Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Md. Tanvir Rahman | + 2444 word(s) | 2444 | 2021-12-07 07:51:13 | | | |
| 2 | Beatrix Zheng | Meta information modification | 2444 | 2021-12-14 02:27:26 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Rahman, M.T. Antimicrobial Resistance in Salmonella in Poultry. Encyclopedia. Available online: https://encyclopedia.pub/entry/17047 (accessed on 25 May 2026).
Rahman MT. Antimicrobial Resistance in Salmonella in Poultry. Encyclopedia. Available at: https://encyclopedia.pub/entry/17047. Accessed May 25, 2026.
Rahman, Md. Tanvir. "Antimicrobial Resistance in Salmonella in Poultry" Encyclopedia, https://encyclopedia.pub/entry/17047 (accessed May 25, 2026).
Rahman, M.T. (2021, December 13). Antimicrobial Resistance in Salmonella in Poultry. In Encyclopedia. https://encyclopedia.pub/entry/17047
Rahman, Md. Tanvir. "Antimicrobial Resistance in Salmonella in Poultry." Encyclopedia. Web. 13 December, 2021.
Copy Citation
Salmonellosis is one of the most common zoonotic diseases globally including in Bangladesh. Every year millions of people, animals, and poultry are affected by salmonella infection. Salmonellosis is frequently associated with high mortality rates that are closely 90%, resulting in significant economic losses.
AMR
poultry
zoonotic
antimicrobials
Bangladesh
MDR
resistance
environment
1. Antimicrobial Resistance Profile of Salmonella spp.
1.1. Resistance to Penicillins
Penicillins are antimicrobials that are classified as β-lactams [1]. Ampicillin, amoxicillin, oxacillin and cloxacillin are broadly utilized semi-synthetic antimicrobials within the penicillin class.
Akond et al. [2] reported 100% penicillin-resistant and 88% ampicillin-resistant Salmonella in hand wash, intestinal fluid, cloacal swab, egg surface and soil samples from a layer farm in Dhaka. Sarker et al. [3] reported similar results ten years later, both in broilers and layers in Rajshahi. Ampicillin was the first broad-spectrum antibiotic of the penicillin group. Ahmed et al. [4] reported that 87.50% of Salmonella exhibited resistance to ampicillin and amoxicillin in egg surface samples from laying hens at different markets in Dhaka city. Additionally, Mahmud et al. [5] and Talukder et al. [6] observed 100% resistance to ampicillin and amoxicillin in Chittagong and Mymensingh. Furthermore, Salmonella was found to be 40% to 92.86% resistant to ampicillin and amoxicillin in broilers and layers [7][8][9][10]. Jahan et al. [11] detected 100% ampicillin- and amoxicillin-resistant Salmonella from dressed broilers, water and device surface samples, whereas Alam et al. [12] and Hossain et al. [13] reported 66.67% to 82.85% ampicillin-resistant Salmonella in cloacal, fecal, litter, feed, water, air and handwashing samples collected from different broiler farms in Mymensingh. Previously, Mir et al. [14] showed that 100% Salmonella were resistant to penicillin and oxacillin, and Sharma et al. [15] found 95.71% ampicillin-resistant Salmonella from poultry samples in India.
Several genes are known to be linked with resistance against penicillin. A study in Bangladesh has recently confirmed the presence of the β-lactam-resistant blaTEM gene in 73.30%, 63.60% and 50% of S. Typhimurium isolates from broilers, sonali and indigenous chickens, respectively [16]. Parvin et al. [17] also detected the blaTEM-1-resistant gene of Salmonella from chicken in Bangladesh. Previously, Alam et al. [12] detected the blaTEM-1 (82.85%) gene in Salmonella from broiler samples in Bangladesh. Likely, in Egypt, Sabry et al. [18] reported the β-lactam-resistant blaTEM gene from healthy and diseased chickens. Earlier, Wajid et al. [19] detected the blaTEM-1 (72.70%) gene in Salmonella from the layers in Pakistan. In addition, Giuriatti et al. [20] detected the blaTEM-1 (83.33%) gene from chickens in Brazil. Similarly, the blaTEM-1-resistant gene of Salmonella from poultry was detected in Brazil and China by Souza et al. [21] and Wang et al. [22], respectively. Therefore, the exhibition of higher resistance patterns of Salmonella to penicillin group of antimicrobials in poultry may well relate to the longtime use of these antimicrobials.
1.2. Resistance to Cephalosporins
Cephalosporins are a class of β-lactam antimicrobials [23] that might be broadly utilized as crucial drugs to treat important bacterial diseases in people and animals [24]. For a long time, there seems to be an increment in records of resistance in Salmonella to cephalosporin in people and food producing animals universally. In Bangladesh, cephalosporins are also used in poultry.
Cephalexin, a class of first-generation cephalosporins, was found to be not utterly effective against Salmonella. For example, in Dhaka city, Salmonella isolated from egg surface, hand wash, cloacal swab, intestinal fluid and soil samples were found about 50% to 65% resistant to cephalexin [4][2]. Similarly, Akond et al. [2] in Dhaka and Chaudhary et al. [25] in Chittagong observed 50.00% to 96.44% resistance of Salmonella to ceftriaxone and cefixime since they are used as the third generation of cephalosporins. Dutil et al. [26] and Jeon et al. [27] recorded ceftiofur-resistant Salmonella from poultry meat in Canada and Korea, respectively. The use of ceftiofur (a third-generation cephalosporin) in farm animals has severe public health concerns since it leads to resistance to extended-spectrum cephalosporins such as ceftriaxone and cephamycins [28]. These findings point to the need for a better monitoring scheme and guidelines for the prudent use of antimicrobial medicines in Bangladesh’s poultry sector.
1.3. Resistance to Carbapenems
Ertapenem, imipenem and meropenem belong to carbapenem group of antimicrobials. Imipenem features a wide range of action against aerobic and anaerobic microbes. Parvin et al. [17] reported 48.60% resistance to imipenem in Salmonella isolates from chicken frozen meat. Tawyabur et al. [29] also observed 40.74% resistance of meropenem in healthy and diseased turkeys. These findings demonstrate that we must be concerned since antibiotics from carbapenem group are frequently used as “last-line agents” to cure diseases caused by MDR Gram-negative bacteria [30][31][32]. Earlier, Wajid et al. [19] also reported resistance of S. Typhimurium for imipenem (79.40%), doripenem (61.70%), and meropenem (54.50%) in poultry in Pakistan. Carbapenems are generally considered to be last-line antimicrobials to treat hospitalized patients affected by different bacterial infections. However, it has been unclear how this sort of resistance has entered the poultry, as these sorts of antimicrobials are not allowed for use in the poultry industry. Higher rates of carbapenem resistance are in poultry is very much alarming, so it is important to ensure quality control and confirmation measures for the poultry processing and production industry.
1.4. Resistance to Fluroquinolones
Fluoroquinolones are a group of antimicrobials that are used universally. Ciprofloxacin, a sort of fluoroquinolone antimicrobials, is commonly used to treat a vast extend infections of humans, poultry, and other animals. As a result, Salmonella isolated from broilers, layers and turkeys showed periodical increase in resistance to ciprofloxacin, ranging from 20% to 100% [2][33][5][25][29][34][16] in different districts of Bangladesh in between 2012 to 2021. The scenario is similar in neighboring countries. Hassan et al. [35] revealed 87.50% resistance of Salmonella to pefloxacin inlayer chickens, whereas Parvin et al. [17] reported 70.30% resistance in the broilers. Sharma et al. [15] observed 82.86% resistance of Salmonella to ciprofloxacin in chickens in India. Similarly, in Pakistan, 92.60% of S. Typhimurium and 100% of S. Enteritidis were resistant to pefloxacin in poultry birds [19]. Furthermore, 60% of S. Typhimurium and 65.85% S. Enteritidis showed resistance in layers in Chittagong, Gazipur, Narsingdi, Tangail, and Brahmanbaria [5][34]. Nalidixic acid (NA) is the first of the synthetic quinolone antibiotics. Various degrees of resistance found against NA have been reported in Salmonella in Bangladesh. About 20% to 100% resistance found in Salmonella to NA secluded from poultry and environmental samples at a different region of Bangladesh [4][2][11][7][36][17][3][16]. Early, Nikolić et al. [37] observed 95.50% resistance of Salmonella to NA in broiler isolates in Serbia. These discoveries highlight the requirement for the execution of reconnaissance frameworks that center on nourishment cleanliness, utilize antimicrobials in poultry farming and continuously check the quality of retail meat items.
1.5. Resistance to Aminoglycosides
Aminoglycosides are antimicrobials that repress bacterial protein synthesis [38]. Streptomycin is one of the primary aminoglycoside antibiotics presented in human medication. It is additionally utilized in animals and poultry. Salmonella resistance to streptomycin has been documented in chicken in Bangladesh, ranging from 38% to 100% [11][39][40][12][16]. Similarly, Souza et al. [21] reported 98.30% resistance in Salmonella to streptomycin from poultry in Brazil. Gentamicin, a broad-spectrum aminoglycoside antibiotic, has long been used to treat Gram-negative and Gram-positive microbes in poultry in Bangladesh. Extremely recently, Siddiky et al. [16] reported 86.70% resistance in Salmonella to gentamicin in the broilers, sonali, and indigenous chickens in Bangladesh. Previously, Wajid et al. [19] observed 64.70% resistance to gentamicin from S. Typhimurium isolates in poultry in Pakistan. Earlier, Hassan et al. [35] and Paul et al. [7] also observed significant amount of resistance to other aminoglycosides in Salmonella such as kanamycin in the layers. Alam et al. [12] reported the aminoglycoside-resistant gene aadA1 (77.10%) in Salmonella isolates from cloacal swabs and a litter of broilers in Mymensingh. Siddiky et al. [16] observed the strA/B (33.33%) resistance gene in S. Typhimurium isolates from broilers ceca at wet markets in Dhaka. Earlier, Wajid et al. [19] reported aminoglycosides aadA1 (35.20%), strA (20.50%) and strB (41.10%) resistance genes, respectively, in S. Typhimurium from poultry in Pakistan.
1.6. Resistance to Macrolides
Macrolides are bacteriostatic, which means that instead of killing bacteria, they limit or restrain their growth [41]. Azithromycin is an azalide, a sort of macrolide antibiotic. Salmonella in Bangladeshi poultry has been found to have varying degrees of azithromycin resistance, ranging from 18.18% to 81.25% [11][42][43][8][40][44][36][10]. Last year, Tîrziu et al. [45] also reported 88.20% resistance in Salmonella to azithromycin was isolated from store raw poultry in Romania. Erythromycin is generally used to cure many diseases of chicken in Bangladesh. About 62.50% to 100.00% resistance found in Salmonella to erythromycin in layer samples [4][2][5][7], while 64.28% to 100.00% resistance observed [11][33][39][43][8][40][44] in case of broiler samples. Cardoso I et al. [46] in Brazil and Sharma et al. [15] in India also reported 100% resistance of avian Salmonella to erythromycin. These higher resistances of Salmonella to macrolides are not unprecedented since in numerous cases, many individuals of Enterobacteriaceae are found resistant to these compounds.
1.7. Resistance to Lincosamides
Clindamycin is an antibiotic that is used to treat a variety of bacterial infections. It belongs to the lincosamides family and operates by preventing bacteria from producing protein [47]. Sultana et al. [42] reported 84% resistance in Salmonella to clindamycin in poultry in Bangladesh. Similarly, Yildirim et al. [48] in Turkey and Mir et al. [14] in India detected 97% and 100% resistance in Salmonella isolated from poultry as resistance to clindamycin, respectively. So, it is obligatory to actualize strict control over the man handle of antimicrobials, especially in the poultry segment. Appropriate logical and open wellbeing controls are required to scrutinize the non-judicial utilization of antimicrobials.
1.8. Resistance to Tetracyclines
One of the heavily used antibiotics in veterinary medication is tetracycline. A variable level of tetracycline resistance has been noticed in Salmonella to tetracycline in poultry in Bangladesh. Several studies had reported about 65% to100% resistance in Salmonella to tetracycline and oxytetracycline in layers and broilers in Bangladesh [11][35][39][5][7][43][8][12][44][36][29][17][6][16]. Recently, Alam et al. [12] and Tawyabur et al. [29] detected tetracycline-resistant phenotype and the tetracycline-resistant gene tetA in Salmonella in poultry in Bangladesh. More specifically, extremely recently, Siddiky et al. [16] identified tetracycline tetAgene 80%, 90.90% and 100% S. Typhimurium isolates of broilers, sonali, and indigenous chickens’ ceca, respectively, in Bangladesh. Earlier, Sharma et al. [15] observed 100% resistance to tetracycline and also detected the tetA-resistant gene in Salmonella in India.
Doxycycline is a broad-spectrum tetracycline class of antibiotic that is commonly used to treat different infections in humans and animals. A significant number of isolates resistant to doxycycline (50.00% to 79.31%) has also been reported in Salmonella in poultry in Bangladesh [4][42][8][10]. Formerly, Waghamare et al. [49] also observed 100% resistance in Salmonella to doxycycline in India. Higher rate of detection for the tetracycline-resistant gene in Salmonella from poultry environment is threat for both animals and humans. The ability of resistance potential of Salmonella to access the food web could expose individuals to life-threatening health risks. To reduce the rise of bacterial resistance in chicken farms in Bangladesh and around the globe, AMR reconnaissance protocols should be implemented.
1.9. Resistance to Phenicols
Chloramphenicol is a broad-spectrum antibiotic not currently used since it is a banned item due to its side effect on the host. However, it has long been used to treat numeric types of bacterial maladies in both individuals and animals [50]. Studies carried out throughout 2012 to 2021 have reported variable degree (20% to 58%) resistance in Salmonella to chloramphenicol in layer birds in Bangladesh [4][7][9][13][16]. In broilers, about 94.28% to 100% resistance was reported in Salmonella to chloramphenicol [11][12]. Alamet al. [12] also detected chloramphenicol resistance floR (94.28%) gene from Salmonella isolates of broilers in Bangladesh. Previously, El-Sharkawy et al. [51] reported 100% resistance to chloramphenicol in S. Typhimurium isolated from chicken in Egypt. These authors also detected the chloramphenicol-resistant gene floR (79.30%) from these isolates [51]. The detection of the chloramphenicol-resistant floR gene of Salmonella in broiler carrying intl1 is of severe general well-being issues because their Salmonella zoonotic type and conceivable outcomes to access into the food web.
1.10. Resistance to Rifampicin
Rifampicin is used for the treatment of a few sorts of bacterial diseases, counting tuberculosis, Mycobacterium avium complex disease, and Legionnaires’ disease [52]. In livestock and poultry, it has been used to some extent experimentally. However, reports are available showing resistance in Salmonella to rifampicin. Akond et al. [2] reported 60% resistance in Salmonella to rifampicin isolated from the egg surface, cloacal swabs, intestinal fluid, soil and hand washing samples of the layers. Later, Sultana et al. [42] also observed 88% resistance in Salmonella to rifampicin isolated from the layers in Bangladesh. Previously Zdragas et al. [53] reported 33.30% rifampicin resistance in avian Salmonella in Greece and Ramatla et al. [54] reported 100% rifampicin resistance in avian Salmonella in South Africa. Horizontal transfer of rifampicin-resistant genes from human isolates to avian species might be linked with this observed resistance in avian isolates.
1.11. Resistance to Glycopeptides
Vancomycin is a glycopeptide antimicrobial useful to treat skin diseases, circulatory system diseases, endocarditis, bone and joint diseases, and meningitis in humans [55]. Although it is not used in poultry, Sultana et al. [42] reported 78% resistance in Salmonella to vancomycin in the layers in Savar. In India, Singh et al. [56] recorded 100% resistance in avian Salmonella to vancomycin. Vancomycin works against the Gram-positive cell wall, so it was not unexpected to see resistance in Salmonella to vancomycin.
1.12. Resistance to Sulpher Drugs
Sulfonamides such as sulfamethoxazole are a widely used group of antimicrobials in poultry [57]. In Bangladesh, variable degrees of sulfamethoxazole resistance in Salmonella in the layers were recorded [7][3][10]. Rahman et al. [8] detected 75.86% resistance to sulfamethoxazole and Parvin et al. [17] also reported 89.20% resistance in Salmonella to sulfamethoxazole in the broilers. In Bangladesh, the percentages of resistance to sulfur medicines detected are close to those reported in other studies in Malaysia, which was 67.50% [58], and Ethiopia [59] where resistance was 60%. Extremely recently, Siddiky et al. [16] detected sulfonamide resistance sul1 gene in 36.40%, 66.70% and 80% S. Typhimurium isolated from broiler, sonali and indigenous hens’ ceca in Bangladesh, respectively. This gene has also been identified in Salmonella in India [60]. The evolution of sulfamethoxazole resistance in hens could be caused by irrational antimicrobial use in the production process or environmental drip. As a result, we should be concerned about developing and implementing an efficient national AMR surveillance strategy in order to assure food safety and market control.
1.13. Resistance to Polymyxins
Colistin is an antibiotic in the reserve group that is used as a last option for curing various MDR bacterial diseases in humans. Despite the fact that there are limits to the utilization of colistin in domesticated fowls in Bangladesh, it has long been used to treat Gram-negative bacterial diseases in this country. A significant level of colistin resistance was observed as ranging from 50% to 92.68% in Salmonella in broilers and layers in Bangladesh [35][33][5][13][34]. Similarly, Phiri et al. [61] also reported 78.70% colistin resistance in Salmonella in Zambia. The main reservoirs for colistin resistance and transmission have been identified as livestock and poultry [62]. Detection of colistin resistance is extremely concerning for public health. In Bangladesh, Uddin et al. [34] detected colistin resistance mcr1 gene in Salmonella in poultry. Earlier, Quesada et al. [63] and Moreno et al. [64] also identified colistin resistancemcr1 gene in poultry in Spain and Brazil, respectively. Globally, increased resistance to colistin is quickly growing, posing a hazard to human health. Colistin-resistant genes are found in the plasmid. There are conceivable outcomes for exchanging these resistance genes from resistant to other sensitive isolates, making the situation more aggravated.
Overall antimicrobial-resistant profile of poultry Salmonella in Bangladesh as reported in the literature on a yearly basis is presented in Figure 1.
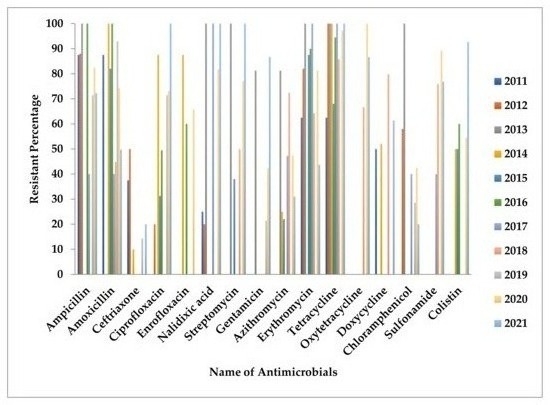
Figure 1. Presents antimicrobial-resistant profiles of Salmonella based on year of publications.
References
- Kardos, N.; Demain, A.L. Penicillin: The medicine with the greatest impact on therapeutic outcomes. Appl. Microbiol. Biotechnol. 2011, 92, 677–687.
- Akond, M.A.; Shirin, M.; Alam, S.; Hassan, S.M.; Rahman, M.M.; Hoq, M. Frequency of drug resistant Salmonella spp. isolated from poultry samples in Bangladesh. Stamford J. Microbiol. 2012, 2, 15–19.
- Sarker, B.R.; Ghosh, S.; Chowdhury, S.; Dutta, A.; Chandra Deb, L.; Krishna Sarker, B.; Sultana, T.; MozafforHossain, K.M. Prevalence and antimicrobial susceptibility profiles of non-typhoidal Salmonella isolated from chickens in Rajshahi, Bangladesh. Vet. Med. Sci. 2021, 7, 820–830.
- Ahmed, M.M.; Rahman, M.M.; Mahbub, K.R. Characterization of antibiotic resistant Salmonella spp. isolated from chicken eggs of Dhaka city. J. Sci. Res. 2011, 3, 191–196.
- Mahmud, T.; Hassan, M.M.; Alam, M.; Khan, M.M.; Bari, M.S.; Islam, A. Prevalence and multidrug-resistant pattern of Salmonella from the eggs and egg-storing trays of retail markets of Bangladesh. Int. J. One Health 2016, 2, 7–11.
- Talukder, M.; Islam, M.S.; Ievy, S.; Sobur, M.A.; Ballah, F.M.; Najibullah, M.; Rahman, M.B.; Rahman, M.T.; Khan, M.F.R. Detection of multidrug resistant Salmonella spp. from healthy and diseased broilers having potential public health significance. J. Adv. Biotechnol. Exp. Ther. 2021, 4, 248–255.
- Paul, P.; Akther, S.; Ali, M.Z.; Banu, H.; Khan, M.S.; Khatun, M.M. Isolation, identification and antibiogram study of Salmonella spp. from poultry farm environment. Int. J. Anim. Biol. 2017, 3, 5–11.
- Rahman, M.A.; Rahman, A.K.; Islam, M.A.; Alam, M.M. Detection of multi-drug resistant Salmonella from milk and meat in Bangladesh. Bangladesh J. Vet. Med. 2018, 16, 115–120.
- Hossain, M.S.; Hossain, K.M.; Sarker, M.M.; Hamid, S.A. Prevalence and antibiotic susceptibility of Salmonella from chicken eggs in Naogaon district of Bangladesh. J. Adv. Microbiol. 2019, 28, 1–6.
- Haque, A.K.M.Z.; Akter, M.R.; Islam, S.S.; Alam, J.; Neogi, S.B.; Yamasaki, S.; Kabir, S.M.L. Salmonella Gallinarum in Small-Scale Commercial Layer Flocks: Occurrence, Molecular Diversity and Antibiogram. Vet. Sci. 2021, 8, 71.
- Jahan, F.; Kabir, S.L.; Amin, M.M. Identification and antimicrobial resistance profiles of Salmonellae isolated from the broiler dressing plants associated with their environments. Adv. Res. J. Microbiol. 2013, 1, 1–9.
- Alam, S.B.; Mahmud, M.; Akter, R.; Hasan, M.; Sobur, A.; Nazir, K.N.H.; Noreddin, A.; Rahman, T.; El Zowalaty, M.E.; Rahman, M. Molecular Detection of Multidrug Resistant Salmonella Species Isolated from Broiler Farm in Bangladesh. Pathogens. 2020, 9, 201.
- Hossain, M.J.; Islam, M.S.; Sobur, M.A.; Zaman, S.B.; Nahar, A.; Rahman, M.; Rahman, M.T. Exploring Poultry Farm Environment for Antibiotic Resistant Escherichia coli, Salmonella spp., and Staphylococcus spp. Having Public Health Significance. J. Bangladesh Agric. Univ. 2020, 18, 615–622.
- Mir, I.A.; Kashyap, S.K.; Maherchandani, S. Isolation, serotype diversity and antibiogram of Salmonella enterica isolated from different species of poultry in India. Asian Pac. J. Trop. Biomed. 2015, 5, 561–567.
- Sharma, J.; Kumar, D.; Hussain, S.; Pathak, A.; Shukla, M.; Kumar, V.P.; Anisha, P.N.; Rautela, R.; Upadhyay, A.K.; Singh, S.P. Prevalence, antimicrobial resistance and virulence genes characterization of nontyphoidal Salmonella isolated from retail chicken meat shops in Northern India. Food Control 2019, 102, 104–111.
- Siddiky, N.A.; Sarker, M.S.; Khan, M.S.R.; Begum, R.; Kabir, M.E.; Karim, M.R.; Rahman, M.T.; Mahmud, A.; Samad, M.A. Virulence and Antimicrobial Resistance Profiles of Salmonella enterica Serovars Isolated from Chicken at Wet Markets in Dhaka, Bangladesh. Microorganisms 2021, 9, 952.
- Parvin, M.S.; Hasan, M.M.; Ali, M.Y.; Chowdhury, E.H.; Rahman, M.T.; Islam, M.T. Prevalence and Multidrug Resistance Pattern of Salmonella Carrying Extended-Spectrum β-Lactamase in Frozen Chicken Meat in Bangladesh. J. Food Prot. 2020, 83, 2107–2121.
- Sabry, M.A.; Abdel-Moein, K.A.; Abdel-Kader, F.; Hamza, E. Extended-spectrum β-lactamase-producing Salmonella serovars among healthy and diseased chickens and their public health implication. J. Glob. Antimicrob. Resist. 2020, 22, 742–748.
- Wajid, M.; Awan, A.B.; Saleemi, M.K.; Weinreich, J.; Schierack, P.; Sarwar, Y.; Ali, A. Multiple drug resistance and virulence profiling of Salmonella enterica serovars Typhimurium and Enteritidis from poultry farms of Faisalabad, Pakistan. Microb. Drug Resist. 2019, 25, 133–142.
- Giuriatti, J.; Stefani, L.M.; Brisola, M.C.; Crecencio, R.B.; Bitner, D.S.; Faria, G.A. Salmonella Heidelberg: Genetic profile of its antimicrobial resistance related to extended spectrum β-lactamases (ESBLs). Microb. Pathog. 2017, 109, 195–199.
- Souza, A.I.; Saraiva, M.M.; Casas, M.R.; Oliveira, G.M.; Cardozo, M.V.; Benevides, V.P.; Barbosa, F.O.; FreitasNeto, O.C.; Almeida, A.M.; Berchieri, A. High occurrence of β-lactamase-producing Salmonella Heidelberg from poultry origin. PLoS ONE 2020, 15, e0230676.
- Wang, W.; Peng, Z.; Baloch, Z.; Hu, Y.; Xu, J.; Zhang, W.; Fanning, S.; Li, F. Genomic characterization of an extensively-drug resistance Salmonella enterica serotype Indiana strain harboring blaNDM-1 gene isolated from a chicken carcass in China. Microbiol. Res. 2017, 204, 48–54.
- Loren, G.; Yamamoto, M.D. Case Based Pediatrics for Medical Students and Residents; Department of Pediatrics, University of Hawaii John, A. Burns School of Medicine: Honolulu, HI, USA, 2003.
- Kim, J.S.; Yun, Y.S.; Kim, S.J.; Jeon, S.E.; Lee, D.Y.; Chung, G.T.; Yoo, C.K.; Kim, J. Rapid emergence and clonal dissemination of CTX-M-15–producing Salmonella enterica serotype Virchow, South Korea. Emerg. Infect. Dis. 2016, 22, 68.
- Chaudhary, P.K.; Salam, S.A.; Reza, M.A.; Ahaduzzaman, M. High prevalence of ciprofloxacin and ceftriaxone resistance Salmonella in the retail chicken market of Chattogram, Bangladesh. Turk. J. Vet. Res. 2019, 3, 51–55.
- Dutil, L.; Irwin, R.; Finley, R.; Ng, L.K.; Avery, B.; Boerlin, P.; Bourgault, A.M.; Cole, L.; Daignault, D.; Desruisseau, A.; et al. Ceftiofur resistance in Salmonella enterica serovar Heidelberg from chicken meat and humans, Canada. Emerg. Infect. Dis. 2010, 16, 48.
- Jeon, H.Y.; Kim, Y.B.; Lim, S.K.; Lee, Y.J.; Seo, K.W. Characteristics of cephalosporin-resistant Salmonella isolates from poultry in Korea, 2010–2017. Poult. Sci. 2019, 98, 957–965.
- Dunne, E.F.; Fey, P.D.; Kludt, P.; Reporter, R.; Mostashari, F.; Shillam, P.; Wicklund, J.; Miller, C.; Holland, B.; Stamey, K.; et al. Emergence of domestically acquired ceftriaxone-resistant Salmonella infections associated with AmpCβ-lactamase. JAMA 2000, 28, 3151–3156.
- Tawyabur, M.; Islam, M.S.; Sobur, M.A.; Hossain, M.J.; Mahmud, M.M.; Paul, S.; Hossain, M.T.; Ashour, H.M.; Rahman, M.T. Isolation and Characterization of Multidrug-Resistant Escherichia coli and Salmonella spp. from Healthy and Diseased Turkeys. Antibiotics 2020, 9, 770.
- Zhanel, G.G.; Wiebe, R.; Dilay, L.; Thomson, K.; Rubinstein, E.; Hoban, D.J.; Noreddin, A.M.; Karlowsky, J.A. Comparative review of the carbapenems. Drugs 2007, 67, 1027–1052.
- Nordmann, P.; Naas, T.; Poirel, L. Global spread of carbapenemase-producing Enterobacteriaceae. Emerg. Infect. Dis. 2011, 17, 1791.
- Patel, G.; Bonomo, R. “Stormy waters ahead”: Global emergence of carbapenemases. Front. Microbiol. 2013, 4, 48.
- Aditya, A. Drug resistant Salmonella in broiler chicken sold at local market in Bangladesh and its public health significance. Afr. J. Biotechnol. 2015, 14, 2995–3000.
- Uddin, M.B.; Hossain, S.M.B.; Hasan, M.; Alam, M.N.; Debnath, M.; Begum, R.; Roy, S.; Harun-Al-Rashid, A.; Chowdhury, M.S.R.; Rahman, M.M.; et al. Multidrug Antimicrobial Resistance and Molecular Detection of mcr-1 Gene in Salmonella Species Isolated from Chicken. Animals 2021, 11, 206.
- Hassan, M.M.; Amin, K.B.; Ahaduzzaman, M.; Alam, M.; Faruk, M.S.; Uddin, I. Antimicrobial resistance pattern against E. coli and Salmonella in layer poultry. Res. J. Vet. Pract. 2014, 2, 30–35.
- Karim, S.J.; Islam, M.; Sikder, T.; Rubaya, R.; Halder, J.; Alam, J. Multidrug-resistant Escherichia coli and Salmonella spp. isolated from pigeons. Vet. World 2020, 13, 2156.
- Nikolić, A.; Baltić, T.; Velebit, B.; Babić, M.; Milojević, L.; Đorđević, V. Antimicrobial resistance among Salmonella entericaserovarInfantis from broiler carcasses in Serbia. IOP Conf. Ser. Earth Environ. Sci. 2017, 85, 012077.
- Mingeot-Leclercq, M.P.; Glupczynski, Y.; Tulkens, P.M. Aminoglycosides: Activity and resistance. Antimicrob. Agents Chemother. 1999, 43, 727–737.
- Al-Salauddin, A.S.; Hossain, M.F.; Dutta, A.; Mahmud, S.; Islam, M.S.; Saha, S.; Kabir, S.L. Isolation, identification, and antibiogram studies of Salmonella species and Escherichia coli from boiler meat in some selected areas of Bangladesh. Int. J. Basic Clin. Pharmacol. 2015, 4, 1000.
- Islam, M.K.; Kabir, S.L.; Haque, A.Z.; Sarker, Y.A.; Sikder, M.H. Molecular detection and characterization of Escherichia coli, Salmonella spp. and Campylobacter spp. isolated from broiler meat in Jamalpur, Tangail, Netrokona and Kishoreganj districts of Bangladesh. Afr. J. Microbiol. Res. 2018, 12, 761–770.
- Giguere, S.; Prescott, J.F.; Baggot, J.D.; Walker, R.D.; Dowling, P.M. Antimicrobial Therapy in Veterinary Medicine, 4th ed.; Wiley-Blackwell: Hoboken, NJ, USA, 2006; ISBN 978-0-8138-0656-3.
- Sultana, M.; Bilkis, R.; Diba, F.; Hossain, M.A. Predominance of multidrug resistant zoonotic Salmonella Enteritidis genotypes in poultry of Bangladesh. J. Poult. Sci. 2014, 51, 424–434.
- Mamun, M.A.; Kabir, S.L.; Islam, M.M.; Lubna, M.; Islam, S.S.; Akhter, A.T.; Hossain, M.M. Molecular identification and characterization of Salmonella species isolated from poultry value chains of Gazipur and Tangail districts of Bangladesh. Afr. J. Microbiol. Res. 2017, 11, 474–481.
- Mridha, D.; Uddin, M.N.; Alam, B.; Akhter, A.T.; Islam, S.S.; Islam, M.S.; Khan, M.S.; Kabir., S.L. Identification and characterization of Salmonella spp. from samples of broiler farms in selected districts of Bangladesh. Vet. World 2020, 13, 275.
- Tîrziu, E.; Bărbălan, G.; Morar, A.; Herman, V.; Cristina, R.T.; Imre, K. Occurrence and antimicrobial susceptibility profile of Salmonella spp. in raw and ready-to-eat foods and Campylobacter spp. in retail raw chicken meat in Transylvania, Romania. Foodborne Pathog. Dis. 2020, 17, 479–484.
- CardosoI, M.O.; RibeiroI, A.R.; Santos, L.R.; PilottoI, F.; Moraes, I.H.; Salle, I.C.T.P.; Rocha, I.L.S.; Nascimento, I.V.P. Resistência antimicrobiana em Salmonella Enteritidis isoladas de carcaças de frango. Braz. J. Microbiol. 2006, 37, 368–371.
- “Clindamycin Hydrochloride”. The American Society of Health-System Pharmacists. Archived from the Original on 5 September 2015. Retrieved 4 September 2015. Available online: https://www.drugs.com/monograph/clindamycin-systemic.html (accessed on 17 July 2021).
- Yildirim, Y.; Gonulalan, Z.; Pamuk, S.; Ertas, N. Incidence and antibiotic resistance of Salmonella spp. on raw chicken carcasses. Food Res. Int. 2011, 44, 725–728.
- Waghamare, R.N.; Paturkar, A.M.; Vaidya, V.M.; Zende, R.J.; Dubal, Z.N.; Dwivedi, A.; Gaikwad, R.V. Phenotypic and genotypic drug resistance profile of Salmonella serovars isolated from poultry farm and processing units located in and around Mumbai city, India. Vet. World 2018, 11, 682.
- “Chloramphenicol”. The American Society of Health-System Pharmacists. Archived from the Original on 25 June 2015. Retrieved 1 August 2015. Available online: https://www.drugs.com/monograph/chloramphenicol.html (accessed on 17 July 2021).
- El-Sharkawy, H.; Tahoun, A.; El-Gohary, A.E.; El-Abasy, M.; El-Khayat, F.; Gillespie, T.; Kitade, Y.; Hafez, H.M.; Neubauer, H.; El-Adawy, H. Epidemiological, molecular characterization and antibiotic resistance of Salmonella enterica serovars isolated from chicken farms in Egypt. Gut Pathog. 2017, 9, 1–2.
- “Rifampin”. The American Society of Health-System Pharmacists. Archived from the Original on 7 September 2015. Retrieved 1 August 2015. Available online: https://www.drugs.com/monograph/rifampin.html (accessed on 17 July 2021).
- Zdragas, A.; Mazaraki, K.; Vafeas, G.; Giantzi, V.; Papadopoulos, T.; Ekateriniadou, L. Prevalence, seasonal occurrence and antimicrobial resistance of Salmonella in poultry retail products in Greece. Lett. Appl. Microbiol. 2012, 55, 308–313.
- Ramatla, T.; Taioe, M.O.; Thekisoe, O.M.; Syakalima, M. Confirmation of antimicrobial resistance by using resistance genes of isolated Salmonella spp. in chicken houses of north west, South Africa. World 2019, 9, 158–165.
- “Vancomycin”. Drugs.com. 2 December 2019. Retrieved 24 December 2019. Available online: https://www.drugs.com/international/vancomycin.html (accessed on 17 July 2021).
- Singh, R.; Yadav, A.S.; Tripathi, V.; Singh, R.P. Antimicrobial resistance profile of Salmonella present in poultry and poultry environment in north India. Food Control 2013, 33, 545–548.
- “Co-trimoxazole”. The American Society of Health-System Pharmacists. Archived from the Original on 6 September. Retrieved 1 August 2015. Available online: https://www.drugs.com/monograph/co-trimoxazole.html (accessed on 17 July 2021).
- Chuah, L.O.; Syuhada, A.K.S.; Suhaimi, I.M.; Hanim, T.F.; Rusul, G. Genetic relatedness, antimicrobial resistance and biofilm formation of Salmonella isolated from naturally contaminated poultry and their processing environment in northern Malaysia. Food Res. Int. 2018, 105, 743–751.
- Tibaijuka, B.; Molla, B.; Hildebrandt, G.; Kleer, J.; Salah, W. Antimicrobila resistance to Salmonellae isolated from retail raw chicken meat and giblets in Ethiopia. Bull. Anim. Health Prod. Afr. 2002, 50, 86–95.
- Adesiji, Y.O.; Deekshit, V.K.; Karunasagar, I. Antimicrobial-resistant genes associated with Salmonella spp. isolated from human, poultry, and seafood sources. Food Sci. Nutr. 2014, 2, 436–442.
- Phiri, N.; Mainda, G.; Mukuma, M.; Sinyangwe, N.N.; Banda, L.J.; Kwenda, G.; Muligisa-Muonga, E.; Flavien, B.N.; Mwansa, M.; Yamba, K.; et al. Antibiotic-resistant Salmonella species and Escherichia coli in broiler chickens from farms, abattoirs and open markets in selected districts of Zambia. J. Epidemiol. Res. 2020, 6, 1.
- Hoelzer, K.; Wong, N.; Thomas, J.; Talkington, K.; Jungman, E.; Coukell, A. Antimicrobial drug use in food-producing animals and associated human health risks: What, and how strong, is the evidence? BMC Vet. Res. 2017, 13, 1–38.
- Quesada, A.; Ugarte-Ruiz, M.; Iglesias, M.R.; Porrero, M.C.; Martínez, R.; Florez-Cuadrado, D.; Campos, M.J.; García, M.; Píriz, S.; Sáez, J.L.; et al. Detection of plasmid mediated colistin resistance (MCR-1) in Escherichia coli and Salmonella enterica isolated from poultry and swine in Spain. Res. Vet. Sci. 2016, 105, 134–135.
- Moreno, L.Z.; Gomes, V.T.; Moreira, J.; de Oliveira, C.H.; Peres, B.P.; Silva, A.P.S.; Thakur, S.; La Ragione, R.M.; Moreno, A.M. First report of mcr-1-harboring Salmonella enterica serovar Schwarzengrund isolated from poultry meat in Brazil. Diagn. Microbiol. Infect. Dis. 2019, 93, 376–379.
More
Information
Subjects:
Agriculture, Dairy & Animal Science
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.2K
Revisions:
2 times
(View History)
Update Date:
14 Dec 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





