Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Suresh Panthee | + 1990 word(s) | 1990 | 2021-12-03 02:15:02 | | | |
| 2 | Bruce Ren | Meta information modification | 1990 | 2021-12-14 02:29:46 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Panthee, S. Novel Pathogenic Mucorales Identified by Silkworm. Encyclopedia. Available online: https://encyclopedia.pub/entry/17038 (accessed on 08 May 2026).
Panthee S. Novel Pathogenic Mucorales Identified by Silkworm. Encyclopedia. Available at: https://encyclopedia.pub/entry/17038. Accessed May 08, 2026.
Panthee, Suresh. "Novel Pathogenic Mucorales Identified by Silkworm" Encyclopedia, https://encyclopedia.pub/entry/17038 (accessed May 08, 2026).
Panthee, S. (2021, December 13). Novel Pathogenic Mucorales Identified by Silkworm. In Encyclopedia. https://encyclopedia.pub/entry/17038
Panthee, Suresh. "Novel Pathogenic Mucorales Identified by Silkworm." Encyclopedia. Web. 13 December, 2021.
Copy Citation
Mucormycosis, a rare but highly fatal infection, is caused by fungi of the order Mucorales. Due to their ubiquitous nature, reduced susceptibility to antifungals, acid tolerance, and ability to infect immunocompromised patients through rapid dissemination, these fungi have been frequently reported to infect the COVID-19 patients. In order to develop strategies to overcome mucormycosis, it is essential to understand and identify novel Mucorales present in the environment.
Mucorales
COVID-19
opportunistic infection
silkworm
animal-model
1. Introduction
Fungi help maintain the diversity of the ecosystem and are critical to nutrient-cycling by degrading dead organic materials [1]. In nature, fungi with agricultural, ecological, economic, biotechnological, and medical importance have been identified. On the other hand, fungi can also cause human infections. Invasive fungal diseases account for a majority of complications among immunocompromised patients worldwide [2]. Fungi of the order Mucorales cause mucormycosis, a rare but highly fatal fungal infection. They can cause cutaneous, rhino-orbital, pulmonary, rhino-cerebral, and disseminated bloodstream infections; the severity and prognosis largely depend upon the infection type. The fatality rate is very high [3] with 96% among patients with disseminated infections [4]. The incidence of mucormycosis is rapidly increasing, especially in developing countries like India and Nepal, where Mucorales were mostly found to cause rhino-orbito-cerebral infections. The most recent incidences are observed in COVID-19 patients [5][6][7] or those who recently recovered from COVID-19. Given that the incidence of mucormycosis has been associated with preexisting conditions such as uncontrolled diabetes mellitus, malignancies, trauma, or extended corticosteroids use [4], it is likely that the number of cases with mucormycosis will increase further due to the current COVID-19 pandemic. Primarily found in soil and decaying vegetation, Mucorales are ubiquitous, have reduced susceptibility to most clinically used antifungal agents, and thrive under high acid conditions. Their thermotolerance, however, is greatly varying; some, such as Rhizopus microspores thrive at temperatures as high as 50 °C [8], while some, such as R. sexualis cannot grow above 25 °C [8]. Based on their thermotolerance, it was previously thought that Mucorales that cannot grow at 37 °C were medically not important. However, case reports and incidences of cutaneous infections caused by M. hiemalis [9][10][11][12], which cannot grow at 37 °C [13][14], have been observed. These cases of infections suggested that the fungi that do not grow at 37 °C are capable of causing superficial infections. Furthermore, invasive mucormycosis rapidly disseminates within the host tissues. Depending upon the infection site, Mucorales interact with specific host receptors and take advantage of the host physiological conditions to derive nutrients such as iron [15]. Furthermore, Mucorales can store iron in ferritins besides siderophores, further depleting the host of iron and proliferating within the host rapidly [16]. Therefore, patients with diabetes mellitus and COVID-19 are more prone to mucormycosis as these patients have a higher level of free iron in their blood [17][18]. As mucormycosis has been increasingly common, studies on the understanding of their pathogenesis as well as identifying emerging virulent strains are of top priority.
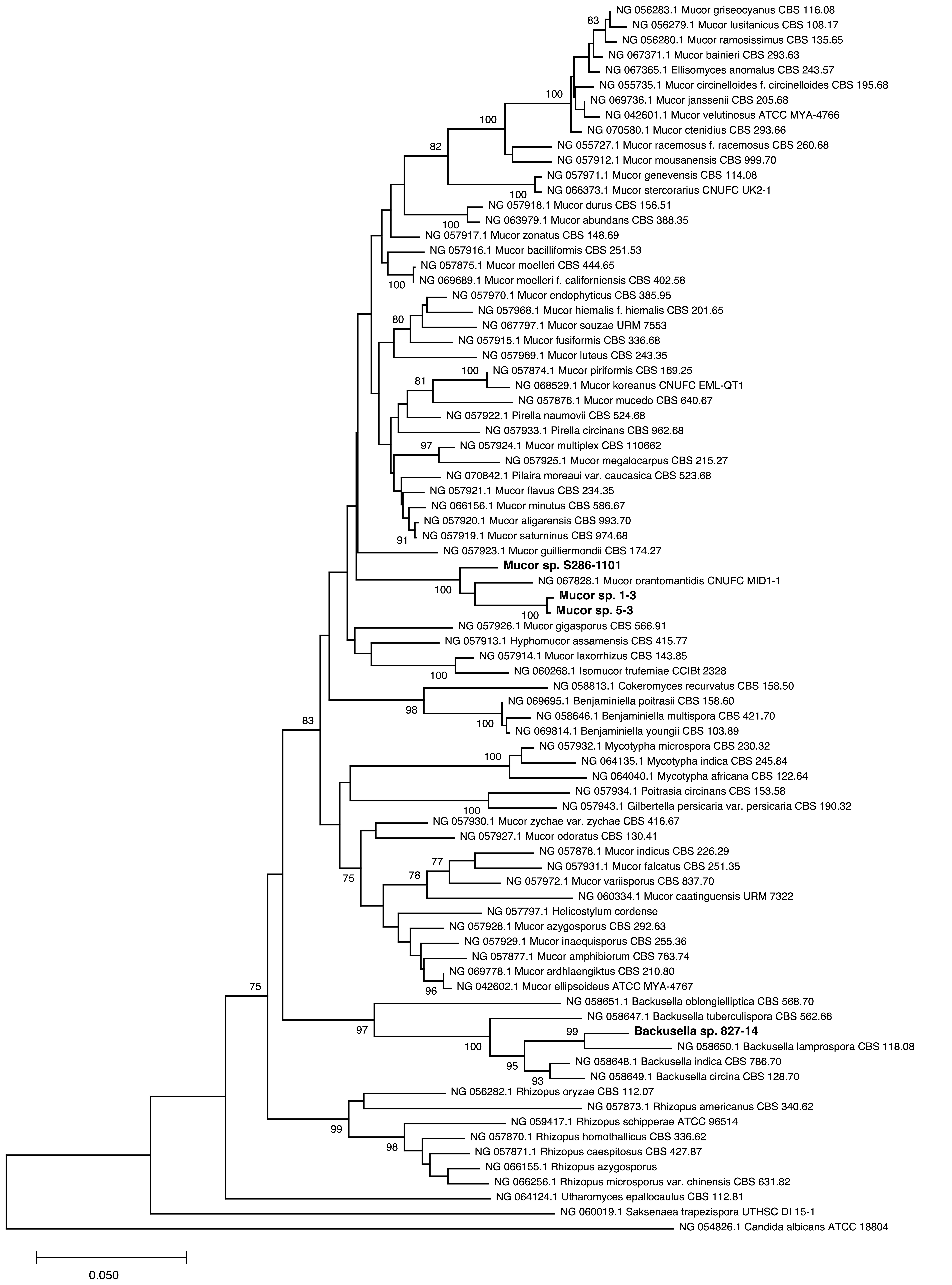
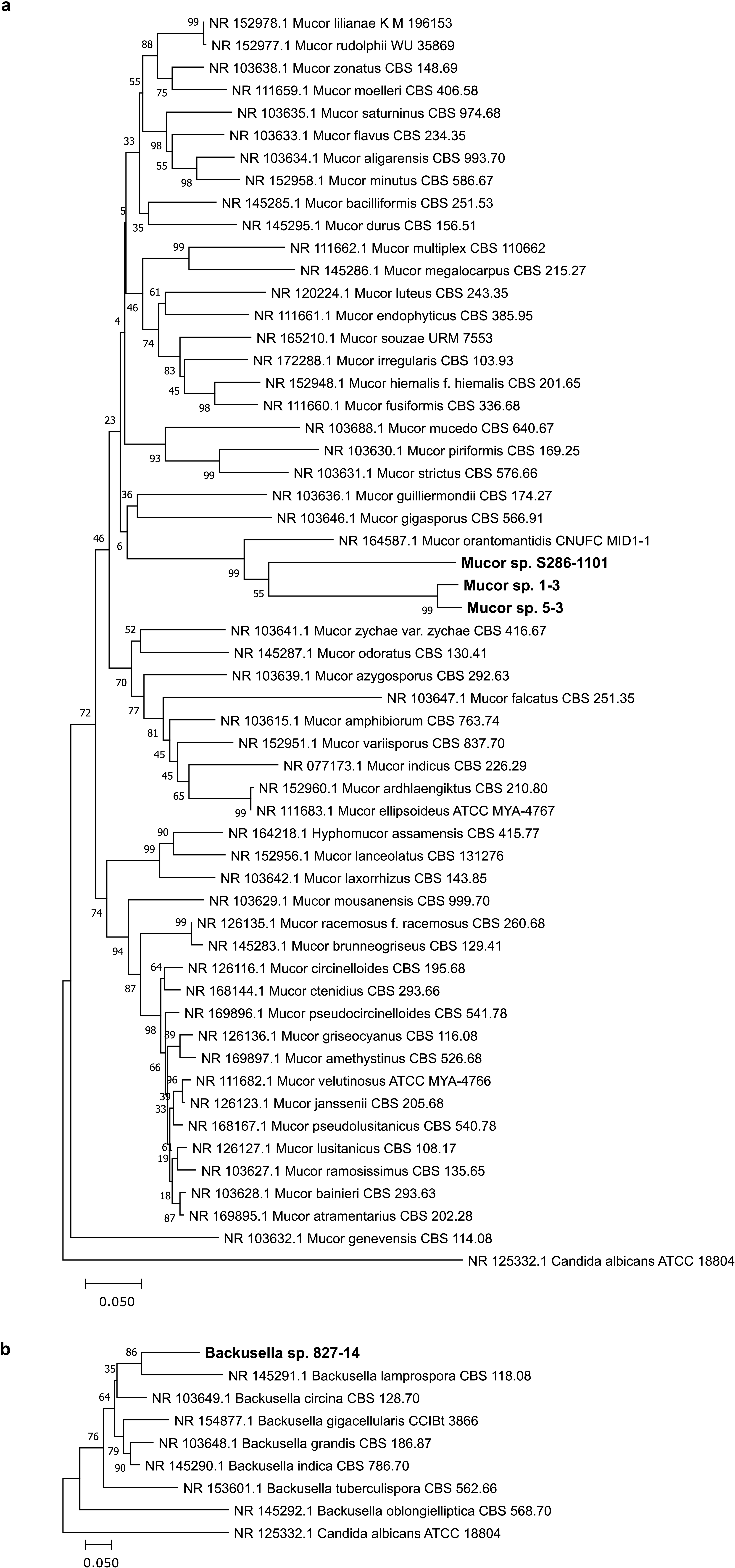

2. Taxonomy and Phylogenetic Analysis
We isolated four fungal strains from plant sources and attempted to identify them (Table 1). BLAST analysis of the LSU rRNA region showed that strains 1-3, 5-3, and S286-1101 were related to Mucor orantomantidis (accession no: NG_067828.1) with the percentage identity of 95.4% (641/672 bp), 95.4% (641/672 bp), and 97.2% (651/670 bp), respectively. Strain 827-14 was 96% (624/650) identical to Backusella lamprospora CBS 118.08 (accession no: NG_058650.1). This result was consistent with the constructed phylogenetic tree using MEGA X [19], where we found that strains 1-3, 5-3, and S286-1101 were claded with M. orantomantidis, and strain 827-14 was claded with B. lamprospora (Figure 1). These findings suggested that these strains were novel species belonging to Mucor (1-3, 5-3, and S286-1101) and Backusella (827-14). The taxonomic assignment of these strains was further confirmed by the phylogenic tree using ITS sequences, another locus used frequently for the taxonomic demarcation of Mucorales [13]. ITS analysis showed that Mucor strains were claded with M. orantomantidis (Figure 2a) and Backusella sp. 827-14 claded with B. lamprospora (Figure 2b). The phylogenetic tree was reconstructed using MAFFT [20] and RAxML [21] to reveal similar results. Among four strains, we randomly selected Mucor sp. 5-3 for whole genome sequencing and SEM analysis.

Figure 1. LSU rRNA-based phylogenetic analysis of Mucorales. The LSU rRNA sequence was utilized to infer the evolutionary relationship of the strains. The optimal tree, drawn to scale, with branch lengths in the same units as those of evolutionary distances, with the sum of branch length = 2.44139183, is shown. The percentage of replicate trees in which the associated taxa clustered together in the bootstrap test (500 replicates) are shown next to the branches. After the ambiguous positions were removed for each sequence pair, there were a total of 1552 positions in the final dataset. The novel strains are written in boldface with larger font size.

Figure 2. ITS-based phylogenetic analysis of Mucorales. The ITS sequence was utilized to infer the evolutionary relationship of the strains. The optimal tree, drawn to scale, with branch lengths in the same units as those of the evolutionary distances, with the sum of branch lengths of (a) 4.16205042 and (b) 1.37992789 for Mucor and Backusella, respectively, is shown. The percentage of replicate trees in which the associated taxa clustered together in the bootstrap test (500 replicates) are shown next to the branches. After the ambiguous positions were removed for each sequence pair, there were a total of 1064 (a) and 975 (b) positions in the final dataset. The novel strains are written in boldface with larger font size.
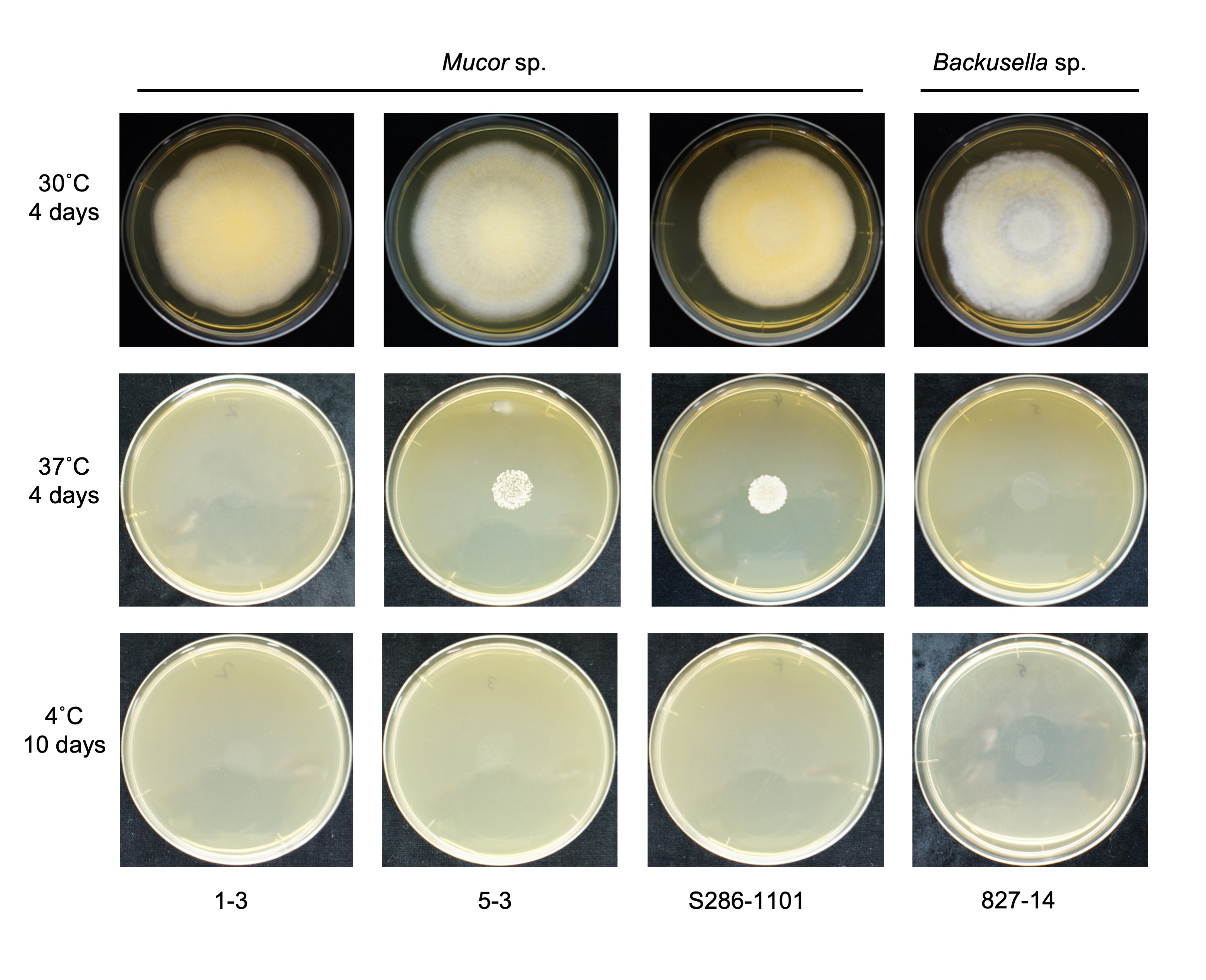
Table 1. Source and the growth of novel fungi at different temperatures.
| Strain | Source (Location) | Growth at | ||
|---|---|---|---|---|
| 37 °C (4 Days) | 30 °C (4 Days) | 4 °C (10 Days) | ||
| Mucor sp. 1-3 | Plant bud (Chiba, Japan) | − | +++ | − |
| Mucor sp. 5-3 | Plant seed (Chiba, Japan) | + | +++ | − |
| Mucor sp. S286-1101 | Plant leaf (Saitama, Japan) | + | +++ | − |
| Backusella sp. 827-14 | Dead plant leaf (Chiba, Japan) | − | +++ | − |
3. Analysis of the Mucor Genome Assembly
We have previously used the next-generation sequencing tool for the genomic analysis of various bacteria [22][23] and fungi [24] and have found that compared to the bacterial genome, the fungal genome contains a large number of repeats and is difficult to assemble. We sequenced the Mucor sp. 5-3 genome using the Ion PGM System and found that its genome size was 30.8 Mb in size, had a G+C content of 39.26%, and divided into about 17,079 contigs (Table 2). This indicated a presence of a large number of repeat elements in the genome. Of note, the same sequencing approach, used for Candida albicans TIMM1768 resulted in about 3400 contigs for a 14.5 Mb genome [24]. Besides, combined with a previous study [25], it can be expected that the Mucor genome is diverse with a genome size ranging between 30–47 Mb.
Table 2. General features of Mucor sp. 5-3 draft genome.
| Features | Characteristics |
|---|---|
| Total reads | 5,016,222 |
| Average length (bp) | 270 |
| Coverage | 44× |
| Genome size (bp) | 30,813,178 |
| Contigs | 17,079 |
| Contigs > 1000 bp | 6792 |
| Longest contig (bp) | 37,105 |
| N50 | 4436 |
| L50 | 2075 |
| G + C (%) | 39.26 |
4. Morphological Studies
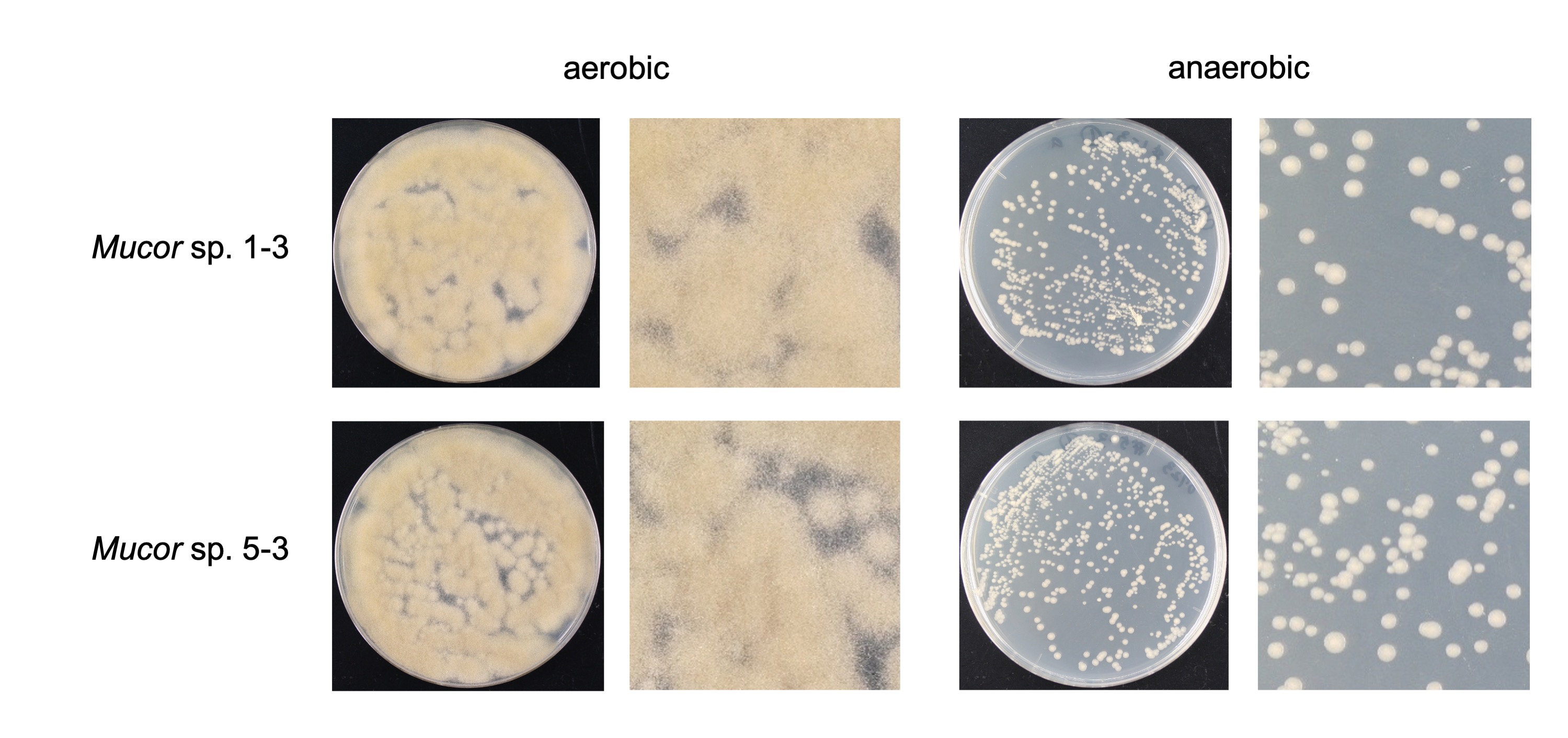
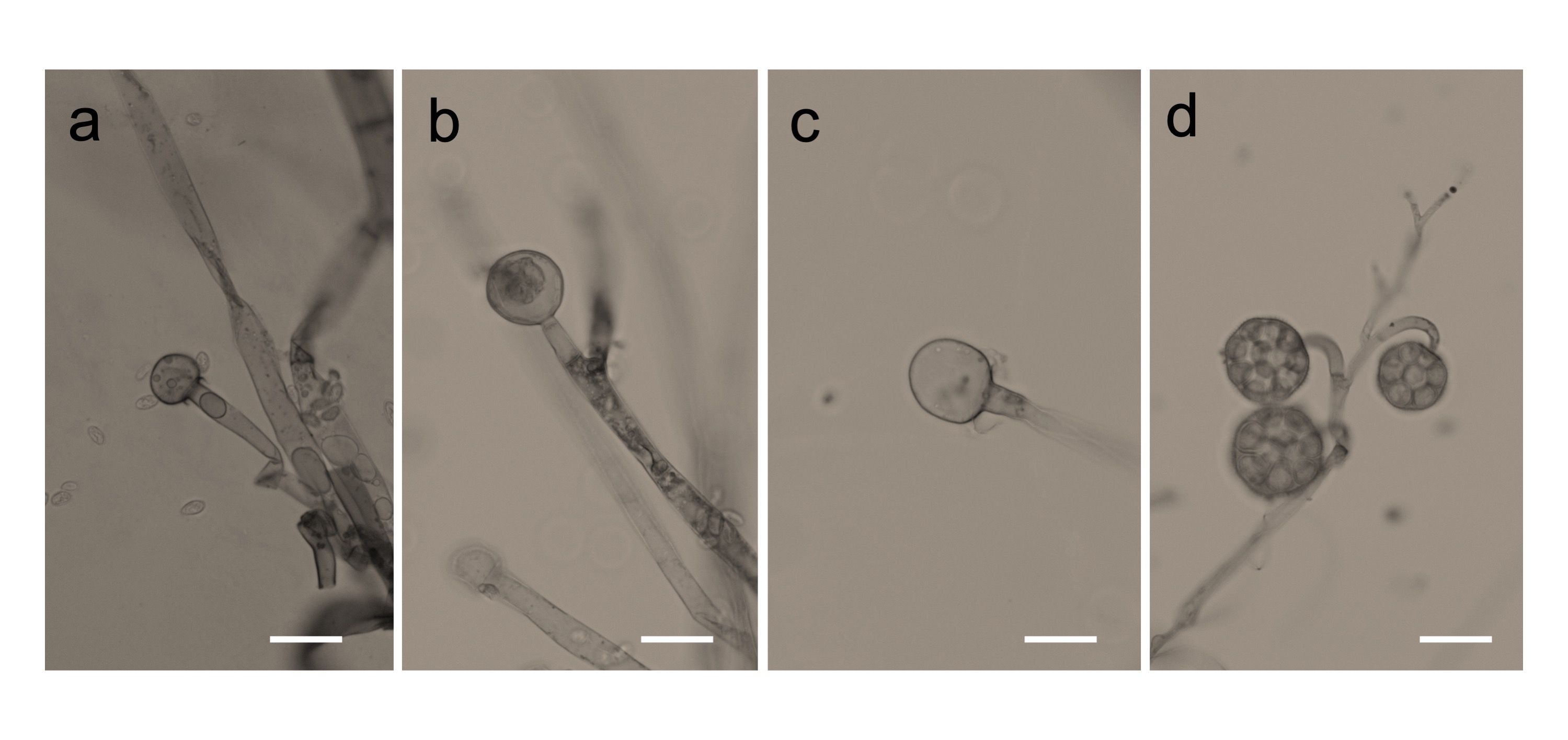
We found that all the strains grew well on the YPD agar plate at 30 °C when grown under aerobic conditions. However, they had a difference in their growth status at 4 °C and 37 °C. Mucor sp. 5-3, and S286-1101 could grow, although slowly, while the other two strains could not grow at 37 °C. At 4 °C, Mucor spp. did not show a visible sign of growth; however, Backusella showed a faint growth (Figure 3). Next, we used Mucor sp. 1-3 and Mucor sp. 5-3 to check their ability to grow under anaerobic conditions. Incubation for two days at 30 °C after streaking on YPD agar showed that the cells grew as yeast under anaerobic conditions; however, mycelial growth was observed under aerobic conditions (Figure 4). Besides, we found that the colony morphology—when visualized with the naked eye—of these two strains on agar plates was similar. When sporangia were observed under a light microscope, a difference was observed among Mucor spp. and Backusella spp. (Figure 5a–d).

Figure 3. Temperature-specific growth of novel Mucorales. 4.0 × 104 spores contained in 40 µL of normal saline were spotted at the center of the YPD agar plates, dried, and incubated at different temperatures for the designated duration before taking a picture.

Figure 4. Aerobic and anaerobic growth of Mucor sp. 1-3 and 5-3.

Figure 5. Photomicrographs of Mucorales grown on slide culture. Mucor sp. 1-3 (a), 5-3 (b), S286-1101 (c), and Backusella sp. 827-14 (d). Mucorales were grown aerobically for 4 days. Bars = 20 µm.
5. Mucor Morphology under High Resolution
Using Mucor sp. 5-3, cell morphology of mycelial growth under aerobic conditions and yeast growth under anaerobic conditions were studied by Scanning Electron Microscope (SEM). Aerobic culture resulted in hyphal growth, where long elongated aseptate hyphae and sporangium were observed (Figure 6a,b). Inside the matured sporangium, a large number of sporangiospores were observed (Figure 6c). The tip of sporangiophore harbored columella and collarette (Figure 6d). The yeast colonies formed under anaerobic growth were made up of spherical cells of various sizes ranging from 2 to 30 µm in diameter (Figure 6e). Multipolar budding cells were observed in large cells (Figure 6e,f). Some budding cells had smooth surfaces, whereas some had small golf ball-like shapes (Figure 6f). Furthermore, strange cells with cleaved cell surfaces were often observed in large cells (Figure 6g). The existence of cells with cleaved surface layers has never been reported before. The significance of these cells in the growth of yeast cells needs to be investigated in more detail in the future.

Figure 6. SEM images of aerobic (a–d) and anaerobic (e–g) culture of Mucor sp. 5-3. (a) Low magnification image of mycelium; aseptate hyphae and sporangia. (b) Sporangia formed at the tip of the sporangiophore. (c) Sporangiospores inside the sporangia. (d) Columella (open star) and collarette (filled arrow) were observed at the tip of the sporangiophore. (e) Yeast-like colonies are composed of spherical cells of various sizes. (f) Multipolar budding found in large cells. (g) Cleavage of surface structure is observed in large cells. Mucor sp. 5-3 was grown aerobically (a–d) and anaerobically (e–g) on a YPD agar plate for 2 days. Bars: (a) = 100 µm, (b–g) = 10 µm.
6. Pathogenicity of Newly Isolated Mucorales
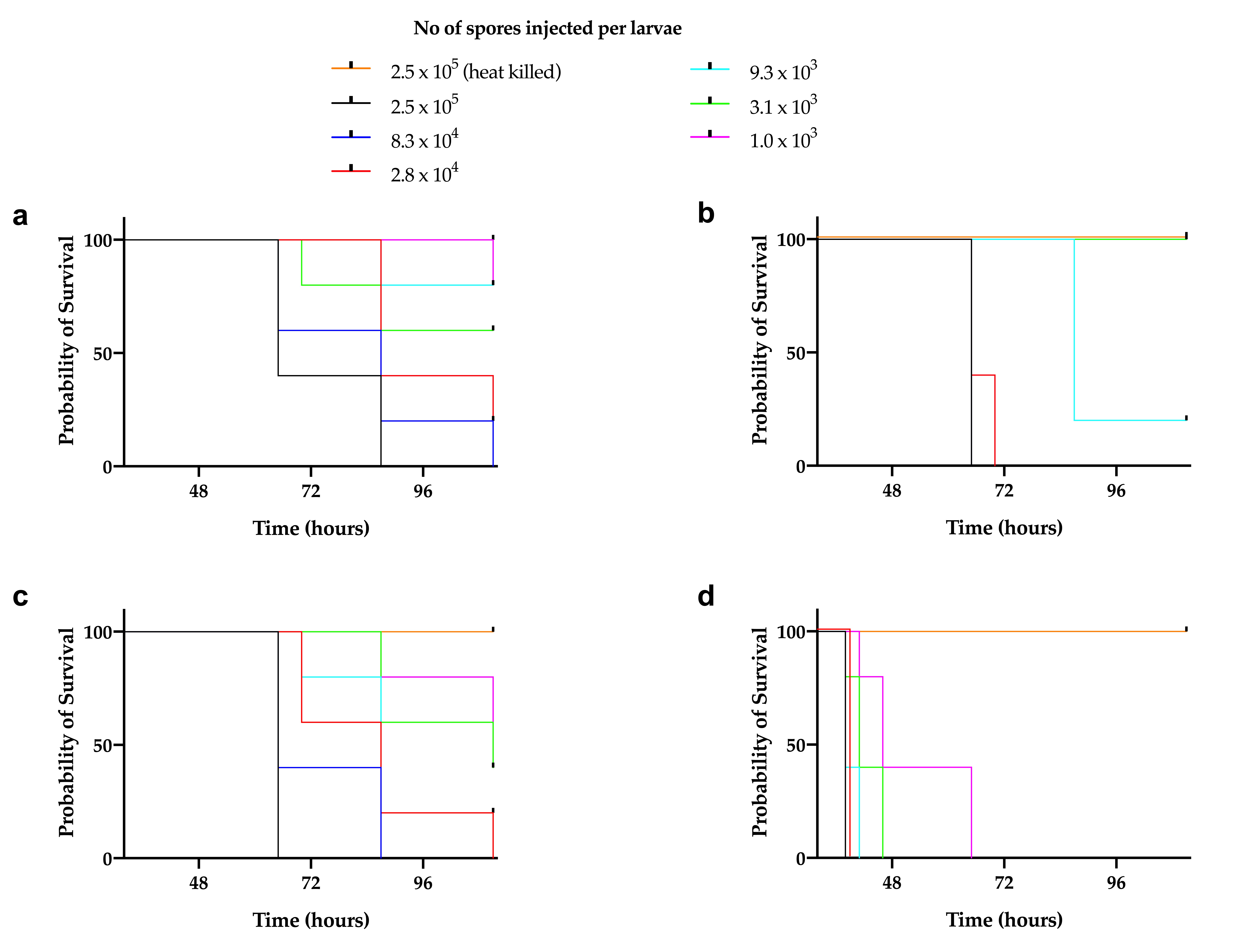
Although various vertebrates and invertebrates are used as animal models to study mucormycosis [26], silkworms were not used to test the pathogenicity of mucoralean fungi so far. Given that the use of the silkworm infection model for the test of pathogenicity of several bacteria and fungi is well established [27][28][29][30], we aimed to evaluate the pathogenicity of novel Mucorales using a silkworm infection model. First, we prepared a suspension of spores and examined the pathogenicity of the suspension without dilution. We found that all silkworms died at 20 h. Next, we prepared a 1/8—fold dilution of spore suspension, injected it into the silkworm, and checked the survival at 15 and 20 h. We found that at 15 h, most of the silkworms were surviving; however, the lethality of these Mucorales was high at 20 h, indicating the ability of these fungi to rapidly kill silkworms. These results indicated the pathogenicity of the spores of newly identified fungi against silkworms. Next, for a quantitative evaluation of pathogenicity, we serially diluted the spores and injected them into the silkworm hemolymph so that each silkworm received 2.5 × 105–1.0 × 103 spores. To confirm the establishment of infection, silkworms were inoculated with 2.5 × 105 heat-killed spores of each strain. Whereas the heat-killed spores were nonpathogenic to silkworms, a dose-dependent killing by live spores was observed (Figure 7a–d). We found that with the same dose injected, Backusella were more pathogenic compared to Mucor.

Figure 7. Pathogenicity of Mucorales spores in silkworm. Fifty microliters serially diluted spore suspensions of Mucor sp. 1-3 (a), Mucor sp. 5-3 (b), Mucor sp. S286-1101 (c), and Backusella sp. 827-14 (d) were injected into the silkworm hemolymph (n = 5). Silkworms were incubated at 27 °C and survival was recorded at various time points.
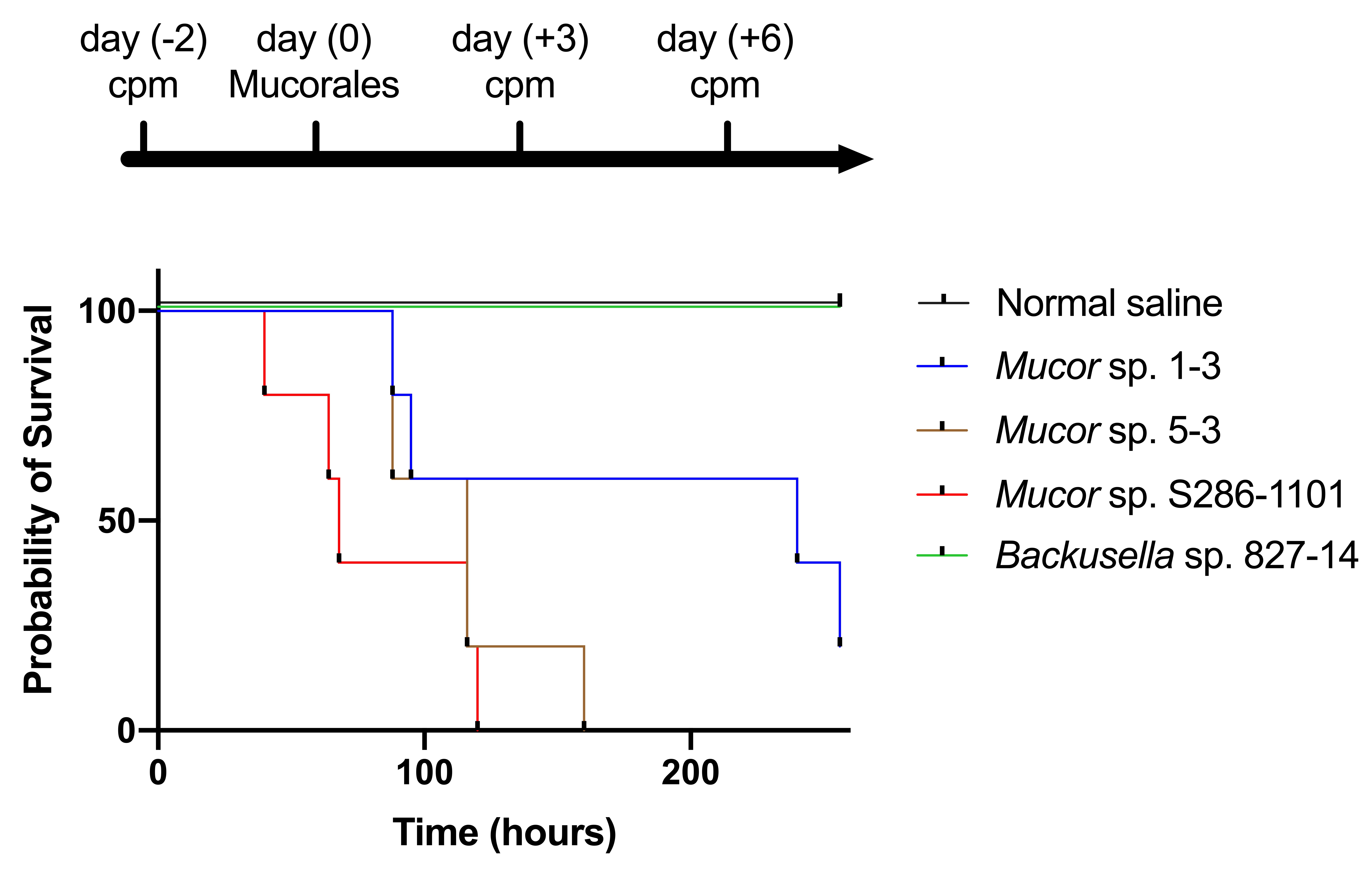
Next, we tested the ability of these strains to infect mammalian hosts using an immunocompromised mouse model. The result showed that three out of four strains could kill the mouse within four days of the infection suggesting their pathogenicity to mammals (Figure 8).

Figure 8. Pathogenicity of Mucorales spores in mice. Four weeks old ICR female mice (n = 5) were immunocompromised by injecting cyclophosphamide (cpm) and injected with 2.0 × 108 colony-forming units of Mucorales spores through the intraperitoneal route and survival was recorded at various time points.
References
- Phookamsak, R.; Hyde, K.D.; Jeewon, R.; Bhat, D.J.; Jones, E.B.G.; Maharachchikumbura, S.S.N.; Raspé, O.; Karunarathna, S.C.; Wanasinghe, D.N.; Hongsanan, S.; et al. Fungal diversity notes 929–1035: Taxonomic and phylogenetic contributions on genera and species of fungi. Fungal Divers. 2019, 95, 1–273.
- Sanguinetti, M.; Posteraro, B.; Beigelman-Aubry, C.; Lamoth, F.; Dunet, V.; Slavin, M.; Richardson, M.D. Diagnosis and treatment of invasive fungal infections: Looking ahead. J. Antimicrob. Chemother. 2019, 74, ii27–ii37.
- Roden, M.M.; Zaoutis, T.E.; Buchanan, W.L.; Knudsen, T.A.; Sarkisova, T.A.; Schaufele, R.L.; Sein, M.; Sein, T.; Chiou, C.C.; Chu, J.H.; et al. Epidemiology and outcome of zygomycosis: A review of 929 reported cases. Clin. Infect. Dis. 2005, 41, 634–653.
- Petrikkos, G.; Skiada, A.; Lortholary, O.; Roilides, E.; Walsh, T.J.; Kontoyiannis, D.P. Epidemiology and clinical manifestations of mucormycosis. Clin. Infect. Dis. 2012, 54 (Suppl. 1), S23–S34.
- Honavar, S.G. Code mucor: Guidelines for the diagnosis, staging and management of rhino-orbito-cerebral mucormycosis in the setting of COVID-19. Indian J. Ophthalmol. 2021, 69, 1361–1365.
- Rao, R.; Shetty, A.P.; Nagesh, C.P. Orbital infarction syndrome secondary to rhino-orbital mucormycosis in a case of COVID-19: Clinico-radiological features. Indian J. Ophthalmol. 2021, 69, 1627–1630.
- Sarkar, S.; Gokhale, T.; Choudhury, S.S.; Deb, A.K. COVID-19 and orbital mucormycosis. Indian J. Ophthalmol. 2021, 69, 1002–1004.
- Kaerger, K.; Schwartze, V.U.; Dolatabadi, S.; Nyilasi, I.; Kovacs, S.A.; Binder, U.; Papp, T.; Hoog, S.; Jacobsen, I.D.; Voigt, K. Adaptation to thermotolerance in Rhizopus coincides with virulence as revealed by avian and invertebrate infection models, phylogeny, physiological and metabolic flexibility. Virulence 2015, 6, 395–403.
- Desai, R.P.; Joseph, N.M.; Ananthakrishnan, N.; Ambujam, S. Subcutaneous zygomycosis caused by Mucor hiemalis in an immunocompetent patient. Australas. Med. J. 2013, 6, 374–377.
- Prevoo, R.L.; Starink, T.M.; de Haan, P. Primary cutaneous mucormycosis in a healthy young girl. Report of a case caused by Mucor hiemalis Wehmer. J. Am. Acad. Derm. 1991, 24, 882–885.
- De Oliveira-Neto, M.P.; Da Silva, M.; Fialho Monteiro, P.C.; Lazera, M.; de Almeida Paes, R.; Novellino, A.B.; Cuzzi, T. Cutaneous mucormycosis in a young, immunocompetent girl. Med. Mycol. 2006, 44, 567–570.
- Costa, A.R.; Porto, E.; Tayah, M.; Valente, N.Y.; Lacaz Cda, S.; Maranhao, W.M.; Rodrigues, M.C. Subcutaneous mucormycosis caused by Mucor hiemalis Wehmer f. luteus (Linnemann) Schipper 1973. Mycoses 1990, 33, 241–246.
- Walther, G.; Wagner, L.; Kurzai, O. Updates on the taxonomy of mucorales with an emphasis on clinically important taxa. J. Fungi 2019, 5, 106.
- Ribes, J.A.; Vanover-Sams, C.L.; Baker, D.J. Zygomycetes in human disease. Clin. Microbiol. Rev. 2000, 13, 236–301.
- Alqarihi, A.; Gebremariam, T.; Gu, Y.; Swidergall, M.; Alkhazraji, S.; Soliman, S.S.M.; Bruno, V.M.; Edwards, J.E., Jr.; Filler, S.G.; Uppuluri, P.; et al. GRP78 and integrins play different roles in host cell invasion during mucormycosis. mBio 2020, 11, e01087-20.
- Ibrahim, A.S.; Spellberg, B.; Edwards, J., Jr. Iron acquisition: A novel perspective on mucormycosis pathogenesis and treatment. Curr. Opin. Infect. Dis. 2008, 21, 620–625.
- Canturk, Z.; Cetinarslan, B.; Tarkun, I.; Canturk, N.Z. Serum ferritin levels in poorly- and well-controlled diabetes mellitus. Endocr. Res. 2003, 29, 299–306.
- Cavezzi, A.; Troiani, E.; Corrao, S. COVID-19: Hemoglobin, iron, and hypoxia beyond inflammation. A narrative review. Clin. Pr. 2020, 10, 1271.
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547–1549.
- Katoh, K.; Rozewicki, J.; Yamada, K.D. MAFFT online service: Multiple sequence alignment, interactive sequence choice and visualization. Brief. Bioinform. 2019, 20, 1160–1166.
- Edler, D.; Klein, J.; Antonelli, A.; Silvestro, D. raxmlGUI 2.0: A graphical interface and toolkit for phylogenetic analyses using RAxML. Methods Ecol. Evol. 2021, 12, 373–377.
- Panthee, S.; Paudel, A.; Blom, J.; Hamamoto, H.; Sekimizu, K. Complete genome sequence of weissella hellenica 0916-4-2 and its comparative genomic analysis. Front. Microbiol. 2019, 10, 1619.
- Panthee, S.; Paudel, A.; Hamamoto, H.; Ogasawara, A.A.; Iwasa, T.; Blom, J.; Sekimizu, K. Complete genome sequence and comparative genomic analysis of Enterococcus faecalis EF-2001, a probiotic bacterium. Genomics 2021, 113, 1534–1542.
- Panthee, S.; Hamamoto, H.; Ishijima, S.A.; Paudel, A.; Sekimizu, K. Utilization of hybrid assembly approach to determine the genome of an opportunistic pathogenic fungus, candida albicans TIMM 1768. Genome Biol. Evol. 2018, 10, 2017–2022.
- Lebreton, A.; Corre, E.; Jany, J.L.; Brillet-Gueguen, L.; Perez-Arques, C.; Garre, V.; Monsoor, M.; Debuchy, R.; Le Meur, C.; Coton, E.; et al. Comparative genomics applied to Mucor species with different lifestyles. BMC Genom. 2020, 21, 135.
- Jacobsen, I.D. Animal models to study mucormycosis. J. Fungi 2019, 5, 27.
- Paudel, A.; Panthee, S.; Hamamoto, H.; Grunert, T.; Sekimizu, K. YjbH regulates virulence genes expression and oxidative stress resistance in Staphylococcus aureus. Virulence 2021, 12, 470–480.
- Paudel, A.; Hamamoto, H.; Panthee, S.; Matsumoto, Y.; Sekimizu, K. Large-Scale screening and identification of novel pathogenic Staphylococcus aureus genes using a silkworm infection model. J. Infect. Dis. 2020, 221, 1795–1804.
- Panthee, S.; Paudel, A.; Hamamoto, H.; Sekimizu, K. Advantages of the silkworm as an animal model for developing novel antimicrobial agents. Front. Microbiol. 2017, 8, 373.
- Kaito, C.; Akimitsu, N.; Watanabe, H.; Sekimizu, K. Silkworm larvae as an animal model of bacterial infection pathogenic to humans. Microb. Pathog. 2002, 32, 183–190.
More
Information
Subjects:
Mycology; Microbiology; Biology
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.2K
Revisions:
2 times
(View History)
Update Date:
14 Dec 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





