| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Nameer Khairullah Mohammed | + 2179 word(s) | 2179 | 2020-08-27 06:14:15 | | | |
| 2 | Bruce Ren | + 2 word(s) | 2181 | 2020-09-03 03:05:37 | | | | |
| 3 | Bruce Ren | + 2 word(s) | 2181 | 2020-09-03 03:06:31 | | | | |
| 4 | Bruce Ren | -1 word(s) | 2180 | 2020-09-03 11:21:33 | | |
Video Upload Options
This is a entry focused on the application of spray drying technology for the edible oils encapsulation. This entry covered most of the relevant works have done recently on the encapsulation of oils by spray drying. The factors affecting the process of encapsulation and the application in food with the properties of the final product.The application of the spray drying technique in the food industry for the production of a broad range of ingredients has become highly desirable compared to other drying techniques. Recently, the spray drying technique has been applied extensively for the production of functional foods, pharmaceuticals and nutraceuticals. Encapsulation using spray drying is highly preferred due to economic advantages compared to other encapsulation methods. Encapsulation of oils using the spray drying technique is carried out in order to enhance the handling properties of the products and to improve oxidation stability by protecting the bioactive compounds. Encapsulation of oils involves several parameters—including inlet and outlet temperatures, total solids, and the type of wall materials—that significantly affect the quality of final product.
1. Introduction
Modern consumers are concerned about their health, and this concern leads to a high demand for foods that contain bioactive or functional ingredients (especially natural ones) that increase the nutritional value and health status of food [1]. In particular, there is a growing demand for nutritious and healthy oils in the food, pharmaceutical, and cosmetic industries due to their multi-functional properties. Nevertheless, due to their high level of unsaturation, vegetable and marine oils are prone to oxidation deterioration in addition to the resulting production of unpleasant taste. Moreover, oils are unstable under processing and storage conditions due to their sensitivity to light and heat, which limits their application in the food industry. Hence, it is necessary to protect the oils to improve their stability during handling, processing, and storage [2][3]. These fluctuations negatively affect the developed products in terms of their shelf-life, sensory properties, and overall acceptability [4]. Encapsulation is a promising approach that is widely used to overcome the above-mentioned problems by protecting the core materials from heat, light and oxygen, thus promoting stability, increasing bioavailability, flavour-masking, and controlled release, while maintaining the oils’ functional properties and increasing their ease of handling [5]. Encapsulation can be defined as a process for entrapping one substance (termed the core material or active agent) within another substance (the coating, shell, or carrier/wall material). Encapsulation using spray drying is a reliable technique in the food industry that has been successfully used to overcome these challenges [6][7]. Microencapsulation technology is becoming a common technology used with oils around the world, such as microencapsulated palm oil, microencapsulated fish oil, and microencapsulated coconut oil, which are used as food ingredients.
Encapsulation may enhance ease of handling, adequacy of concentration and uniformity of dispersion [8]. Despite the rapid drying contact time (several seconds) of the core materials in the drying chamber, spray dryers have high drying temperatures, typically with an input air temperature of 150 to 250 °C, and an outlet air temperature of 50–80 °C [9]. The resulting particles range from a few nanometers to a few hundred micrometers [10]. The structural type and desired size generally depend on the preparation method and the wall materials applied [11]. The application of encapsulation in food production requires the wall materials to be of food grade and have protective properties for the core materials against external factors. The carrier materials typically used for oil encapsulation include synthetic polymers and natural biomaterials (commonly carbohydrates and proteins) [12].
2. Oil Encapsulation Benefits
Encapsulation of oils improves the oxidative stability of their lipids and protects sensitive constituents (core) such as active compounds, oils, flavour compounds, and vitamins from environmental factors. This facilitates high solubility and easy mixing of the core material, and manages the release of the core material in order to attain an appropriate delay until the appropriate stimulus. Moreover, it reduces the evaporation of volatile compounds in the core material, masking or covering any unpleasant tastes associated with the core material [13][14][15][16]. The reasons for applying encapsulation for oils can be summarized as follows:
a) Improvement of the oxidative stability of lipids by applying the encapsulation process to produce powdered edible oil products, helping to prolong shelf-life by protecting oils from oxidation [14].
b) Shielding of core materials, which are usually sensitive compounds such as oils, flavours, and vitamins, from oxygen, light, or water. In general, food oils exhibit significant susceptibility to light, temperature, air, and irradiation [17][18].
c) Alteration of the oils from a liquid to a dry form in order to produce powder with high solubility and acceptable mixing properties of the core materials. Conversion of the fluid feed (flavours and edible oils) into powders in solid form with desirable handling properties is an important use of encapsulation in the food industry [19].
d) Managing the release of the core material in order to achieve a suitable delay for the proper stimulus; a vital benefit of the encapsulation of oils and flavours is control over the release time of the active ingredients until they have arrived at their target [20].
e) Preventing evaporation of volatile compounds in the core materials. The dry powder obtained through the encapsulation process possesses high oxidation stability and reduced volatility. The encapsulation of oils results in a dry powder with improved oxidation and less volatility, which simplifies its application in various end-products such as cakes and beverages [21].
f) Masking or covering the unpleasant tastes of the core material. The incorporation of some edible oils, such as vegetable oils or marine oils, in food products is in high demand due to the nutritional value of these products . In addition, encapsulation can also help to overcome the main problems associated with food containing 3 PUFA, the unpalatable “fishy” flavour of fish oil, and the susceptibility of polyunsaturated fatty acids to oxidation, has all of which have a negative impact on food acceptability [22]. Encapsulation positively affects the stability and resistance of oils under storage conditions in comparison to oils without encapsulation [23].
-
Encapsulation Using the Spray Drying Technique
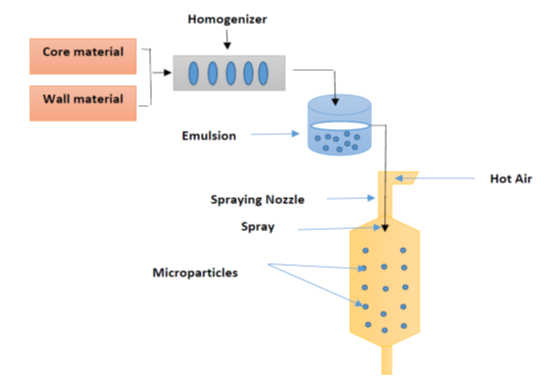
Encapsulation is a technique whereby small particles or droplets of active substances that have several attractive characteristics are enclosed within a coating in order to protect them against environmental factors such as oxygen, light, moisture and interactions with other compounds. Encapsulation is typically used to obtain particles with a diameter of 1–1000 μm of food ingredients or other materials. In addition, this technique can enable the controlled release of the encapsulated core when certain conditions are met [24]. Encapsulation using a spray dryer has frequently been used for food production on an industrial scale since the late 1950s, primarily for fats and oils, and flavourings and colourings [25]; see Table 1. Moreover, encapsulation is widely used in the food industry to incorporate oil aromas in a spray-dried form, because it is inexpensive, flexible, can be used in continuous operation, and produces particles of good quality . The process can be conducted by changing the slurry emulsion from a liquid form into a powder in a continuously operating procedure. The basic principle of this method is to dissolve the core/wall materials in water to prepare an emulsion in liquid form, and then to feed this emulsion into a hot medium (100–300 °C) to evaporate the water. The final dried product can be collected in powder form, or as agglomerated particles, depending on the nature of the materials used in the feed, the design of the dryer’s operation, and the operating conditions. The high temperature of the drying chamber facilitates the water evaporation from the droplets [26]. Despite the energy used in the spray-drying process in terms of heat, spray drying has been shown to be more efficient than freeze drying for the encapsulation of oils, with a 30–50 times lower cost [27]. This technique does, however, require high-temperature conditions and access to air. Although the temperature of the spray dryer is high, the wet-bulb only requires a short duration of exposure (few seconds), and water vaporization will take place in the range of 30 to 50 °C [28]. A schematic diagram of the encapsulation process using the spray drying technique is presented in Figure 1. According to Bakry et al. , encapsulation using spray-drying consists of four main stages: (i) preparation of a stable emulsion; (ii) homogenization of the dispersion; (iii) atomization of the emulsion; and (iv) dehydration of the atomized particles. Typically, the first stage is carried out by dissolving the wall materials in distilled water and emulsifying, or dispersing using a magnetic stirrer overnight at 25 °C to ensure full saturation of the polymer molecules, and to prevent any variations caused by temperature changes. Before starting the second stage, core materials mixed with an aqueous solution of the wall materials and then the emulsifying agent can be added, depending on the emulsifying characteristics of the wall materials. The formed emulsion containing the wall materials and the core substances must be stable until the drying stage [29].
3. Unit Operation of Spray Drying
The process of spray drying is illustrated in Figure 1. The steps of the spray drying process involve: a) atomization of fluid feed; b) drying of the medium; c) drying of the feed and spray contact; and d) separation of the product from air. The properties of the final product are directly influenced by all of the steps, including their operational parameters. Spray drying is a very rapid and reproducible drying method, due to the very large surface area created by the atomization of the liquid feed [30].
3.1. Feed Atomization
Atomization involves converting the fluid feed (emulsion) into small droplets of uniform size resulting in a balance of heat and mass transfer in the drying stage, as well as increasing the surface area and allowing a good distribution of the feed within the dryer chamber. The size enlargement of the particles (surface area) leads to fast water evaporation and formation of a crust, drying the feed in seconds. Different types of atomizers, including rotary atomizers, pressure nozzles, pneumatic nozzles and sonic nozzles, exist to accomplish feed liquid collapse [30]. To choose the atomizer type based on the feed needing to be converted to dried powder and also on the desired particle size of the final product.
3.2. Air Flow Contact
Air flow contact is influenced by the fluid spray and the air contact time, because both factors determine the drying rate and the intensity of the drying. At this stage, the air flow contact starts from the drying step and continues during atomization. The atmosphere inside the heating chamber of the spray dryer is controlled by the operating parameters of the air heating and filtration system. The gas used during the process is usually nitrogen or another gas chosen based on the sensitivity or instability of the feed in the presence of oxygen [30]. In general, two types of feed-drying air flow conditions are available:
-
a) The co-current drying design: in this configuration, the atomizer is located in the top of the drying chamber along with the drying gas stream inlet. The feed is sprayed in the same direction as the hot air, and thus it is preferred for heat sensitive composites. The inlet temperature is usually 150–220 °C and rapid vaporization takes place; the outlet temperature is 50–80 °C, which limits the thermal degradation. This design exposes heat-sensitive materials to the lower exit air temperature only.
-
b) The counter-current drying design: this system is not popular and has limited applications for dry products. The emulsion and the drying air are introduced at opposite ends of the dryer. Although this configuration is more thermally efficient than the co-current drying design as it exposes the dried powders to high temperatures, it has limited use for products that are sensitive to heat. As a result, of the complicated flow, the drying phenomena are not clear in these systems [31].
3.3. Drying and Particle Formation
The third step is drying and particle formation by convection. The heat is transferred from the drying medium to the droplets, where it is converted to latent heat during evaporation of moisture content from the droplets. The temperature and mass transfer rate are based on the diameter of the droplets and the relative velocity of the air and the droplets. Quick moisture evaporation immediately happens when the droplets come into contact with the drying air. Initially, the droplet surface exhibits a continuous mass transfer of moisture from within the droplet. At last, a dried shell is formed and moisture evaporation continues at a slower rate until the final product is formed [30].
3.4. Separation of Product from the Drying Air
The last step involves the separation of the product from the drying air. The spray drying begins when the dried particles fall to the bottom of the drying chamber and are collected through gravitational effects; while fines entrained in the drying air are parted from the atmosphere and separation occurs after the dried particles recovered in a cyclone filter that is located outside the dryer with collection bottle . The liquid emulsion consists of the wall and core materials and is converted through the spray drying process into dried particles in powder form. The dried particles are spherical in shape with a size range from 10 to 100 micrometres [32].
Figure 1. Schematic representation of the encapsulation process by spray-drying
References
- Marrelli, M.; Statti, G.; Conforti, F. A Review of Biologically Active Natural Products from Mediterranean Wild Edible Plants: Benefits in the Treatment of Obesity and Its Related Disorders. Molecules 2020, 25, 649.
- Di Giorgio, L.; Salgado, P.R.; Mauri, A.N. Encapsulation of Fish Oil in Soybean Protein Particles by Emulsification and Spray Drying. Food Hydrocoll. 2019, 87, 891–901.
- Eratte, D.; Dowling, K.; Barrow, C.J.; Adhikari, B. Recent Advances in the Microencapsulation of Omega-3 Oil and Probiotic Bacteria through Complex Coacervation: A Review. Trends Food Sci. Technol. 2018, 71, 121–131.
- Ojagh, S.M.; Hasani, S. Characteristics and Oxidative Stability of Fish Oil Nano-Liposomes and Its Application in Functional Bread. J. Food Meas. Charact. 2018, 12, 1–9.
- Fang, Z.; Bhandari, B. Encapsulation of Polyphenols–A Review. Trends Food Sci. Technol. 2010, 21, 510–523.
- Agnihotri, N.; Mishra, R.; Goda, C.; Arora, M. Microencapsulation–A Novel Approach in Drug Delivery: A Review. Indo Glob. J. Pharm. Sci. 2012, 2, 1–20.
- Aguiar, J.; Estevinho, B.N.; Santos, L. Microencapsulation of Natural Antioxidants for Food Application–The Specific Case of Coffee Antioxidants–A Review. Trends Food Sci. Technol. 2016, 58, 21–39.
- Lozano-Vazquez, G.; Lobato-Calleros, C.; Escalona-Buendia, H.; Chavez, G.; Alvarez-Ramirez, J.; Vernon-Carter, E.J. Effect of the Weight Ratio of Alginate-Modified Tapioca Starch on the Physicochemical Properties and Release Kinetics of Chlorogenic Acid Containing Beads. Food Hydrocoll. 2015, 48, 301–311.
- Geranpour, M.; Assadpour, E.; Jafari, S.M. Recent Advances in the Spray Drying Encapsulation of Essential Fatty Acids and Functional Oils. Trends Food Sci. Technol. 2020, 102, 71–90
- Amstad, E. Capsules: Their Past and Opportunities for Their Future; ACS Publications: Washington, DC, USA, 2017.
- Estevinho, B.N.; Rocha, F. Application of Biopolymers in Microencapsulation Processes. In Biopolymers for Food Design; Elsevier: Amsterdam, The Netherlands, 2018; pp. 191–222.
- Zuidam, N.J.; Nedović, V. Encapsulation Technologies for Active Food Ingredients and Food Processing; Springer: Berlin/Heidelberg, Germany, 2010.
- Zheng, X.; Wu, F.; Hong, Y.; Shen, L.; Lin, X.; Feng, Y. Developments in Taste-Masking Techniques for Traditional Chinese Medicines. Pharmaceutics 2018, 10, 157.
- Ahn, J.-H.; Kim, Y.-P.; Lee, Y.-M.; Seo, E.-M.; Lee, K.-W.; Kim, H.-S. Optimization of Microencapsulation of Seed Oil by Response Surface Methodology. Food Chem. 2008, 107, 98–105.
- Jafari, S.M.; Assadpoor, E.; He, Y.; Bhandari, B. Encapsulation Efficiency of Food Flavours and Oils during Spray Drying. Dry. Technol. 2008, 26, 816–835.
- Kolanowski, W.; Ziolkowski, M.; Weißbrodt, J.; Kunz, B.; Laufenberg, G. Microencapsulation of Fish Oil by Spray Drying--Impact on Oxidative Stability. Part 1. Eur. Food Res. Technol. 2006, 222, 336–342.
- Bilia, A.R.; Guccione, C.; Isacchi, B.; Righeschi, C.; Firenzuoli, F.; Bergonzi, M.C. Essential Oils Loaded in Nanosystems: A Developing Strategy for a Successful Therapeutic Approach. Evid. Based Complement. Altern. Med. 2014, 2014, 14.
- Bakry, A.M.; Abbas, S.; Ali, B.; Majeed, H.; Abouelwafa, M.Y.; Mousa, A.; Liang, L. Microencapsulation of Oils: A Comprehensive Review of Benefits, Techniques, and Applications. Compr. Rev. Food Sci. Food Saf. 2016, 15, 143–182.
- Gupta, S.; Khan, S.; Muzafar, M.; Kushwaha, M.; Yadav, A.K.; Gupta, A.P. Encapsulation: Entrapping Essential Oil/Flavors/Aromas in Food. In Encapsulations; Elsevier: Amsterdam, The Netherlands, 2016; pp. 229–268.
- Manaf, M.A.; Subuki, I.; Jai, J.; Raslan, R.; Mustapa, A.N. Encapsulation of Volatile Citronella Essential Oil by Coacervation: Efficiency and Release Study. In IOP Conference Series: Materials Science and Engineering; IOP Publishing: Bristol, UK, 2018; p. 12072.
- Encina, C.; Márquez-Ruiz, G.; Holgado, F.; Giménez, B.; Vergara, C.; Robert, P. Effect of Spray-Drying with Organic Solvents on the Encapsulation, Release and Stability of Fish Oil. Food Chem. 2018, 263, 283–291.
- Melgosa, R.; Benito-Román, Ó.; Sanz, M.T.; de Paz, E.; Beltrán, S. Omega–3 Encapsulation by PGSS-Drying and Conventional Drying Methods. Particle Characterization and Oxidative Stability. Food Chem. 2019, 270, 138–148.
- Mohammed, N.K.; Meor Hussin, A.S.; Tan, C.P.; Abdul Manap, M.Y.; Alhelli, A.M. Quality Changes of Microencapsulated Nigella Sativa Oil upon Accelerated Storage. Int. J. Food Prop. 2017, 20, S2395–S2408.
- Veiga, R.D.S. Da; Aparecida Da Silva-Buzanello, R.; Corso, M.P.; Canan, C. Essential Oils Microencapsulated Obtained by Spray Drying: A Review. J. Essent. Oil Res. 2019, 31, 457–473.
- Pellicer, J.A.; Fortea, M.I.; Trabal, J.; Rodríguez-López, M.I.; Gabaldón, J.A.; Núñez-Delicado, E. Stability of Microencapsulated Strawberry Flavour by Spray Drying, Freeze Drying and Fluid Bed. Powder Technol. 2019, 347, 179–185.
- Costa, S.S.; Machado, B.A.S.; Martin, A.R.; Bagnara, F.; Ragadalli, S.A.; Alves, A.R.C. Drying by Spray Drying in the Food Industry: Micro-Encapsulation, Process Parameters and Main Carriers Used. African J. Food Sci. 2015, 9, 462–470.
- Ng, S.; Jessie, L.L.; Tan, C.; Long, K.; Nyam, K. Effect of Accelerated Storage on Microencapsulated Kenaf Seed Oil. J. Am. Oil Chem. Soc. 2013, 90, 1023–1029.
- Schuck, P.; Dolivet, A.; Méjean, S.; Zhu, P.; Blanchard, E.; Jeantet, R. Drying by Desorption: A Tool to Determine Spray Drying Parameters. J. Food Eng. 2009, 94, 199–204.
- Gharsallaoui, A.; Roudaut, G.; Chambin, O.; Voilley, A.; Saurel, R. Applications of Spray-Drying in Microencapsulation of Food Ingredients: An Overview. Food Res. Int. 2007, 40, 1107–1121.
- Sosnik, A.; Seremeta, K.P. Advantages and Challenges of the Spray-Drying Technology for the Production of Pure Drug Particles and Drug-Loaded Polymeric Carriers. Adv. Colloid Interface Sci. 2015, 223, 40–54.
- Edris, A.E.; Kalemba, D.; Adamiec, J.; Piątkowski, M. Microencapsulation of Nigella Sativa Oleoresin by Spray Drying for Food and Nutraceutical Applications. Food Chem. 2016, 204, 326–333.
- Munin, A.; Edwards-Lévy, F. Encapsulation of Natural Polyphenolic Compounds; A Review. Pharmaceutics 2011, 3, 793–829.