
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Ana Cristina Silvestre Ferreira | + 1657 word(s) | 1657 | 2021-12-13 03:48:00 | | | |
| 2 | Catherine Yang | -1 word(s) | 1656 | 2021-12-28 03:42:15 | | | | |
| 3 | Catherine Yang | Meta information modification | 1656 | 2022-01-10 03:13:13 | | | | |
| 4 | Catherine Yang | Meta information modification | 1656 | 2022-01-10 03:14:15 | | |
Video Upload Options
The AB blood group system has been identified in wild felids, as well as in the domestic cat. In both, type A blood seems to be the most common, although the majority of wild felid species exhibit one single blood type, showing that there seems to be variation between species, but not within species, and no evidence of geographical variation was yet found, showing apparently no genetic variability.
1. Introduction
Knowledge of blood type systems in animals is of paramount importance mainly for blood transfusion medicine, phylogenetic studies, and reproductive compatibilities. Concerning phylogenetic studies, the analysis of the phylogenetic tree for the primate ABO blood group allowed for an estimation of the divergence time between human and nonhuman primates [1]. Although sharing a similar nomenclature, the human ABO blood group and the feline AB blood group do not share the same glycolipid antigenic determinants of the erythrocyte membrane [2]. Recently, in order to avoid misinterpretations, it was proposed that the feline blood group system should be renamed to ABC, where C corresponds to the blood type AB [3]. In felids, the divergence is estimated to have occurred over 9000 years ago, where the domestic cat was considered a member of the subspecies Felis silvestris catus, derived from the domestication of the Libyan cat (Felis silvestris lybica). Genetic studies show that, in some European regions, most European wildcats are hybrids between the wild and domestic subspecies [4]. However, the phylogenetic relationships among the felids need to be further studied, and it is also unknown how the feline blood type systems have been inhered among the Felidae family.
Blood transfusions can be life-saving when required [5]. In wild felids medicine, there are common problems like run overs, parasitism, blood loss, and hypotension associated with surgical procedures, where a blood transfusion can be a life-saving procedure [6][7]. Reports of blood transfusions in species other than dogs and cats are very scarce in the literature, even more so in regard to wild felids [6]. In order to perform a blood transfusion in a safe manner, there is a need for knowledge of feline blood groups; however, blood groups remain mostly unstudied or unknown for wild felids [5][6]. Mismatched transfusions can produce immune reactions that result in red blood cells hemolysis and recipient death; blood typing and crossmatching ensure compatible and effective transfusions [8][9]. Xenotransfusions have been tried, both between domestic cats and wild cats, or even between dogs and domestic cats, but the results do not always lead to an effective result [10][11][12][13][14].
2. Wild Felids AB Blood System
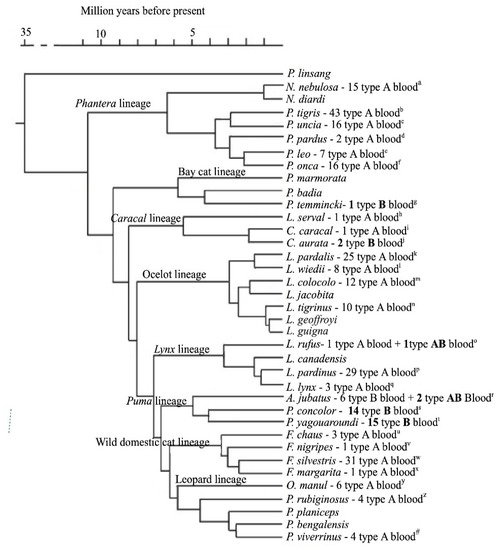
A study on 131 wild felids of 26 different species from zoos and wild animal parks in the USA and Dubai identified for the first time the AB blood group system in the species studied [15]. The authors also demonstrated, by high performance thin layer chromatography, that erythrocyte membrane glycolipids of the wild cats correspond to the major disialoganglioside patterns observed in domestic cats, with type B cells expressing exclusively N-acetylneuraminic acid (NeuAc) and type A cells predominantly N-glycolylneuraminic acid, with small amounts of NeuAc [2][15][16][17]. Thus, it has been assumed that the traditional and the commercial feline blood typing methods could also be applied for wild felids blood typing [18]. Since then, a few new studies on wild felids blood group system have been performed in different species and with different blood typing methods (Table 1). In Figure 1, according to a study on phylogeny of Felidae based on a data set of 22,789 base pairs of DNA, including autosomal, Y-linked, X-linked, and mitochondrial gene segments [19], we added to the phylogenetic tree data showing the number of animals studied, their blood type and place of origin. Naturally occurring alloantibodies were also studied [15][20][21] and identified [20][21].
| Lineage | Nº Animals | Blood Type | Typing Method | Alloantibodies | Country | ||
|---|---|---|---|---|---|---|---|
| A | B | AB | - | ||||
| Ocelot | 15 | 15 | Tube hemagglutination | Not detected | USA/Dubai [23] | ||
| Caracal | 8 | 6 | 2 | ||||
| Asian leopard | 4 | 4 | |||||
| Puma | 23 | 21 | 2 | ||||
| Lynx | 5 | 4 | 1 | ||||
| Bay cat | 1 | 1 | |||||
| Wild domestic cats | 17 | 17 | |||||
| Panthera | 58 | 58 | |||||
| Felis silvestris | 25 | 25 | Tube hemagglutination | Detected | Spain [24] | ||
| Lynx pardinus | 29 | 29 | Tube hemagglutination | Detected | Spain [25] | ||
| Lynx pardinus | 111 | 111 | Immunochromatographic | Not determined | Spain * | ||
| Panthera tigris tigris | 30 | 30 | Slide agglutination test | Not determined | Thailand [6] | ||
| Ocelot | 15 | 15 | Tube hemagglutination | Not determined | Brazil ** [7] | ||
| Puma | 8 | 8 | |||||
| Ocelot | 25 | 25 | Tube hemagglutination and card *** | Not determined | Brazil **** [26] | ||
| Puma | 6 | 6 | |||||
| Panthera | 11 | 11 | |||||
Type A blood seems to be the most common among wild species as it is in domestic
cats [27][28][29][30][31][32][33][34][35][36][37][38][39][40][41][42][43][44][45][46][47][48][49][50][51][52][53][22] (Table 1 and Figure 1). The overall presentation of wild felids blood types show that there is variation among breeds, but not within breeds, and no evidence of geographical variation has yet been found, showing apparently no genetic variability and probably reflecting a relatively minor gene pool within wild felid species in contrast to domestic cats. In domestic cats, according to a recent study, there is no evidence of the relationship between blood types and phylogenetic origin, despite some similarities found in blood types prevalences between Asian and American breeds and European and Oceanian breeds that could be explained by geographical proximity and trade relations in the first case and colonization policies in the second [54]. Recently, five feline erythrocyte antigens were identified [55], raising the suspicion that other blood group erythrocyte antigens might exist either for domestic cats or wild felids. Genetic and molecular studies in a larger number of animals are needed in order to unravel the presence of new blood groups.
In domestic cats, blood loss is the most common indication for blood transfusion, followed by ineffective erythropoiesis and red blood cells destruction [56]. Little is known about the incidence of such diseases in wild felids. Blood loss in wild felids may be related to run overs, surgical procedures, or parasitism [6][7]. A study in Mato Grosso, Brazil, describes that from 211 run overs, 59 were mammals, mostly represented by carnivores, with 52 roadkilled individuals (24% of all roadkilled animals), including some rare or endangered species such as Puma yagouaroundi, Leopardus colocolo, Puma concolor, and ocelot (Leopardus pardalis) [57], but no reports of blood transfusion were found. As far as we know, no erythropoiesis disorders have been described in wild felids, but there has been a report of red blood cells destruction related to a rattlesnake bite in a caracal [58]. As a major cause of blood transfusion related to hemolytic anemia in domestic cats, erythrocyte parasitism, like hemoplasmosis, babesiosis, or cytauxzoonosis, has also been reported in wild felids [59][60][61][62][63][64][23], and is sometimes related to anemia [59][61][64], but there have been no reports of animals receiving blood transfusions. As in domestic cats, in wild felids, to ensure compatible transfusions, blood typing and crossmatching should be performed. As mismatched transfusions can produce immune reactions that might result in red blood cells hemolysis and recipient death [8][9], a major and minor crossmatch prior to blood transfusion should always be performed and, whenever possible, a blood group determination should be conducted [65][25]. However, the clinical relevancy of the AB blood group system in wild felids is still not clear. The finding of only one blood type within the majority of wild felids species evaluated can allow, in those species where blood groups have not been defined, reliance on a single transfusion that must always be preceded by a crossmatch. Sometimes, upon compatibility studies, the use of domestic cat blood can lead to a safe blood transfusion. However, because of the presence of naturally occurring alloantibodies, post transfusion reactions or neonatal isoerythrolysis cannot be completely ruled out. As blood groups are not well defined in wild felids, leaning on crossmatching to ensure a safe blood transfusion may be an alternative to identify circulating blood group antibodies, however it cannot determine blood groups [5].
Although blood transfusions can be life-saving, in domestic cats and dogs blood transfusion medicine there are descriptions of risks associated with the transmission of infectious and transmissible parasitic diseases that should also be taken in account when performing blood transfusions in wild cats [26][66][67][68][69][70].
3. Conclusions
Unfortunately, the amount of research conducted on this topic is scarce, however, although an absence of genetic variability has been observed, as wild felid species seem to exhibit one single blood type, the information presented here and the recent knowledge on domestic cat blood types leads us to suspect the possibility that blood groups other than AB exist in wild felids. Particular attention should be paid to the presence of naturally occurring alloantibodies. In endangered wild felids in captivity, common problems in clinical and surgical management might include blood transfusions for its resolution; however, the number of blood donors is extremely limited and are generally not available. In the case of an urgent need for a blood transfusion, upon a crossmatch test, domestic cat blood could be used, but more studies are required before attempting xenotransfusion among felid groups. Taking into account our data, wild felids blood types knowledge could be useful not only for zoo medicine, but also for phylogenetic studies for reintroduction projects in order to preserve genetic diversity. More studies, with more species and a greater number of animals, are needed in order to clarify the importance of the AB blood group system in wild felids and its clinical relevance. In addition, because the majority of the European wildcats are hybrids between wild and domestic subspecies, we are currently left to wonder what mutations occurred in the CMHA 9000 years ago when the divergence from the wild cat to the domestic cat took place.
References
- Saitou, N.; Yamamoto, F. Evolution of primate ABO blood group genes and their homologous genes. Mol. Biol. Evol. 1997, 14, 399–411.
- Andrews, G.A.; Chavey, P.S.; Smith, J.E.; Rich, L. N-glycolylneuraminic acid and N-acetylneuraminic acid define feline blood group A and B antigens. Blood 1992, 79, 2485–2491.
- Kehl, A.; Truchet, L.; Langbein-Detsch, I.; Müller, E.; Giger, U. Updates on practical ABC blood compatibility testing in cats. Tierärztl. Prax. Ausg. K Kleintiere Heimtiere 2019, 47, 425–438.
- Driscoll, C.A.; Menotti-Raymond, M.; Roca, A.L.; Hupe, K.; Johnson, W.E.; Geffen, E.; Harley, E.H.; Delibes, M.; Pontier, D.; Kitchener, A.C.; et al. The near eastern origin of cat domestication. Science 2007, 317, 519–523.
- Gamble, K.C.; Moyse, J.A.; Lovstad, J.N.; Ober, C.B.; Thompson, E.E. Blood groups in the species survival Plan®, European Endangered species program, and managed in situ populations of bonobo (Pan paniscus), common chimpanzee (Pan troglodytes), gorilla (Gorilla ssp.), and orangutan (Pongo pygmaeus ssp.). Zoo Biol. 2011, 30, 427–444.
- Thengchaisri, N.; Sinthusingha, C.; Arthitwong, S.; Sattasathuchana, S. Comparative serological investigation between cat and tiger blood for transfusion. J. Vet. Med. Sci. 2017, 79, 1081–1085.
- Silva, T.D.P.; Dreyer, M.O.; Back, F.P.; Lacerda, L.A.; Damasceno, A.D.; Araújo, L.B.M.; Sant’Ana, F.J.F.; Fioravanti, M.C.S. Sistema de grupos sanguíneos AB em felídeos neotropicais e compatibilidade com gatos domésticos [AB blood group system in neotropical felids and compatibility with domestic cats]. Arq. Bras. Med. Vet. Zootec. 2017, 69, 889–895. (In Portuguese)
- Knottenbelt, C.M.; Day, M.J.; Cripps, P.; Mackin, A.J. Measurement of titres of naturally occurring alloantibodies against feline blood group antigens in the UK. J. Small Anim. Pract. 1999, 40, 365–370.
- Auer, L.; Bell, K. Transfusion reactions in cats due to AB blood group incompatibility. Res. Vet. Sci. 1983, 35, 145–152.
- Hessler, J.; Davis, L.E.; Dale, H.E. Effect of repeated transfusions of dog blood to cats. Small Anim. Clin. 1962, 2, 684–687.
- Bovens, C.; Gruffydd-Jones, T. Xenotransfusion with canine blood in the feline species: Review of the literature. J. Feline Med. Surg. 2013, 15, 62–67.
- Adamantos, S.; Smith, C. Alternative transfusion methods. In Manual of Veterinary Transfusion Medicine and Blood Banking; Kenichiro, Y., Holowaychuck, M.K., Eds.; John Wiley & Sons: Hoboken, NJ, USA, 2016; pp. 296–297.
- Euler, C.C.; Raj, K.; Mizukami, K.; Murray, L.; Chen, C.; Mackin, A.; Giger, U. Xenotransfusion of anemic cats with blood compatibility issues: Pre- and post-transfusion laboratory diagnostic and crossmatching studies. Vet. Clin. Pathol. 2016, 45, 244–253.
- Le Gal, A.; Thomas, E.K.; Hamm, K.R. Xenotransfusion of canine blood to cats: A review of 49 cases and their outcome. J. Small Anim. Pract. 2019, 61, 156–162.
- Griot-Wenk, M.E.; Giger, U. The AB blood group system in wild felids. Anim. Genet. 1999, 30, 144–147.
- Griot-Wenk, M.; Pahlsson, P.; Chisholm-Chait, A.; Spitalnik, F.; Spitalnik, S.L.; Giger, U. Biochemical characterization of the feline AB blood group system. Anim. Genet. 1993, 24, 401–407.
- Silvestre-Ferreira, A.C.; Masso, O.; Pastor, J. High-performance liquid chromatography ganglioside pattern of the AB feline blood group. Comp. Clin. Pathol. 2011, 20, 597–605.
- Silvestre-Ferreira, A.C.; Pastor, J. Feline neonatal isoerythrolysis and the importance of feline blood types. Vet. Med. Int. 2010, 2010, 753726.
- Werdelin, L.; Yamaguchi, N.; Johnson, W.E.; O’Brien, S.J. Phylogeny and evolution of cats (Felidae). In Biology and Conservation of Wild Felids; Macdonald, D.W., Loveridge, A.J., Eds.; Oxford University: Oxford, UK, 2010; pp. 59–82.
- Silvestre-Ferreira, A.C.; Marco, I.; Daussa, B.; Piñol, C.; Lavin, S.; Pastor, J. Blood group system in a captive population of European wildcats (Felis silvestris). Vet. Rec. 2006, 159, 567–568.
- Silvestre-Ferreira, A.C.; Bach-Raich, E.; Mesalles, M.; Vargas, A.; Martinez, F.; Cuenca, R.; Pastor, J. Blood group system in the iberian lynx (Lynx pardinus). In Proceedings of the 12th International Society of Animal Biochemistry Congress, Istanbul, Turkey, 22–26 May 2006.
- Bisca, J.M. Prevalência dos Tipos Sanguíneos A, B e AB em Felinos Selvagens Neotropicais Nativos do Brasil. Master’s Thesis, Universidade Estadual Paulista, São Paulo, Brasil, 2017.
- Sacristán, I.; Acuña, F.; Aguilar, E.; García, S.; López, M.J.; Cevidanes, A.; Cabello, J.; Hidalgo-Hermoso, E.; Johnson, W.E.; Poulin, E.; et al. Assessing cross-species transmission of hemoplasmas at the wild-domestic felid interface in Chile using genetic and landscape variables analysis. Sci. Rep. 2019, 9, 16816.
- Giger, U. Blood typing and crossmatching to ensure compatible transfusions. In Kirk’s Current Veterinary Therapy. XIII Small Animal Practice; Bonagura, J.D., Ed.; WB Saunders: Philadelphia, PA, USA, 2000; pp. 396–399.
- Knottenbelt, C.M. The feline AB blood group system and its importance in transfusion medicine. J. Feline Med. Surg. 2002, 4, 69–76.
- Marenzoni, M.L.; Antognoni, M.T.; Baldelli, F.; Miglio, A.; Stefanetti, V.; Desario, C.; Di Summa, A.; Buonavoglia, C.; Decaro, N. Detection of parvovirus and herpesvirus DNA in the blood of feline and canine blood donors. Vet. Microbiol. 2018, 224, 66–69.
- Giger, U.; Bucheler, J.; Diserens, D.; Hale, A.; Griot-Wenk, M. Geographical variation of the feline blood type frequencies in the United States. Feline Pract. 1991, 19, 21–26.
- Hubler, M.; Arnold, S.; Casal, M.; Fairburn, A.; Nussbaumer, M.; Rüsch, P. The blood group distribution in domestic cats in Switzerland. Schweiz. Arch.r Tierheilkd. 1993, 135, 231–235. (In German)
- Jensen, A.L.; Olesen, A.B.; Arnbjerg, J. Distribution of feline blood types detected in the Copenhagen area of Denmark. Acta Vet. Scand. 1994, 35, 121–124.
- Knottenbelt, C.M.; Addie, D.D.; Day, M.J.; Mackin, A.J. Determination of the prevalence of feline blood types in the UK. J. Small Anim. Pract. 1999, 40, 115–118.
- Mylonakis, M.E.; Koutinas, A.F.; Saridomichelakis, M.; Leontidis, L.; Papadogiannakis, E.; Plevraki, K. Determination of the prevalence of blood types in the non-pedigree feline population in Greece. Vet. Rec. 2001, 149, 213–214.
- Bagdi, N.; Magdus, M.; Leidinger, E.; Leidinger, J.; Vörös, K. Frequencies of feline blood types in Hungary. Acta Vet. Hung. 2001, 49, 369–375.
- Silvestre-Ferreira, A.C.; Pastor, J.; Almeida, O.; Montoya, A. Frequencies of feline blood types in northern Portugal. Vet. Clin. Pathol. 2004, 33, 240–243.
- Silvestre-Ferreira, A.C.; Pastor, J.; Sousa, A.P.; Pires, M.J.; Morales, M.; Abreu, Z.; Montoya, J.A. Blood types in the non-pedigree cat population of Gran Canaria. Vet. Rec. 2004, 155, 778–779.
- Ruiz de Gopegui, R.; Velasquez, M.; Espada, Y. Survey of feline blood types in the Barcelona area of Spain. Vet. Rec. 2004, 154, 794–795.
- Malik, R.; Griffin, D.L.; White, J.D.; Rozmanec, M.; Tisdall, P.L.C.; Fosters, S.F.; Bell, K.; Nicholas, F.W. The prevalence of feline A/B blood types in the Sydney region. Aust. Vet. J. 2005, 83, 38–44.
- Arikan, S.; Gurkan, M.; Ozaytekim, E.; Dodurka, T.; Giger, U. Frequencies of blood types A, B, and AB in non-pedigree domestic cats in Turkey. J. Small Anim. Pract. 2006, 47, 10–13.
- Forcada, Y.; Guitian, J.; Gibson, G. Frequencies of feline blood types at a referral hospital in the south east of England. J. Small Anim. Pract. 2007, 48, 570–573.
- Medeiros, M.A.S.; Soares, A.M.; Alviano, D.S.; Ejzemberg, R.; da Silva, M.H.; Almosny, N.R. Frequencies of feline blood types in the Rio de Janeiro area of Brazil. Vet. Clin. Pathol. 2008, 37, 272–276.
- Arikan, S.; Guzel, M.; Ozturk, A.S.; Simsek, O. Frequencies of blood type A, B and AB in cats from the mediterranean sea coast of the Turkey. Rev. Med. Vet. 2010, 161, 322–325.
- Juvet, F.; Brennan, S.; Mooney, C.T. Assessment of feline blood for transfusion purposes in the Dublin area of Ireland. Vet. Rec. 2011, 168, 352.
- Marques, C.; Ferreira, M.; Gomes, J.F.; Leita ̄o, N.; Costa, M.; Serra, P.; Duarte Correia, J.H.; Pomba, C.F. Frequency of blood type A, B, and AB in 515 domestic shorthair cats from the Lisbon area. Vet. Clin. Pathol. 2011, 40, 185–187.
- Zheng, L.; Zhong, Y.; Shi, Z.; Giger, U. Frequencies of blood types A, B, and AB in non-pedigree domestic cats in Beijing. Vet. Clin. Pathol. 2011, 40, 513–517.
- Spada, E.; Miglio, A.; Proverbio, D.; Antognoni, M.T.; Bagnagatti De Giorgi, G.; Ferro, E.; Mangili, V. Signalment and blood types in cats being evaluated as blood donors at two Italian university blood banks. Vet. Med. Int. 2014, 2014, 704836.
- Fosset, F.T.; Blais, M.C. Prevalence of feline blood groups in the Montreal area of Quebec, Canada. Can. Vet. J. 2014, 55, 1225–1228.
- Karadjole, T.; Kovacˇevic ́, I.; Samardžija, M.; Babic ́, T.; Kreszinger, M.; Radišic ́, B.; Harapin, I.; Bedrica, L. Blood groups in cats in the city of Zagreb. Vet. Arh. 2016, 86, 209–216.
- Cattin, R. Distribution of blood types in a sample of 245 New Zealand non-purebred cats. N. Z. Vet. J. 2016, 64, 154–157.
- Barrot, A.C.; Buttin, R.; Linsart, A.; Bachy, V.; Guidetti, M.; Blais, M.C. Frequency of feline blood types in non-pedigree cats in France. Rev. Med. Vet. 2017, 168, 235–240.
- Vieira, S.M.; Ferreira, R.R.F.; de Matos, A.J.; Cardoso, I.M.; Graça, R.M.C.; Soares, A.R.; Blasi-Brugué, C.; Sánchez, I.M.; Gopegui, R.R. Distribution of feline AB blood types: A review of frequencies and its implications in the Iberian Peninsula. J. Feline Med. Surg. Open Rep. 2017, 3, 2055116917727693.
- Sorgatto, S.; Brito De Oliveira, B.; Cristina, K.; Godoy, S.; Antunes, T.R.; Almeida Lacerda, L.; De Souza, A.I. Frequência dos tipos sanguíneos de gatos domésticos mestiços no município de Campo Grande, Mato Grosso do Sul, Brasil. Med. Vet. 2017, 11, 172–178.
- Nectoux, A.; Guidetti, M.; Barthélemy, A.; Pouzot-Nevoret, C.; Hoareau, G.L.; Goy-Thollot, I. Assessment of risks of feline mismatched transfusion and neonatal isoerythrolysis in the Lyon (France) area. J. Feline Med. Surg. Open Rep. 2019, 5, 205511691986317.
- Spada, E.; Perego, R.; Baggiani, L.; Salatino, E.; Priolo, V.; Mangano, C.; Pennisi, M.G.; Proverbio, D. Prevalence of blood types and alloantibodies of the AB blood group system in non-pedigree cats from Northern (Lombardy) and Southern (Sicily) Italy. Animals 2020, 10, 1129.
- McDermott, F.M.; Maloney, S.; McMillan, C.; Snead, E. The prevalence of blood groups in Domestic cats in the Saskatoon and Calgary areas of Saskatchewan and Alberta, Canada. Front. Vet. Sci. 2020, 7, 160.
- Gavazza, A.; Rossi, G.; Antognoni, M.T.; Cerquetella, M.; Miglio, A.; Mangiaterra, S. Feline blood groups: A systematic review of phylogenetic and geographical origin. Animals 2021, 11, 3339.
- Binvel, M.; Arsenault, J.; Depré, B.; Blais, M.C. Identification of 5 novel feline erythrocyte antigens based on the presence of naturally occurring alloantibodies. J. Vet. Intern. Med. 2021, 35, 234–244.
- Martinez-Sogues, L.; Blois, S.L.; Manzanilla, E.G.; Abrams-Ogg, A.O.; Cosentino, P. Exploration of risk factors for non-survival and for transfusion-associated complications in cats receiving red cell transfusions: 450 cases (2009 to 2017). J. Small Anim. Pract. 2020, 61, 177–184.
- Melo, E.S.; Santos-Filho, M. Efeitos da BR-070 na Província Serrana de Cáceres, Mato Grosso, sobre a comunidade de vertebrados silvestres. Rev. Bras. Zoociências 2007, 9, 185–192. (In Portuguese)
- Singleton, C.L.; Oosterhuis, J.E.; Seibold, K.; Lamberski, N. Successful treatment of a southern Pacific rattlesnake (Crotalus viridis helleri) bite in a caracal (Caracal caracal). J. Zoo Wildl. Med. 2009, 40, 378–381.
- Haefner, M.; Burke, T.J.; Kitchell, B.E.; Lamont, L.A.; Schaeffer, D.J.; Behr, M.; Messick, J.B. Identification of Haemobartonella felis (Mycoplasma haemofelis) in captive nondomestic cats. J. Zoo Wildl. Med. 2003, 34, 139–143.
- Yabsley, M.J.; Murphy, S.M.; Mark, W. Cunningham molecular detection and characterization of Cytauxzoon felis and a Babesia species in cougars from Florida. J. Wildl. Dis. 2006, 42, 366–374.
- Harvey, J.W.; Dunbar, M.R.; Norton, T.M.; Yabsley, M.J. Laboratory findings in acute Cytauxzoon felis infection in cougars (Puma concolor couguar) in Florida. J. Zoo Wildl. Med. 2007, 38, 285–291.
- Birkenheuer, A.J.; Marr, H.S.; Warren, C.; Acton, A.E.; Mucker, E.M.; Humphreys, J.G.; Tucker, M.D. Cytauxzoon felis infections are present in bobcats (Lynx rufus) in a region where cytauxzoonosis is not recognized in domestic cats. Vet. Parasitol. 2008, 153, 126–130.
- Williams, B.M.; Berentsen, A.; Shock, B.C.; Teixiera, M.; Dunbar, M.R.; Becker, M.S.; Yabsley, M.J. Prevalence and diversity of Babesia, Hepatozoon, Ehrlichia, and Bartonella in wild and domestic carnivores from Zambia, Africa. Parasitol. Res. 2014, 113, 911–918.
- Zaeemi, M.; Razmi, G.R.; Khoshnegah, J. The first detection of Cytauxzoon felis in a wild cat (Felis silvestris) in Iran. Comp. Clin. Pathol. 2015, 24, 181–184.
- Feldman, B.F. In-house canine and feline blood typing. J. Am. Anim. Hosp. Assoc. 1999, 35, 455–456.
- Reine, N.J. Infection and blood transfusion: A guide to donor screening. Clin. Tech. Small Anim. Pract. 2004, 19, 68–74.
- Satake, M.; Hoshi, Y.; Taira, R.; Momose, S.Y.; Hino, S.; Tadokoro, K. Symptomatic parvovirus B19 infection caused by blood component transfusion. Transfusion. 2011, 51, 1887–1895.
- Pennisi, M.G.; Hartmann, K.; Addie, D.D.; Lutz, H.; Gruffydd-Jones, T.; Boucraut-Baralon, C.; Egberink, H.; Frymus, T.; Horzinek, M.C.; Hosie, M.J.; et al. Blood transfusion in cats: ABCD guidelines for minimising risks of infectious iatrogenic complications. J. Feline Med. Surg. 2015, 17, 588–593.
- Stefanetti, V.; Miglio, A.; Cappelli, K.; Capomaccio, S.; Sgariglia, E.; Marenzoni, M.L.; Antognoni, M.T.; Coletti, M.; Mangili, V.; Passamonti, F. Detection of bacterial contamination and DNA quantification in stored blood units in 2 veterinary hospital blood banks. Vet. Clin. Pathol. 2016, 45, 406–410.
- Wardrop, K.J.; Birkenheuer, A.; Blais, M.C.; Callan, M.B.; Kohn, B.; Lappin, M.R.; Sykes, J. Update on canine and feline blood donor screening for blood-borne pathogens. J. Vet. Intern. Med. 2016, 30, 15–35.





