| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | di costanzo Luigi | + 1911 word(s) | 1911 | 2020-08-11 08:58:50 | | | |
| 2 | Bruce Ren | Meta information modification | 1911 | 2020-10-27 09:55:27 | | |
Video Upload Options
Scientists are designing new ways to combine proteins and carbon-based nanomomecules. We review strategies of selecting proteins able to interact with proteins and typical van der Waals interactions. Proteins and carbon based nanomomecules can form ordered clusters of hybrid materials and will guide new projects for bioimaging tools and tuning of intrinsically disordered proteins.
Since the discovery of fullerene, carbon-based nanomolecules sparked a wealth of research across biological, medical and material sciences. Understanding the interactions of these materials with biological samples at the atomic level is crucial for improving the applications of nanomolecules and address safety aspects concerning their use in medicine. Protein crystallography provides the interface view between proteins and carbon-based nanomolecules. We review forefront structural studies of nanomolecules interacting with proteins and the mechanism underlying these interactions. We provide a systematic analysis of approaches used to select proteins interacting with carbon-based nanomolecules explored from the worldwide Protein Data Bank (wwPDB) and scientific literature. The analysis of van der Waals interactions from available data provides important aspects of interactions between proteins and nanomolecules with implications on functional consequences. Carbon-based nanomolecules modulate protein surface electrostatic and, by forming ordered clusters, could modify protein quaternary structures. Lessons learned from structural studies are exemplary and will guide new projects for bioimaging tools, tuning of intrinsically disordered proteins, and design assembly of precise hybrid materials.
1. Introduction
Since the discovery of buckminsterfullerene in 1985, a discrete molecule made of 60 atoms of carbon arranged to form a Ih symmetrical hollow sphere with surprising properties, it has become a favorite subject in nanotechnology and related disciplines [1]. The truncated icosahedral fullerene sphere has a van der Waals diameter of about one nanometer and several variants of smaller and larger diameters are known including elongated-shaped molecules recognized as nanotubes [2][3]. The chemical structure of these carbon-only molecules make them better conductors of electricity than common metals on much smaller scale [4]. The interest for these nanomolecules is consequence of their light weights, making them ideal for technological applications as well as biology and related fields applications [5][6]. By the 2000s, a number of studies had explored chemical strategies to link proteins and nucleic acids to nanomolecules, including metal clusters, with the aim to engineer devices for medical and biotechnological applications [7].
Among emerging carbon-made nanostructures, graphene, a single carbon sheet derived from graphite, and its related graphene oxide are very promising materials for tissue engineering, drug delivery, nerve tissue regeneration and biosensing [8][9][10][11].
Major obstacles with use of carbon-only nanomolecules for biological and medical purposes include their very poor solubility in water and poor affinity for a given protein target.
The coupling of fullerene with chemical polar groups or using water soluble capsules hosting fullerene facilitated preparation of water fullerene solutions [12][13][14]. Another aspect to consider for graphene based materials is represented by their variable bonding arrangement with not well-defined stoichiometry [15]. Therefore, well characterized protein/carbon-based nanomolecule complexes are really sought for stimulating studies involving hybrid materials.
Since 2008, protein crystallography has been instrumental in understanding the nature of interactions between proteins and carbon-made nanomolecules and provided insights for chemical modifications of these materials [16][17][18]. Although structure determination of these complexes is not trivial considering the size and often the scarce solubility of these carbon-based nanomolecules, [16] use of nanomolecules sparked a strategy for crystallization of difficult biological macromolecules [19][20]. For instance, graphene wrapped protein crystals protect from dehydration and stabilize disordered surface solvent molecules, therefore improving crystal diffraction [21]. A number of fullerene derivatives were reported to inhibit important drug target enzymes as HIV-1 protease and acetylcholinesterase a key enzyme for nervous system activity [22][23][24]. A metal hydroxylated form of fullerene was designed as a potential anti-metastatic agent for pancreatic cancer [25][26]. In addition, a number of chemical approaches are available to functionalize fullerenes or nanotubes and alter their proteins binding ability [27][28][29][30][31][32][33].
Precise understanding of interactions between proteins/nucleic acids and carbon-based nanomolecules provide crucial insights to improve safety aspects concerning the use of these materials [33][34][35][36][37].
Beginning with the pioneering studies of Brian Matthews with lysozyme phage T4 mutants, aimed to understand the minimal requirements for protein folding stability and how it can be rescued by binding to nonpolar or very slightly polar ligands such as benzene molecule [38], the selection of a stable protein with high affinity for a given nano-particles became a very attractive topic [39][40]. Typical nanomolecules involve protein interfacing interactions in much larger numbers than smaller probes.
The search for proteins as a binder of a given nanomolecule is not trivial because of many factors to balance between van der Waals and solvation interactions [41]. The first well-characterized synthetic protein aimed to solubilize a carbon nanotube through its coating consists of an amphiphilic α-helix containing hydrophobic and aromatic residues used to pack against the nanotube wall to improve interface affinity [42][43]. Aromatic amino acids are known to play a central role for protein tertiary structure stability and facilitate protein folding by reducing intramolecular hydrogen bonds around large aromatic residues [44][45][46]. Peptide sequences with high affinity for carbon nanomolecules show aromatic residues such as histidine or tryptophan residues and are characterized by flexible segments [47][48]. Similar considerations can be drawn for the affinity between nucleic acid and a nanotube [49].
The search for a protein as a good nanomolecule binder is reversed with respect to a search for a small ligand binder for which libraries are available [50]. Protein selection for a nanomolecule using high-throughput virtual screening or cheminformatics analysis are available [51][52].
Earlier crystallographic studies with cyclodextrins, a family of macro-cyclic oligosaccharides, and other studies fullerenes interacting with proteins inspired use of hollow shaped host-guest carbon-based nanomolecules that could potentially bind to proteins: cryptophanes, calixarenes, cucurbiturils, tweezers, etc. [53]. For these classes of organic molecules, besides aromatic residues hydrophilic and charged residues like arginine, lysine is important for protein–nanomolecule interactions [54][55][56][57][58][59]
2. All-Carbon Nanomolecules Interacting with Proteins
A widespread method used by researchers to promote protein affinity towards fullerenes (or nanotubes) is the use of covalently linked pyrenyl group anchored to the protein through surface lysines. Pyrenyl behaves as a molecular “glue”, able to stick to the nanotube wall via non-covalent π-stacking interactions [60].
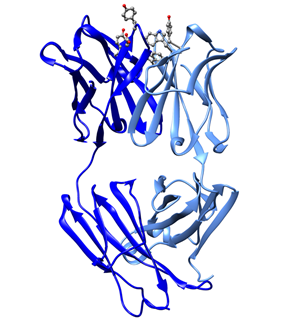
Immunization of mice with fullerene derivatives represents another method of producing in vivo IgG antibodies with high affinity towards fullerene (or nanotubes) [61][62]. With this approach, papain-cleaved Fab-IgG chains were obtained and purified and they showed high affinity for fullerene, measured in 22 nM [63]. The unbound Fab-IgG chains structure was solved by X-ray crystallography (pdb entry ID 1emt) [63]. Similarly, a recent antifullerene antibody Fab-C60 was obtained from mouse immunization and the structure of the complex of heavy (H) and light (L) chains solved by X-ray crystallography (Figure 1, pdb entry 6h3h) [64]. The structure of Fab-C60 shows a binding pocket consisting of a canonical CDR region that contains various aromatic residues (Tyr50 (H), Tyr101 (H), Tyr34 (L), Trp93 (L), and Trp98 (L)) and an aspartate residue (Asp100 (L)) [64]. The segment Asp100-Tyr101 solvent exposed the conformational disorder and is postulated to facilitate fullerene binding [64].
Figure 1. Ribbon drawing of mouse antifullerene antibody Fab-C60 (pdb entry 6h3h). The structure of Fab-C60 (complex of heavy (H) and light (L) chains) shows a fullerene binding pocket consisting of a canonical CDR region that contains various aromatic residues and an aspartate residue highlighted in balls-and-sticks (O red, N blue, C gray).
This approach was also used to select Fab chains with a high affinity towards a nanotube [65]. Another in vivo approach is the phage display technique that allows peptide selection from a library in presence of a nanotube used as target. During rounds of evolution while bacteria is infected, a peptide gene of higher binding affinity is isolated.
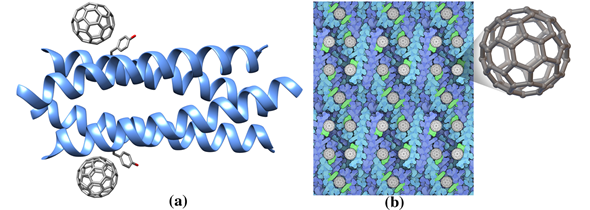
Among methods to select proteins with good affinity for nanotubes de novo design offered an exemplary strategy. Researchers noticed that the geometry of an ideal alpha helix matches the honeycomb geometry of graphene. So, they positioned alanine amino acids along an alpha helix to match the center of the repeating hexagonal unit of the graphene sheet. Then, they engineered interactions based on a previously designed four-helix bundle in order to wrap helices around the nanotube. As expected, the designed peptide composed by the following thirty amino acids sequence —AEAESALEYAQQALEKAQLALQAARQALKA—binds to nanotubes and its structure solved by X-ray crystallography shows an Ala-rich surface in agreement with the designed peptide (named Hexcoil-Ala, pdb entry 3s0r) [66]. Serendipitously, the designed alpha helix, called COP (C60-organizing peptide), forms a crystalline complex also when mixed with buckminsterfullerene. The crystal structure of COP (Figure 2, Table 1, pdb entry ID 5et3) shows how the peptide, organized in a four-helix bundle motif (Figure 2), recognizes the fullerene with their tyrosine amino acids. Each nanomolecule is sandwiched by two four-helix bundles forming a large superstructure (Figure 2b) [67]. Astonishingly, when tested, fullerenes or COP proteins by themselves are not conductive, but the hybrid material with this 3D lattice does conduct electricity.
Figure 2. (a) Ribbon drawing of the de novo designed protein COP (C60-organizing peptide) in complex with fullerene (pdb entry 5et3). (b) COP protein in complex with fullerene forms a large superstructure. Each fullerene molecule is bound to two four helix bundles through the side chain of a Tyr residue (green). This figure is obtained from the Molecule of the Month column “Proteins and Nanoparticles” (pdb101.rcsb.org/motm/222). Inset: fullerene molecule from the crystal structure of COP-fullerene complex (gray spheres, cif code 60C).
In summary, different strategies from in vivo selection or through de novo design are available to produce artificial proteins/peptides with high affinity to fullerenes and nanotubes. Fullerene does interact with Tyr and the methyl group of an Ala, or other aromatic residues properly placed in a protein sequence, resembling nanoparticle geometrical periodic features.
3. Conclusion
A recent nanomaterial database resource (PubVINAS) archives a total of 705 unique nanomaterials corresponding to twelve materials types [68]. At the time of this writing (July 2020), eighty of these nanomaterials are represented by carbon nanotubes, forty-eight by C60 fullerene derivatives, and twenty by carbon nanoparticles. Carbon-based nanomolecules research is rapidly growing due to potential applications ranging across biological, medical, and material sciences [69][70]. Applications involving multifunctional cyclodextrins, used for molecules delivery, received a widespread interest and are already in use for clinical purposes [71]. The gathering of carbon-based nanomolecules with biological samples has the potential for trending areas of medical chemistry including protein–protein interactions and conformational flexibility of disordered proteins for which metal based nanomolecules were explored [72].
In order to improve the property of carbon-based nanomolecules and address their safety for medical use, it is crucial to have a clear understanding of their interactions with a target protein. X-ray crystallography proved instrumental to understand the key interactions of proteins and carbon-based nanomolecules and inspired many of the studies we reviewed. Although these interactions are similar to those involving typical small molecules, the presence of a larger number of aromatic groups in carbon-based nanomolecules implies an important role of π-interactions (see Table 1). Despite the binding of large size carbon-based nanomolecules, these ligands often have a negligible effect on protein overall shape. Carbon-based nanomolecules have the propensity to cluster because of their significant radii and rigid skeletons with resulting effect on protein quaternary structure. Carbon-based nanomolecules coupled with charged or other chemical groups could change protein electrostatic surface. Carbon-based nanomolecules can be used as framework to tune crystalline porosity by simple use of common buffer molecule as an additive.
Therefore, new chemical modifications of carbon-based nanomolecules have potential as creative ways to address specific questions involving targeted proteins. Lessons learned from structural studies examined here are exemplary for the future use of carbon-based nanomolecules to stoichiometrically combine a number of protein entities to build functional hybrid materials [73][74].
References
- Kroto, H.W.; Heath, J.R.; O’Brien, S.C.; Curl, R.F.; Smalley, R.E. C60: Buckminsterfullerene. Nature 1985, 318, 162–163.
- Iijima, S. Helical microtubules of graphitic carbon. Nature 1991, 354, 56–58.
- Iijima, S.; Ichihashi, T. Single-shell carbon nanotubes of 1-nm diameter. Nature 1993, 363, 603–605.
- Ragab, T.; Basaran, C. Joule heating in single-walled carbon nanotubes. J. Appl. Phys. 2009, 106, e063705.
- Cai, L.; Wang, C. Carbon nanotube flexible and stretchable electronics. Nanoscale Res. Lett. 2015, 10, 320.
- Park, S.; Vosguerichian, M., Bao, Z. A review of fabrication and applications of carbon nanotube film-based flexible electronics. Nanoscale 2013, 5, 1727–1752.
- Niemeyer, C.M. Nanoparticles, proteins, and nucleic acids: Biotechnology meets materials science. Angew. Chemie Int. Ed. 2001, 40, 4128–4158.
- Stankovich, S.; Dikin, D.A.; Dommett, G.H.B.; Kohlhaas, K.M.; Zimney, E.J.; Stach, E.A.; Piner, R.D.; Nguyen, S.B.T.; Ruoff, R.S. Graphene-based composite materials. Nature 2006, 442, 282–286.
- Singh, V.; Joung, D.; Zhai, L.; Das, S.; Khondaker, S.I.; Seal, S. Graphene based materials: Past, present and future. Prog. Mater. Sci. 2011, 56, 1178–1271.
- Reina, G.; González-Domínguez, J.M.; Criado, A.; Vázquez, E.; Bianco, A.; Prato, M. Promises, facts and challenges for graphene in biomedical applications. Chem. Soc. Rev. 2017, 46, 4400–4416.
- Convertino, D.; Luin, S.; Marchetti, L.; Coletti, C. Peripheral neuron survival and outgrowth on graphene. Front. Neurosci. 2018, 12, 1.
- Andersson, T.; Nilsson, K.; Sundahl, M.; Westman, G.; Wennerström, O. C60 embedded in γ-cyclodextrin: A water-soluble fullerene. J. Chem. Soc. Chem. Commun. 1992,1992, 694–606.
- Wharton, T.; Kini, V.U.; Mortis, R.A.; Wilson, L.J. New non-ionic, highly water-soluble derivatives of C60 designed for biological compatibility. Tetrahedron Lett. 2001, 42, 5133–5134.
- Zakharian, T.Y.; Christianson, D.W. Design and synthesis of C60-benzenesulfonamide conjugates. Tetrahedron Lett. 2010, 51, 3599–3696.
- Pumera, M.; Sofer, Z. Towards stoichiometric analogues of graphene: Graphane, fluorographene, graphol, graphene acid and others. Chem. Soc. Rev. 2017, 46, 4450–4463.
- Aaron, J.A.; Chambers, J.M.; Jude, K.M.; Di Costanzo, L.; Dmochowski, I.J.; Christianson, D.W. Structure of a 129Xe-cryptophane biosensor complexed with human carbonic anhydrase II. J. Am. Chem. Soc. 2008, 130, 6942–6943.
- Calvaresi, M.; Zerbetto, F. The devil and holy water: Protein and carbon nanotube hybrids. Acc. Chem. Res. 2013, 46, 2454–2463.
- Patra, J.K.; Das, G.; Fraceto, L.F.; Campos, E.V.R.; Rodriguez-Torres, M. del P.; Acosta-Torres, L.S.; Diaz-Torres, L.A.; Grillo, R.; Swamy, M.K.; Sharma, S.; et al. Nano based drug delivery systems: Recent developments and future prospects. J. Nanobiotechnology 2018, 16, 71.
- Morgan, H.P.; McNae, I.W.; Hsin, K.Y.; Michels, P.A.M.; Fothergill-Gilmore, L.A.; Walkinshaw, M.D. An improved strategy for the crystallization of Leishmania mexicana pyruvate kinase. Acta Crystallogr. Sect. F Struct. Biol. Cryst. Commun. 2010, 66, 215–218.
- Goel, M.; Jain, D.; Kaur, K.J.; Kenoth, R.; Maiya, B.G.; Swamy, M.J.; Salunke, D.M. Functional equality in the absence of structural similarity. J. Biol. Chem. 2001, 276, 39277–39281.
- Warren, A.J.; Crawshaw, A.D.; Trincao, J.; Aller, P.; Alcock, S.; Nistea, I.; Salgado, P.S.; Evans, G. In vacuo X-ray data collection from graphene-wrapped protein crystals. Acta Crystallogr. Sect. D Biol. Crystallogr. 2015, 71, 2079–2088.
- Friedman, S.H.; DeCamp, D.L.; Kenyon, G.L.; Sijbesma, R.P.; Srdanov, G.; Wudl, F. Inhibition of the HIV-1 protease by fullerene derivatives: Model building studies and experimental verification. J. Am. Chem. Soc. 1993, 52, 2090–2100.
- Pastorin, G.; Marchesan, S.; Hoebeke, J.; Da Ros, T.; Ehret-Sabatier, L.; Briand, J.-P.; Prato, M.; Bianco, A. Design and activity of cationic fullerene derivatives as inhibitors of acetylcholinesterase. Org. Biomol. Chem. 2006, 4, 2556–2562.
- Qian, M.; Shan, Y.; Guan, S.; Zhang, H.; Wang, S.; Han, W. Structural basis of fullerene derivatives as novel potent inhibitors of protein tyrosine phosphatase 1B: Insight into the inhibitory mechanism through molecular modeling studies. J. Chem. Inf. Model. 2016, 56, 2024–2034.
- Kang, S.; Zhou, G.; Yang, P.; Liu, Y.; Sun, B.; Huynh, T.; Meng, H.; Zhao, L.; Xing, G.; Chen, C.; et al. Molecular mechanism of pancreatic tumor metastasis inhibition by Gd@C82(OH)22 and its implication for de novo design of nanomedicine. Proc. Natl. Acad. Sci. 2012, 109, 15431–15436.
- Kang, S.; Araya-Secchi, R.; Wang, D.; Wang, B.; Huynh, T.; Zhou, R. Dual inhibitory pathways of metallofullerenol Gd@C82(OH)22 on matrix metalloproteinase-2: Molecular insight into drug-like nanomedicine. Sci. Rep. 2014, 4, 4775.
- Basu-Dutt, S.; Minus, M.L.; Jain, R.; Nepal, D.; Kumar, S. Chemistry of carbon nanotubes for everyone. J. Chem. Educ. 2012, 89, 221–229.
- Marchesan, S.; Prato, M. Under the lens: Carbon nanotube and protein interaction at the nanoscale. Chem. Commun. 2015, 51, 4347–4359.
- Tasis, D.; Tagmatarchis, N.; Bianco, A.; Prato, M. Chemistry of carbon nanotubes. Chem. Rev. 2006, 3, 1105–1136.
- Maggini, M.; Scorrano, G.; Prato, M. Addition of azomethine ylides to C60: Synthesis, characterization, and functionalization of fullerene pyrrolidines. J. Am. Chem. Soc. 1993, 115, 9798–9799.
- Yang, J.; Alemany, L.B.; Driver, J.; Hartgerink, J.D.; Barron, A.R. Fullerene-derivatized amino acids: Synthesis, characterization, antioxidant properties, and solid-phase peptide synthesis. Chem.-A Eur. J. 2007, 13, 2530–2545.
- Pochkaeva, E.I.; Podolsky, N.E.; Zakusilo, D.N.; Petrov, A.V.; Charykov, N.A.; Vlasov, T.D.; Penkova, A.V.; Vasina, L.V.; Murin, I.V.; Sharoyko, V.V.; et al. Fullerene derivatives with amino acids, peptides and proteins: From synthesis to biomedical application. Prog. Solid State Chem. 2020, 57, 100255.
- Gao, J.; Wang, Y.; Folta, K.M.; Krishna, V.; Bai, W.; Indeglia, P.; Georgieva, A.; Nakamura, H.; Koopman, B.; Moudgil, B. Polyhydroxy fullerenes (fullerols or fullerenols): Beneficial effects on growth and lifespan in diverse biological models. PLoS ONE 2011, 6, e0019976.
- Jimenez-Cruz, C.A.; Kang, S. gu; Zhou, R. Large scale molecular simulations of nanotoxicity. Wiley Interdiscip. Rev. Syst. Biol. Med. 2014, 6, 329–343.
- Hernández-Cancel, G.; Suazo-Dávila, D.; Ojeda-Cruzado, A.J.; García-Torres, D.; Cabrera, C.R.; Griebenow, K. Graphene oxide as a protein matrix: Influence on protein biophysical properties. J. Nanobiotechnology 2015, 13, 70.
- Rahman, M.; Laurent, S.; Tawil, N.; Yahia, L.; Mahmoudi, M. Nanoparticle and protein corona. In Protein-Nanoparticle Interactions (The Bio-Nano Interface); Springer: London, UK, 2013; Volume 15, pp. 21–44.
- Othman, Z.; Cillero Pastor, B.; van Rijt, S.; Habibovic, P. Understanding interactions between biomaterials and biological systems using proteomics. Biomaterials 2018, 167, 191–204.
- Baase, W.A.; Liu, L.; Tronrud, D.E.; Matthews, B.W. Lessons from the lysozyme of phage T4. Protein Sci. 2010, 19, 631–641.
- Vaitheeswaran, S.; Garcia, A.E. Protein stability at a carbon nanotube interface. J. Chem. Phys. 2011, 134, 125101.
- Asuri, P.; Bale, S.S.; Karajanagi, S.S.; Kane, R.S. The protein-nanomaterial interface. Curr. Opin. Biotechnol. 2006, 17, 562–568.
- Trozzi, F.; Marforio, T.D.; Bottoni, A.; Zerbetto, F.; Calvaresi, M. Engineering the fullerene-protein interface by computational design: The sum is more than its parts. Isr. J. Chem. 2017, 57, 547–552.
- Dieckmann, G.R.; Dalton, A.B.; Johnson, P.A.; Razal, J.; Chen, J.; Giordano, G.M.; Muñoz, E.; Musselman, I.H.; Baughman, R.H.; Draper, R.K. Controlled assembly of carbon nanotubes by designed amphiphilic peptide helices. J. Am. Chem. Soc. 2003, 125, 1770–1777.
- Xie, H.; Ortiz-Acevedo, A.; Zorbas, V.; Baughman, R.H.; Draper, R.K.; Musselman, I.H.; Dalton, A.B.; Dieckmann, G.R. Peptide cross-linking modulated stability and assembly of peptide-wrapped single-walled carbon nanotubes. J. Mater. Chem. 2005, 15, 1734–1741.
- Burley, S.K.; Petsko, G.A. Aromatic-aromatic interaction: A mechanism of protein structure stabilization. Science 1985, 229, 23–28.
- Bignucolo, O.; Leung, H.T.A.; Grzesiek, S.; Berneìche, S. Backbone hydration determines the folding signature of amino acid residues. J. Am. Chem. Soc. 2015, 137, 4300–4303.
- Ferreira De Freitas, R.; Schapira, M. A systematic analysis of atomic protein-ligand interactions in the PDB. Medchemcomm 2017, 8, 1970–1981.
- Wang, S.; Humphreys, E.S.; Chung, S.Y.; Delduco, D.F.; Lustig, S.R.; Wang, H.; Parker, K.N.; Rizzo, N.W.; Subramoney, S.; Chiang, Y.M.; et al. Peptide with selective activity for carbon nanotube Nat. Mater. 2003, 2, 196–200.
- Ge, C.; Du, J.; Zhao, L.; Wang, L.; Liu, Y.; Li, D.; Yang, Y.; Zhou, R.; Zhao, Y.; Chai, Z.; et al. Binding of blood proteins to carbon nanotubes reduces cytotoxicity. Proc. Natl. Acad. Sci. 2011, 108, 16968–16973.
- Johnson, R.R.; Johnson, A.T.C.; Klein, M.L. Probing the structure of DNA-carbon nanotube hybrids with molecular dynamics. Nano Lett. 2008, 8, 69–75.
- Calvaresi, M.; Zerbetto, F. Fullerene sorting proteins. Nanoscale 2011, 3, 2873–2881.
- Di Giosia, M.; Valle, F.; Cantelli, A.; Bottoni, A.; Zerbetto, F.; Fasoli, E.; Calvaresi, M. High-throughput virtual screening to rationally design protein-carbon nanotube interactions. Identification and preparation of stable water dispersions of protein-carbon nanotube hybrids and efficient design of new functional materials. Carbon 2019, 147, 112–119.
- Fjodorova, N.; Novič, M.K.; Venko, A.; Rasulev, A. A comprehensive cheminformatics analysis of structural features affecting the binding activity of fullerene derivatives. Nanomater. 2020, 10, 90.
- Mikami, B.; Hehre, E.J.; Sato, M.; Katsube, Y.; Hirose, M.; Morita, Y.; Sacchettini, J.C. The 2.0-A resolution structure of soybean beta-amylase complexed with alpha-cyclodextrin. Biochemistry 1993, 32, 6836–6845.
- Alex, J.M.; Brancatelli, G.; Volpi, S.; Bonaccorso, C.; Casnati, A.; Geremia, S.; Crowley, P.B. Probing the determinants of porosity in protein frameworks: Co-crystals of cytochrome: C and an octa-anionic calix[4]arene. Org. Biomol. Chem. 2020, 18, 211–214.
- Brancatelli, G.; Dalcanale, E.; Pinalli, R.; Geremia, S. Probing the structural determinants of amino acid recognition: X-ray studies of crystalline ditopic host-guest complexes of the positively charged amino acids, ARg, Lys, and His with a cavitand molecule. Molecules 2018, 23, 3368.
- Pinalli, R.; Brancatelli, G.; Pedrini, A.; Menozzi, D.; Hernández, D.; Ballester, P.; Geremia, S.; Dalcanale, E. The origin of selectivity in the complexation of N-methyl amino acids by tetraphosphonate cavitands. J. Am. Chem. Soc. 2016, 138, 8569–8580.
- Westbrook, J.D.; Shao, C.; Feng, Z.; Zhuravleva, M.; Velankar, S.; Young, J. The chemical component dictionary: Complete descriptions of constituent molecules in experimentally determined 3D macromolecules in the Protein Data Bank. Bioinformatics 2015, 31, 1274–1278.
- Goodsell, D.S.; Zardecki, C.; Di Costanzo, L.; Duarte, J.M.; Hudson, B.P.; Persikova, I.; Segura, J.; Shao, C.; Voigt, M.; Westbrook, J.D.; et al. RCSB Protein Data Bank: Enabling biomedical research and drug discovery. Protein Sci. 2020, 29, 52–65.
- Sobolev, V.; Sorokine, A.; Prilusky, J.; Abola, E.E.; Edelman, M. Automated analysis of interatomic contacts in proteins. Bioinformatics 1999, 15, 327–332.
- Chen, R.J.; Zhang, Y.; Wang, D.; Dai, H. Noncovalent sidewall functionalization of single-walled carbon nanotubes for protein immobilization J. Am. Chem. Soc. 2001, 123, 3838–3839.
- Chen, B.X.; Wilson, S.R.; Das, M.; Coughlin, D.J.; Erlanger, B.F. Antigenicity of fullerenes: Antibodies specific for fullerenes and their characteristics. Proc. Natl. Acad. Sci. 1998, 95, 10809–10813.
- Hendrickson, O.; Fedyunina, N.; Zherdev, A.; Solopova, O.; Sveshnikov, P.; Dzantiev, B. Production of monoclonal antibodies against fullerene C 60 and development of a fullerene enzyme immunoassay. Analyst 2012, 137, 98–105.
- Braden, B.C.; Goldbaum, F.A.; Chen, B.X.; Kirschner, A.N.; Wilson, S.R.; Erlanger, B.F. X-ray crystal structure of an anti-Buckminsterfullerene antibody Fab fragment: Biomolecular recognition of C60. Proc. Natl. Acad. Sci. 2000, 97, 12193–12197.
- Osipov, E.M.; Hendrickson, O.D.; Tikhonova, T.V.; Zherdev, A.V.; Solopova, O.N.; Sveshnikov, P.G.; Dzantiev, B.B.; Popov, V.O. Structure of the anti-C60 fullerene antibody fab fragment: Structural determinants of fullerene binding. Acta Naturae 2019, 11, 58–65.
- Erlanger, B.F.; Chen, B.X.; Zhu, M.; Brus, L. Binding of an anti-fullerene IgG monoclonal antibody to single wall carbon nanotubes. Nano Lett. 2001, 9, 465–467.
- Grigoryan, G.; Kim, Y.H.; Acharya, R.; Axelrod, K.; Jain, R.M.; Willis, L.; Drndic, M.; Kikkawa, J.M.; DeGrado, W.F. Computational design of virus-like protein assemblies on carbon nanotube surfaces. Science 2011, 332, 1071–1076.
- Kim, K.H.; Ko, D.K.; Kim, Y.T.; Kim, N.H.; Paul, J.; Zhang, S.Q.; Murray, C.B.; Acharya, R.; Degrado, W.F.; Kim, Y.H.; et al. Protein-directed self-assembly of a fullerene crystal. Nat. Commun. 2016, 7, 11429.
- Yan, X.; Sedykh, A.; Wang, W.; Yan, B.; Zhu, H. Construction of a web-based nanomaterial database by big data curation and modeling friendly nanostructure annotations. Nat. Commun. 2020, 11, 2519.
- Shi, J.; Votruba, A.R.; Farokhzad, O.C.; Langer, R. Nanotechnology in drug delivery and tissue engineering: From discovery to applications. Nano Lett. 2010, 10, 3223–3230.
- Malik, S.A.; Mohanta, Z.; Srivastava, C.; Atreya, H.S. Modulation of protein-graphene oxide interactions with varying degrees of oxidation. Nanoscale Adv. 2020, 2, 1904–1912.
- Hu, Q. Da; Tang, G.P.; Chu, P.K. Cyclodextrin-based host-guest supramolecular nanoparticles for delivery: From design to applications. Acc. Chem. Res. 2014, 47, 2017–2025.
- Vitali, M.; Rigamonti, V.; Natalello, A.; Colzani, B.; Avvakumova, S.; Brocca, S.; Santambrogio, C.; Narkiewicz, J.; Legname, G.; Colombo, M.; et al. Conformational properties of intrinsically disordered proteins bound to the surface of silica nanoparticles. Biochim. Biophys. Acta-Gen. Subj. 2018, 1862, 1556–1564.
- Kobayashi, N.; Arai, R. Design and construction of self-assembling supramolecular protein complexes using artificial and fusion proteins as nanoscale building blocks. Curr. Opin. Biotechnol. 2017, 46, 57–65.
- Garcia-Seisdedos, H.; Empereur-Mot, C.; Elad, N.; Levy, E.D. Proteins evolve on the edge of supramolecular self-assembly. Nature 2017, 548, 244–247.