Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Dalen Zuidema | + 3640 word(s) | 3640 | 2021-12-20 05:02:47 | | | |
| 2 | Peter Tang | Meta information modification | 3640 | 2021-12-22 02:59:16 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Zuidema, D. Livestock Artificial Insemination. Encyclopedia. Available online: https://encyclopedia.pub/entry/17416 (accessed on 25 May 2026).
Zuidema D. Livestock Artificial Insemination. Encyclopedia. Available at: https://encyclopedia.pub/entry/17416. Accessed May 25, 2026.
Zuidema, Dalen. "Livestock Artificial Insemination" Encyclopedia, https://encyclopedia.pub/entry/17416 (accessed May 25, 2026).
Zuidema, D. (2021, December 21). Livestock Artificial Insemination. In Encyclopedia. https://encyclopedia.pub/entry/17416
Zuidema, Dalen. "Livestock Artificial Insemination." Encyclopedia. Web. 21 December, 2021.
Copy Citation
Artificial insemination is a crucial technology for livestock producers world-wide. This technology has afforded producers access to premier genetics without having to physically own superior sires.
artificial insemination
sperm sexing
livestock andrology
semen analysis
sperm selecting
semen storage
1. Introduction
The world-wide consumption of animal products has grown by over 20% in the past ten years [1]. The increase in meat and dairy consumption is expected to rise worldwide, with the majority of growth taking place in developing countries [1]. The rise in demand will be met by applying cutting-edge technology to agricultural production and by improving on the current best practices; artificial insemination (AI) is one such technology in need of improvement. When used by the livestock industry, AI can improve both quality and quantity of animal products.
AI has been commercially available to livestock producers for over sixty years [2]. The primary benefit of utilizing AI is the ability to rapidly improve the genetic quality of a herd using a premier male animal’s genetics without a producer having to purchase that specific male. Rather, a producer can buy semen doses from several premier sires with valuable genomes and production traits to improve their herd’s genetic and phenotypic profile in an affordable manner without having to buy or house the sires. AI is used to increase herd productivity by using sires that provide superior production traits to their offspring. Other benefits of AI are the reduced risk of disease transmission between animals, and reduced risk of other animals and workers being injured by male animals. AI maximizes the use of a genetically superior males, enabling them to sire hundreds or thousands of offspring by extending and shipping their semen for wider use.
2. Improving AI Technology
Despite its massive contributions to livestock production, there are numerous ways in which AI can be improved. These improvements are driven by new technologies and deeper understanding of reproductive physiology. The AI industry has many promising technologies which are being investigated and adopted for use. These technologies utilize new paradigms related to reproductive physiology to drive improved AI outcomes. Such technologies have been applied to semen analysis, sperm selection, and semen storage.
Improving semen quality control for use in AI has been studied using many different approaches. The conventional method for semen quality analysis is the use of a light microscope to evaluate sperm count, motility, and morphology. The use of subjective motility, and morphology as selection criteria is somewhat reliable but is associated with variable fertility rates, especially in horses [3]. Analyzing semen using more accurate and objective sperm quality parameters will improve sire AI fertility [4]. Semen analysis with such parameters can be applied as selection criteria for sires and used to eliminate those sires which lack an objective threshold of fertilization potential. Objective, biomarker-based semen analysis can also be used to evaluate samples of individual semen collections to ensure the quality of each ejaculation in sires with acceptable, but varied semen quality and AI fertility.
Finally, semen analysis parameters can be used in conjunction with semen purification methods during processing to improve the quality of sperm within each dose of semen, particularly when processing semen of high value sires with superior genetic traits. Additionally, using sperm quality parameters that are correlated with better storability and higher tolerance to cryopreservation will improve semen shelf-life, further benefitting the AI industry [5]. All these improvements can be achieved using cutting-edge semen analysis and sperm selection methods.
3. Semen Analysis
Two standout technologies have been developed to accurately and objectively analyze sperm quality. Both implement computer software which can calculate sperm quality based on objective measurements. Computer-assisted semen analysis (CASA) is a system which analyzes sperm count (thus concentration), morphology, and motility objectively and accurately [6]. CASA evaluates these quality measures more accurately than conventional light microscopy and has been enhanced with the ability to analyze sperm viability and DNA fragmentation, which are markers of sperm quality; these advancements will further improve the utility of the CASA system [7].
The second system being developed is even more powerful than CASA in terms of its throughput and ability to analyze semen quality based on objective, machine-measurable parameters. Flow cytometry (FC) based semen analysis is an approach that works with specific probes to evaluate sperm viability, DNA content, acrosomal integrity, mitochondrial membrane potential, and various determinants of sperm quality including surface proteins, glycans, and select ions [8][9] linked to sperm function and quality [10][11][12]. This approach is termed biomarker-based sperm analysis [5]. Fluorescent probes are used to detect specific sperm quality biomarkers. These probes induce a level of fluorescence to be emitted by every single spermatozoon positive for the biomarker used and the emission is measured by a light detector in the flow cytometer and used to evaluate the quality of the entire semen sample [7]. This technology can be further refined to sort spermatozoa based on the different fluorescent levels emitted by each spermatozoon, which is how sexed semen is currently sorted based on quantification of the fluorescence emitted by a DNA stain. Similar to sperm sexing, the biomarker-based fluorescence activated cell sorting would require a modified cell sorter to sort the spermatozoa within a semen sample for sperm quality characteristics. Such an approach may be accelerated by the development of so-called gentle, low-pressure cell sorters currently introduced to market.
Currently, CASA is the simpler and more affordable option, that allows quick and repeatable sperm motility and morphometry assessment. FC-based semen analysis adds additional analysis power because identifying specific biomarkers provides a more in-depth understanding of the sperm quality of individual sires and ejaculates. Despite the greater cost, FC-based semen analysis has begun to be adopted within the cattle AI industry [13][14][15]. As flow cytometry continues to grow in popularity and the technology is further developed, these systems will become more affordable and be further adopted for use in the AI industry at large, accelerating a trend that started a decade ago. This biomarker-based flow cytometric semen analysis allows for the detection of subtle differences in sperm quality that cannot be measured by CASA alone. As the AI industries begin to adopt more objective semen analysis tool such as CASA, the next step will be to include biomarker-based analysis in order to further ensure best possible semen quality. In terms of the swine industry specifically, the use of biomarker-based semen analysis will help reduce the use of semen pooling practices and reserve this practice only when necessary. Thus, males with inferior semen quality can more quickly be removed from the breeding herd. We acknowledge pooling might not be completely eliminated in the swine industry because these analyses are impractical in some settings. Additionally, swine producers are more concerned about genetic lines and the herd at large than the individual animals. This is different than how the other livestock industries are currently managed because of the differences in genetic seedstock and commodity production flow. However, even with this caveat, ensuring that the boar herd uses males with the best semen quality possible based on objective measurements including both CASA and biomarker-based analysis will help the level of boar fertility achieved even when pooled and can be pushed to a higher quality with these tools.
Furthermore, the potential for biomarker-based analysis is extensive as new sperm quality biomarkers continue to be identified [5][7][8][9][16][17]. New advancements to flow cytometry also allow for the dissemination of image-based flow cytometry (IBFC); this technology captures images of each single cell at high speeds (500 to 2000 sperm/second) and allows for characterizing biomarker localization at subcellular level (e.g., between sperm head and tail or within their respective sub-compartments), which is not possible in regular flow cytometry [18][19]. Coupled with artificial intelligence methods such as machine learning, IBFC in animal andrology may eventually lead to development of label-free approaches to semen analysis [20]. In this scenario, images from IBFC could have features extracted from ground truth images from biomarker characterization to train neural networks for differentiating fertile and infertile spermatozoa within an ejaculate based solely on bright field data with no need for sample labeling and fluorescence detection, an example is provided in Figure 1 [16]. Algorithms created by this type of research could then be utilized on bare bone image-based flow cytometers with no need of expensive lasers or on CASAs used in andrology labs daily.
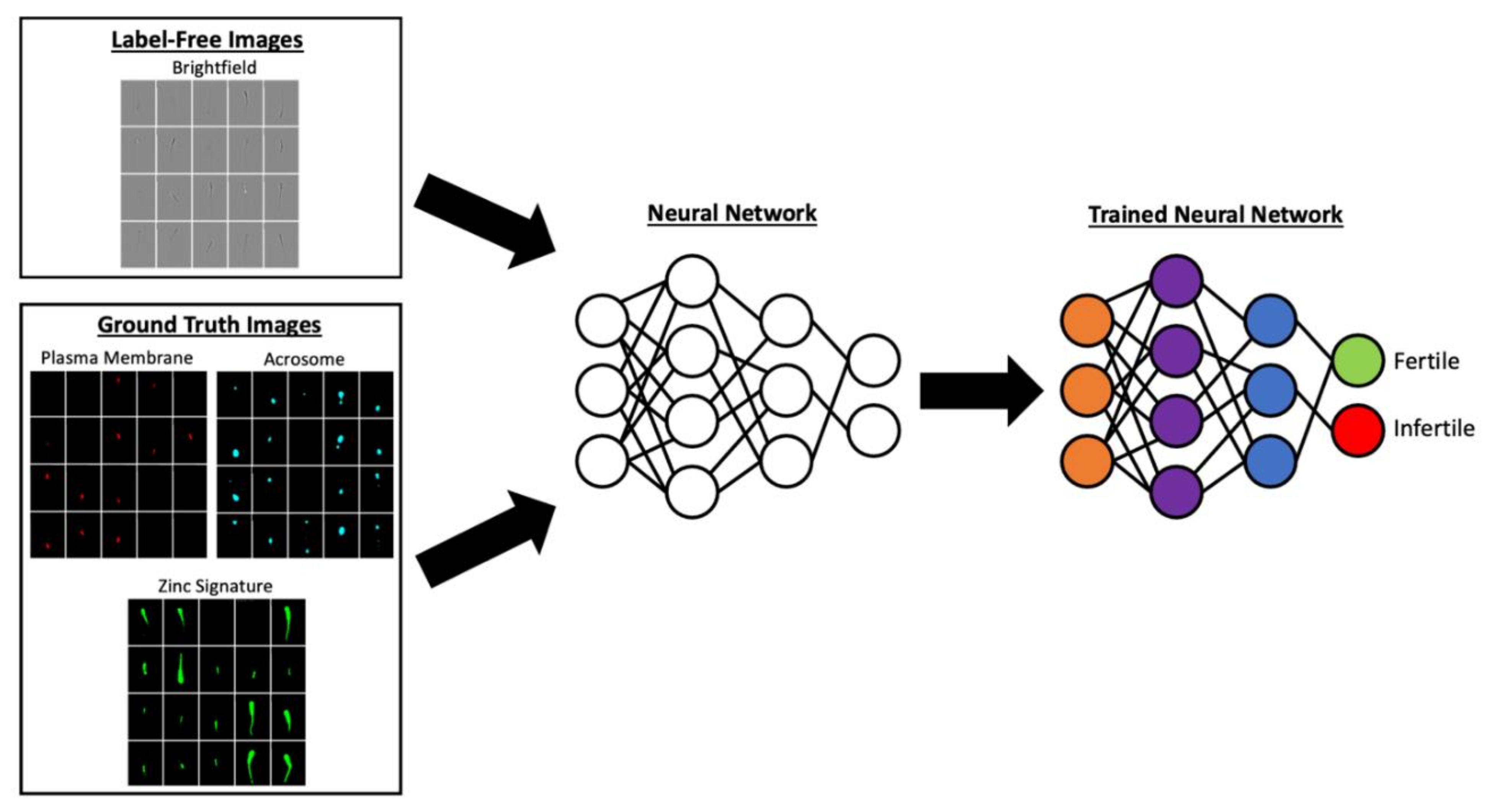
Figure 1. Biomarker sperm neural network training example. Images reflecting the biomarker status such as plasma membrane integrity (detected by propidium iodide), acrosome integrity (detected by lectin PNA conjugated to Cy5), and the sperm zinc signature (detected by FluoZin™-3 AM, ThermoFisher Scientific, Waltham, MA, USA) could be used to train neural networks to detect the percent of fertile and infertile sperm in a sample.
4. Sperm Selection
The analysis of semen samples provides baseline data which is invaluable to the AI industry to improve the quality of sires and the semen they provide. The ability to use sperm quality parameters and biomarkers as targets to purify a semen sample is highly advantageous as well. By utilizing the same sperm quality parameters and sperm surface determinants previously mentioned, the quality of spermatozoa within each semen sample can be improved by rejecting defective spermatozoa and seminal debris. Sperm selection used in conjunction with more stringent sire and ejaculate selection will lead to an even greater increase in fertility. Additionally, the use of sperm selection methods can rescue a semen sample from a genetically valuable sire that produces substandard semen collections [7]; this application is the opposite of increasing male fertility at large but is still of economic value. Sires with valuable production traits but lower fertility will continue to be sought after; by purifying their semen collections, their AI fertility can be manually boosted.
Beyond the previously mentioned sperm sorting using biomarker-based flow cytometry, there are other promising sperm selection methods that are currently less laborious and require less expensive equipment. Colloid centrifugation is one such selection method. It selects spermatozoa based on motility, morphology, viability, and membrane integrity. The most common form of colloid centrifugation for sperm analysis is Single Layer Centrifugation (SLC) [21], which uses a single filter layer made from silica nanoparticles. SLC effectively purifies semen samples using a gentle centrifugation. Motile spermatozoa can line up in the direction of the centrifugation forces and pass through the silica layer. Additionally, those spermatozoa which are morphologically normal and have intact plasma membranes and chromatin pass through the colloid layer more easily [21]. This purification method has been studied in swine [22], sheep [23], cattle [24], and horses [25]. In all species, it was able to reliably improve the quality of the purified semen samples and in horses SLC has successfully increased AI conception rates in field trials by nearly 14% [25]. SLC purification can improve sperm quality in semen samples while remaining relatively cheap and simple to apply. Additionally, SLC selected sperm samples have maintained post-thaw viability better than non-selected samples when used in conjunction with semen freezing protocols [26][27]. This technique seems promising; however, field AI trials must still be conducted in other species. Furthermore, the method may not be conducive to boar semen purification due to large semen volume.
Nanopurification is another selection method which selects spermatozoa based on some of the same sperm quality biomarkers used in FC-based semen analysis. Specific probes can identify abnormal spermatozoa based on surface determinants (proteins, glycans) that relate to sperm quality [28]. Nanopurification uses these same negative biomarkers to remove abnormal or defective spermatozoa. Magnetic nanoparticles are coated with probes such as lectins, recombinant proteins and antibodies, that are known to bind negative biomarkers found on abnormal spermatozoa’s surface. These bound spermatozoa can then be removed from a sample using a magnet, without the need for sperm-damaging centrifugation or filtration [28]. The nanoparticles and the spermatozoa bound to them are drawn to the bottom of the tube by a magnet to form a plaque/pellet of bound, abnormal spermatozoa. The particle free spermatozoa are then pipetted out with the rest of the supernatant and ready for use in standard freezing, extending, or AI protocols [28]. Developed originally for bull spermatozoa and validate by AI field trials, the technique has been more recently adapted for boar [29][30] and even human semen purification [31].
This technique is quite simple once the coated nanoparticles are made. Nanoparticles are incubated with the semen samples for fifteen minutes at room temperature, and then, the sample is placed on a magnet for fifteen minutes. PNA-lectin is a probe which reliably binds spermatozoa with compromised acrosomes; a half-dose of semen purified with PNA-lectin coated nanoparticles was able to yield conception rates equal to a full dose of non-purified semen from the same bull. This demonstrates that the improvement of quality of semen acts in a compensatory manner for semen quantity. This technology has yielded calves that are appropriately developed and healthy [28]. Additionally, nanopurification has been experimentally implemented in swine utilizing negative biomarker probes for reacted acrosomes and apoptotic spermatozoa and was found to result in an equivalent fertility rate to non-selected spermatozoa. The resulting piglets developed normally and were healthy [29]. Nanopurification has encouraging outcomes and possibilities; it can easily be applied using other probes, which may be even more useful than PNA-lectin. Additionally, more than one type of probe can be used in a nanopurification at the same time, which would further purify the semen. Nanopurification is easily adaptable across species; it is a cheap and effective way to ameliorate sperm quality within a semen dose [28][29].
In summary, selection methods based on gradient separation and nanopurification are cheap, simple, and modifiable. Both methods can be species-optimized and further improved with better filters/gradients or probes. Furthermore, both methods have been linked to better AI fertility and semen quality [22][23][24][25][28][29][32]. Either method can be used to improve semen quality and potentially rescue semen samples from sub-fertile males. Though these selection methods are relatively new, they are promising and will be further optimized in the near future.
5. Sperm Sexing
A specialized form of semen selection which has been greatly improved upon in the past decade and continues to be optimized is sperm sexing. This is a method of selection which targets either X or Y chromosome bearing spermatozoa and effectively “purifies” a sample to contain a high percentage of spermatozoa with the desired sex chromosome. The most popular method of sexing and the method which has been utilized for commercial application is known as the Beltsville Sperm Sexing Technology. This technology was commercialized in 2004 by Sexing Technologies and has been adopted for use in the cattle industry. It is most substantially used by the dairy cattle industry, where a heifer calf is of far greater value than a bull calf, as females are necessary to produce milk, the intended product of the dairy industry.
Sex-sorted semen is sorted using a modified high-speed flow cytometer, which separate spermatozoa based on DNA content [33]. The flow cytometer measures DNA content based off the DNA-binding fluorescent probe Hoechst 33342. Spermatozoa containing the male, Y, chromosome lacking one arm have measurably less fluorescence than the spermatozoa containing X chromosomes, thereby allowing the flow cytometer to sort the sperm into X and Y groups based on the amount of fluorescence that the spermatozoa emit when excited by a laser within the flow cytometer [33]. According to Sexing Technologies, they are able to sex cattle semen with 93% gender accuracy. Though sexed semen has been available for cattle producers for over a decade, this product is still in its infancy across all other livestock species. The first goat kids were born using sexed semen in 2013 [34]. As previously mentioned, the first small ruminant sexed semen company was launched as of August 2020, by Animal Breeding Europe and Cogent. The swine industry experimented with sexed semen earlier and produced the first sexed semen piglets in 1998 [35]. As previously mentioned, Sexing Technologies is actively attempting to commercialized sex boar semen; however, no swine-sexed semen products are available for routine use at this time [36]. Additionally, the cost of sexed semen is higher than that of conventional semen. Doses of sexed cattle semen cost anywhere from USD 15 to USD 30 US more per dose [37]. This increase in cost will be seen in sexed semen products for the other livestock species as well. Though sexed semen is only routinely used in cattle right now, this area of the industry is continuing to grow; the efficient sexing of spermatozoa in all species is of great interest for the livestock industry.
Currently, cattle-sexed semen has a fertility rate of 71.5% to 78.5% that of whole semen [38]. Though these reduced rates are acceptable for dairy producers and some beef producers to use sexed semen, fertility rates are still the foremost issue facing sexed semen. Part of the issue is the large amount of handling the sexed sperm undergoes, as well as high pressure, speed and shear force, and speed of sheath fluid carrying through the cell sorter. The sexing process involves nearly 30 steps of processing, and the spermatozoa are exposed to several different media through-out the processing [39]. Furthermore, the actual sexing, though becoming faster all the time, is still quite slow (100 to 200 million spermatozoa/hour). Increased handling and slow processing reduce the life span of spermatozoa and can result in damaged spermatozoa [40][41], which makes the stable storage of the spermatozoa more difficult.
6. Semen Storage
Liquid extenders are currently the most popular method for storing semen in all the livestock species besides cattle. Though liquid extenders are widely used, they leave much to be desired. Boar extenders have been a subject of extensive research to improve storage efficiency [42]. Both equine and small ruminant extenders are currently lagging behind and have a shorter shelf life than extended boar semen [43]. Many different extender supplementations have been attempted and have resulted in improvements [42]; however, the shelf life of liquid extended semen is still much shorter than frozen, or cryopreserved, semen. There is considerable effort being made to fine-tune the efficiency of freezing semen for all species. Though there are currently protocols that attempt to maximize the use of liquid stored sperm, successful semen cryopreservation protocols are a priority for all livestock species [44].
Liquid extenders include antibiotics, as oftentimes the storage environment (e.g., temperature, energy substrates, pH, etc.) support bacterial growth. Antibiotic resistance is an increasing concern. To date, there are primarily two ways around this. One is to make use of hypothermic storage as alluded to in the Horse section. In boar semen storage, hypothermic storage consists of storing extended semen at 4–5 °C [45][46]. This brings about added stress to sperm cells and the inclusion of additional compounds that allow the cells to survive at this temperature. Storage at this temperature helps decrease the ability of bacteria to multiply. Alternatively, others have suggested use of bacteriostatic agents in the plastic of dose containers. These bacteriostatic agents, in practice, do not kill the bacteria, but rather prevent them from multiplying. Follow up studies should compare these two methods.
Cryopreservation is more effective for the long-term storage of spermatozoa than liquid extender storage [47]. The main hurdle for cryopreservation of semen is the retention of acceptable viability and fertility from the semen dose upon thawing [42]. Cattle semen can be cryopreserved effectively using a standardized slow-freezing protocol [48]. This is not the case with other species. Boar spermatozoa in particular are very susceptible to cold shock [47], and as a result, the spermatozoa that undergo freezing are often weakened [49]. These weakened spermatozoa can suffer from DNA fragmentation, degradation of proteins and RNAs contained within the spermatozoa, disruption of the acrosome and sperm membranes, as well as reduction in mitochondrial activity and sperm motility [47]. All these issues can result in decreased fertility from a semen dose [49].
Freezing semen is further complicated by the variable tolerance to freezing between different sires and even different collections from the same sire [44]. This variability has prompted researchers to explore the possibility of biomarkers which can determine the ability of a particular sire or ejaculate to be frozen [50]. If such biomarkers were identified and used in conjunction with FC-based semen analysis and nanopurification, it would be feasible to devise a method to analyze and purify semen for maximum freezing potential, and even select sires with highest sperm cryotolerance.
Correlating freezability itself and these biomarkers to specific genes may help researchers further grasp the intricacies between variable tolerances to sperm freezing in males. Additionally, the practice of diagnosing semen samples’ cryotolerance and assessing the level of cryodamage after thawing will improve the outcomes of freezing in the non-cattle livestock species. Altogether, these approaches will further improve frozen semen within cattle AI also.
Sperm lyophilization/freeze-drying has been considered as an alternative storage method based on live births with lyophilized mouse and rabbit spermatozoa following the transfer of embryos conceived by intracytoplasmic sperm injection (ICSI) [51][52]. However, this approach may not be suitable for commercial embryo transfer in livestock species because of the high equipment and labor cost of ICSI. Furthermore, lyophilization is likely to cause sperm-borne centriole damage in non-rodent species (rodent spermatozoa do not carry this otherwise essential microtubule organizing organelle), and sperm DNA damage by lyophilization is a concern in all species [53]. Although some progress has been made toward understanding the centrosomal inheritance issue after sperm lyophilization in livestock species [54][55], DNA damage and poor developmental potential after freeze-dried ICSI are yet to be addressed.
Both liquid extenders and frozen semen protocols can benefit from improvements in analysis and selection technologies. As previously stated, much of the work carried out in this field of study revolves around the ingredients used within the extenders [42]; however, focusing on the spermatozoa which are actually extended or frozen is of importance. The previously discussed analysis and selection methods will produce semen samples with more robust spermatozoa capable of withstanding extension and freezing protocols better. Such results have already been shown using SLC selection protocols [26][27]. Improving semen storage will greatly benefit the AI industry and using more effective sperm analysis and selection methods will improve semen storage outcomes. Additional advances in frozen semen storage and distribution could stem from ongoing research on sperm cryoprotectant improvement and sperm preservation at temperatures above the temperature of liquid nitrogen-dependent sperm storage [56][57].
References
- Ahmed, J.; Lorch, J.; Ong, L.; Wolfgram, J. How the Global Supply Landscape for Meat Protein Will Evolve. Available online: https://www.mckinsey.com/industries/agriculture/our-insights/how-the-global-supply-landscape-for-meat-protein-will-evolve (accessed on 20 December 2020).
- Ombelet, W.; Van Robays, J. Artificial insemination history: Hurdles and milestones. Facts Views Vis. ObGyn 2015, 7, 137–143.
- Amann, R.P. Weaknesses in reports of “fertility” for horses and other species. Theriogenology 2005, 63, 698–715.
- Flowers, W. Factors Affecting the Efficient Production of Boar Sperm. Reprod. Domest. Anim. 2015, 50, 25–30.
- Sutovsky, P. New Approaches to Boar Semen Evaluation, Processing and Improvement. Reprod. Domest. Anim. 2015, 50 (Suppl. 2), 11–19.
- Farrell, P.; Trouern-Trend, V.; Foote, R.H.; Douglas-Hamilton, D. Repeatability of measurements on human, rabbit, and bull sperm by computer-assisted sperm analysis when comparing individual fields and means of 12 fields. Fertil. Steril. 1995, 64, 208–210.
- Sutovsky, P.; Aarabi, M.; Miranda-Vizuete, A.; Oko, R. Negative biomarker based male fertility evaluation: Sperm phenotypes associated with molecular-level anomalies. Asian J. Androl. 2015, 17, 554–560.
- Kerns, K.; Zigo, M.; Drobnis, E.Z.; Sutovsky, M.; Sutovsky, P. Zinc ion flux during mammalian sperm capacitation. Nat. Commun. 2018, 9, 2061.
- Kerns, K.; Sharif, M.; Zigo, M.; Xu, W.; Hamilton, L.E.; Sutovsky, M.; Ellersieck, M.; Drobnis, E.Z.; Bovin, N.; Oko, R.; et al. Sperm Cohort-Specific Zinc Signature Acquisition and Capacitation-Induced Zinc Flux Regulate Sperm-Oviduct and Sperm-Zona Pellucida Interactions. Int. J. Mol. Sci. 2020, 21, 2121.
- Sutovsky, P.; Lovercamp, K. Molecular markers of sperm quality. Biosci. Proc. 2019.
- Sutovsky, P.; Moreno, R.; Ramalho-Santos, J.; Dominko, T.; Thompson, W.E.; Schatten, G. A putative, ubiquitin-dependent mechanism for the recognition and elimination of defective spermatozoa in the mammalian epididymis. J. Cell Sci. 2001, 114, 1665–1675.
- Ibrahim, N.M.; Gilbert, G.R.; Loseth, K.J.; Crabo, B.G. Correlation between clusterin-positive spermatozoa determined by flow cytometry in bull semen and fertility. J. Androl. 2000, 21, 887–894.
- Christensen, P.; Hansen, C.; Liboriussen, T.; Lehn-Jensen, H. Implementation of flow cytometry for quality control in four Danish bull studs. Anim. Reprod. Sci. 2005, 85, 201–208.
- Hossain, S.; Johannisson, A.; Wallgren, M.; Nagy, S.; Siqueira, A.P.; Rodriguez-Martinez, H. Flow cytometry for the assessment of animal sperm integrity and functionality: State of the art. Asian J. Androl. 2011, 13, 406–419.
- Petrunkina, A.; Harrison, R. Fluorescence Technologies for Evaluating Male Gamete (Dys) Function. Reprod. Domest. Anim. 2013, 48, 11–24.
- Kerns, K.; Jankovitz, J.; Robinson, J.; Minton, A.; Kuster, C.; Sutovsky, P. Relationship between the Length of Sperm Tail Mitochondrial Sheath and Fertility Traits in Boars Used for Artificial Insemination. Antioxidants 2020, 9, 1033.
- Sutovsky, P.; Kerns, K.; Zigo, M.; Zuidema, D. Boar semen improvement through sperm capacitation management, with emphasis on zinc ion homeostasis. Theriogenology 2019, 137, 50–55.
- Kennedy, C.E.; Krieger, K.B.; Sutovsky, M.; Xu, W.; Vargovič, P.; Didion, B.A.; Ellersieck, M.R.; Hennessy, M.E.; Verstegen, J.; Oko, R.; et al. Protein expression pattern of PAWP in bull spermatozoa is associated with sperm quality and fertility following artificial insemination. Mol. Reprod. Dev. 2014, 81, 436–449.
- Sironen, A.; Uimari, P.; Nagy, S.; Paku, S.; Andersson, M.; Vilkki, J. Knobbed acrosome defect is associated with a region containing the genes STK17b and HECW2 on porcine chromosome 15. BMC Genom. 2010, 11, 699.
- Rubessa, M.; Feugang, J.M.; Kandel, M.E.; Schreiber, S.; Hessee, J.; Salerno, F.; Meyers, S.; Chu, I.; Popescu, G.; Wheeler, M.B. High-throughput sperm assay using label-free microscopy: Morphometric comparison between different sperm structures of boar and stallion spermatozoa. Anim. Reprod. Sci. 2020, 219, 106509.
- Jane, M.; Morrell, H.R.-M. Colloid Centrifugation of Semen: Applications in Assisted Reproduction. Am. J. Anal. Chem. 2016, 7, 597–610.
- Sjunnesson, Y.C.B.; Morrell, J.M.; Gonzalez, R. Single layer centrifugation-selected boar spermatozoa are capable of fertilization in vitro. Acta Veter-Scand. 2013, 55, 20.
- Šterbenc, N.; Morrell, J.M.; Kosec, M.; Rath, D.; Klein, S.; Klinc, P. Single layer colloid centrifugation technique improves motility, viability and chromatin integrity of ram spermatozoa after thawing. Cryobiology 2018, 86, 77–83.
- Morrell, J.; Rodriguez-Martinez, H.; Andersson, M. Colloid Centrifugation Selects Normal Spermatozoa from Polymorphic Bull Ejaculates: A Case Study. Reprod. Domest. Anim. 2014, 49, 281–284.
- Morrell, J.; Richter, J.; Martinsson, G.; Stuhtmann, G.; Hoogewijs, M.; Roels, K.; Dalin, A.-M. Pregnancy rates after artificial insemination with cooled stallion spermatozoa either with or without Single Layer Centrifugation. Theriogenology 2014, 82, 1102–1105.
- Hoogewijs, M.; Morrell, J.; Van Soom, A.; Govaere, J.; Johannisson, A.; Piepers, S.; De Schauwer, C.; De Kruif, A.; De Vliegher, S. Sperm selection using single layer centrifugation prior to cryopreservation can increase thawed sperm quality in stallions. Equine Veter-J. 2011, 43, 35–41.
- Martinez-Alborcia, M.; Morrell, J.; Parrilla, I.; Barranco, I.; Vazquez, J.M.; Martinez, E.A.; Roca, J. Improvement of boar sperm cryosurvival by using single-layer colloid centrifugation prior freezing. Theriogenology 2012, 78, 1117–1125.
- Odhiambo, J.; DeJarnette, J.; Geary, T.W.; Kennedy, C.E.; Suarez, S.S.; Sutovsky, M.; Sutovsky, P. Increased Conception Rates in Beef Cattle Inseminated with Nanopurified Bull Semen1. Biol. Reprod. 2014, 91, 97.
- Durfey, C.L.; Swistek, S.E.; Liao, S.F.; Crenshaw, M.A.; Clemente, H.J.; Thirumalai, R.V.K.G.; Steadman, C.S.; Ryan, P.L.; Willard, S.T.; Feugang, J.M. Nanotechnology-based approach for safer enrichment of semen with best spermatozoa. J. Anim. Sci. Biotechnol. 2019, 10, 14.
- Feugang, J.M.; Rhoads, C.E.; Mustapha, P.A.; Tardif, S.; Parrish, J.J.; Willard, S.T.; Ryan, P.L. Treatment of boar sperm with nanoparticles for improved fertility. Theriogenology 2019, 137, 75–81.
- MCRM Fertility. Sperm Selection. Available online: https://www.mcrmfertility.com/treatment-options/in-vitro-fertilization-ivf/sperm-selection/#nanobead (accessed on 1 January 2021).
- Morrell, J. Effect of colloid centrifugation on boar sperm quality during storage and function in in vitro fertilization. Theriogenology 2019, 137, 122–126.
- Johnson, L.; Welch, G. Sex preselection: High-speed flow cytometric sorting of X and Y sperm for maximum efficiency. Theriogenology 1999, 52, 1323–1341.
- Bathgate, R.; Mace, N.; Heasman, K.; Evans, G.; Maxwell, W.; De Graaf, S. Birth of Kids after Artificial Insemination with Sex-Sorted, Frozen-Thawed Goat Spermatozoa. Reprod. Domest. Anim. 2013, 48, 893–898.
- Abeydeera, L.; Johnson, L.; Welch, G.; Wang, W.; Boquest, A.; Cantley, T.; Rieke, A.; Day, B. Birth of piglets preselected for gender following in vitro fertilization of in vitro matured pig oocytes by X and Y chromosome bearing spermatozoa sorted by high speed flow cytometry. Theriogenology 1998, 50, 981–988.
- Pig Progress. Producing Sex-Sorted Sperm with 99% Accuracy. Available online: https://www.pigprogress.net/Sows/Articles/2017/4/Producing-sex-sorted-sperm-with-99-accuracy-120805E/ (accessed on 14 April 2017).
- Thomas, J.; Locke, J.; Vishwanath, R.; Hall, J.; Ellersieck, M.; Smith, M.; Patterson, D. Effective use of SexedULTRA™ sex-sorted semen for timed artificial insemination of beef heifers. Theriogenology 2017, 98, 88–93.
- Joezy-Shekalgorabi, S.; Maghsoudi, A.; Mansourian, M.R. Reproductive performance of sexed versus conventional semen in Holstein heifers in various semiarid regions of Iran. Ital. J. Anim. Sci. 2017, 16, 666–672.
- Brito, L.; Ramakrishnan, V.; Heuer, C.; Evans, K. Bovine sexed semen production and utilization. Clin. Theriogenol. 2019, 11, 297–315.
- Gosálvez, J.; Ramirez, M.; López-Fernández, C.; Crespo, F.; Evans, K.; Kjelland, M.; Moreno, J. Sex-sorted bovine spermatozoa and DNA damage: I. Static features. Theriogenology 2011, 75, 197–205.
- Gosálvez, J.; Ramirez, M.; López-Fernández, C.; Crespo, F.; Evans, K.; Kjelland, M.; Moreno, J. Sex-sorted bovine spermatozoa and DNA damage: II. Dynamic features. Theriogenology 2011, 75, 206–211.
- Yeste, M. State-of-the-art of boar sperm preservation in liquid and frozen state. Anim. Reprod. 2017, 14, 69–81.
- Morrell, J.M. Artificial Insemination: Current and Future Trends. In Artificial Insemination in Farm Animals; InTech: London, UK, 2011.
- Rodriquez-Martinez, H. Sperm biotechnologies in domestic species: State of the art. Anim. Reprod. 2013, 10, 268–276.
- Jäkel, H.; Henning, H.; Luther, A.; Rohn, K.; Waberski, D. Assessment of chilling injury in hypothermic stored boar spermatozoa by multicolor flow cytometry. Cytom. Part A 2021, 99, 1033–1041.
- Jäkel, H.; Scheinpflug, K.; Mühldorfer, K.; Gianluppi, R.; Lucca, M.S.; Mellagi, A.P.G.; Bortolozzo, F.P.; Waberski, D. In vitro performance and in vivo fertility of antibiotic-free preserved boar semen stored at 5 °C. J. Anim. Sci. Biotechnol. 2021, 12, 9.
- Yeste, M. Sperm cryopreservation update: Cryodamage, markers, and factors affecting the sperm freezability in pigs. Theriogenology 2016, 85, 47–64.
- Rodriguez-Martinez, H.; Barth, A.D. In vitro evaluation of sperm quality related to in vivo function and fertility. Soc. Reprod. Fertil. Suppl. 2019, 64, 39.
- Rodriguez-Martinez, H. Cryopreservation of Porcine Gametes, Embryos and Genital Tissues: State of the Art; InTech: London, UK, 2012; pp. 231–260.
- Ugur, M.R.; Saber Abdelrahman, A.; Evans, H.C.; Gilmore, A.A.; Hitit, M.; Arifiantini, R.I.; Purwantara, B.; Kaya, A.; Memili, E. Advances in Cryopreservation of Bull Sperm. Front. Vet. Sci. 2019, 6, 268.
- Liu, J.-L.; Kusakabe, H.; Chang, C.-C.; Suzuki, H.; Schmidt, D.W.; Julian, M.; Pfeffer, R.; Bormann, C.L.; Tian, X.C.; Yanagimachi, R.; et al. Freeze-Dried Sperm Fertilization Leads to Full-Term Development in Rabbits1. Biol. Reprod. 2004, 70, 1776–1781.
- Wakayama, T.; Yanagimachi, R. Development of normal mice from oocytes injected with freeze-dried spermatozoa. Nat. Biotechnol. 1998, 16, 639–641.
- Palazzese, L.; Gosalvez, J.; Anzalone, D.A.; Loi, P.; Saragusty, J. DNA fragmentation in epididymal freeze-dried ram spermatozoa impairs embryo development. J. Reprod. Dev. 2018, 64, 393–400.
- Hara, H.; Abdalla, H.; Morita, H.; Kuwayama, M.; Hirabayashi, M.; Hochi, S. Procedure for Bovine ICSI, not Sperm Freeze-drying, Impairs the Function of the Microtubule-organizing Center. J. Reprod. Dev. 2011, 57, 428–432.
- Ressaissi, Y.; Anzalone, D.A.; Palazzese, L.; Czernik, M.; Loi, P. The impaired development of sheep ICSI derived embryos is not related to centriole dysfunction. Theriogenology 2020, 159, 7–12.
- Grötter, L.G.; Cattaneo, L.; Marini, P.E.; Kjelland, M.E.; Ferré, L.B. Recent advances in bovine sperm cryopreservation techniques with a focus on sperm post-thaw quality optimization. Reprod. Domest. Anim. 2019, 54, 655–665.
- Kuiper, M.; Spencer, M.; Kanyima, B.M.; Ng, C.H.; Newell, M.; Turyahikayo, S.; Makoni, N.; Madan, D.; Lieberman, D.H. Using on-demand dry ice production as an alternative cryogenic cold chain for bovine artificial insemination outreach in low-resource settings1. Transl. Anim. Sci. 2020, 4, 1196–1205.
More
Information
Subjects:
Agriculture, Dairy & Animal Science
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
857
Revisions:
2 times
(View History)
Update Date:
22 Dec 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





