2. Chemical Identification of Isolated Compounds
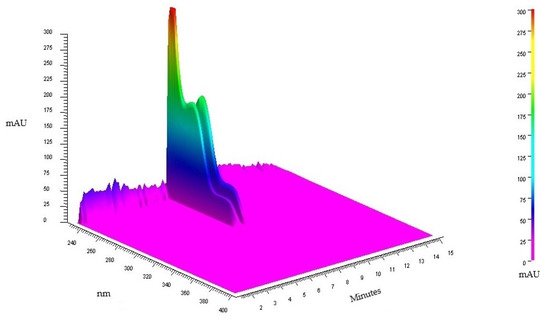
The qualitative analysis by HPLC-DAD of TAF-Ap indicated the presence of two alkaloids. The acquisition of the 3D chromatogram with scanning from 200 to 400 nm (Figure 1) pointed the presence of the alkaloids with absorption greater than 200 mAU and retention times of 7.68 and 7.78 min, respectively.
Figure 1. 3D chromatogram of an exploratory analysis of TAF-Ap.
The mass spectra of
1 and
2 showed molecular ion peaks at
m/
z = 385.44 (calcd
m/
z 384.241) and
m/
z = 415.45 (calcd
m/
z 414.252), which allowed us to manage the following structural formulas: C
23H
32N
2O
3 and C
24H
34N
2O
4, respectively. The UV spectrum showed three absorption bands at λ
max. 222, 256 and 285 nm (
Figure S1), suggestive of indoline chromophore
[11].
The
1H NMR data (500 MHz, acetone-d
6) (
Table 1) (
Figures S2–S5) showed four signals aromatic hydrogen at δ
H 7.16 (t,
J = 7.8, 1H), δ
H 7.06 (d,
J = 8.2 Hz, 1H), δ
H 7.00 (d,
J = 8.2 Hz, 1H) and δ
H 6.82 (dd,
J = 8.3 and 1.9 Hz, 2H). In addition to these signals, others were visualized between δ
H 3.86 and 0.68, suggestive of aliphatic hydrogens
[9][11].
Table 1. 1H and 13C NMR data of 15-methoxyaspidospermine (1) and 15-methoxypyrifolidine (2) (δ, acetone-d6, 500 and 125 MHz).
| Position |
1 |
2 |
| C |
δC |
δH |
δC |
δH |
| 2 |
67.67 |
4.93 (dd, J = 6.2 and 10.7 Hz, 1H) |
67.90 |
4.85 (dd, J = 6.1 and 10.6 Hz, 1H) |
| 3 |
52.83 |
3.26 (brd, J = 8.2 Hz, 1H) and 2.17 (m, 1H) |
52.70 |
3.26 (brd, J = 8.2 Hz, 1H) and 2.17 (m, 1H) |
| 5 |
52.80 |
3.37 (d, J = 6.8 Hz, 1H) and 2.36 (m, 1H) |
52.70 |
3.37 (d, J = 6.8 Hz, 1H) and 2.36 (m, 1H) |
| 6 |
37.71 |
2.06 (brt, J = 2.2 Hz, 1H) and 1.92 (dd, J = 3.8 and 15.2 Hz, 1H) |
37.35 |
(brt, J = 2.2 Hz, 1H) and 1.92 (dd, J = 3.8 and 15.2 Hz, 1H) |
| 7 |
52.80 |
- |
52.83 |
- |
| 8 |
141.65 |
- |
141.65 |
- |
| 9 |
110.38 |
6.82 (dd, J = 8.3 and 1.9 Hz, 2H) |
117.39 |
7.06 (d, J = 8.2 Hz, 1H) |
| 10 |
127.42 |
7.16 (t, J = 7.8, 1H) |
113.45 |
7.00 (d, J = 8.2 Hz, 1H) |
| 11 |
110.09 |
6.82 (dd, J = 8.3 and 1.9 Hz, 2H) |
154.66 |
- |
| 12 |
141.68 |
- |
150.10 |
- |
| 13 |
133.50 |
- |
134.33 |
- |
| 14 |
24.44 |
2.03 (m, 1H) and 1.59 (brd, J = 3.8 Hz, 1H) |
24.33 |
2.03 (m, 1H) and 1.59 (brd, J = 4.5 Hz, 1H) |
| 15 |
74.02 |
3.26 (brd, J = 9.0 Hz, 1H) |
74.50 |
3.17 (brd, J = 14.3 Hz, 1H) |
| 16 |
24.77 |
2.00 (m, 1H) and 1.35 (m, 1H) |
24.60 |
2.00 (m, 1H) and 1.35 (m, 1H) |
| 17 |
24.33 |
2.03 (m, 1H) and 1.37 (m, 1H) |
24.44 |
2.03 (m, 1H) and 1.37 (m, 1H) |
| 18 |
6.77 |
0.69 (t, J = 7.5 Hz, 3H) |
6.84 |
0.68 (t, J = 7.5 Hz, 3H) |
| 19 |
30.45 |
1.05 (q, J = 7.3 Hz, 2H) |
29.98 |
1.00 (q, J = 7.3 Hz, 2H) |
| 20 |
36.59 |
- |
36.57 |
- |
| 21 |
71.17 |
3.80 (s, 1H) |
71.15 |
3.81 (s, 1H) |
| 11-OCH3 |
- |
- |
56.53 |
3.83 (s, 3H) |
| 12-OCH3 |
56.01 |
3.84 (s, 3H) |
56.53 |
3.86 (s, 3H) |
| 15-OCH3 |
56.74 |
3.31 (s, 3H) |
56.74 |
3.31 (s, 3H) |
| NCOCH3 |
169.59 |
|
170.26 |
|
| NCOCH3 |
22.93 |
2.18 (s, 3H) |
22.98 |
2.19 (s, 3H) |
The
13C-NMR spectrum (125 MHz, acetone-d
6) (
Table 1) (
Figures S6 and S7) showed 31 signals: 9 from unhydrogenated carbons, 7 from CH carbons, 7 from CH
2 carbons and 8 from CH
3 carbons. The signals at δ
C 110.38, 110.09, 127.42, 117.39 and 113.45 are characteristic of aromatic methinic carbons. In addition, the signals at δ
C 141.65, 141.68, 133.50, 154.66, 150.10 and134.33 are consistent with non-hydrogenated aromatic carbons
[9][12].
The heteronuclear correlation map-HSQC (
Figures S8 and S9) showed the following correlations: δ
H 6.82/δ
C 110.38 and 110.09 (C-9 and C-11) and δ
H 7.16/δ
C 127.42 (C-10), suggestive of a tri-substituted aromatic nucleus. The correlations of δ
H 7.06/δ
C 117.39 (C-9) and δ
H 7.00/δ
C 113.45 (C-10) are suggestive of another tetra-substituted aromatic ring
[9]. The correlations between δ
H 3.84/δ
C 141.68 e δ
H 3.83/δ
C 154.66 e δ
H 3.86/δ
C 150.10, seen in the heteronuclear correlation map-HMBC (
Figures S10–S12), confirm the presence of methoxyl groups in C-11, C-11 and C-12 in the two aromatic nuclei, respectively. According to
[9], the signals at δ
C 169.59 and δ
C 170.26 suggested the presence of
N-acethyl carbonyl groups, in addition to a signal at δ
C 52.80 and 52.83, characteristic of C-7 indolic-ring quaternary carbon. These positions were confirmed by long-range heteronuclear correlation at δ
H 2.18/δ
C 169.59; δ
H 2.19/δ
C 170.26 and δ
H 6.82/δ
C 52.80; and δ
H 7.06/δ
C 52.83.
All the methylene carbons were displayed by 13C NMR and experiment APT and the hydrogens attached to them were revealed by chemical shifts, coupling constants and comparison with data from previous research. The signals at δC 71.17 and 71.15 were attributed to the methinic carbons of the position 21, respectively.
A signal at δ
H 3.31 (s, 6H) showed a correlation in the HSQC with δ
C 56.74 and suggested the presence of other methoxyl groups in the compounds. Methoxyl insertion at C-15 was reinforced by
α deprotection in this carbon,
β deprotection at C-14 and C-20 and
γ protection at C-19 and C-3, when compared to a structure without this substituent and confirmed by correlations of δ
H 3.31/δ
C 74.02 and 74.50, seen in the HMBC. In addition, the coupling constants for the two broad doublets at δ
H 3.26 (
J = 9.0 Hz) and 3.17 (
J = 14.3 Hz) were in accordance with the expected values when the hydrogens H-15 were located in the
α position in relation to ring
D. Therefore, it can be deduced that OCH
3-15 features pseudo-equatorial stereochemistry
[13]. The correlation between δ
H 2.03/δ
H 3.17 and 3.26, observed in the NOESY spectrum (
Figure S13), reinforce this argument.
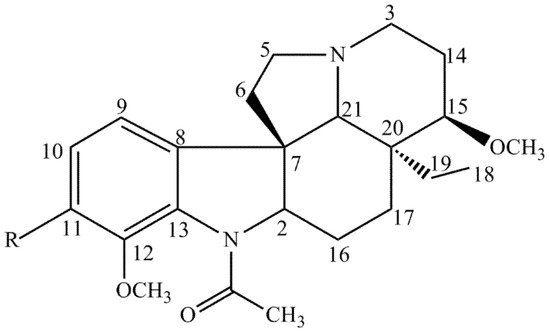
The interpretation of spectral data, in addition to the comparison with the previous research data, made it possible to identify two monoterpenoid indole alkaloids of the plumeran class. Compound 1 was identified as 15-methoxyaspidospermine. The presence of a methoxyl group at position C-11 in compound 2 allowed us to identify it as being t 15-methoxypyrifolidine (Figure 2).
Figure 2. Compounds isolated from A. pyrifolium present in TAF-Ap. 1—R=H, 2—R=OCH3.
3. Acute Toxicity
The TAF-Ap dose of 200 mg kg−1 was 100% lethal to animals within 30 min of its administration and death was preceded by severe tremors. The 100 mg kg−1 dose caused death in a female one hour after administration; by the fourth hour, all the animals showed poor responses to tail pinching, low auricular reflex and impaired posture, as well as tremors and signs of sedation and forced breathing. Regarding the dose of 50 mg kg−1, no animal died; however, the animals presented signs of mild sedation and forced breathing in the first 24 h. The lethal dose capable of killing 50% of animals (LD50) was estimated to be 160 mg kg−1.
The mean values obtained for each group in the weight evolution of the animals showed significant differences for the dose of 100 mg kg−1; although they gained weight, the values for body mass increase in the animals of this group were lower than those of the negative control group and of the groups receiving the 50 mg kg−1 dose (Table 2).
Table 2. Effect of oral administration of the total alkaloid fraction from A. pyrifolium (TAF-Ap) on weight evolution (change in body mass) by water and feed intake in 14 day-old male and female Swiss mice.
| Parameter |
Sex |
Saline Solution |
TAF-Ap (50 mg kg−1) |
TAF-Ap (100 mg kg−1) |
| Initial W. (g) |
M |
28.83 ± 1.32 |
27.67 ± 2.58 |
30.67 ± 0.81 |
| Final W. (g) |
33.17 ± 1.31 |
34.50 ± 3.72 |
31.50 ± 3.72 |
| Gain (%) |
4.34 |
6.83 |
0.83 *** |
| Initial W. (g) |
F |
27.50 ± 0.83 |
25.00 ± 1.41 |
25.33 ± 3.14 |
| Final W. (g) |
32.00 ± 1.26 |
32.17 ± 1.54 |
26.31 ± 3.18 |
| Gain (%) |
4.50 |
7.17 |
0.98 *** |
| Feed intake per day (g) |
M |
34.57 ± 2.92 |
37.29 ± 3.42 |
25.50 ± 3.25 *** |
| |
F |
36.71 ± 2.94 |
35.86 ± 2.65 |
23.07 ± 2.30 *** |
| Water consumption per day (mL) |
M |
52.14 ± 4.25 |
52.86 ± 4.68 |
56.43 ± 2.25 *** |
| |
F |
46.79 ± 4.64 |
50.00 ± 3.39 *** |
60.29 ± 3.93 *** |
Regarding the daily feed intake, those animals that received the 100 mg kg−1 dose consumed a smaller amount of food when compared to the negative control groups and those receiving the 50 mg kg−1 dose (Table 2). Still, as shown in Table 2, the groups that received the 100 mg kg−1 dose consumed a larger amount of water than the others. In addition, it was found that there was no significant difference when comparing the water and feed intake of animals receiving the 50 mg kg−1 dose in relation to the negative control.
Table 3 shows that there was a significant decrease in the relative weight of the livers of the animals receiving the 100 mg kg−1 dose compared to those receiving the 50 mg kg−1 dose and the negative control, which corroborates the results of the weight evolution shown in Table 2; these showed a lower weight gain in the animals that received the highest dose.
Table 3. Effect of oral administration of the total alkaloid fraction from A. pyrifolium (TAF-Ap) on relative organ weight of male and female Swiss mice at the end of the experiment.
| Relative Weight of Organs (g/100 g) |
Sex |
Negative Control |
TAF-Ap (50 mg kg−1) |
TAF-Ap (100 mg kg−1) |
| Liver |
M |
5.11 ± 0.65 |
5.06 ± 0.55 |
4.13 ± 0.54 * |
| Spleen |
0.59 ± 0.32 |
0.73 ± 0.47 |
0.80 ± 0.21 |
| Heart |
0.47 ± 0.02 |
0.50 ± 0.05 |
0.52 ± 0.11 |
| Kidneys |
1.23 ± 0.64 |
1.37 ± 0.11 |
1.24 ± 0.16 |
| Liver |
F |
4.98 ± 0.20 |
4.95 ± 0.47 |
4.03 ± 0.53 * |
| Spleen |
0.59 ± 0.11 |
0.45 ± 0.11 |
0.48 ± 0.25 |
| Heart |
0.48 ± 0.03 |
0.45 ± 0.04 |
0.36 ± 0.18 |
| Kidneys |
1.08 ± 0.11 |
0.97 ± 0.08 |
0.85 ± 0.44 |
In addition to body weight changes, the individual weight of each organ can also be considered as an indicator of drug-induced adverse effects, both of which are indicative of toxicity
[14]. Regarding the macroscopic characteristics, no alteration in the color or shape of the organs was observed between the studied groups.
The acute toxicity test was useful to establish the doses employed in the anti-inflammatory and anti-nociceptive activity assays. The reason why doses above 50 mg kg−1 exerted harmful effects on the animals in the present study is that TAF-Ap is a concentrated fraction of alkaloids, enabling a greater chance of producing toxicity than crude extract.
For this same species, some experimental studies have already been carried out to prove toxicity. In an experimental study of goats at different gestational stages and fed with freshly harvested green leaves from
A. pyrifolium (4 g kg
−1 dose), during 19 days of consumption, miscarriage and embryonic losses were observed. Desiccated leaves were not able to cause reproductive changes, although the ingestion of the plant in the first 34 days of gestation was shown to cause mortality
[15]. In a survey of 60 respondents residing in the Western and Eastern Seridó of Rio Grande do Norte, including 17 producers and technicians, abortions or the births of weak animals were reported in 16 goats that ingested
A. pyrifolium leaves during the drought period. Nine respondents also reported the occurrence of poisoning, which was characterized by hind limb stiffness and locomotion difficulty
[16].
In a toxicity study of male and female Wistar rats with ethanolic extracts from
A. pyrifolium, the females demonstrated reduced fetal weight and strong indications of maternal toxicity, in addition to motor disturbances and death at higher concentrations. The male rats were more resistant than the females. In the same study, it was found that
A. pyrifolium extract promoted hemolysis and was lethal to the
Artemia salina organism in an in vitro cytotoxicity test
[17].