
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Ivan Alekseev | + 3072 word(s) | 3072 | 2020-08-13 07:34:41 | | | |
| 2 | Rita Xu | -792 word(s) | 2280 | 2020-08-19 05:41:17 | | |
Video Upload Options
Although ice-free areas cover only about 0.1% of Antarctica and are characterized by harsh environmental conditions, these regions provide quite diverse conditions for the soil-forming process, having various physical and geochemical properties, and also assuring different conditions for living organisms. This study is aimed to determine existing soil microbial communities, their relationship with soil parameters and the influence of anthropogenic activity in Larsemann Hills, Eastern Antarctica. The soil microbiome was investigated at different locations using 16S rRNA gene pyrosequencing. The taxonomic analysis of the soil microbiomes revealed 12 predominant bacterial and archaeal phyla—Proteobacteria, Actinobacteria, Acidobacteria, Chloroflexi, Gemmatimonadetes, Verrucomicrobia, Planctomycetes, Bacteroidetes, Armatimonadetes, Firmicutes, Cyanobacteria, Thaumarchaeota. Some specific phyla have been also found in sub-surface horizons of soils investigated, thus providing additional evidence of the crucial role of gravel pavement in saving the favorable conditions for both soil and microbiome development. Moreover, our study also revealed that some bacterial species might be introduced into Antarctic soils by human activities. We also assessed the effect of different soil parameters on microbial community in the harsh environmental conditions of Eastern Antarctica. pH, carbon and nitrogen, as well as fine earth content, were revealed as the most accurate predictors of soil bacterial community composition.
1. Introduction
Although ice-free areas cover only about 0.1% of Antarctica, these regions provide quite diverse conditions for the soil-forming process, having various physical and geochemical properties, and also providing different conditions for living organisms. In Antarctica, microbial communities are generally the dominant biomass component of terrestrial ecosystems, also they control most of the biological flux of carbon, nutrients and energy [1][2]. Soil is an important component of Antarctic ecosystems, since it determines their sustainability and serves as a habitat for living organisms. Soil microbiomes play an essential role in the development of soil profiles and the implementation of soil biochemical processes. Soil genesis is closely related to the activity and development of its microbiome [3]. Microorganisms play a key role in ensuring the cycling of the main nutrients during the decomposition of organic material and in the formation of organic matter in the soil.
Antarctica weakly obeys the general geographic law of latitudinal zoning. Remote ice-free areas (oases) are isolated from each other and have no biological or even climatic connection, so they are more like islands in the ocean. According to Bockheim and Hall [4], three climatic zones can be distinguished in Antarctica: Subantarctic (including the South Shetland Islands), Antarctic coastal and Antarctic continental zones.
Eastern Antarctica oases are characterized by climatic extremality, which in turn determines the specificity of soil formation. Acute lack of moisture, ultraviolet radiation, sharp temperature changes and strong winds significantly reduce the primary production of organic matter and the formation of organogenic horizons on the surfaces of loose and rocky substrates that prevail in Eastern Antarctica oases [5]. An important feature of the landscapes of the oases is that most of the biomass is concentrated beneath the mineral surface, which greatly changes the vertical organization of the soil profile. In these extreme habitats, the leading role in soil formation belongs to the most adapted fungi and bacteria [6].
Investigation of microbial communities in extreme environments have been actively discussed in recent decades. Many researchers indicated a significant diversity of microorganisms in Antarctic soils and rocks [1][7][8][9] Special attention has been given to investigation of cryptoendolithic systems to reveal fungal and bacterial community structure in harsh environments of different regions of Antarctica [10][11][12][13]. Previous studies of microbial communities in soils have been mainly conducted in the Transantarctic region of the Victoria Land, Antarctic Peninsula and surrounding Islands. According to these investigations, microbial communities in Western Antarctica are dominated by several bacterial phyla, such as Proteobacteria, Bacteroidetes, Actinobacteria, Cyanobacteria, Verrucomicrobia, Thermotogae, Fibrobacteres, Deinococcus-Thermus and Chlorobi [14][15][16][17]. At the same time, the microbial communities of Eastern Antarctica coastal oases remain less investigated. Moreover, relatively little attention has been shown towards soil microbial communities, although they could serve to enhance our knowledge on life adaptation strategies under such harsh environmental conditions. It has been shown that soil bacterial communities in Eastern Antarctica contain a significant amount of small filtering bacterial forms, which is considered as an adaptation to the harsh climatic conditions of Antarctica [6][8]. A role of human impact on soil bacterial diversity in Eastern Antarctica has been rarely investigated as well. Previously, anthropogenic factor were found to affect both soil geochemical properties and soil bacterial diversity from sites around Casey Station [7]. Nowadays, anthropogenic impact on the natural ecosystems of both Western and Eastern Antarctica is expressed not only in contamination, but also in various physical disturbances, such as soil over-compaction caused by mechanical impact, soil surface consumption by a waste disposal and changes in thermal regime (especially in the areas of polar stations and their surroundings).
The human disturbance footprint for the entire Antarctic continent has recently been calculated [18][19][20]. Effective realization of environmental protection measures is vital for saving unique ecosystems of the sixth continent as well as for appropriate compliance of the Antarctic Treaty’s fundamental principles. These measures include environmental impact assessment and permanent monitoring of various components in natural ecosystems, such as soil. Due to the unique geological structure, biological diversity and increasing rates of human activity (e.g., tourism activity, chemical contamination and physical disturbance of surface and permafrost), the Antarctic Specially Managed Area (ASMA-6) has been designated in the Larsemann Hills. Additionally, the Antarctic Specially Protected Area (ASPA № 174) has been established in the area of Stornes Peninsula to “protect the outstanding geological features of this area, specifically the rare mineral occurrences and the highly unusual host rocks in which they occur” [21].
It should be concluded that soil microbiological studies in Antarctica are still lacking in regard to investigation of taxonomic composition and functional diversity of microbiome. Although some research revealed the Antarctic microbiome as unique [22][23], other studies showed microbiome composition similar to other regions of the Earth [15][24]. Moreover, increased rates of anthropogenic forcing (tourism activities, logistic load, functioning of the numerous scientific stations) require more attention to be paid, with specific studies dedicated to revealing the possible changes in microbiome due to anthropogenic transformation. Modern pyrosequencing techniques allow not only to identify the culturable bacteria in various Antarctic environments, but also delineate the wide spectra of unculturable bacteria associated with humans. In this respect, using the next-generation sequencing techniques to study the microbiome of soils in vicinities of Antarctic stations are especially relevant, since these data are still poor [25].
Moreover, comprehensive knowledge of the microbial community is needed for studying the fundamental problem of mineral substrate transformation under extreme climatic conditions of Antarctica. This is especially relevant in regards to studying the initial soil-forming process and finding the modern analogues of Precambrian soils [26].
2. Soil Physical–Chemical Properties
The main physical–chemical parameters of investigated soils are summarized in Table 1. Values of pH in all studied soils were moderately acidic to slightly neutral. This is due to the absence of the salt accumulation from the ocean acid to the neutral nature of parent materials. The soils investigated contained different amounts of total organic carbon (TOC), which ranged from 0.54 to 1.64%, being the highest in sub-surface algal–mineral and moss–algal–mineral horizons in natural sites. Therefore, we can summarize that soils of Larsemann Hills have different levels of carbon and nitrogen accumulation, being lower than reported for Maritime Antarctica [27][28][29][30]. Additionally, carbon to nitrogen (C:N) ratio varied between 10.48 and 16.33. A narrow range observed in some topsoil horizons could be explained by the presence of well-decomposed organic matter (mainly peat material). This has been also described for soils in Eastern Antarctica previously [26]. Basal respiration (BR) rates ranged from 0.006 ± 0.001 to 0.150 ± 0.010 mg g−1 day−1, which is in agreement with results previously published for coarse soils of Antarctica [31][32]. Higher values of BR were observed at sub-surface horizons with hypolithic algae–bacterial communities. However, it should be noted that higher basal respiration rates could be found in soils under strong influence of birds (penguin rockeries, nesting sites of penguins and flying birds), which were described previously for both Western and Eastern Antarctica [33][34][35]. The percentage of fine earth ranged between 18.99 to 31.98% in studied soils of Larsemann Hills, with highest values in sub-surface horizon with more favorable conditions for fine earth formation.
Table 1. Soil physical–chemical parameters ± SD.
| Site | pHH2O | pHKCl | TOC, % | N, % | C:N | Basal Respiration, mg g−1 day−1 | Fine Earth (<2 mm), % |
|---|---|---|---|---|---|---|---|
| Anthropogenically-Affected Sites | |||||||
| Progress 1 | 5.85 ± 0.53 | 5.23 ± 0.42 | 0.87 ± 0.10 | 0.083± 0.005 | 10.48 ± 1.27 | 0.009 ± 0.001 | 18.99 ± 1.23 |
| Progress 2 | 5.32 ± 0.56 | 4.89± 0.32 | 0.76 ±0.16 | 0.062 ±0.005 | 12.26 ± 2.09 | 0.008 ± 0.001 | 21.91 ± 1.97 |
| Near-Natural Sites | |||||||
| Lars 1 (0–1 cm) |
6.42 ± 0.56 | 5.76 ± 0.45 | 0.89 ± 0.12 | 0.08 ± 0.010 | 11.13 ± 1.56 | 0.010 ± 0.002 | 21.78 ± 1.86 |
| Lars 1 (1–5 cm) |
6.01 ±0.43 | 5.54 ± 0.31 | 1.50 ± 0.12 | 0.12± 0.01 | 12.50 ± 1.23 | 0.101 ± 0.006 | 31.98 ± 1.98 |
| Lars 2 (0–1 cm) |
6.21 ± 0.42 | 5.86 ± 0.23 | 0.98 ± 0.12 | 0.06 ± 0.01 | 16.33 ± 1.86 | 0.009 ± 0.001 | 22.03 ± 1.54 |
| Lars 2 (1–5 cm) |
5.98 ± 0.32 | 5.54 ± 0.27 | 1.65 ± 0.17 | 0.15 ± 0.03 | 10.84 ± 0.87 | 0.150 ± 0.010 | 29.87 ± 1.50 |
| Lars 2 (15–25 cm) |
6.03 ± 0.24 | 5.65 ± 0.23 | 0.54 ± 0.08 | 0.04 ± 0.01 | 13.50 ± 1.24 | 0.006 ± 0.001 | 19.87 ± 1.65 |
| Lars 3 | 6.04 ± 0.21 | 5.76 ± 0.21 | 0.87 ± 0.10 | 0.08 ± 0.02 | 10.88 ± 1.26 | 0.007 ± 0.001 | 21.09 ± 1.78 |
The main limiting factor for soil formation in Eastern Antarctica oases is the lack of moisture. Melting snow in the summer months is the determining regulator of bio- and pedological diversity [5][36]. In conditions of very low liquid precipitation, low air humidity existence of living organisms is supported mostly by melting water from snow patches. However, usually soil parameters of Eastern Antarctica do not show any clear trend in profile distribution and could be largely affected by microclimate effect and local-scale variability [37].
3. Microbiome Composition of Studied Soils
The taxonomic analysis of the soil microbiomes revealed 12 major bacterial and archaeal phyla-Proteobacteria, Actinobacteria, Acidobacteria, Chloroflexi, Gemmatimonadetes, Verrucomicrobia, Planctomycetes, Bacteroidetes, Armatimonadetes, Firmicutes, Cyanobacteria and Thaumarchaeota, which constituted the majority (>96% of sequences in amplicon libraries) (Figure 1). The rest of the identified phylum accounted for less than 0.01% of the total.
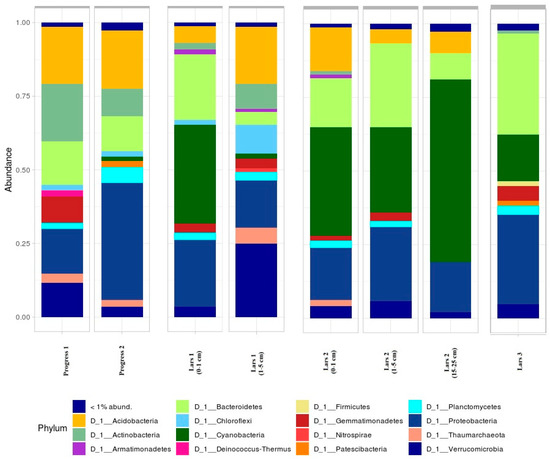
Figure 1. Taxonomic composition at phylum level of soil microbial communities of investigated soil samples.
To evaluate the alpha-diversity of the soil microbiomes, OTUs were identified and several indices (Shannon, Simpson and Phylogenetic diversity) were calculated (Table 2). For OTUs that significantly differed between the samples, further taxonomic identification using the RDP and GenBank databases were conducted. Only the OTUs that had more than 98% identity to the nearest homologous sequence in GenBank (the sequences with known taxonomic names were used) were included in the analysis. A significant proportion of these OTUs were homologous to species found in polar environments (both in Arctic and Antarctic regions), among which were samples from glacial forefield in Larsemann Hills [22] and Fildes Peninsula, King George Island [25].
Table 4. Alpha-diversity parameters of investigated soils microbiomes.
| Site | OTUs | PD_Whole_Tree | Simpson Index | Shannon Index |
|---|---|---|---|---|
| Anthropogenically-Affected Sites | ||||
| Progress 1 | 504 ± 32 | 40 ± 3.5 | 0.985 ± 0.12 | 5.02 ± 0.78 |
| Progress 2 | 760 ± 28 | 58 ± 4.1 | 0.981 ± 0.15 | 5.31 ± 0.85 |
| Near-Natural Sites | ||||
| Lars 1 (0–1 cm) | 566 ± 26 | 45 ± 3.5 | 0.984 ± 0.17 | 5.10 ± 1.02 |
| Lars 1 (1–5 cm) | 513 ± 25 | 46 ± 5.2 | 0.967 ± 0.11 | 4.85 ± 0.65 |
| Lars 2 (0–1 cm) | 476 ± 22 | 40 ± 4.2 | 0.981 ± 0.14 | 4.85 ± 0.98 |
| Lars 2 (1–5 cm) | 590 ± 29 | 46 ± 6.1 | 0.959 ± 0.15 | 4.75 ± 0.76 |
| Lars 2 (15–25 cm) | 310 ± 21 | 28 ± 5.3 | 0.945 ± 0.23 | 4.02 ± 0.73 |
| Lars 3 | 403 ± 22 | 41.5 ± 4.5 | 0.979 ± 0.25 | 4.63 ± 0.56 |
OTUs number varied significantly among studied soil samples (from 310 in transitional horizon of Lars 2 site to 760 in surface Progress 2 sample). Shannon’s index mean values ranged from 4.02 to 5.31 in studied samples. Similar values has been reported previously for different glacier forefield areas of Larsemann Hills [22]. Lower values of Shannon’s index have been observed in soils (3.3) and endoliths (2.8) of McKelvey Valley, McMurdo Valley [38]. Other investigations on endolithic microbial communities also reported lower values of this index [39][40]. However, significantly higher values of the Shannon’s diversity index have been reported for the Maritime Antarctica soil microbiome [25].
The highest diversity was found in the Progress 2 sample, which corresponds to the topsoil from the field base facilities. The soil surface is experiencing an active physical disturbance during the summer season, when numerous scientific operations are carried out. The site is also affected by vehicles since the site is used for logistical support for Progress airfield. High alpha-diversity at this site might be also caused by high amount of available moisture, since the site is located in wet valley accumulating water from melting snow patches and precipitation. The highest phylogenetic diversity (PD_tree) was also found in topsoil sample from Progress 2 site. Therefore, we can suppose that the higher Shannon index here is caused by anthropogenic inputs. Simpson’s diversity index varied less significantly among studied soil samples (from 0.945 in Lars 2 transitional horizon to 0.985 in topsoil from anthropogenically-affected site at Progress station). These values are higher compared to previously reported data reported for different endolith types from the McKelvey Valley [38], Miers Valley [39], different locations in McMurdo Valleys [40] as well as for soils of the glacier forefield in Larsemann Hills [22].
A clear trend of decreased alpha-diversity in deep soil horizons (where no features of macroscopic life is observed) was revealed for near-natural sites. The lowest alpha-diversity values were observed for transitional BC horizons of Lars 2 profile (Figure 2). However, this is not always true for soil profiles in Eastern Antarctica—especially in bottoms of wet valleys, where cryogenic processes (cryoturbation, patterned ground formation, frost sorting) are very active. A characteristic feature of Eastern Antarctica—presence of life mainly in sub-surface soil layers, which are not under direct influence of harsh conditions (UV-radiation, strong winds, low temperatures)—lead to heterogeneity of profile distribution of soil organisms. The mineral substrate in soils serves as a protector for microorganisms.
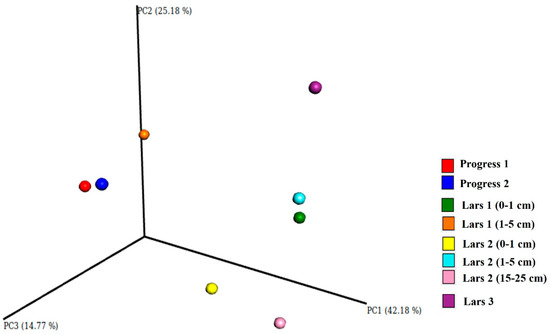
Figure 2. PCoA analysis of weighted UniFrac distance matrix representing β-diversity of the prokaryotic communities. PC1, PC2 and PC3 are the main coordinate axes of the multidimensional space, with the values of the explained variance (percentage).
In general, soils of sub-surface layers at both anthropogenic and near-natural sites showed greater evenness and richness compared to surface and deep soil horizons. At the same time, the area under anthropogenic influence (Progress 2 site) showed the highest richness, which indicates that human impact could lead to introducing some bacterial species into Antarctic soils. Ordination analyses of weighted UniFrac distances for bacteria supported these trends in alpha diversity (Figure 2).
The content of microorganisms in soils of Eastern Antarctica was previously counted [6]. Authors investigated microbial biomass in soils of Thala and Larsemann Hills and noticed a significant amount of bacterial filtering forms, which by their small size might indicate an adaptation to severe environmental conditions of Eastern Antarctica coastal oases. It was also reported less biomass in soils of the Thala Hills, which were caused by less developed moss cover on the soil surface. This coincides with our data of higher bacteria abundance in soils with developed moss cushion under gravel pavement in near-natural profiles, where sub-surface horizons were examined.
References
- Vincent, W. Evolutionary origins of Antarctic microbiota: Invasion, selection, and endemism. Antarct. Sci. 2000, 12, 374–385, doi:10.1017/S0954102000000420.
- Wynn-Williams, D. Antarctic microbial diversity: The basis of polar ecosystem processes. Biodivers. Conserv. 1996, 5, 1271–1293.
- Wolińska, A.; Gałazka, A.; Kuzniar, A.; Goraj, W.; Jastrzębska, N.; Grzadziel, J.; Stepniewska, Z. Catabolic fingerprinting and diversity of bacteria in Mollic Gleysol contaminated with petroleum substances. Appl. Sci. 2018, 8, 1–12.
- Bockheim, J.; Hall, K. Permafrost, active-layer dynamics and periglacial environments of continental Antarctica. S. Afr. J. Sci. 2002, 98, 82–90.
- Mergelov, N.; Konyushkov, D.; Lupachev, A.; Goryachkin, S. Soils of Mac Robertson land. In The Soils of Antarctica; Bockheim, G., Ed.; Springer International Publishing: Cham, Switzerland, 2015, pp. 65–86, doi:10.1007/978-3-319-05497-1-5.
- Lysak, V.; Maksimova, I.A.; Nikitin, D.A.; Ivanova, A.E.; Kudinova, A.G.; Soina, V.S.; Marfenina, O.E. Soil microbial communities of eastern Antarctica. Mosc. Univ. Biol. Sci. Bull. 2018, 73, 104–112, doi:10.3103/s0096392518030124.
- Friedman, E.I. Endolithic microorganisms in the Antarctic cold desert. Science 1982, 215, 1045–1053.
- Kudinova, A.G.; Lysak, L.V.; Soina, V.S.; Mergelov, N.S.; Dolgikh, A.V.; Shorkunov, I.G. Bacterial communities in the soils of cryptogamic barrens of East Antarctica (the Larsemann Hills and Thala Hills oases). Eurasian Soil Sci. 2015, 48, 276–287.
- Wang, N.F.; Zhang, T.; Zhang, F.; Wang, E.T.; He, J.F.; Ding, H.; Zhang, B.T.; Liu, J.; Ran, X.B.; Zan, J.Y. Diversity and structure of soil bacterial communities in the Fildes Region (maritime Antarctica) as revealed by 454 pyrosequencing. Front. Microbiol. 2015, 6, 1188, doi:10.3389/fmicb.2015.01188.
- Coleine, C.; Pombubpa, N.; Zucconi, L.; Onofri, S.; Turchetti, B.; Buzzini, P.; Stajich, J.E.; Selbmann, L. Uncovered Microbial Diversity in Antarctic Cryptoendolithic Communities Sampling three Representative Locations of the Victoria Land. Microorganisms 2020, 8, 942.
- Coleine, C.; Pombubpa, N.; Zucconi, L.; Onofri, S.; Stajich, J.E.; Selbmann, L. Endolithic Fungal Species Markers for Harshest Conditions in the McMurdo Dry Valleys, Antarctica. Life 2020, 10, 13.
- De Los Rios, A.; Wierzchos, J.; Sancho, L.; Ascaso, C. Acid microenvironments in microbial biofilms of Antarctic endolithic microecosystems. Environ. Microbiol. 2003, 5, 231–237.
- De Los Ríos, A.; Grube, M.; Sancho, L.; Ascaso, C. Ultrastructural and genetic characteristics of endolithic cyanobacterial biofilms colonizing Antarctic Granite Rocks. FEMS Microbiol. Ecol. 2007, 59, 386–395, doi:10.1111/j.1574-6941.2006.00256.x.
- Bottos, E.; Scarrow, J.; Archer, S.; McDonald, I.; Cary, S. Bacterial community structures of Antarctic soils. In Antarctic Terrestrial Microbiology: Physical and Biological Properties of Antarctic Soils; Cowan, D., Ed.; Springer: Heidelberg, Germany, 2014, pp. 9–33.
- Foong, C.; Ling, C.; Gonzalez, M. Metagenomic analyses of the dominant bacterial community in the Fildes Peninsula, King George Island (South Shetland Islands). Polar Sci. 2010, 4, 263–273, doi:10.1016/j.polar.2010.05.010.
- Pan, Q.; Wang, G.; Zhang, Y.; Cai, M.; He, J.; Yang, H. Denaturing gradient gel electrophoresis fingerprinting of soil bacteria in the vicinity of the Chinese Great Wall Station, King George Island, Antarctica. J. Environ. Sci, 2013, 25, 1649–1655, doi:10.1016/S1001-0742(12)60229-0.
- Chong, C.W.; Pearce, D.A.; Convey, P.; Tan, G.Y.A.; Wong, R.C.S.; Tan, I.K.P. High levels of spatial heterogeneity in the biodiversity of soil prokaryotes on Signy Island, Antarctica. Soil Biol. Biochem. 2010, 42, 601–610.
- Brooks, S.T.; Jabour, J.; Bergstrom, D. What is ‘footprint’ in Antarctica: Proposing a set of definitions. Antarctic Sci. 2018, 30, 227–235.
- Brooks, S.T.; Jabour, J.; Van Den Hoff, J.; Bergstrom, D. Our footprint on Antarctica competes with nature for rare ice-free land. Nat. Sustain. 2019, 2, 185–190, doi:10.1038/s41893-019-0237-y.
- Pertierra, L.; Hughes, K.; Vega, G.; Ollala-Tarraga, M. High Resolution Spatial Mapping of Human Footprint across Antarctica and Its Implications for the Strategic Conservation of Avifauna. PLoS ONE 2017, 12, e0168280.
- Secretariat of the Antarctic Treaty. The Protocol on Environmental Protection to the Antarctic Treaty. Available online: https://www.ats.aq/e/ep.html (accesed on 30, June, 2020)
- Bajerski, F.; Wagner, D. Bacterial succession in Antarctic soils of two glacier forefields on Larsemann Hills, East Antarctica. FEMS Microbiol. Ecol. 2013, 85, 128–142, doi:10.1111/1574-6941.12105.
- Teixeira, L.C.; Peixoto, R.S.; Cury, J.C.; Sul, W.J.; Pellizari, V.H.; Tiedje, J.; Rosaldo, A.S. Bacterial diversity in rhizosphere soil from Antarctic vascular plants of Admiralty Bay, maritime Antarctica. ISME J. 2010, 4, 989–1001, doi:10.1038/ismej.2010.35.
- Fierer, N.; Jackson, R.B. The diversity and biogeography of soil bacterial communities. Proc. Natl. Acad. Sci. USA 2006, 103, 626–631.
- Pershina, E.; Ivanova, E.; Abakumov, E.; Andronov, E. The impacts of deglaciation and human activity on the taxonomic structure of prokaryotic communities in Antarctic soils on King George Island. Antarct. Sci. 2018, 30, 278–288, doi:10.1017/S095410201800024X.
- Mergelov, N.; Mueller, C.W.; Prater, I.; Shorkunov, I.; Dolgikh, A.; Zazovskaya, E.; Shishkov, V.; Krupskaya, V.; Abrosimov, K.; Cherkinsky, A.; et. al. Alteration of rocks by endolithic organisms is one of the pathways for the beginning of soils on Earth. Sci. Rep. 2018, 8, 3367.
- Abakumov, E.; Alekseev, I. Stability of soil organic matter in Cryosols of the maritime Antarctic: Insights from 13C NMR and electron spin resonance spectroscopy. Solid Earth 2018, 9, 1329–1339.
- Michel, R.F.M.; Schaefer, C.E.; Dias, L.E.; Simas, F.; De Melo Benites, V.; Mendonca, E.S. Ornithogenic gelisols (cryosols) from Maritime Antarctica: Pedogenesis, vegetation and carbon studies. Soil Sci. Soc. Am. J. 2006, 70, 1370–1376.
- Simas, N.; Schaefer, C.; Mendonca, E.; Silva, I.; Ribeiro, A. Organic carbon stocks in permafrost-affected soils from Admiralty Bay, Antarctica. J. Res. U.S. Geol. Surv. 2007, 1047, 76–79.
- Schaefer, C.; Simas, F.; Gilkes, R.; Mathison, C.; Costa, L.; Albuquerque, M. Micromorphology and microchemistry of selected Cryosols from Maritime Antarctica. Geoderma 2008, 144, 104–115.
- Abakumov, E.; Mukhametova, N. Microbial biomass and basal respiration of selected Sub-Antarctic and Antarctic soils in the areas of some Russian polar stations. Solid Earth 2014, 5, 705–712.
- Gilichinsky, D.; Abakumov, E.; Abramov, A.; Fyodorov-Davydov, D.; Goryachkin, S.; Lupachev, A.; Mergelov, N.; Zazovskaya, E. Soils of mid and low antarctic: Diversity, geography, temperature regime. In Proceedings of the 19th World Congress of Soil Science, Soil Solutions for a Changing World, Brisbane, Australia, 1–6 August 2010; pp. 32–35.
- Espejo, W.; Celis, J.; Sandoval, M.; Gonzalez-Acuna, D.; Barra, D.; Capulin, J. The Impact of Penguins on the Content of Trace Elements and Nutrients in Coastal Soils of North Western Chile and the Antarctic Peninsula Area. Water Air Soil Pollut. 2017, 228, 116.
- Metcheva, R.; Yurukova, L.; Teodorova, S. Biogenic and toxic elements in feathers, eggs, and excreta of Gentoo penguin (Pygoscelis papua ellsworthii) in the Antarctic. Environ. Monit. Assess. 2011, 182, 571–585.
- Santamans, A.C.; Boluda, R.; Picazo, A.; Gil, C.; Ramos-Miras, J.; Tejedo, P.; Pertierra, L.R.; Benayas, J.; Camacho, A. Soil features in rookeries of Antarctic penguins reveal sea to land biotransport of chemical pollutants. PLoS ONE 2017, 12, e0181901.
- Schutte, U.M.E.; Abdo, Z.; Bent, S.J.; Williams, C.J.; Schneider, G.M.; Solheim, B.; Forney, L.J. Bacterial succession in a glacier foreland of the High Arctic. ISME J. 2009, 3, 1258–1268.
- Cannone, N.; Wagner, D.; Hubberten, H.W.; Guglielmin, M. Biotic and abiotic factors influencing soil properties across a latitudinal gradient in Victoria Land, Antarctica. Geoderma 2008, 144, 50–65.
- Pointing, S.B.; Chan, Y.; Lacap, D.C.; Lau, M.C.Y.; Jurgens, J.A.; Farrel, R.L. Highly specialized microbial diversity in hyper-arid polar desert. Proc. Natl. Acad. Sci. USA 2009, 106, 19964–19969.
- Yung, C.C.M.; Chan, Y.; Lacap, D.C.; Perez-Ortega, S.; de los Rios-Murillo, A.; Lee, C.K.; Cary, S.K.; Pointing, S.B. Characterization of chasmoendolithic community in Miers Valley, McMurdo DryValleys, Antarctica. Microb. Ecol. 2014, 68, 351–359.
- Archer, D.; de los Rios, A.; Lee, K.; Niederberger, T.; Cary, S.; Coyne, K.; Douglas, S.; Lacap-Bugler, D.; Pointing, S. Endolithic microbial diversity in sandstone and granite from the McMurdo Dry Valleys, Antarctica. Polar Biol. 2016, 40, 997–1006.





