
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Qi Li | + 1088 word(s) | 1088 | 2021-12-09 08:52:23 | | | |
| 2 | Yvaine Wei | -4 word(s) | 1084 | 2021-12-17 10:18:56 | | |
Video Upload Options
Flavor is the main quality characteristic for beer and requires the flavor-active components presented as raw materials or developed by yeast metabolism to be controlled within a certain range in order to maintain flavor balance.
1. Introduction
During the shelf life of beer, numerous reactions take place, resulting in a decrease of fresh flavor notes and the appearance of typical aged flavors [1][2][3]. Hence, improving the flavor stability of beer during its shelf life is of great concern for brewers as it is important for a commercial beer to have a consistent sensory experience and satisfy the expectations of consumers at all times.
Aldehydes such as (E)-2-nonenal, 5-hydroxymethylfurfural, hexanal, and acetaldehyde are characterized as the aged flavor components in beer, and higher concentrations of these aldehydes greatly impair flavor stability [4][5][6]. Thus, many strategies have been applied to reduce the production of these aldehydes during industrial manufacturing, for example, reducing the aeration levels of wort, limiting the time of boiling and cooling, and increasing the inoculation volume of cells [4]. Notably, the natural reducing activity of yeast can also facilitate the reduction of these components [7].
There are many powerful methods to create microbial mutations, of which atmospheric and room temperature plasma (ARTP) mutagenesis is the new developed technology with the advantages of high efficiency, consistency, nontoxicity, environmental-friendliness, and low cost [8][9]. Therefore, generating non-genetically modified mutant yeast cells with this method would be a good choice. In addition, prior findings suggested that the uncoupler 2,4-dinitrophenol (DNP) could block nicotinamide adenine dinucleotide hydride (NADH) oxidation and affect cell growth [10][11]; therefore, it might be a promising selective marker for NADH perturbation varients.
2. DNP Serves as a Selective Marker for the NADH Perturbation Variants
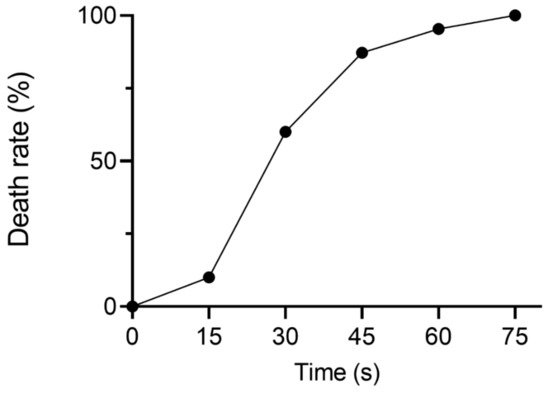
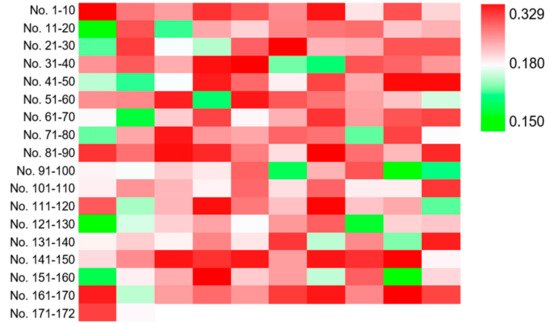
3. Screening the Optimal Strain with Industrial Potential
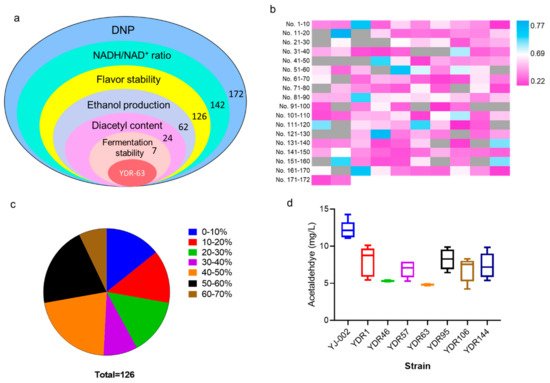
4. Screening Strains with Hgher NADH Levels
References
- Vanderhaegen, B.; Neven He Verachtert, H.; Derdelinckx, G. The chemistry of beer aging—A critical review. Food Chem. 2006, 95, 357–381.
- Gil, M.I.; Tomás-Barberán, F.A.; Hess-Pierce, B.; Holcroft, D.M.; Kader, A.A. Antioxidant activity of pomegranate juice and its relationship with phenolic composition and processing. J. Agric. Food Chem. 2000, 48, 4581–4589.
- Rakete, S.; Klaus, A.; Glomb, M.A. Investigations on the Maillard reaction of dextrins during aging of Pilsner type beer. J. Agric. Food Chem. 2014, 62, 9876–9884.
- Baert, J.J.; De Clippeleer, J.; Hughes, P.S.; De Cooman, L.; Aerts, G. On the origin of free and bound staling aldehydes in beer. J. Agric. Food Chem. 2012, 60, 11449–11472.
- Bustillo Trueba, P.; Jaskula-Goiris, B.; Ditrych, M.; Filipowska, W.; De Brabanter, J.; De Rouck, G.; De Clippeleer, J. Monitoring the evolution of free and cysteinylated aldehydes from malt to fresh and forced aged beer. Food Res. Int. 2021, 140, 110049.
- Saison, D.; Schutter, D.; Uyttenhove, B.; Delvaux, F.; Delvaux, F.R. Contribution of staling compounds to the aged flavour of lager beer by studying their flavour thresholds. Food Chem. 2009, 114, 1206–1215.
- Saison, D.; De Schutter, D.P.; Vanbeneden, N.; Daenen, L.; Delvaux, F.; Delvaux, F.R. Decrease of aged beer aroma by the reducing activity of brewing yeast. J. Agric. Food Chem. 2010, 58, 3107–3115.
- Lv, Q.; Hu, M.; Tian, L.; Liu, F.; Wang, Q.; Xu, M.; Rao, Z. Enhancing l-glutamine production in Corynebacterium glutamicum by rational metabolic engineering combined with a two-stage pH control strategy. Bioresour. Technol. 2021, 341, 125799.
- Li, J.; Guo, S.; Hua, Q.; Hu, F. Improved AP-3 production through combined ARTP mutagenesis, fermentation optimization, and subsequent genome shuffling. Biotechnol. Lett. 2021, 43, 1143–1154.
- Grundlingh, J.; Dargan, P.I.; El-Zanfaly, M.; Wood, D.M. 2,4-dinitrophenol (DNP): A weight loss agent with significant acute toxicity and risk of death. J. Med. Toxicol. 2011, 7, 205–212.
- Ludwig, N.; Yerneni, S.S.; Menshikova, E.V.; Gillespie, D.G.; Jackson, E.K.; Whiteside, T.L. Simultaneous inhibition of glycolysis and oxidative phosphorylation triggers a multi-fold increase in secretion of exosomes: Possible Role of 2′3′-cAMP. Sci. Rep. 2020, 10, 6948.
- Lei, H.; Feng, L.; Peng, F.; Xu, H. Amino acid supplementations enhance the stress resistance and fermentation performance of lager yeast during high gravity fermentation. Appl. Biochem. Biotechnol. 2019, 187, 540–555.
- Piddocke, M.P.; Kreisz, S.; Heldt-Hansen, H.P.; Nielsen, K.F.; Olsson, L. Physiological characterization of brewer’s yeast in high-gravity beer fermentations with glucose or maltose syrups as adjuncts. Appl. Microbiol. Biotechnol. 2009, 84, 453–464.
- Xu, X.; Song, Y.; Guo, L.; Cheng, W.; Niu, C.; Wang, J.; Li, Q. Higher NADH availability of lager yeast increases the flavor stability of beer. J. Agric. Food Chem. 2020, 68, 584–590.
- Wang, J.; Shen, N.; Yin, H.; Liu, C.; Li, Y.; Li, Q. Development of industrial brewing yeast with low acetaldehyde production and improved flavor stability. Appl. Biochem. Biotechnol. 2013, 169, 1016–1025.
- Liu, C.; Li, Q.; Niu, C.; Tian, Y.; Zhao, Y.; Yin, X. The use of atmospheric and room temperature plasma mutagenesis to create a brewing yeast with reduced acetaldehyde production. J. Inst. Brew. 2018, 124, 236–243.
- Shen, N.; Wang, J.; Liu, C.; Li, Y.; Li, Q. Domesticating brewing yeast for decreasing acetaldehyde production and improving beer flavor stability. Eur. Food Res. Technol. 2014, 238, 347–355.
- Bamforth, C.W. A critical control point analysis for flavor stability of beer. Tech. Q. Mbaa Commun. 2004, 14, 97–103.
- Yasui, T.; Matsuura, C.; Hori, T.; Ogawa, Y.; Ohkochi, M. Importance of raw materials and the wort production process on trans-2-nonenal formation in beer. In EBC Symposium “Flavour and Flavour Stability”; Fachverlag Hans Carl: Nuürnberg, Germany, 2001; Volume 8, pp. 1–12.
- Issekutz, B., Jr. Effect of propranolol in dinitrophenol poisoning. Arch. Int. Pharm. 1984, 272, 310–319.
- Rognstad, R.; Katz, J. The effect of 2,4-dinitrophenol on adipose-tissue metabolism. Biochem. J. 1969, 111, 431–444.
- Colman, E. Dinitrophenol and obesity: An early twentieth-century regulatory dilemma. Regul. Toxicol. Pharm. 2007, 48, 115–117.





