182 | 2022-02-09 10:01:10 | | | |
| 2 | Yvaine Wei | Meta information modification | 1182 | 2022-02-21 04:14:31 | | | | |
| 3 | Lasse Cramer Ahrens | + 213 word(s) | 1395 | 2022-02-26 13:44:05 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Poulsen, F.; Ahrens, L. Effect of Sodium Fluorescein on High-Grade Gliomas. Encyclopedia. Available online: https://encyclopedia.pub/entry/19659 (accessed on 10 May 2026).
Poulsen F, Ahrens L. Effect of Sodium Fluorescein on High-Grade Gliomas. Encyclopedia. Available at: https://encyclopedia.pub/entry/19659. Accessed May 10, 2026.
Poulsen, Frantz, Lasse Ahrens. "Effect of Sodium Fluorescein on High-Grade Gliomas" Encyclopedia, https://encyclopedia.pub/entry/19659 (accessed May 10, 2026).
Poulsen, F., & Ahrens, L. (2022, February 20). Effect of Sodium Fluorescein on High-Grade Gliomas. In Encyclopedia. https://encyclopedia.pub/entry/19659
Poulsen, Frantz and Lasse Ahrens. "Effect of Sodium Fluorescein on High-Grade Gliomas." Encyclopedia. Web. 20 February, 2022.
Copy Citation
High-grade gliomas (HGG) are the most common primary malignant brain cancer in adults. Complete surgical removal of HGG is known to increase the overall survival and progression-free survival. Fluorescence-guided surgery with 5-aminolevulinic acid (5-ALA) increases gross total resection considerably compared to white light surgery (65% vs. 36%). Recently, an off-label fluorophore, sodium fluorescein (SF), has become popular in fluorescence-guided surgery due to numerous utility benefits compared to 5-ALA, including lower cost, non-toxicity, easy administration during surgery and a wide indication for other CNS tumors.
fluorescence-guided surgery
5-ALA
sodium fluorescein
resection
glioblastoma
glioma
brain metastases
1. Introduction
High-grade gliomas (HGGs) arise from astrocytes or oligodendrocytes. They are classified as WHO grade III–IV [1], with an aggressive growth pattern [2][3], a poor prognosis and a median overall survival of approximately 12 to 15 months from diagnosis [4][5]. The current treatment consists of the maximum safe resection followed by chemotherapy and radiotherapy [6]. It is well-documented that the extent of resection (EOR) correlates positively with improved overall survival (OS) and progression-free survival (PFS) [7][8]. Surgery is commonly aimed at gross total resection (GTR) with removal of all contrast-enhancing tumor. However, a recent study by Glenn et al. [9] showed that supramaximal resection (SMR), i.e., resection beyond the boundaries of contrast enhancement, further improved PFS and OS for the 32 glioma patients included in the study compared to GTR and subtotal resection (STR), respectively, with PFS = 15 months vs. 7 months vs. 6 months (p < 0.003) and OS = 24 months vs. 11 months vs. 9 months (p < 0.004). At the same time, the authors found no statistically significant differences in the postoperative rate of complications between the groups [9]. These findings support the notion that glioma surgery should aim for resection to the functional limit of the peritumoral region. Although an interesting prospect, this concept is associated with technical challenges as access to functional intraoperative mapping techniques is limited in some centers. Furthermore, it can be difficult to distinguish healthy brain tissue from tumor-infiltrated tissue when surgery is performed under white light, which is the most common technique in many centers (Figure 1A). These issues result in suboptimal EOR with historical GTR rates of approximately 36–52% [10][11] despite the use of supportive technologies such as neuronavigation systems and intraoperative MRI.
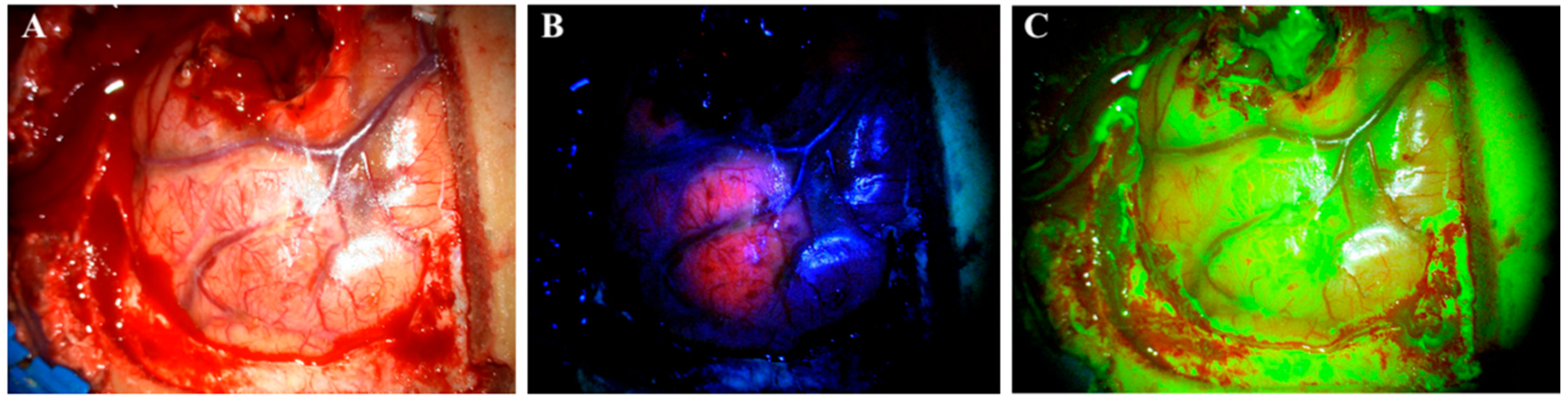
Figure 1. Visualization of a glioblastoma under a surgical microscope with different techniques. (A) white light. (B) 5-aminolevulinic acid. (C) sodium fluorescein.
Recently, fluorescence-guided surgery (FGS) has been becoming increasingly popular among neurosurgeons as a tool to distinguish between pathological and nonpathological tissue intraoperatively [12][13]. The only approved drug for fluorescence-guided glioma surgery is 5-aminolevulinic acid (5-ALA), although sodium fluorescein (SF) has been used increasingly (off-label) by many neurosurgeons due to several benefits. The use of 5-ALA is supported by multiple studies demonstrating superior evidence level compared to SF [14].
2. Agents for FGS (5-ALA and SF)
Currently, 5-ALA is the only approved drug for FGS. It is administrated orally three hours before surgery in doses of 20 mg/kg [10][11]. However, a recent study indicated that the maximum fluorescence occurs 7–8 h after administration and even later in marginal tumor tissue (8–9 h) [15]. One study even reported fluorescence of 5-ALA 28 h after administration [16]. In addition to glioma surgery, 5-ALA also has other medical applications, such as photodynamic therapy of actinic keratosis, superficial basal cell carcinomas [17][18] and urothelial carcinomas [19]; 5-ALA is a natural precursor molecule to the hemeprotein of hemoglobin synthesis. The compound undergoes a number of intracellular transformations in the cytosol and is transferred to the mitochondria to complete the synthesis. Protoporphyrin IX (PPIX) is the last compound in the synthesis pathway before the enzyme ferrochelatase adds the Fe2+ ion and transforms it into a heme. By similar mechanisms, exogenous 5-ALA is taken up by cells and then transformed into PPIX. Due to the downregulated level of ferrochelatase in glioma cells, PPIX accumulates in them instead of in normal cells [20]. PPIX has fluorescent properties and absorbs light in the blue spectrum (375–440 nm) to emit a red–violet light (640–710 nm) upon fluorophore relaxation. This enables neurosurgeons to visualize glioma tissue with accumulated PPIX under a surgical microscope using a red light filter [21] (Figure 1B).
Contrary to 5-ALA, SF is an unspecific fluorescent marker of a disrupted blood–brain barrier (BBB). The mechanism of action for SF is different from 5-ALA as it is not accumulated intracellularly in tumor cells, but rather is distributed and accumulated in the extracellular space throughout brain areas where the BBB is disrupted, e.g., by tumor infiltration [22]. Thereby, SF is not selectively used for HGGs, but rather accumulates in all tumors with contrast enhancement on CT or MR [13]. When first introduced, SF was administrated intravenously in high doses (20 mg/kg) and visualized under a surgical microscope illuminating white light. Recently, the surgical procedure changed and SF is now administrated at the beginning of an operation in low doses of 3–5 mg/kg [13]. SF has the light absorption maximum at 494 nm and emits green fluorescence at 540–690 nm. It is visualized under a surgical microscope with a yellow (560 nm) filter (Figure 1C). SF can be visualized up to 4 h after administration [23].
Contrary to 5-ALA, SF is an unspecific fluorescent marker of a disrupted blood–brain barrier (BBB). The mechanism of action for SF is different from 5-ALA as it is not accumulated intracellularly in tumor cells, but rather is distributed and accumulated in the extracellular space throughout brain areas where the BBB is disrupted, e.g., by tumor infiltration [22]. Thereby, SF is not selectively used for HGGs, but rather accumulates in all tumors with contrast enhancement on CT or MR [13]. When first introduced, SF was administrated intravenously in high doses (20 mg/kg) and visualized under a surgical microscope illuminating white light. Recently, the surgical procedure changed and SF is now administrated at the beginning of an operation in low doses of 3–5 mg/kg [13]. SF has the light absorption maximum at 494 nm and emits green fluorescence at 540–690 nm. It is visualized under a surgical microscope with a yellow (560 nm) filter (Figure 1C). SF can be visualized up to 4 h after administration [23].
3. White Light Surgery Compared to 5-ALA-Guided Surgery in HGGs
Many studies have found increased GTR, OS and PFS with 5-ALA-guided HGG resection compared to conventional white light surgery. Specifically, a recent meta-analysis found GTR rates of 79.1% with 5-ALA use compared to 52.8% without 5-ALA [10]. The OS was increased by approximately three months (95% CI, 2.43–3.68 months; p < 0.001), PFS—by approximately one month (95% CI, 0.61–1.45 months; p < 0.001) when using 5-ALA compared to conventional surgery.
In 2006, a pivotal randomized controlled trial (RCT) was performed to evaluate the efficacy of 5-ALA for HGG surgery [11]. The study found a significant difference in GTR rates between the groups, with GTR = 65% in the 5-ALA group and 36% in the white light group (p < 0.0001).
In 2006, a pivotal randomized controlled trial (RCT) was performed to evaluate the efficacy of 5-ALA for HGG surgery [11]. The study found a significant difference in GTR rates between the groups, with GTR = 65% in the 5-ALA group and 36% in the white light group (p < 0.0001).
4. SF-Guided Surgery Compared to White Light Surgery in HGGs
In recent years, SF has become the preferred agent for fluorescence-guided surgery for HGG in some centers, although the evidence is less compared to 5-ALA.
Several studies report an increased GTR rate in SF-guided tumor resection compared to conventional white light surgery (73.4–85.7% vs. 30.1–62.5%, respectively)[24][25][26][27][28][29]. However, it is important to note that the level of evidence in the available literature is class III or below as all the studies have important caveats that must be considered, such as lack of randomization and small sample size [30].
5. Direct Comparison of 5-ALA and SF
Until recently, all evidence of SF-guided surgery was based on uncontrolled studies or studies comparing SF to white light surgery alone [24][25][26][27][28][29]. As 5-ALA guidance is a standard practice technology for HGG resection in many centers, studies that directly compare SF with 5-ALA are necessary to investigate the superiority or potential equivalence of either compound based on GTR or survival endpoints. In addition, such studies would also clarify the potential practical and health economic benefits of one agent over the other.
In 2019, some of these aspects were investigated in a retrospective cohort study by Hansen et al.[31]. The authors found no significant difference in GTR or CRET between 5-ALA and SF (64% vs. 62% (OR, 0.90, p = 0.76) and 29.5% vs. 36.2% (OR, 1.34, p = 0.86), respectively).
In 2019, some of these aspects were investigated in a retrospective cohort study by Hansen et al.[31]. The authors found no significant difference in GTR or CRET between 5-ALA and SF (64% vs. 62% (OR, 0.90, p = 0.76) and 29.5% vs. 36.2% (OR, 1.34, p = 0.86), respectively).
6. SF and 5-ALA in Brain Metastases
Although the evidence of 5-ALA is superior in HGG surgery, it is well-known that it does not have an essential part in the removal of brain metastases. In 2019, Marhold et al., administrated 5-ALA to 157 different brain metastases and found that only 66% of the tumors exhibited visible fluorescence [32]. Hence, 5-ALA is not reliable to use in brain metastases and therefore is not routinely used.
Currently, the strongest evidence for FGS in brain metastases is based on the use of SF. Five clinical studies have investigated SF for this indication with the GTR rate as the primary endpoint [33][34][35][36][37]. Only two studies compared SF directly to white light surgery [36][37], while the others did not include a control population. Studies with control group found a significant higher GTR in the SF group compared to the white light group (94% and 62-84%, respectively)
7. Conclusions
It is found that there is generally solid evidence for the use of FGS for multiple indications in neuro-oncology. Although 5-ALA remains the gold standard for FGS in HGG surgery, recent studies have indicated that SF may be a plausible alternative for HGG surgery. Additional comparative studies with 5-ALA and SF are needed to determine if SF is a feasible alternative based on GTR. Furthermore, SF is useful for a multitude of contrast-enhancing CNS tumors, including brain metastases. SF has several benefits over 5-ALA, including a broader spectrum of indications, lower cost and more practical utility features.
References
- Louis, D.N.; Perry, A.; Reifenberger, G.; von Deimling, A.; Figarella-Branger, D.; Cavenee, W.K.; Ohgaki, H.; Wiestler, O.D.; Kleihues, P.; Ellison, D.W. The 2016 World Health Organization Classification of Tumors of the Central Nervous System: A summary. Acta Neuropathol. 2016, 131, 803–820.
- Sanai, N.; Polley, M.Y.; McDermott, M.W.; Parsa, A.T.; Berger, M.S. An extent of resection threshold for newly diagnosed glioblastomas. J. Neurosurg. 2011, 115, 3–8.
- Stupp, R.; Mason, W.P.; van den Bent, M.J.; Weller, M.; Fisher, B.; Taphoorn, M.J.; Belanger, K.; Brandes, A.A.; Marosi, C.; Bogdahn, U.; et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N. Engl. J. Med. 2005, 352, 987–996.
- Koshy, M.; Villano, J.L.; Dolecek, T.A.; Howard, A.; Mahmood, U.; Chmura, S.J.; Weichselbaum, R.R.; McCarthy, B.J. Improved survival time trends for glioblastoma using the SEER 17 population-based registries. J. Neurooncol. 2012, 107, 207–212.
- Tran, B.; Rosenthal, M.A. Survival comparison between glioblastoma multiforme and other incurable cancers. J. Clin. Neurosci. 2010, 17, 417–421.
- Weller, M.; van den Bent, M.; Tonn, J.C.; Stupp, R.; Preusser, M.; Cohen-Jonathan-Moyal, E.; Henriksson, R.; Rhun, E.L.; Balana, C.; Chinot, O.; et al. European Association for Neuro-Oncology (EANO) guideline on the diagnosis and treatment of adult astrocytic and oligodendroglial gliomas. Lancet Oncol. 2017, 18, e315–e329.
- Lacroix, M.; Abi-Said, D.; Fourney, D.R.; Gokaslan, Z.L.; Shi, W.; DeMonte, F.; Lang, F.F.; McCutcheon, I.E.; Hassenbusch, S.J.; Holland, E.; et al. A multivariate analysis of 416 patients with glioblastoma multiforme: Prognosis, extent of resection, and survival. J. Neurosurg. 2001, 95, 190–198.
- Brown, T.J.; Brennan, M.C.; Li, M.; Church, E.W.; Brandmeir, N.J.; Rakszawski, K.L.; Patel, A.S.; Rizk, E.B.; Suki, D.; Sawaya, R.; et al. Association of the Extent of Resection With Survival in Glioblastoma: A Systematic Review and Meta-analysis. JAMA Oncol. 2016, 2, 1460–1469.
- Glenn, C.A.; Baker, C.M.; Conner, A.K.; Burks, J.D.; Bonney, P.A.; Briggs, R.G.; Smitherman, A.D.; Battiste, J.D.; Sughrue, M.E. An Examination of the Role of Supramaximal Resection of Temporal Lobe Glioblastoma Multiforme. World Neurosurg. 2018, 114, e747–e755.
- Gandhi, S.; Tayebi Meybodi, A.; Belykh, E.; Cavallo, C.; Zhao, X.; Syed, M.P.; Borba Moreira, L.; Lawton, M.T.; Nakaji, P.; Preul, M.C. Survival Outcomes Among Patients With High-Grade Glioma Treated With 5-Aminolevulinic Acid-Guided Surgery: A Systematic Review and Meta-Analysis. Front. Oncol. 2019, 9, 620.
- Stummer, W.; Pichlmeier, U.; Meinel, T.; Wiestler, O.D.; Zanella, F.; Reulen, H.J. Fluorescence-guided surgery with 5-aminolevulinic acid for resection of malignant glioma: A randomised controlled multicentre phase III trial. Lancet Oncol. 2006, 7, 392–401.
- Senders, J.T.; Muskens, I.S.; Schnoor, R.; Karhade, A.V.; Cote, D.J.; Smith, T.R.; Broekman, M.L. Agents for fluorescence-guided glioma surgery: A systematic review of preclinical and clinical results. Acta Neurochir. 2017, 159, 151–167.
- Schipmann, S.; Schwake, M.; Suero Molina, E.; Stummer, W. Markers for Identifying and Targeting Glioblastoma Cells during Surgery. J. Neurol. Surg. A Cent. Eur. Neurosurg. 2019, 80, 475–487.
- Kaneko, S.; Eljamel, M.S. Fluorescence image-guided neurosurgery. Future Oncol. 2017, 13, 2341–2348.
- Kaneko, S.; Suero Molina, E.; Ewelt, C.; Warneke, N.; Stummer, W. Fluorescence-Based Measurement of Real-Time Kinetics of Protoporphyrin IX After 5-Aminolevulinic Acid Administration in Human In Situ Malignant Gliomas. Neurosurgery 2019, 85, E739–E746.
- Maragkos, G.A.; Schüpper, A.J.; Lakomkin, N.; Sideras, P.; Price, G.; Baron, R.; Hamilton, T.; Haider, S.; Lee, I.Y.; Hadjipanayis, C.G.; et al. Fluorescence-Guided High-Grade Glioma Surgery More Than Four Hours After 5-Aminolevulinic Acid Administration. Front. Neurol. 2021, 12, 644804.
- Patel, G.; Armstrong, A.W.; Eisen, D.B. Efficacy of photodynamic therapy vs other interventions in randomized clinical trials for the treatment of actinic keratoses: A systematic review and meta-analysis. JAMA Dermatol. 2014, 150, 1281–1288.
- Hauschild, A.; Stockfleth, E.; Popp, G.; Borrosch, F.; Bruning, H.; Dominicus, R.; Mensing, H.; Reinhold, U.; Reich, K.; Moor, A.C.; et al. Optimization of photodynamic therapy with a novel self-adhesive 5-aminolaevulinic acid patch: Results of two randomized controlled phase III studies. Br. J. Dermatol. 2009, 160, 1066–1074.
- Inoue, K. 5-Aminolevulinic acid-mediated photodynamic therapy for bladder cancer. Int. J. Urol. 2017, 24, 97–101.
- Lakomkin, N.; Hadjipanayis, C.G. Fluorescence-guided surgery for high-grade gliomas. J. Surg. Oncol. 2018, 118, 356–361.
- Tonn, C.J.; Stummer, W. Fluorescence-guided resection of malignant gliomas using 5-aminolevulinic acid: Practical use, risks, and pitfalls. Clin. Neurosurg. 2008, 55, 20–26.
- Bömers, J.P.; Danielsen, M.E.; Schulz, M.K.; Halle, B.; Kristensen, B.W.; Sørensen, M.D.; Poulsen, F.R.; Pedersen, C.B.; Sodium fluorescein shows high surgeon-reported usability in glioblastoma surgery. Surgeon 2020, 18, 344–348, https://doi.org/10.1016/j.surge.2020.01.003.
- Schupper, A.J.; Rao, M.; Mohammadi, N.; Baron, R.; Lee, J.Y.K.; Acerbi, F.; Hadjipanayis, C.G; Fluorescence-Guided Surgery: A Review on Timing and Use in Brain Tumor Surgery.. Front. Neurol 2021, 12, 682151, https://doi.org/10.3389/fneur.2021.682151.
- Shinoda, J.; Yano, H.; Yoshimura, S.; Okumura, A.; Kaku, Y.; Iwama, T.; Sakai, N. Fluorescence-guided resection of glioblastoma multiforme by using high-dose fluorescein sodium: Technical note. J. Neurosurg. 2003, 99, 597–603.
- Koc, K.; Anik, I.; Cabuk, B.; Ceylan, S. Fluorescein sodium-guided surgery in glioblastoma multiforme: A prospective evaluation. Br. J. Neurosurg. 2008, 22, 99–103.
- Chen, B.; Wang, H.; Ge, P.; Zhao, J.; Li, W.; Gu, H.; Wang, G.; Luo, Y.; Chen, D. Gross total resection of glioma with the intraoperative fluorescence-guidance of fluorescein sodium. Int. J. Med. Sci. 2012, 9, 708–714.
- Catapano, G.; Sgulò, F.G.; Seneca, V.; Lepore, G.; Columbano, L.; di Nuzzo, G. Fluorescein-Guided Surgery for High-Grade Glioma Resection: An Intraoperative “Contrast-Enhancer”. World Neurosurg. 2017, 104, 239–247.
- Katsevman, G.A.; Turner, R.C.; Urhie, O.; Voelker, J.L.; Bhatia, S. Utility of sodium fluorescein for achieving resection targets in glioblastoma: Increased gross- or near-total resections and prolonged survival. J. Neurosurg. 2019, 132, 1–7.
- Hong, J.; Chen, B.; Yao, X.; Yang, Y. Outcome comparisons of high-grade glioma resection with or without fluorescein sodium-guidance. Curr. Probl. Cancer 2019, 43, 236–244.
- Burns, P.B.; Rohrich, R.J.; Chung, K.C. The levels of evidence and their role in evidence-based medicine. Plast. Reconstr. Surg. 2011, 128, 305–310.
- Hansen, R.W.; Pedersen, C.B.; Halle, B.; Korshoej, A.R.; Schulz, M.K.; Kristensen, B.W.; Poulsen, F.R.; Comparison of 5-aminolevulinic acid and sodium fluorescein for intraoperative tumor visualization in patients with high-grade gliomas: A single-center retrospective study.. J. Neurosurg 2019, 133, 1-8, https://thejns.org/doi/abs/10.3171/2019.6.JNS191531.
- Marhold, F.; Mercea, P.A.; Scheichel, F.; Berghoff, A.S.; Heicappell, P.; Kiesel, B.; Mischkulnig, M.; Borkovec, M.; Wolfsberger, S.; Woehrer, A.; et al. Detailed analysis of 5-aminolevulinic acid induced fluorescence in different brain metastases at two specialized neurosurgical centers: Experience in 157 cases. J. Neurosurg. 2019, 133, 1–12.
- Okuda, T.; Kataoka, K.; Yabuuchi, T.; Yugami, H.; Kato, A. Fluorescence-guided surgery of metastatic brain tumors using fluorescein sodium. J. Clin. Neurosci. 2010, 17, 118–121.
- Schebesch, K.M.; Hoehne, J.; Hohenberger, C.; Proescholdt, M.; Riemenschneider, M.J.; Wendl, C.; Brawanski, A. Fluorescein sodium-guided resection of cerebral metastases-experience with the first 30 patients. Acta Neurochir. 2015, 157, 899–904.
- Hohne, J.; Hohenberger, C.; Proescholdt, M.; Riemenschneider, M.J.; Wendl, C.; Brawanski, A.; Schebesch, K.M. Fluorescein sodium-guided resection of cerebral metastases-an update. Acta Neurochir. 2017, 159, 363–367.
- Xiao, S.Y.; Zhang, J.; Zhu, Z.Q.; Li, Y.P.; Zhong, W.Y.; Chen, J.B.; Pan, Z.Y.; Xia, H.C. Application of fluorescein sodium in breast cancer brain-metastasis surgery. Cancer Manag. Res. 2018, 10, 4325–4331.
- Kofoed, M.S.; Pedersen, C.B.; Schulz, M.K.; Kristensen, B.W.; Hansen, R.W.; Markovic, L.; Halle, B.; Poulsen, F.R. Fluorescein-guided resection of cerebral metastases is associated with greater tumor resection. Acta Neurochir. 2021.
- Lee, C.H.; Kim, D.G.; Kim, J.W.; Han, J.H.; Kim, Y.H.; Park, C.K.; Kim, C.Y.; Paek, S.H.; Jung, H.W. The role of surgical resection in the management of brain metastasis: A 17-year longitudinal study. Acta Neurochir. 2013, 155, 389–397.
More
Information
Subjects:
Surgery
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.1K
Revisions:
3 times
(View History)
Update Date:
28 Feb 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





