
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | John Matsoukas | + 1965 word(s) | 1965 | 2021-12-06 07:01:54 | | | |
| 2 | Jessie Wu | + 284 word(s) | 2249 | 2021-12-10 02:29:23 | | |
Video Upload Options
Multiple Sclerosis (MS) is a serious autoimmune disease. The patient in an advanced state of the disease has restrained mobility and remains handicapped. It is therefore understandable that there is a great need for novel drugs and vaccines for the treatment of MS.
1. Introduction
Multiple Sclerosis (MS) is a serious systemic demyelinating disease that leads to the partial paralysis of the patient who needs the assistance of medicare in order to survive with low quality of life. The need for an effective treatment in the form of medication or vaccine is more urgent than ever before [1][2][3][4][5]. This chronic inflammatory and neurodegenerative disease is initiated by autoreactive T helper (Th) cells and affects approximately 2.5 million people worldwide. Thus, there is an urgent need to develop effective treatments. Advances in the immunotherapy of MS have been recently reported in excellent reviews [6][7][8][9]. The pathogenesis of MS has been extensively studied over the last years, which shows a complex immunological involvement. Myelin epitopes have been identified as a target for autoimmune CD4+ T cells and antibodies, and much focus has been around the modulation of Th1 pro-inflammatory autoreactive CD4+ T cells against myelin epitopes, namely myelin basic protein (MBP), proteolipid protein (PLP), and myelin oligodendrocyte glycoprotein (MOG) [10][11][12][13][14][15][16][17][18].
2. Applied Strategies Utilized against MS
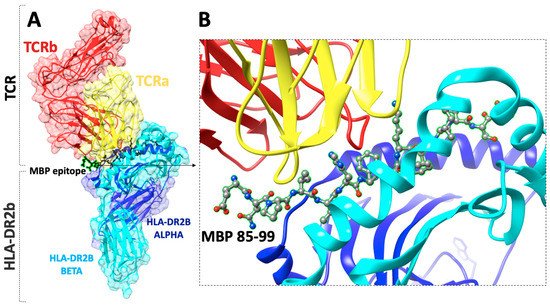
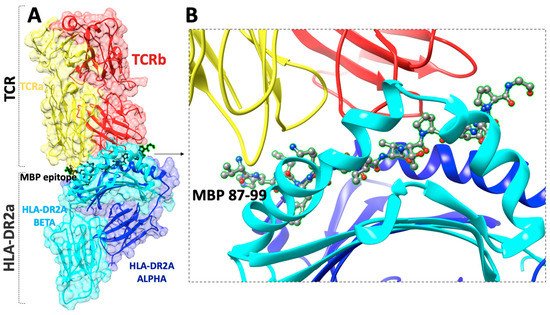
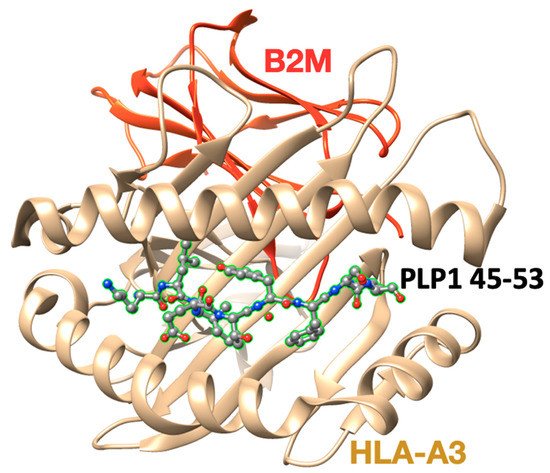
2.1. Linear Epitopes and Derivatives of Selective MBP Epitopes
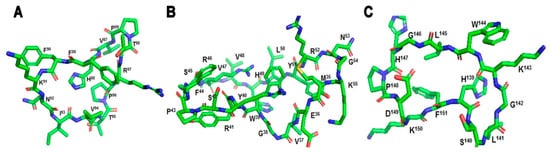
2.2. Cyclization of Selective Linear Epitopes and Their Derivatives
2.3. Conjugation of Selective Epitopes with Mannan
| Peptide Analog [Reference] | Major Effects |
|---|---|
| MBP83–99 and PLP139–151 [10][11][12][13][14][15][16][17][18] | These agonist peptides are involved in the pathophysiology of MS and also induce EAE in animal models. |
| MBP82–98 [20][21] | Dirucotide in animal models inhibits disease and in early human clinical trials showed efficacy; however, the peptide did not meet primary endpoints in phase III-trials. |
| cyclic(87–99)[MBP87–99] [27] | Stimulates Th2 cytokines and inhibits EAE in mice. |
| MBP87–99(R91,A96), MBP87–99(A91,A96) [26] | Induces IL-4 and antagonizes IFNγ responses in mice. |
| MBP72–85 [22] | These agonist peptides induce EAE in mice and Th1 responses in humans. |
| MBP72–85(A79) [22] | Suppresses EAE in mice. |
| PLP139–151(L144, R147) [23] | Antagonizes PLP-specific T-clones in vitro. |
| cyclic-MBP82–98 | Exerts strong binding to the HLA-DR2 and lowers binding to the HLA-DR4 allele in vitro |
| cyclic-MBP87–99(A96) or (R91A96) [26][19] | Suppresses proliferation of CD4+ T cells and exerts IL-10 selectivity in vitro. Binds to HLA-DR4 and is stable to lysosomal enzymes and cathepsins B, D, and H. |
| cyclic-MOG35–55 [31] cyclic-PLP131–151 [23] linear and cyclic-MBP83–99(A91,A96) [69] Mannan-linear and cyclic-MBP83–99(A91,A96) [69] |
Reduces EAE, demyelination, and chronic axonopathy in acute and chronic phases of EAE in mice. Low disease burden in regards to EAE in mice with minimal inflammatory, demyelinating, and axonopathic pathology compared to its linear counterpart. Decreases IFNγ responses in mice. Diverts the “bad” IFNγ to “good” IL-4 cytokine in mice. |
| Mannan-MOG35–55 [70] | Protects mice against EAE in prophylactic and therapeutic protocols, with oxidized-conjugated peptides giving the best results. |
| Cyclo(87–99)MBP87-99(A91,A96) [69][19] Mannan-cyclo(87–99)MBP87-99(A91,A96) [69][19] |
Decreases Th1 responses. Shifts Th1 responses to Th2 responses. |
| MBP87–99[Cit91,A96,Cit97] [29] cyclic-MBP87–99[Cit91,A96,Cit97] [29] |
Induces T-cell proliferation and IFNγ secretion in mice. Activates T cells and increases IFNγ secretion in mice. |
References
- Compston, A.; Coles, A. Multiple sclerosis. Lancet 2002, 359, 1221–1231.
- Steinman, M.D.L. Multiple Sclerosis: A Coordinated Immunological Attack against Myelin in the Central Nervous System. Cell 1996, 85, 299–302.
- Grytten, N.; Torkildsen, Ø.; Myhr, K.M. Time trends in the incidence and prevalence of multiple sclerosis in Norway during eight decades. Acta Neurol. Scand. 2015, 132, 29–36.
- Polman, C.H.; Reingold, S.C.; Banwell, B.; Clanet, M.; Cohen, J.A.; Filippi, M.; Fujihara, K.; Havrdova, E.; Hutchinson, M.; Kappos, L.; et al. Diagnostic criteria for multiple sclerosis: 2010 Revisions to the McDonald criteria. Ann. Neurol. 2011, 69, 292–302.
- Eckstein, C.; Bhatti, M.T. Currently approved and emerging oral therapies in multiple sclerosis: An update for the ophthalmologist. Surv. Ophthalmol. 2016, 61, 318–332.
- Dargahi, N.; Katsara, M.; Tselios, T.; Androutsou, M.E.; de Courten, M.; Matsoukas, J.; Apostolopoulos, V. Multiple Sclerosis: Immunopathology and Treatment Update. Brain Sci. 2017, 7, 78.
- Florou, D.; Katsara, M.; Feehan, J.; Dardiotis, E.; Apostolopoulos, V. Anti-CD20 Agents for Multiple Sclerosis: Spotlight on Ocrelizumab and Ofatumumab. Brain Sci. 2020, 10, 758.
- Kammona, O.; Kiparissides, C. Recent Advances in Antigen-Specific Immunotherapies for the Treatment of Multiple Sclerosis. Brain Sci. 2020, 10, 333.
- Metaxakis, A.; Petratou, D.; Tavernarakis, N. Molecular Interventions towards Multiple Sclerosis Treatment. Brain Sci. 2020, 10, 299.
- Dai, H.; Ciric, B.; Zhang, G.-X.; Rostami, A. Interleukin-10 plays a crucial role in suppression of experimental autoimmune encephalomyelitis by Bowman–Birk inhibitor. J. Neuroimmunol. 2012, 245, 1–7.
- Rostami, A.; Gregorian, S.K. Peptide 53–78 of myelin P2 protein is a T cell epitope for the induction of experimental autoimmune neuritis. Cell. Immunol. 1991, 132, 433–441.
- Rostami, A.; Gregorian, S.K.; Brown, M.J.; Pleasure, D.E. Induction of severe experimental autoimmune neuritis with a synthetic peptide corresponding to the 53–78 amino acid sequence of the myelin P2 protein. J. Neuroimmunol. 1990, 30, 145–151.
- Martin, R.; McFarland, H.F.; McFarlin, D.E. Immunological Aspects of Demyelinating Diseases. Annu. Rev. Immunol. 1992, 10, 153–187.
- Hafler, D.A.; Weiner, H.L. Immunologic Mechanisms and Therapy in Multiple Sclerosis. Immunol. Rev. 1995, 144, 75–107.
- Kappos, L.; Comi, G.; Panitch, H.; Oger, J.; Antel, J.; Conlon, P.; Steinman, L.; Comi, G.; Kappos, L.; Oger, J.; et al. Induction of a non-encephalitogenic type 2 T helper-cell autoimmune response in multiple sclerosis after administration of an altered peptide ligand in a placebo-controlled, randomized phase II trial. Nat. Med. 2000, 6, 1176–1182.
- Bielekova, B.; Goodwin, B.; Richert, N.; Cortese, I.; Kondo, T.; Afshar, G.; Gran, B.; Eaton, J.; Antel, J.; Frank, J.A.; et al. Encephalitogenic potential of the myelin basic protein peptide (amino acids 83–99) in multiple sclerosis: Results of a phase II clinical trial with an altered peptide ligand. Nat. Med. 2000, 6, 1167–1175.
- Anagnostouli, M.; Artemiadis, A.; Gontika, M.; Skarlis, C.; Markoglou, N.; Katsavos, S.; Kilindireas, K.; Doxiadis, I.; Stefanis, L. HLA-DPB1*03 as Risk Allele and HLA-DPB1*04 as Protective Allele for Both Early- and Adult-Onset Multiple Sclerosis in a Hellenic Cohort. Brain Sci. 2020, 10, 374.
- Vergelli, M.; Hemmer, B.; Utz, U.; Vogt, A.; Kalbus, M.; Tranquill, L.; Conlon, P.; Ling, N.; Steinman, L.; McFarland, H.F.; et al. Differential activation of human autoreactive T cell clones by altered peptide ligands derived from myelin basic protein peptide (87–99). Eur. J. Immunol. 1996, 26, 2624–2634.
- Katsara, M.; Deraos, G.; Tselios, T.; Matsoukas, J.; Apostolopoulos, V. Design of Novel Cyclic Altered Peptide Ligands of Myelin Basic Protein MBP83−99That Modulate Immune Responses in SJL/J Mice. J. Med. Chem. 2008, 51, 3971–3978.
- Warren, K.G.; Catz, I.; Ferenczi, L.Z.; Krantz, M.J. Intravenous synthetic peptide MBP8298 delayed disease progression in an HLA Class II-defined cohort of patients with progressive multiple sclerosis: Results of a 24-month double-blind placebo-controlled clinical trial and 5 years of follow-up treatment. Eur. J. Neurol. 2006, 13, 887–895.
- Freedman, M.S.; Bar-Or, A.; Oger, J.; Traboulsee, A.; Patry, D.; Young, C.; Olsson, T.; Li, D.; Hartung, H.P.; Krantz, M.; et al. A phase III study evaluating the efficacy and safety of MBP8298 in secondary progressive MS. Neurology 2011, 77, 1551–1560.
- Tselios, T.; Probert, L.; Daliani, I.; Matsoukas, E.; Troganis, A.; Gerothanassis, I.P.; Mavromoustakos, T.; Moore, G.J.; Matsoukas, J.M. Design and Synthesis of a Potent Cyclic Analogue of the Myelin Basic Protein Epitope MBP72-85: Importance of the Ala81 Carboxyl Group and of a Cyclic Conformation for Induction of Experimental Allergic Encephalomyelitis. J. Med. Chem. 1999, 42, 1170–1177.
- Lourbopoulos, A.; Matsoukas, M.-T.; Katsara, M.; Deraos, G.; Giannakopoulou, A.; Lagoudaki, R.; Grigoriadis, N.; Matsoukas, J.; Apostolopoulos, V. Cyclization of PLP139-151 peptide reduces its encephalitogenic potential in experimental autoimmune encephalomyelitis. Bioorganic Med. Chem. 2018, 26, 2221–2228.
- Katsara, M.; Tselios, T.; Deraos, S.; Deraos, G.; Matsoukas, M.T.; Lazoura, E.; Matsoukas, J.; Apostolopoulos, V. Round and round we go: Cyclic peptides in disease. Curr. Med. Chem. 2006, 13, 2221–2232.
- Katsara, M.; Deraos, G.; Tselios, T.; Matsoukas, M.-T.; Friligou, I.; Matsoukas, J.; Apostolopoulos, V. Design and Synthesis of a Cyclic Double Mutant Peptide (cyclo(87−99)MBP87−99) Induces Altered Responses in Mice after Conjugation to Mannan: Implications in the Immunotherapy of Multiple Sclerosis. J. Med. Chem. 2008, 52, 214–218.
- Katsara, M.; Yuriev, E.; Ramsland, P.A.; Deraos, G.; Tselios, T.; Matsoukas, J.; Apostolopoulos, V. A double mutation of MBP83–99 peptide induces IL-4 responses and antagonizes IFN-γ responses. J. Neuroimmunol. 2008, 200, 77–89.
- Matsoukas, J.; Apostolopoulos, V.; Kalbacher, H.; Papini, A.-M.; Tselios, T.; Chatzantoni, K.; Biagioli, T.; Lolli, F.; Deraos, S.; Papathanassopoulos, P.; et al. Design And Synthesis of a Novel Potent Myelin Basic Protein Epitope 87−99 Cyclic Analogue: Enhanced Stability and Biological Properties of Mimics Render Them a Potentially New Class of Immunomodulators. J. Med. Chem. 2005, 48, 1470–1480.
- Deraos, G.; Rodi, M.; Kalbacher, H.; Chatzantoni, K.; Karagiannis, F.; Synodinos, L.; Plotas, P.; Papalois, A.; Dimisianos, N.; Papathanasopoulos, P.; et al. Properties of myelin altered peptide ligand cyclo(87-99)(Ala91,Ala96)MBP87-99 render it a promising drug lead for immunotherapy of multiple sclerosis. Eur. J. Med. Chem. 2015, 101, 13–23.
- Deraos, G.; Chatzantoni, K.; Matsoukas, M.-T.; Tselios, T.; Deraos, S.; Katsara, M.; Papathanasopoulos, P.; Vynios, D.; Apostolopoulos, V.; Mouzaki, A.; et al. Citrullination of Linear and Cyclic Altered Peptide Ligands from Myelin Basic Protein (MBP87−99) Epitope Elicits a Th1 Polarized Response by T Cells Isolated from Multiple Sclerosis Patients: Implications in Triggering Disease. J. Med. Chem. 2008, 51, 7834–7842.
- Deraos, G.; Kritsi, E.; Matsoukas, M.-T.; Christopoulou, K.; Kalbacher, H.; Zoumpoulakis, P.; Apostolopoulos, V.; Matsoukas, J. Design of Linear and Cyclic Mutant Analogues of Dirucotide Peptide (MBP82–98) against Multiple Sclerosis: Conformational and Binding Studies to MHC Class II. Brain Sci. 2018, 8, 213.
- Lourbopoulos, A.; Deraos, G.; Matsoukas, M.T.; Touloumi, O.; Giannakopoulou, A.; Kalbacher, H.; Grigoriadis, N.; Apostolopoulos, V.; Matsoukas, J. Cyclic MOG35-55 ameliorates clinical and neuropathological features of experimental autoimmune encephalomyelitis. Bioorg. Med. Chem. 2017, 25, 4163–4174.
- Apostolopoulos, V.; Barnes, N.; Pietersz, G.A.; McKenzie, I.F. Ex vivo targeting of the macrophage mannose receptor generates anti-tumor CTL responses. Vaccine 2000, 18, 3174–3184.
- Sheng, K.C.; Kalkanidis, M.; Pouniotis, D.S.; Esparon, S.; Tang, C.K.; Apostolopoulos, V.; Pietersz, G.A. Delivery of antigen using a novel mannosylated dendrimer potentiates immunogenicity in vitro and in vivo. Eur. J. Immunol. 2008, 38, 424–436.
- Sheng, K.C.; Kalkanidis, M.; Pouniotis, D.S.; Wright, M.D.; Pietersz, G.A.; Apostolopoulos, V. The adjuvanticity of a mannosylated antigen reveals TLR4 functionality essential for subset specialization and functional maturation of mouse dendritic cells. J. Immunol. 2008, 181, 2455–2464.
- Sheng, K.C.; Pouniotis, D.S.; Wright, M.D.; Tang, C.K.; Lazoura, E.; Pietersz, G.A.; Apostolopoulos, V. Mannan derivatives induce phenotypic and functional maturation of mouse dendritic cells. Immunology 2006, 118, 372–383.
- Tang, C.K.; Sheng, K.C.; Apostolopoulos, V.; Pietersz, G.A. Protein/peptide and DNA vaccine delivery by targeting C-type lectin receptors. Expert Rev. Vaccines 2008, 7, 1005–1018.
- Tang, C.K.; Sheng, K.C.; Pouniotis, D.; Esparon, S.; Son, H.Y.; Kim, C.W.; Pietersz, G.A.; Apostolopoulos, V. Oxidized and reduced mannan mediated MUC1 DNA immunization induce effective anti-tumor responses. Vaccine 2008, 26, 3827–3834.
- Acres, B.; Apostolopoulos, V.; Balloul, J.M.; Wreschner, D.; Xing, P.X.; Ali-Hadji, D.; Bizouarne, N.; Kieny, M.P.; McKenzie, I.F. MUC1-specific immune responses in human MUC1 transgenic mice immunized with various human MUC1 vaccines. Cancer Immunol. Immunother. 2000, 48, 588–594.
- Apostolopoulos, V.; Lofthouse, S.A.; Popovski, V.; Chelvanayagam, G.; Sandrin, M.S.; McKenzie, I.F. Peptide mimics of a tumor antigen induce functional cytotoxic T cells. Nat. Biotechnol. 1998, 16, 276–280.
- Apostolopoulos, V.; McKenzie, I.F. Role of the mannose receptor in the immune response. Curr. Mol. Med. 2001, 1, 469–474.
- Apostolopoulos, V.; Pietersz, G.A.; Gordon, S.; Martinez-Pomares, L.; McKenzie, I.F. Aldehyde-mannan antigen complexes target the MHC class I antigen-presentation pathway. Eur. J. Immunol. 2000, 30, 1714–1723.
- Apostolopoulos, V.; Pietersz, G.A.; Loveland, B.E.; Sandrin, M.S.; McKenzie, I.F. Oxidative/reductive conjugation of mannan to antigen selects for T1 or T2 immune responses. Proc. Natl. Acad. Sci. USA 1995, 92, 10128–10132.
- Apostolopoulos, V.; Pietersz, G.A.; McKenzie, I.F. Cell-mediated immune responses to MUC1 fusion protein coupled to mannan. Vaccine 1996, 14, 930–938.
- Davis, W.C.; Konzek, R.L.; Haas, K.; Estes, D.M.; Hamilton, M.J.; Call, D.R.; Apostolopoulos, V.; McKenzie, I.F. Use of the mannan receptor to selectively target vaccine antigens for processing and antigen presentation through the MHC class I and class II pathways. Ann. N. Y. Acad. Sci. 2002, 969, 119–125.
- Lees, C.J.; Apostolopoulos, V.; Acres, B.; Ong, C.S.; Popovski, V.; McKenzie, I.F. The effect of T1 and T2 cytokines on the cytotoxic T cell response to mannan-MUC1. Cancer Immunol. Immunother. 2000, 48, 644–652.
- Lees, C.J.; Apostolopoulos, V.; Acres, B.; Ramshaw, I.; Ramsay, A.; Ong, C.S.; McKenzie, I.F. Immunotherapy with mannan-MUC1 and IL-12 in MUC1 transgenic mice. Vaccine 2000, 19, 158–162.
- Lees, C.J.; Apostolopoulos, V.; McKenzie, I.F. Cytokine production from murine CD4 and CD8 cells after mannan-MUC1 immunization. J. Interferon Cytokine Res. 1999, 19, 1373–1379.
- Lofthouse, S.A.; Apostolopoulos, V.; Pietersz, G.A.; Li, W.; McKenzie, I.F. Induction of T1 (cytotoxic lymphocyte) and/or T2 (antibody) responses to a mucin-1 tumour antigen. Vaccine 1997, 15, 1586–1593.
- Tang, C.K.; Lodding, J.; Minigo, G.; Pouniotis, D.S.; Plebanski, M.; Scholzen, A.; McKenzie, I.F.; Pietersz, G.A.; Apostolopoulos, V. Mannan-mediated gene delivery for cancer immunotherapy. Immunology 2007, 120, 325–335.
- Sandrin, M.S.; Vaughan, H.A.; Xing, P.X.; McKenzie, I.F. Natural human anti-Gal alpha(1,3)Gal antibodies react with human mucin peptides. Glycoconj. J. 1997, 14, 97–105.
- Vaughan, H.A.; Ho, D.W.; Karanikas, V.; Sandrin, M.S.; McKenzie, I.F.; Pietersz, G.A. The immune response of mice and cynomolgus monkeys to macaque mucin 1-mannan. Vaccine 2000, 18, 3297–3309.
- Vaughan, H.A.; Ho, D.W.; Karanikas, V.A.; Ong, C.S.; Hwang, L.A.; Pearson, J.M.; McKenzie, I.F.; Pietersz, G.A. Induction of humoral and cellular responses in cynomolgus monkeys immunised with mannan-human MUC1 conjugates. Vaccine 1999, 17, 2740–2752.
- Apostolopoulos, V.; Osinski, C.; McKenzie, I.F. MUC1 cross-reactive Gal alpha(1,3)Gal antibodies in humans switch immune responses from cellular to humoral. Nat. Med. 1998, 4, 315–320.
- Apostolopoulos, V.; Pietersz, G.A.; Tsibanis, A.; Tsikkinis, A.; Drakaki, H.; Loveland, B.E.; Piddlesden, S.J.; Plebanski, M.; Pouniotis, D.S.; Alexis, M.N.; et al. Pilot phase III immunotherapy study in early-stage breast cancer patients using oxidized mannan-MUC1 . Breast Cancer Res. 2006, 8, R27.
- Apostolopoulos, V.; Pietersz, G.A.; Tsibanis, A.; Tsikkinis, A.; Stojanovska, L.; McKenzie, I.F.; Vassilaros, S. Dendritic cell immunotherapy: Clinical outcomes. Clin. Transl. Immunology 2014, 3, e21.
- Karanikas, V.; Hwang, L.A.; Pearson, J.; Ong, C.S.; Apostolopoulos, V.; Vaughan, H.; Xing, P.X.; Jamieson, G.; Pietersz, G.; Tait, B.; et al. Antibody and T cell responses of patients with adenocarcinoma immunized with mannan-MUC1 fusion protein. J. Clin. Investig. 1997, 100, 2783–2792.
- Karanikas, V.; Lodding, J.; Maino, V.C.; McKenzie, I.F. Flow cytometric measurement of intracellular cytokines detects immune responses in MUC1 immunotherapy. Clin. Cancer Res. 2000, 6, 829–837.
- Karanikas, V.; Thynne, G.; Mitchell, P.; Ong, C.S.; Gunawardana, D.; Blum, R.; Pearson, J.; Lodding, J.; Pietersz, G.; Broadbent, R.; et al. Mannan Mucin-1 Peptide Immunization: Influence of Cyclophosphamide and the Route of Injection. J. Immunother. 2001, 24, 172–183.
- Loveland, B.E.; Zhao, A.; White, S.; Gan, H.; Hamilton, K.; Xing, P.X.; Pietersz, G.A.; Apostolopoulos, V.; Vaughan, H.; Karanikas, V.; et al. Mannan-MUC1-pulsed dendritic cell immunotherapy: A phase I trial in patients with adenocarcinoma. Clin. Cancer Res. 2006, 12, 869–877.
- Mitchell, P.L.; Quinn, M.A.; Grant, P.T.; Allen, D.G.; Jobling, T.W.; White, S.C.; Zhao, A.; Karanikas, V.; Vaughan, H.; Pietersz, G.; et al. A phase 2, single-arm study of an autologous dendritic cell treatment against mucin 1 in patients with advanced epithelial ovarian cancer. J. Immunother. Cancer 2014, 2, 16.
- Prince, H.M.; Wall, D.M.; Ritchie, D.; Honemann, D.; Harrrison, S.; Quach, H.; Thompson, M.; Hicks, R.; Lau, E.; Davison, J.; et al. In vivo tracking of dendritic cells in patients with multiple myeloma. J. Immunother. 2008, 31, 166–179.
- Vassilaros, S.; Tsibanis, A.; Tsikkinis, A.; Pietersz, G.A.; McKenzie, I.F.; Apostolopoulos, V. Up to 15-year clinical follow-up of a pilot Phase III immunotherapy study in stage II breast cancer patients using oxidized mannan-MUC1. Immunotherapy 2013, 5, 1177–1182.
- Agnes, M.C.; Tan, A.; Jordens, R.; Geluk, A.; Roep, B.O.; Ottenhoff, T.; Drijfhout, J.W.; Koning, F. Strongly increased efficiency of altered peptide ligands by mannosylation. Int. Immunol. 1998, 10, 1299–1304.
- Chen, J.; Fang, H.; Hu, Y.; Wu, J.; Zhang, S.; Feng, Y.; Lin, L.; Tian, H.; Chen, X. Combining mannose receptor mediated nanovaccines and gene regulated PD-L1 blockade for boosting cancer immunotherapy. Bioact. Mater. 2022, 7, 167–180.
- Mommaas, A.M.; Mulder, A.A.; Jordens, R.; Out, C.; Tan, M.C.; Cresswell, P.; Kluin, P.M.; Koning, F. Human epidermal Langerhans cells lack functional mannose receptors and a fully developed endosomal/lysosomal compartment for loading of HLA class II molecules. Eur. J. Immunol. 1999, 29, 571–580.
- Motoyama, K.; Mitsuyasu, R.; Akao, C.; Abu, H., II; Sato, N.; Tanaka, T.; Higashi, T.; Arima, H. Potential Use of Thioalkylated Mannose-Modified Dendrimer (G3)/alpha-Cyclodextrin Conjugate as an NF-kappaB siRNA Carrier for the Treatment of Fulminant Hepatitis. Mol. Pharm. 2015, 12, 3129–3136.
- Motoyama, K.; Mitsuyasu, R.; Akao, C.; Tanaka, T.; Ohyama, A.; Sato, N.; Higashi, T.; Arima, H. Design and evaluation of thioalkylated mannose-modified dendrimer (G3)/alpha-cyclodextrin conjugates as antigen-presenting cell-selective siRNA carriers. AAPS J. 2014, 16, 1298–1308.
- Tan, M.C.; Mommaas, A.M.; Drijfhout, J.W.; Jordens, R.; Onderwater, J.J.; Verwoerd, D.; Mulder, A.A.; van der Heiden, A.N.; Ottenhoff, T.H.; Cella, M.; et al. Mannose receptor mediated uptake of antigens strongly enhances HLA-class II restricted antigen presentation by cultured dendritic cells. Adv. Exp. Med. Biol. 1997, 417, 171–174.
- Katsara, M.; Yuriev, E.; Ramsland, P.A.; Deraos, G.; Tselios, T.; Matsoukas, J.; Apostolopoulos, V. Mannosylation of mutated MBP83–99 peptides diverts immune responses from Th1 to Th2. Mol. Immunol. 2008, 45, 3661–3670.
- Tseveleki, V.; Tselios, T.; Kanistras, I.; Koutsoni, O.; Karamita, M.; Vamvakas, S.-S.; Apostolopoulos, V.; Dotsika, E.; Matsoukas, J.; Lassmann, H.; et al. Mannan-conjugated myelin peptides prime non-pathogenic Th1 and Th17 cells and ameliorate experimental autoimmune encephalomyelitis. Exp. Neurol. 2015, 267, 254–267.
- Dagkonaki, A.; Avloniti, M.; Evangelidou, M.; Papazian, I.; Kanistras, I.; Tseveleki, V.; Lampros, F.; Tselios, T.; Jensen, L.T.; Möbius, W.; et al. Mannan-MOG35-55 Reverses Experimental Autoimmune Encephalomyelitis, Inducing a Peripheral Type 2 Myeloid Response, Reducing CNS Inflammation, and Preserving Axons in Spinal Cord Lesions. Front. Immunol. 2020, 11.
- Degano, M.; Garcia, K.C.; Apostolopoulos, V.; Rudolph, M.G.; Teyton, L.; Wilson, I.A. A functional hot spot for antigen recognition in a superagonist TCR/MHC complex. Immunity 2000, 12, 251–261.





