| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Gun Wirtanen | + 4317 word(s) | 4317 | 2021-10-11 11:41:06 | | | |
| 2 | Catherine Yang | Meta information modification | 4317 | 2022-03-22 09:17:28 | | |
Video Upload Options
High quality colostrum is a valuable raw material in food products. The quality of colostrum is classified by low bacterial counts and adequate immunoglobulin (Ig) concentrations. Bacterial contamination can contain a variety of high counts of spoilage bacteria or pathogens. The supply of good quality bovine colostrum is required. The microbial safety of bovine colostrum is increased using an appropriate processing, e.g. heat treatment, which does not destroy the high nutritional value of colostrum. The heat treatment is cost-effective compared to other methods and heat treatment can be performed in both small- and large-scale production.
1. Background on Bovine Colostrum, Contaminants, and Processing
Bovine colostrum is the first milk given by a cow after parturition as nutrition to the newborn calf. This liquid is essential for the conferring of passive immunity to the newborn calves. The newborn calves lack immunization at birth and require the uptake of immunoglobulins (Igs) within 24–36 h after birth. Initial milk is considered bovine colostrum up to 3 days postpartum. Specifically, an elevated concentration of immunoglobulin G (IgG) is characteristic for bovine colostrum, as it is of significance in the transfer of passive immunity [1]. Besides these Igs there are other immune components present, e.g., enzymes or lactoferrin (LF), which act as nonspecific antibacterial factors [2]. Furthermore, healthy cows produce colostrum in excess of the calf’s need, which means that the ethical aspects for calves are not impacted [3]. Therefore, there is an increasing interest for the human use of bovine colostrum as a nutraceutical food [4]. Several human studies provide information on treatment or prevention effectiveness in bone development, respiratory, inflammatory, and gastrointestinal diseases, e.g., inflammatory bowel syndrome and Escherichia coli induced diarrhea [5][6][7]. Additionally, improvements in athletes’ performances have been confirmed [8].
The microbiology in raw colostrum is expected to be highly diverse. There are risks for the growth of both spoilage and pathogenic bacteria. Therefore, the consumption of raw contaminated colostrum may lead to illnesses in the calves due to spoiled nutrition and to intoxication or infections of Staphylococcus spp., Bacillus spp., or Salmonella spp., etc. in humans [9][10]. When the calves do not get enough good quality colostrum, the calves can develop microbial diseases due to inadequate passive immunity [11][12][13]. Specific microbes, e.g., Mycoplasma bovis or Staphylococcus aureus, in the colostrum provoke the mastitis [14]. The harvesting procedure of bovine colostrum is a critical control point, when the occurrence of contamination is to be reduced [15][16][17]. Raw dairy products such as bovine colostrum can be contaminated by several human pathogens during harvesting, which means that there is a need for treatment before consumption [18].
The processing techniques for an efficient inactivation of pathogenic/spoilage bacteria must be applied to obtain health promoting colostrum of good quality. The regulations for marketing dairy products require heat treatment or an equivalently effective treatment to improve the shelf life before selling the product [19]. The design of a heat treatment process for bovine colostrum will be introduced in this review. However, besides a mandatory efficient reduction of the bacterial count, the beneficial constituents in colostrum have to be preserved, not diminished, through the processing. Bioactive components, e.g., Igs, with nutraceutical value for humans are degrading through the high temperature heat treatment of the colostrum [4][20], which means that other feasible methods for the bacterial reduction are of interest to the industry.
2. Bioactive Components in Colostrum
The bioactive compounds in bovine colostrum play a key role in its high nutritional value for human consumers. The list of components with immunomodulatory capabilities comprises direct and indirect powerful mechanisms as well as the adaption of the host’s immune response [21][22]. The worldwide market of colostrum is expected to further increase. This can be explained by a rising request for health promoting foods, linked to emerging illnesses and health risks due to improper nutrition[23].
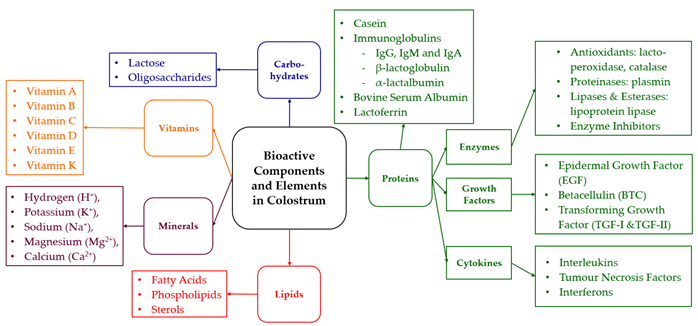
The list of these bioactive compounds consists of carbohydrates, proteins, growth factors, cytokines, lipids, enzymes, vitamins, and minerals [2][22][24].
Figure 1. Overview of bioactive components and elements in bovine colostrum based on McGrath et al. [1] and Korhonen [2][24].
The amount of bioactive compounds in bovine colostrum is significantly higher than in milk [1]. This is proven by an elevated protein concentration, which is 15.9 g/100 g within 24 h postpartum and 3.3 g/100 g after 5 months [25]. Approximately 70–80% of this total protein content in colostrum are Igs, prevalent in concentrations of 30–200 g/L. The Ig concentration declines soon after parturition, being considerably lower in milk with 0.4–1 g/L [1][5]. The subcategory IgG1 accounts for 75% of the antibody content in colostrum, followed by IgM, IgA, and IgG2 [26].
Additionally, it has been reported that bovine colostrum IgG can retain biological functionality with human digestion. After colostrum consumption, immunological activity in the ileum effluents of adults was discovered [26][27]. Therefore, the ingesting of the colostrum derived IgG can prevent both respiratory and gastrointestinal infections, and is enhanced through improved barrier integrity. By the prevention of respiratory infections, the development of allergies such as allergic asthma can be prevented [28][29]. According to Fox and Kelly [30][31], about 70 indigenous enzymes have been identified in bovine milk. Both ribonucleases and lysozyme [EC 3.2.1.17] (LZM) are present in higher concentrations in colostrum than in milk with an extensively elevated concentration of enzymes in the early postpartum period [1][31][32]. Enzymes have several purposes, e.g., they work synergic with other proteins to fulfil antimicrobial activities. LZM supports the digestive system or catalyzes other important reactions [2][33]. and plays an undoubtable role in the body’s immune system by elevating antiviral and antineoplastic activities [34]. It hydrolyzes the β (1→4)-bond between muramic acid and N-acetylglucosamine in the cell wall [31], which leads to the lysis of bacterial cells [32] and is effective against Escherichia coli and Pseudomonas aeruginosa [35]. Lactoperoxidase [EC 1.11.1.7] (LPO) is one of the major antimicrobial agents in colostrum, while having a broad substrate specificity [36].
Lactoferrin (LF), being an iron-binding glycoprotein, is categorized as a multifunctional compound [36]. LF is reported to have proteolytic broad substrate specificity [37] .while inhibiting the growth of both E. coli and Listeria monocytogenes. The presence of LF enhances the antimicrobial effects of LZM [32]. Due to the beneficial health effects, bovine LF is gaining attention for being used in functional foods [36].
3. Microbiological Quality of Colostrum
Bacteria in raw colostrum and milk is of vast concern. Pathogenic bacteria present in milk and milk products have been reported to account for 1–5% of bacterial foodborne disease outbreaks in humans in industrialized countries [18]. Bovine milk and colostrum serve as nutritious growth media for bacteria. Furthermore, the storage conditions on the dairy farm as well as before and after processing affect the bacterial loads within the colostrum. At refrigerated temperatures, most bacterial species grow slower, and the storage can be prolonged [17]. Bacteria can grow, digest, and harm colostrum by generating toxic agents or spoilage by-products, which possibly can prevent the beneficial effects of colostrum components [38]. The bacterial count increases rapidly when the colostrum is kept warm. High bacterial counts can lead to a decrease in protein content [38][39].
However, the probiotic Lactobacillus casei strains secrete beneficial compounds, e.g. heteropolysaccharides. Probiotics synthesize exopolysaccharides or indirectly enzymes synthesizing polysaccharides, which have many health benefits for consumers. Lactic acid bacteria (LAB) generates lactic acid. They have anticancer, antimicrobial, immunomodulatory, and anti-inflammatory activity [40][41][42][43][44].
For the overall assessment of colostrum quality, the standard plate count (SPC) of raw colostrum samples is of significance. The regulations are the same as for raw bovine milk given for the SPC, the total coliform count (TCC), and stating the absence of other infectious bacteria [19]. Regulation (EC) 853/2004 states that raw milk and colostrum must have a SPC at 30 °C of ≤105 CFU/mL [18][19][45].
The major zoonotic bacterial species cause diseases in both animals and humans. These are causatives of foodborne diseases and they include S. aureus, Salmonella spp., Campylobacter spp., L. monocytogenes, E. coli, and Bacillus spp. [46][47]. Zoonotic bacteria derived from non-human origin can infect humans with a transferable disease [47]. The microbial contamination has to be monitored to prevent transfers to the human population [10]. In general, diseases caused by bacteria in food can be divided into infections and intoxications. Intoxications are evoked by secretion of toxins of specific pathogens, causing food poisoning [47]. These bacterial species include Staphylococcus spp. [48], E. coli [49], and B. cereus [50]. Infections, on the contrary, are induced by the ingestion of food containing living pathogenic cells [47].
Mastitis is the inflammation of the mammary gland parenchyma and is considered a highly prevalent infectious disease in dairy cowherds, affecting 95% of American dairy herds [14]. Infection leads to reduced milk yield and changes in milk composition. Furthermore, it shortens the productive life of affected cows [45]. Economically, mastitis is considered a significant burden for the dairy farms [14]. Mastitis causing bacteria include Streptococcus uberis [51], S. agalactiae [10], S. dysgalactiae [51], S. aureus [52], Corynebacterium spp. [15], M. bovis [53], E. coli [49], and Trueperella pyogenes [54]. All above have been reported to be present in bovine colostrum and are described below.
Streptococcus colonies have been found in colostrum [9][40]. Humans should avoid the intake of S. bovis, because it has been associated with bacteremia, meningitis, endocarditis, and colorectal cancer [46]. Its effect is reinforced by the chronic interaction between S. bovis and human immune response especially in susceptible hosts [55]. Furthermore, S. agalactiae is known to cause several diseases in humans including gastrointestinal infections in infants, septicemia, urinary tract infections (UTIs), and mastitis in adults [49][56]. S. dysgalactiae is regarded as a contagious environmental Streptococcus strain not yet reported to be harmful to humans. The environmental pathogen S. uberis causes mastitis in animals, but it has not been found to be harmful to humans [49].
Staphylococcus spp. have been reported in colostrum several times [14][15][57]. S. aureus of bovine origin, a major zoonotic pathogen, can lead to a wide range of infectious diseases also in humans. Due to its risk to develop antimicrobial resistance this risk increases [47][49].
L. monocytogenes is a Gram-positive bacterium causing an infection called listeriosis has been described in bovine colostrum. It seriously affects various human groups with reduced resistance. The fatality rate in hospitalized patients is reported to be as high as 30% [58].
Bacillus spp. are also important zoonotic pathogens in milk [46]. The presence of Bacillus spp. were detected to be above 1000 CFU/mL in 15% of more than 230 colostrum samples [9].
Coliforms, a group of bacteria prevalently appearing in human and animal feces, are used as indicators for the occurrence of fecal contamination in milk products [59] and as signs of poor teat treatment or inadequate refrigeration [60]. The presence of Enterobacteriaceae in colostrum has been confirmed [15][61]. Only the Shiga-toxin-producing E. coli (STEC) strains cause severe diseases [49].
Salmonella spp. are involved in many foodborne outbreaks [47][62] through infections in several hosts [51]. The emerging risk of multidrug resistant Salmonella spp. of bovine origin makes it a threat to human health [62].
Mycoplasma spp. has been reported in bovine colostrum [14]. M. bovis is a pathogen, which causes respiratory disease, mastitis, and pneumonia in cows [52].
The general occurrence of Corynebacterium spp. in bovine colostrum have been described [9][14][15]. C. bovis is regarded as a causative agent of mastitis and is described to be a rare human pathogen [63]. In one study, two-thirds of the Corynebacterium spp. in milk from mastitis-infected cows were identified as C. bovis [64].
T. pyogenes causes both mastitis and other diseases in cows [54]. In humans, it can cause endocarditis. Antibiotic treatment at dairy farms enables the emergence of multidrug-resistant strains also in hosts [65].
Mycobacterium avium ssp. paratuberculosis (MAP) causes paratuberculosis in cows. Paratuberculosis is referred to as Johne’s disease. It is characterized as a chronic granulomatous infection of ruminant intestines [66] presenting an economic burden in the dairy industry [67]. The shedding of the bacteria by infected cows mainly happens through feces, but it can also be excreted in colostrum [68]. Besides the threat for bovine health, a likely connection between MAP and Crohn’s disease in humans is suspected, but not proven, to be of zoonotic risk [66][67]. In humans, MAP can also cause tuberculosis infection [46][68].
Both Pseudomonas spp. and Acinetobacter spp. have been found in bovine colostrum using 16S rRNA analysis [14][15]. Pseudomonas spp. are non-fermentative Gram-negative rods [9], which can cause pneumonia, UTI, meningitis, and enterocolitis in humans [48]. Acinetobacter junii has been reported to cause septicemia [69]. Additional bacteria, reported to occur in raw colostrum, have been summarized in Fasse et al. [70].
Probiotic bacteria are beneficial viable microorganisms employed in food, drinks and medical health products [7][40]. They mainly consist of LAB, Bifidobacterium spp., and Enterococcus spp. [48]. Bifidobacterium spp. are commonly used as a probiotic strain due to acclaimed health benefits and their presence in the gastrointestinal tract. There is rising interest especially towards Bifidobacterium spp., as it is a protective agent against infectious diseases. It helps to improve the immune response and to reduce symptoms of irritable bowel syndrome (IBS), ulcerative colitis (UC), allergic diseases, and atopic dermatitis associated to immunoglobulin E [42].
The presence of L. casei and Bifidobacterium pseudolongum in bovine colostrum has been confirmed [57]. Their presence in the human intestinal microflora is presumably beneficial in digestion [40], which can be explained with the production of conjugated linoleic acid (CLA) isomers important in physiological properties in humans. These applications are valuable in probiotic foods and food supplements [43]. These probiotic bacteria can be cultured and added in suitable amounts to dairy products [41][44]. Furthermore, Prevotella spp. and Ruminococcaceae bacteria have been discovered in bovine colostrum using 16S rRNA analysis [14][15]. In the human gastrointestinal microbiota, they enable the digestion of plant polysaccharides [71]. LAB strains, which are generally regarded as safe (GRAS) organisms, inhibit growth of coliforms and other bacterial pathogens [18][39].
4. Contamination Control On-Farm and in Processing of Bovine Colostrum Products
Nowadays, bovine colostrum as food is available on the market. Bovine colostrum for human consumption is collected and frozen on the individual farms and thereafter shipped frozen to processing facilities, where it is pasteurized and further treated through optional fat and lactose removal before spray- or freeze-drying to powder [7]. Available colostrum products include: (1) raw whole colostrum powder, (2) raw skim colostrum powder, and (3) industrially produced colostrum milk protein concentrate [29]. Currently, bovine colostrum is available in liquid form or as spray- or freeze-dried colostrum powder [8][72]. It has also been used as nutraceutical. Whey formulations with high concentrations of bioactive proteins and peptides are accumulating immense interest among human health specialists [73]. The powder can also be marketed as a dietary supplement in the form of sachets, capsules, or chewable tablets [7]. The fractionation of bioactive components out of bovine milk and colostrum yielding health-promoting foods is also gaining attention. This entails the fractionation of caseins or whey proteins as well as the isolation of LF, LPO, or especially bovine colostrum’s Igs [74]. There are also dairy products, in which bovine colostrum is used as an additive in cheeses, butter, yogurts, kefir, fermented milk, milk powdered beverages, ice cream, jellies, nutritional bars, and ready-to-drink beverages [6][7].
The harvesting and storage of colostrum are the main factors in determining the microbiological quality during primary production [75]. Pathogenic and spoilage microbes can enter the colostrum directly from the udder due to contamination, the environment, workers, and contaminated equipment and contamination during milking, processing, packaging, storage, and transport can be observed [76][77][78].
Bacterial counts of colostrum derived aseptically from the udder contain relatively low counts of bacteria. The harvesting process is regarded as a critical control point in colostrum production [17]. Transmission of bovine infections like mastitis, commonly caused by S. uberis or paratuberculosis infection by MAP, is described to occur due to the shedding of bacteria from feces [51]. Stewart et al. [17] reported the TCC to be quite low directly on the udder, with the average being 8 CFU/mL in 39 samples. The numbers vastly increase up to 50,000 CFU/mL during further storage without processing. Besides the harvesting, the storage conditions are also critical [79].
The following steps affect the colostrum quality prior to and during processing: (1) cow’s health status, (2) milking hygiene, (3) chilling practices and efficiency, (4) cleanliness of milking equipment including equipment design, maintenance, cleaning, and disinfection, (5) personnel hygiene, (6)–(8) chill temperatures during packaging, storage, and transportation, as well as (9) heat treatment taking into account the soiling of the heat exchanger plates during use [78][80].
The colostrum must be treated either decentral at the dairy farm or central at a dairy processing site (Figure 2). There are several techniques utilized to improve the colostrum quality, e.g., pasteurization either batch wise or continuously with high temperature short time (HTST) or low temperature short time (LTST) pasteurization [20].
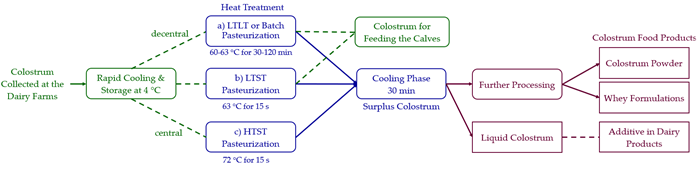
Figure 2. Feasible colostrum heat treatment procedures are either decentral on-farm or central at the dairy. The steps within the boxes display options for heat treatment of colostrum, which can be: (a) pasteurization in batches, i.e., low temperature long time (LTLT) pasteurization, (b) continuous low temperature short time (LTST) pasteurization, or (c) continuous high temperature short time (HTST) pasteurization. A part of the heat-treated colostrum is feed for the calf, whereas the rest is used in food.
Different industrial standards include batch pasteurization in which the colostrum is heated to 63 °C for 30 min, also called LTLT pasteurization [82]. HTST pasteurization is commonly replacing the batch method. It gives similar effects but more efficiently, e.g. at 72 °C for 15 s (Table 1). LTLT heat treatment is the most convenient treatment for farms with small amounts of colostrum [18][20][81]. These treatment options are also described in Regulation (EC) No. 852/2004, Annex II, Chapter XI for the treatment of raw milk or colostrum [76]. With pasteurization, most of the harmful organisms in milk will be inactivated or reduced to a secure microbial level in the product [20]. The feeding of heat-treated colostrum leads to an increased IgG absorption rate in calves [82]. While decreasing IgG concentrations in heat-treated colostrum have also been observed. The heating can inactivate bioactive compounds in bovine colostrum [20]. Fasse et al. [83] described details in various pasteurization methods.
Table 1 Summary of the effects of processing methods reported as microbial inactivation or microbial inactivation rate in bovine colostrum. The inactivation rates are given based on the measured reduction as standard plate counts (SPCs) before and after treatment (in log CFU/mL). The detected loss in antibody concentration (IgG) is given in percentage (%) [84][85].
|
Method |
Inactivation Rate (log CFU/mL) |
IgG Concentration Loss (%) |
|
Low temperature long time (LTLT) batch pasteurization, 30 min at 60 °C [84] |
Inactivation of Mycoplasma bovis, Listeria monocytogenes, Escherichia coli O157:H7 & Salmonella enterica Enteritidis, but Mycobacterium avium ssp. paratuberculosis (MAP) was detected |
No significant loss detected |
|
LTLT batch pasteurization, 60 min at 60 °C [84] |
Inactivation of M. bovis, L. monocytogenes, E. coli O157:H7, S. Enteritidis, and in three of four batches no MAP |
No significant loss detected |
|
LTLT batch pasteurization, 120 min at 60 °C [85] |
Viable M. bovis, L. monocytogenes, E. coli O157:H7, S. Enteritidis & MAP were not detected |
No significant loss detected |
|
High temperature short time (HTST) pasteurization, 15 s at 67 °C [85] |
MAP 4 |
22 |
|
HTST pasteurization, 15 s at 72 °C [85] |
Inactivation of MAP in colostrum |
27 |
5. Process Design
Processing is described as a systematic series of actions designed to increase the value-added to the food product [86]. In the process development system, the configuration of processing steps for desired and safe products is included. When determining the colostrum treatment process, attention is focused on ensuring food safety without destroying biological activity. A minimum number of process steps is always the goal in an economically viable, efficient process. There are a number of process options, which certify the safety of heat treatment, and among these the most optimal option should be chosen. The first and simplest flow diagram is the block diagram (Figure 2), in which the various unit operations of the process are represented as simple blocks connected to each other by lines representing the process flow from one operation to another.
The first step is a review of colostrum processing choosing between the batch and continuous processes. The batch process has been part of human activity throughout history. It remains used most of the time on a laboratory scale. The batch process is suitable for small-scale manufacturing in capacity. In the small scale, investment in LTLT pasteurization is a possible method for heat treatment. According to the data in Table 1, no significant loss in IgG concentration has been observed for this method [87].
The bovine colostrum is brought from the farm to the dairy for processing. The collection options include refrigeration (4 °C), longer-term freezing, or freeze-drying [3][88]. Refrigeration at 4 °C in plastic containers maintains the Ig properties up to 1 week [89].
For large-scale production, there are several possible thermal treatment processes available. Both LTST pasteurization and HTST pasteurization can be performed efficiently in a continuous plate heat exchanger. Numerical results obtained by Lazaar et al. [90] point out that the turbulence depends on the angle of plate corrugations’ inclination. Therefore, in the case of colostrum processing, attention must be paid to the selection of the flow forms of plates of the plate heat exchanger and the distance between the plates, and possibly tested experimentally beforehand. The colostrum feed enters the regeneration section, absorbs heat from the pasteurized colostrum stream, then enters the pasteurization section. Process integration can easily reach a 96% regeneration rate [91].
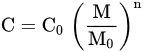
where, C is the equipment cost in capacity M0, and C0 is the equipment cost in capacity M. The scale index, exponent n varies with a type of equipment, i.e., heat exchangers have the value 0.65 for n [94].
The most reliable cost assessment method for food process equipment is to conduct budget intelligence from key equipment manufacturers or suppliers. However, pricing requires accurate and detailed information about the device sizing, material choices, operating and cleaning conditions, etc. Another way of estimating the prices of food process equipment is to extract the unit processes into small parts and to compile transparent price information for the process device based on standardized parts. Investment costs will increase tenfold when the batch heat treatment becomes continuous. The continuous process allows for a high degree of regeneration of thermal energy, resulting in the cost of energy per unit falling to the tenth. If the energy source is steam, there is an energy cost of 4% more compared to the batch heat treatment process.
Data on the previously described processing techniques regarding the bacterial and IgG inactivation rate have been summarized in Table 1. The IgG concentration is used as an immediate comparison as most data are given. However, the given IgG inactivation rates might not equal the amount of active antibodies, as the processing affects their functionality. Regarding most of the other immune components, there is less information available. HTST pasteurization, for example, will lead to the inactivation of 50% of the initial LPO activity [36].
6. Conclusions
Bovine colostrum invigorates newborn calves and supports their immune defense. It contains a variety of different antibodies for conferring passive immunity and for combating microbial infection. Furthermore, about 70 indigenous enzymes have been identified in bovine milk as well as other bioactive components such as carbohydrates, (glycol-) proteins, e.g. lactoferrin and caseins, growth factors, cytokines, lipids, enzymes, vitamins, and minerals.
The amount of bioactive components and elements are significantly higher in colostrum than in milk. Due to the variety of nutritive beneficial components with chemical/functional activities, bovine colostrum is also of interest for human consumption since healthy cows produce colostrum in excess of the calf’s need. The use of bovine colostrum as food and food supplements for human consumers is gaining increasing interest. There are benefits for all persons especially those active in athletic performance and for those wanting to prevent or treat gastrointestinal, respiratory, inflammatory, and bone development diseases.
However, there are risks of the growth of both spoilage and pathogenic bacteria in the bovine colostrum, which can lead to the spread of zoonotic diseases from bovine origin. In addition, the product quality is lowered due to contamination, which can be a result of poor harvesting and subsequent storage conditions. This review outlines the literature on microbial hazards in bovine colostrum, which shows the need for treatment before consumption. There are suitable processing techniques listed in the review. The design of a treatment process contains three options for the heat treatment of bovine colostrum decentral or central. The procedures presented focus on ensuring the food safety and keeping the high nutritional values for consumers. The review pinpoints that the processing of bovine colostrum at the dairy farms with large enough colostrum production can improve the quality and extend the shelf life of it.
On-farm processing would improve the quality of bovine colostrum used both at home and in small processes, e.g. as ingredients. The traditional pasteurization methods are cost-effective compared with newer processing methods, e.g. HPP. The investment costs of the pasteurization process are adjustable in accordance with the production capacity of the bovine colostrum treated. The pasteurization process does not increase the costs by bringing in other consecutive cost-intensive process steps into the production. Beyond this, studies on more optimized industrial scale heat treatment in combination with the maintenance of nutritional values are required.
References
- McGrath, B.A.; Fox, P.F.; McSweeney, P.L.H.; Kelly, A.L. Composition and properties of bovine colostrum: A review. Dairy Sci. Technol. 2016, 96, 133–158.
- Korhonen, H.J. Bioactive milk proteins and peptides: From science to functional applications. Aust. J. Dairy Technol. 2009, 64, 16.
- Foley, J.A.; Otterby, D.E. Availability, storage, treatment, composition, and feeding value of surplus colostrum: A review. J. Dairy Sci. 1978, 61, 1033–1060.
- Gosch, T.; Apprich, S.; Kneifel, W.; Novalin, S. A combination of microfiltration and high pressure treatment for the elimination of bacteria in bovine colostrum. Int. Dairy J. 2014, 34, 41–46.
- El-Loly, M. Bovine milk immunoglobulins in relation to human health. Int. J. Dairy Sci. 2007, 2, 183–195.
- dos Santos Oliveira Silva, E.G.; do Nascimento Rangel, A.H.; Mürmam, L.; Bezerra, M.F.; de Oliveira, J.P.F. Bovine colostrum: Benefits of its use in human food. Food Sci. Technol. 2019, 39, 355–362.
- Playford, R.J.; Weiser, M.J. Bovine colostrum: Its constituents and uses. Nutrients 2021, 13, 265.
- Borad, S.G.; Singh, A.K. Colostrum immunoglobulins: Processing, preservation and application aspects. Int. Dairy J. 2018, 85, 201–210.
- Fecteau, G.; Baillargeon, P.; Higgins, R.; Paré, J.; Fortin, M. Bacterial contamination of colostrum fed to newborn calves in Québec dairy herds. Can. Vet. J. 2002, 43, 523–527.
- Houser, B.A.; Donaldson, S.C.; Kehoe, S.I.; Heinrichs, A.J.; Jayarao, B.M. A survey of bacteriological quality and the occurrence of Salmonella in raw bovine colostrum. Foodborne Pathog. Dis. 2008, 5, 853–858.
- Morrill, K.M.; Conrad, E.; Lago, A.; Campbell, J.; Quigley, J.; Tyler, H. Nationwide evaluation of quality and composition of colostrum on dairy farms in the United States. J. Dairy Sci. 2012, 95, 3997–4005.
- Godden, S.M.; Lombard, J.E.; Woolums, A.R. Colostrum management for dairy calves. Vet. Clinics. N. Am. Food Anim. Pract. 2019, 35, 535–556.
- Lora, I.; Gottardo, F.; Bonfanti, L.; Stefani, A.L.; Soranzo, E.; Dall’Ava, B.; Capello, K.; Martini, M.; Barberio, A. Transfer of passive immunity in dairy calves: The effectiveness of providing a supplementary colostrum meal in addition to nursing from the dam. Animal 2019, 13, 2621–2629.
- Lima, S.F.; Teixeira, A.G.V.; Lima, F.S.; Ganda, E.K.; Higgins, C.H.; Oikonomou, G.; Bicalho, R.C. The bovine colostrum microbiome and its association with clinical mastitis. J. Dairy Sci. 2017, 100, 3031–3042.
- Derakhshani, H.; Plaizier, J.C.; de Buck, J.; Barkema, H.W.; Khafipour, E. Composition of the teat canal and intramammary microbiota of dairy cows subjected to antimicrobial dry cow therapy and internal teat sealant. J. Dairy Sci. 2018, 101, 10191–10205.
- Klein-Jöbstl, D.; Quijada, N.M.; Dzieciol, M.; Feldbacher, B.; Wagner, M.; Drillich, M.; Schmitz-Esser, S.; Mann, E. Microbiota of newborn calves and their mothers reveals possible transfer routes for newborn calves’ gastrointestinal microbiota. PLoS ONE 2019, 14, e0220554.
- Stewart, S.; Godden, S.M.; Bey, R.; Rapnicki, P.; Fetrow, J.; Farnsworth, R.; Scanlon, M.; Arnold, Y.; Clow, L.; Mueller, K.; Ferrouillet, C.; Preventing bacterial contamination and proliferation during the harvest, storage, and feeding of fresh bovine colostrum. J. Dairy Sci. 2005, 88, 2571–2578. [Google Scholar] [CrossRef]
- Claeys, W.L.; Cardoen, S.; Daube, G.; de Block, J.; Dewettinck, K.; Dierick, K.; de Zutter, L.; Huyghebaert, A.; Imberechts, H.; Thiange, P.; Vandenplas, Y.; Herman, L. Raw or heated cow milk consumption: Review of risks and benefits. Food Control 2013, 31, 251–262.
- Fernandes, R. Microbiology Handbook: Dairy Products; Royal Society of Chemistry: Leatherhead, UK, 2009.
- Elizondo-Salazar, J.A.; Heinrichs, A.J. Heat treating bovine colostrum. Appl. Anim. Sci. 2008, 24, 530–538.
- Stelwagen, K.; Carpenter, E.; Haigh, B.; Hodgkinson, A.; Wheeler, T.T. Immune components of bovine colostrum and milk. J. Anim. Sci. 2009, 87 (Suppl. 1), 3–9.
- Park, Y.W.; Nam, M.S. Bioactive peptides in milk and dairy products: A review. Korean J. Food Sci. Anim. Resour. 2015, 35, 831–840.
- Future Market Insights. Colostrum Market to Surge at 6.4% CAGR, Rising Health Concern due to COVID-19 Will Promote Overall Growth, Says FMI. 2020. Available online: https://www.futuremarketinsights.com/press-release/colostrum-market (accessed on 20 December 2020)
- Korhonen, H.J. Bioactive milk proteins, peptides and lipids and other functional components derived from milk and bovine colostrum. In Functional Foods, 2nd ed.; Saarela, M., Ed.; Woodhead Publishing Series in Food Science, Technology and Nutrition; Woodhead Publishing: Cambridge, UK, 2011; pp. 471–511.
- Contarini, G.; Povolo, M.; Pelizzola, V.; Monti, L.; Bruni, A.; Passolungo, L.; Abeni, F.; Degano, L. Bovine colostrum: Changes in lipid constituents in the first 5 days after parturition. J. Dairy Sci. 2014, 97, 5065–5072.
- Korhonen, H.J.; Marnila, P.; Gill, H.S. Milk immunoglobulins and complement factors. Br. J. Nutr. 2000, 84 (Suppl. 1), S75–S80.
- Roos, N.; Mahé, S.; Benamouzig, R.; Sick, H.; Rautureau, J.; Tomé, D. 15N-labeled immunoglobulins from bovine colostrum are partially resistant to digestion in human intestine. J. Nutr. 1995, 125, 1238–1244.
- Ulfman, L.H.; Leusen, J.H.W.; Savelkoul, H.F.J.; Warner, J.O.; van Neerven, R.J.J. Effects of bovine immunoglobulins on immune function, allergy, and infection. Front. Nutr. 2018, 5, 52.
- Anderson, R.C.; Dalziel, J.E.; Haggarty, N.W.; Dunstan, K.E.; Gopal, P.K.; Roy, N.C. Short communication: Processed bovine colostrum milk protein concentrate increases epithelial barrier integrity of Caco-2 cell layers. J. Dairy Sci. 2019, 102, 10772–10778
- Fox, P.F.; Kelly, A.L. Indigenous enzymes in milk: Overview and historical aspects—Part 1. Int. Dairy J. 2006, 16, 500–516.
- Fox, P.F.; Kelly, A.L. Indigenous enzymes in milk: Overview and historical aspects—Part 2. Int. Dairy J. 2006, 16, 517–532.
- Pakkanen, R.; Aalto, J. Growth factors and antimicrobial factors of bovine colostrum. Int. Dairy. J. 1997, 7, 285–297.
- Korhonen, H.J. Antimicrobial factors in bovine colostrum. Agric. Food Sci. 1977, 49, 434–447.
- Lie, O.; Solbu, H.; Syed, M. A genetic association between bovine serum and colostrum lysozyme levels. Anim. Genet. 1986, 17, 39–45.
- Reiter, B. Review of nonspecific antimicrobial factors in colostrum. J. Vet. Res. 1978, 9, 205–224.
- Korhonen, H.J. Production and properties of health-promoting proteins and peptides from bovine colostrum and milk. Cell. Mol. Biol. 2013, 59, 12–24.
- Plaut, A.G.; St. Geme, J. Lactoferrin. In Handbook of Proteolytic Enzymes, 3rd ed.; Rawlings, N.D., Salvesen, G., Eds.; Academic Press: London, UK, 2013; pp. 3635–3640.
- Gelsinger, S.L.; Jones, C.M.; Heinrichs, A.J. Effect of colostrum heat treatment and bacterial population on immunoglobulin G absorption and health of neonatal calves. J. Dairy Sci. 2015, 98, 4640–4645.
- Wittanalai, S.; Tanruean, K.; Mapoong, P. Inhibition of coliform bacteria by lactic acid bacteria isolated from nham hed (fermented mushroom). Appl. Mech. Mater. 2019, 886, 56–60.
- Behera, S.S.; Ray, R.C.; Zdolec, N. Lactobacillus plantarum with functional properties: An approach to increase safety and shelf-life of fermented foods. BioMed Res. Int. 2018, 2018, 9361614.
- Parodi, P.W. The role of intestinal bacteria in the causation and prevention of cancer: Modulation by diet and probiotics. Aust. J. Dairy Technol. 1999, 54, 103–121.
- Morrin, S.T.; Lane, J.A.; Marotta, M.; Bode, L.; Carrington, S.D.; Irwin, J.A.; Hickey, R.M. Bovine colostrum-driven modulation of intestinal epithelial cells for increased commensal colonisation. Appl. Microbiol. Biotechnol. 2019, 103, 2745–2758.
- Kuhl, G.C.; Lindner, J.D.D. Biohydrogenation of linoleic acid by lactic acid bacteria for the production of functional cultured dairy products: A review. Foods 2016, 5, 13.
- Angelin, J.; Kavitha, M. Exopolysaccharides from probiotic bacteria and their health potential. Int. J. Biol. Macromol. 2020, 162, 853–865.
- Janzen, J.J. Economic losses resulting from mastitis: A review. J. Dairy Sci. 1970, 53, 1151–1160.
- Jans, C.; Meile, L.; Kaindi, D.W.M.; Kogi-Makau, W.; Lamuka, P.; Renault, P.; Kreikemeyer, B.; Lacroix, C.; Hattendorf, J.; Zinsstag, J.; Schelling, E.; Fokou, G.; Bonfoh, B. African fermented dairy products–Overview of predominant technologically important microorganisms focusing on African Streptococcus infantarius variants and potential future applications for enhanced food safety and security. Int. J. Food Microbiol. 2017, 250, 27–36.
- Abebe, E.; Gugsa, G.; Ahmed, M. Review on major food-borne zoonotic bacterial pathogens. J. Trop. Med. 2020, 2020, 4674235.
- Fritsche, O. Mikrobiologie; Springer Spektrum: Wiesbaden, Germany, 2016.
- Zadoks, R.N.; Middleton, J.R.; McDougall, S.; Katholm, J.; Schukken, Y.H. Molecular epidemiology of mastitis pathogens of dairy cattle and comparative relevance to humans. J. Mammary Gland Biol. Neoplasia 2011, 16, 357–372.
- Hoorfar, J. Rapid Detection, Characterization, and Enumeration of Foodborne Pathogens; ASM Press: Washington, DC, USA, 2011.
- Gyles, C.L.; Prescott, J.F.; Prescott, J.F.; Songer, J.G.; Thoen, C.O.; Songer, G. Pathogenesis of Bacterial Infections in Animals; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2004.
- Moretti, A.; Pasquali, P.; Mencaroni, G.; Boncio, L.; Piergili Fioretti, D. Relationship between cell counts in bovine milk and the presence of mastitis pathogens (yeasts and bacteria). J. Vet. Med. B 1998, 45, 129–132.
- Maunsell, F.P.; Woolums, A.R.; Francoz, D.; Rosenbusch, R.F.; Step, D.L.; Wilson, D.; Janzen, E. Mycoplasma bovis infections in cattle. J. Vet. Intern. Med./Am. Coll. Vet. Intern. Med. 2011, 25, 772–783.
- Zastempowska, E.; Lassa, H. Genotypic characterization and evaluation of an antibiotic resistance of Trueperella pyogenes (Arcanobacterium pyogenes) isolated from milk of dairy cows with clinical mastitis. Vet. Microbiol. 2012, 161, 153–158.
- Mager, D.L. Bacteria and cancer: Cause, coincidence or cure? A review. J. Transl. Med. 2006, 4, 14.
- Finke, E.-J.; Tomaso, H.; Frangoulidis, D. Bioterrorismus, infektiologische Aspekte. In Lexikon der Infektionskrankheiten des Menschen: Erreger, Symptome, Diagnose, Therapie und Prophylaxe; Darai, G., Handermann, M., Sonntag, H.-G., Zöller, L., Eds.; Springer: Berlin/Heidelberg, Germany, 2012; pp. 76–392.
- Lindner, J.D.D.; Santarelli, M.; Yamaguishi, C.T.; Soccol, C.R.; Neviani, E. Recovery and identification of bovine colostrum microflora using traditional and molecular approaches. Food Technol. Biotechnol. 2011, 49, 364–368.
- Hasegawa, M.; Iwabuchi, E.; Yamamoto, S.; Esaki, H.; Kobayashi, K.; Ito, M.; Hirai, K. Prevalence and characteristics of Listeria monocytogenes in bovine colostrum in Japan. J. Food Prot. 2013, 76, 248–255.
- Gleeson, C.; Gray, N.F. The coliform index and waterborne disease: Problems of microbial drinking water assessment; CRC Press: London, UK, 1997.
- Cockcroft, P. Bovine Medicine, 3rd ed.; Wiley Blackwell: Chichester, UK, 2015.
- dos Santos, G.; da Silva, J.T.; da Rocha Santos, F.H.; Machado Bittar, C.M. Nutritional and microbiological quality of bovine colostrum samples in Brazil. R. Bras. Zootec. 2017, 46, 72–79.
- Lailler, R.; Sanaa, M.; Chadoeuf, J.; Fontez, B.; Brisabois, A.; Colmin, C.; Millemann, Y. Prevalence of multidrug resistant (MDR) Salmonella in bovine dairy herds in western France. Prev. Vet. Med. 2005, 70, 177–189.
- Bernard, K.A.; Munro, C.; Wiebe, D.; Ongsansoy, E. Characteristics of rare or recently described Corynebacterium species recovered from human clinical material in Canada. J. Clin. Microbiol. 2002, 40, 4375–4381.
- Langoni, H.; Polo Camargo da Silva, C.; Troncarelli, M.Z.; Tata, A.; Belaz, K.R.A.; Eberlin, M.N.; Joaquim, S.F.; Guimarães, F.F.; Bonini Pardo, R.; Nardini Gomes, E. Short communication: Identification of Corynebacterium bovis by MALDI-mass spectrometry. J. Dairy Sci. 2017, 100, 4287–4289.
- Dong, W.-L.; Odah, K.A.; Liu, L.; Xu, Q.-J.; Gao, Y.-H.; Kong, L.-C.; Ma, H.-X. Multidrug resistance genes are associated with a 42-kb island TGI1 carrying a complex class 1 integron in Trueperella pyogenes. J. Glob. Antimicrob. Resist. 2020, 22, 1–4.
- Ali, Z.I.; Saudi, A.M.; Albrecht, R.; Talaat, A.M. The inhibitory effect of nisin on Mycobacterium avium ssp. paratuberculosis and its effect on mycobacterial cell wall. J. Dairy Sci. 2019, 102, 4935–4944.
- Peterz, M.; Butot, S.; Jagadeesan, B.; Bakker, D.; Donaghy, J. Thermal inactivation of Mycobacterium avium subsp. paratuberculosis in artificially contaminated milk by direct steam injection. Appl. Environ. Microbiol. 2016, 82, 2800–2808.
- Nielsen, S.S.; Bjerre, H.; Toft, N. Colostrum and milk as risk factors for infection with Mycobacterium avium subspecies paratuberculosis in dairy cattle. J. Dairy Sci. 2008, 91, 4610–4615.
- Kröger, C.; Schauer, K.; Clerkin, S.R.; Märtlbauer, E.; Fleming, A.B. Draft genome sequence and annotation of Acinetobacter junii MHI21018 isolated from bovine colostrum. Microbiol. Resour. Announc. 2019, 8, e01700–e01718.
- Fasse, S.; Alarinta, J.; Frahm, B.; Wirtanen, G. Bovine colostrum for human consumption—Improving microbial quality and maintaining bioactive characteristics through processing. Dairy 2021, 2, 556–575.
- Rajilić-Stojanović, M.; de Vos, W.M. The first 1000 cultured species of the human gastrointestinal microbiota. FEMS Microbiol. Rev. 2014, 38, 996–1047.
- Borad, S.G.; Singh, A.K.; Meena, G.S.; Arora, S.; Raju, P.N.; Sabikhi, L. Optimization of spray drying of colostrum protein ingredients—A rheological approach. J. Food Eng. 2021, 288, 110247.
- Sats, A.; Kaart, T.; Poikalainen, V.; Aare, A.; Lepasalu, L.; Andreson, H.; Jõudu, I. Bovine colostrum whey: Postpartum changes of particle size distribution and immunoglobulin G concentration at different filtration pore sizes. J. Dairy Sci. 2020, 103, 6810–6819.)
- Korhonen, H.J.; Pihlanto, A. Technological options for the production of health-promoting proteins and peptides derived from milk and colostrum. Curr. Pharm. Des. 2007, 13, 829–843.)
- Phipps, A.J.; Beggs, D.S.; Murray, A.J.; Mansell, P.D.; Stevenson, M.A.; Pyman, M.F. Survey of bovine colostrum quality and hygiene on northern Victorian dairy farms. J. Dairy Sci. 2016, 99, 8981–8990.
- European Parliament and the Council of the European Union. Regulation (EC) no 852/2004 of the European Parliament and of the Council of 29 April 2004 on the Hygiene of Foodstuffs. 2004. Available online: https://eur-lex.europa.eu/eli/reg/2004/852/oj. (accessed on 10 June 2021).
- Dewulf, J.; Van Immerseel, F. (Eds.) Biosecurity in Animal Production and Veterinary Medicine from Principles to Practice; Acco: Leuven, Belgium, 2018.
- Food Standards Australia New Zealand. Primary Production and Processing (PPP) Standards. 2020. Available online: https://www.foodstandards.gov.au/foodsafety/standards/Pages/Primary-Production-and-Processing-(PPP)-Standards-(Chapter-4).aspx (accessed on 28 March 2021)
- Cummins, C.; Lorenz, I.; Kennedy, E. Short communication: The effect of storage conditions over time on bovine colostral immunoglobulin G concentration, bacteria, and pH. J. Dairy Sci. 2016, 99, 4857–4863.
- Food Standards Australia New Zealand. Food Safety Hub. 2019. Available online: https://www.foodstandards.gov.au/foodsafety/Pages/default.aspx. (accessed on 28 March 2021).
- Elizondo-Salazar, J.A.; Jayarao, B.M.; Heinrichs, A.J. Effect of heat treatment of bovine colostrum on bacterial counts, viscosity, and Immunoglobulin G concentration. J. Dairy Sci. 2010, 93, 961–967.
- Johnson, J.L.; Godden, S.M.; Molitor, T.; Ames, T.; Hagman, D. Effects of feeding heat-treated colostrum on passive transfer of immune and nutritional parameters in neonatal dairy calves. J. Dairy Sci. 2007, 90, 5189–5198.
- Fasse, S.; Alarinta, J.; Wirtanen, G. 2021. Processing of bovine colostrum to improve microbial quality with sustained level of immune components. In SeAMK Ruoka 2021 - ilmastokestävää ruokaketjua edistämässä, Publications of Seinäjoki University of Applied Sciences B. Reports 162; Lauhanen, R.; Junkkari, T.; Mäki, T.; Saarikoski, S. Eds.; 2021. Seinäjoen ammattikorkeakoulu: Seinäjoki, Finland. 2021; pp. 19 – 30.
- Godden, S.M.; McMartin, S.; Feirtag, J.; Stabel, J.; Bey, R.; Goyal, S.; Metzger, L.; Fetrow, J.; Wells, S.; Chester-Jones, H. Heat-treatment of bovine colostrum. II: Effects of heating duration on pathogen viability and immunoglobulin G. J. Dairy Sci. 2006, 89, 3476–3483.
- Stabel, J.R.; Hurd, S.; Calvente, L.; Rosenbusch, R.F. Destruction of Mycobacterium paratuberculosis, Salmonella spp., and Mycoplasma spp. in raw milk by a commercial on-farm high-temperature, short-time pasteurizer. J. Dairy Sci. 2004, 87, 2177–2183.
- De Haan, A.B. Process Technology—An Introduction; De Gruyter: Eindhoven, The Netherlands, 2015; pp. 11–26.
- Godden, S.M.; Smolenski, D.J.; Donahue, M.; Oakes, J.M.; Bey, R.; Wells, S.; Sreevatsan, S.; Stabel, J.; Fetrow, J. Heat-treated colostrum and reduced morbidity in preweaned dairy calves: Results of a randomized trial and examination of mechanisms of effectiveness. J. Dairy Sci. 2012, 95, 4029–4040.
- McGuirk, S.M.; Collins, M. Managing the production, storage, and delivery of colostrum. Vet. Clin. Food Anim. 2004, 20, 593–603.
- Manohar, A.A.; Williamson, M.; Koppikar, G.V. Effect of storage of colostrum in various containers. Indian Pediatr. 1997, 34, 93–295.
- Lazaar, M.; Boughanmi, H.; Bouadila, S.; Jarraya, M. Parametric study of plate heat exchanger for eventual use in a solar pasteurization process designed for small milk collection centers in Tunisia. Sustain. Energy Technol. Assess. 2021, 45, 101174.
- Tomasula, P.M.; Yee, W.C.F.; MacAloon, A.J.; Nutter, D.W.; Bonnaillie, L.M. Computer simulation of energy use, greenhouse gas emissions, and process economics of the fluid milk process. J. Dairy Sci. 2013, 96, 3350–3368.
- Meraj, M.; Mahmood, S.M.; Khan, M.E.; Azhar, M.; Tiwari, G.N. Effect of N-Photovoltaic thermal integrated parabolic concentrator on milk temperature for pasteurization: A simulation study. Renew. Energy 2021, 163, 2153–2164.
- Al-Hilphy, A.R.; Abdulstar, A.R.; Gavahian, M. Moderate electric field pasteurization of milk in a continuous flow unit: Effects of process parameters, energy consumption, and shelf-life determination. Innov. Food Sci. Emerg. Technol. 2021, 67, 102568.
- Maroulis, Z.B.; Saravacos, G.D. Food Process Design; Marcel Dekker: New York, NY, USA, 2003; pp. 21–57.
- Parin, M.A.; Zugarramurdi, A. Investment and production costs analysis in food processing plants. Int. J. Prod. Econ. 1994, 34, 83–89.
- Couper, J.R.; Hertz, D.W.; Smith, L. Process economics. In Perry’s Chemical Engineers’ Handbook, 8th ed.; Perry, R.H., Green, D.W., Eds.; McGraw-Hill: New York, NY, USA, 2007; 56p.