Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
He, S. Nanomaterials in Cell-Based Drug Delivery Systems. Encyclopedia. Available online: https://encyclopedia.pub/entry/16345 (accessed on 24 May 2026).
He S. Nanomaterials in Cell-Based Drug Delivery Systems. Encyclopedia. Available at: https://encyclopedia.pub/entry/16345. Accessed May 24, 2026.
He, Shun. "Nanomaterials in Cell-Based Drug Delivery Systems" Encyclopedia, https://encyclopedia.pub/entry/16345 (accessed May 24, 2026).
He, S. (2021, November 24). Nanomaterials in Cell-Based Drug Delivery Systems. In Encyclopedia. https://encyclopedia.pub/entry/16345
He, Shun. "Nanomaterials in Cell-Based Drug Delivery Systems." Encyclopedia. Web. 24 November, 2021.
Copy Citation
The combination of nanomaterials with cell-based drug delivery systems has shown tremendous advantages in cancer treatment.
cell therapy
biomimetic strategy
nanomaterial
cancer treatment
1. Introduction
Recent decades have witnessed the tremendous progress of nanobiotechnology in cancer treatment [1][2]. Nano-based drug delivery systems (DDS) are one of the most widely investigated strategies to improve the targetability of therapeutic molecules, increase circulation time, and enhance the total bioavailability [3]. As important constituents of nano-based DDS, various nanomaterials like organic nanomaterials, inorganic nanomaterials, and hybrid nanomaterials have been intensively explored in anticancer drug delivery, owing to their unique properties [4][5][6]. The delivery of therapeutic agents using nanomaterials holds numerous advantages over their free drug counterparts, which can not only protect the encapsulated drugs from degradation or inactivation before reaching sites of action, but also enable the controlled drug release in specific environments. In addition, both passive and active targeting can be achieved via the enhanced permeability and retention (EPR) phenomenon, or by means of extra modification [7][8]. Moreover, owing to the EPR effect, nanosystems possess the ability to improve the accumulation of chemotherapeutics both as single agent and in combination, which largely elevates the amounts of drugs in target tissue [9][10][11][12]. When it comes to active targeting, two main approaches are currently adopted to improve tumor accumulation of nanoparticles. One is to apply targeting molecules to endow the nanosystems with targetability [13][14]. The other is to modulate the protein corona of nanocarriers to provide a “natural targeting” towards TME [15][16][17]. In theory, nano-based DDS can be employed as an ideal vehicle in cancer treatment. However, many obstacles still impede the wide application of nanomedicine. For instance, although the EPR effect and active targeting approaches can modulate the biodistribution of nanomedicine to a certain extent, only a part of nanomedicine can reach the tumor sites while the majority of them are cleared by the reticuloendothelial system (RES) [18]. Moreover, EPR effect tends to be more efficient in some angiogenic tumor models with leaky blood, there are still some cases that are not suitable for EPR effect [19]. Besides, modification using targeting ligands could potentially compromise the stealth ability of the nano-based DDS [20]. Therefore, other novel strategies in combination with nanotechnology is of great necessity to achieve improved therapeutic performance.
Hopefully, cell-mediated drug delivery has become a promising approach in addressing the aforementioned limitations. This innovative strategy takes advantage of the natural properties of various cells such as prolonged circulation time in blood stream, specific targetability to tumor cells, the ability to cross challenging biological barriers, abundant surface ligands, flexible morphology, and cellular signaling or metabolism [21]. Very recently, d’Avanzo et al. reported a kind of peptide-functionalized-liposomes that were able to selectively bind breast cancer cells, which in vitro demonstrated the ability of the resultant liposomes to target M2-macrophages to exploit a potential hitchhiking effect in vivo, bridging the gap between conventional nanosystems and cell-derived ones [22]. Therefore, cooperating advanced nanomaterials with cell-based therapies can largely strengthen the total therapeutic efficacy through maximizing the advantages of both, while minimizing their inherent shortcomings. In this review, a number of novel DDSs, based on various cell types including leucocytes, erythrocytes, platelets, stem cells, and adipocytes will be highlighted (Figure 1). Different cell types possess distinctive properties, which enables their multifunctional application in personalized cancer treatment. It is discussed how nanomaterials empower the field of cell-based treatment and how cellular characteristics improve the performance of nanomedicine. Diverse delivery strategies that utilize living cell internalization or cell membrane-cloaking in cooperation with multiple treatment modalities including chemotherapy, phototherapy, gene therapy and immunotherapy will be introduced in detail. Table 1 is a summary of nanomaterials in cell-based drug delivery for cancer treatment. In addition, compared to the existing review articles about cell-based therapies, the novelty of this review is that we comprehensively summarize the most widely used cell types in cyto-pharmaceuticals, which highlights the combinational strategy of innovative nanomaterials and cell-derived vectors with very latest research examples [21][23]. Moreover, the current limitations and future orientations for cell-based therapies are also discussed in this paper to provide more detailed instruction for the development of cell therapies.
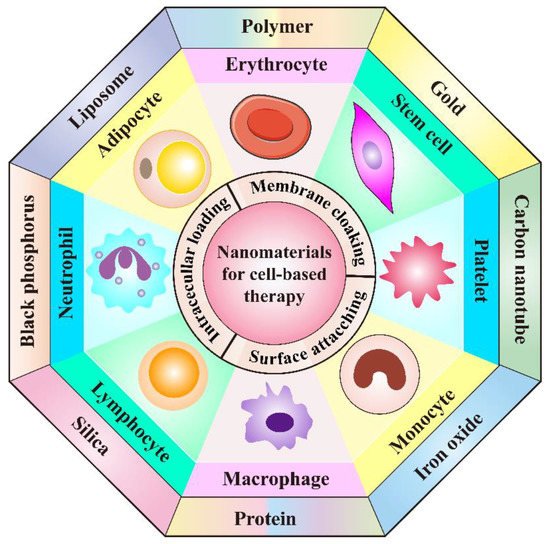
Figure 1. Schematic illustration of the cooperation of cell-based therapies with novel nanomaterials in cancer treatment.
Table 1. Overview of the combination of nanomaterials in cell-based drug delivery systems for cancer treatment.
| Type of Living Cells/Cell Membranes | Combined Nanomaterials/Applied Targeting Molecules | Advantages | Therapeutic Agents | Tumor Model | Therapeutic Performance | Refs. |
|---|---|---|---|---|---|---|
| Neutrophil membrane | PLGA | Achieve efficient tumor targeting, prolong circulation time and promote cellular internalization | Paclitaxel | Human ovarian adenocarcinoma | Inhibit tumor growth and prolong the survival rate | [24] |
| Neutrophil | PEG-b-PLGA and bacteria-secreted outer membrane vesicles | Improve tumor targeting, combine chemotherapy with PTT | Cisplatin | Murine breast cancer | Completely eradicate tumors | [25] |
| T cell | Gold nanospheres | Improve tumor targeting through the recognition of tumor-associated antigens | AuNPs | Human lymphoma | Achieve specific tumor AuNPs accumulation | [26] |
| Lipid nanocapsules | Achieve lymphoid organ-specific targeting | SN-38 | Murine lymphoma | Reduce tumor burden significantly | [27] | |
| CAR T cell membrane | Mesoporous silica | Improve tumor targeting, prolong circulation time | IR780 | Human hepatocellular carcinoma | Possess significant photothermal antitumor effect and tumor imaging | [28] |
| CAR NK cell | Cross-linked multilamellar liposomal vesicles | Improve tumor targeting | Paclitaxel | Human ovarian cancer | Inhibit tumor growth | [29] |
| NK cell membrane | Liposome | Improve tumor targeting, prolong circulation time | Doxorubicin | Human breast cancer | Inhibit tumor growth | [30] |
| Macrophage | Liposome | Improve tumor targeting, promote cellular internalization, recruited to tumor sites by CCL-2 | Resveratrol and Paclitaxel | Murine breast cancer | Inhibit tumor recurrence | [31] |
| Engineered macrophage | Lipopolysaccharide | Improve tumor targeting, induce secretion of TNF-α | Doxorubicin | Human lung cancer | Increase the inhibitory effects on tumor growth and metastasis | [32] |
| Monocyte | Polymer | Improve tumor targeting | Conjugated polymer NPs (CPNs) | Murine glioblastoma | Efficiently deliver CPNs into glioblastoma sites and improve PDT effect | [33] |
| N/A | Cross endothelial barriers and improve tumor targeting | Doxorubicin | Human glioblastoma | Induce cancer cell damage | [34] | |
| Chitosan polymeric micelles | Improve tumor targeting | N/A | Murine breast cancer | Increase NPs accumulation within tumor sites, enhance antitumor efficacy | [35] | |
| Gold-silver nanorods (AuNRs) | Improve tumor targeting, promote immunostimulation | AuNRs and CpG | Murine lymphoma | Ablate primary tumors and elicit a potent immunity to prevent tumors from metastasis and recurrence | [36] | |
| Erythrocyte | PLGA | Enable lung physiology-assisted shear-responsive targeted delivery | Doxorubicin | Murine melanoma | Inhibit tumor growth and metastasis | [37] |
| Iron oxide-based super-paramagnetic NPs | Improve tumor targeting, prolong circulation time | Monoclonal antibody mAb198.3 | Human colon-rectal cancer | Inhibit tumor growth | [38] | |
| Erythrocyte membrane | DSPE-PEG-mannose | Improve tumor targeting, possess outstanding mobility | Antigen peptides self-assembled NPs | Human breast cancer | Promote DC maturation and CTL activation, achieving broad-spectrum breast cancer inhibition | [39] |
| PLGA | Prolong circulation time, efficiently load and deliver oxygen to hypoxic tumor | Perfluorocarbon | Murine breast cancer | Promote cancer radiotherapy | [40] | |
| Erythrocyte-cancer cell hybrid membrane | N/A | Prolong blood circulation, improve targetability and PTT effect | Melanin nanoparticle | Human breast cancer | Inhibit tumor growth | [41] |
| Platelet | Anti-CD22 antibody | Prolong circulation time and achieve previse delivery of DOX to tumor cells | Doxorubicin | Human lymphoma | Inhibit tumor growth and attenuate cardiotoxicity of DOX | [42] |
| Anti-PD-L1 antibody | Excellent inflammatory targeting ability | N/A | Murine breast cancer | Reduce residual tumor growth and metastasis | [43] | |
| Transferrin | Effectively target melanoma | Doxorubicin | Murine melanoma | Reduce melanoma cell growth and inhibit tumor progression | [44] | |
| Platelet membrane | DSPE-PEOz liposome | Enhance tumor affinity and achieve selective drug release in acidic microenvironment | Doxorubicin | Murine colon cancer, breast cancer and pancreatic carcinoma | Inhibit tumor growth | [45] |
| PLGA | Achieve active targeting and immune evasion abilities | Doxorubicin | Murine breast cancer | Eliminate tumor completely and enhance multimodal imaging | [46] | |
| Fe3O4 NPs | Promote targetability to tumor metastasis | Sulfasalazine | Murine breast cancer | Inhibit the metastatic tumor growth | [47] | |
| BPQDs | Improve drug loading efficiency, enhance biocompatibility and targetability | Hederagenin | Human breast cancer | Inhabit tumor growth and decrease the side effects of myelosuppression | [48] | |
| Mesenchymal stem cell | Carbon nanotubes | Improve tumor homing ability | Doxorubicin | Human lung cancer | Promote lung cancer cell apoptosis and eliminate lung tumor after treatment | [49] |
| Liposome | Enhance the intercellular delivery of DOX and improve tumor targeting ability | Doxorubicin | Murine colon adenocarcinoma | Significantly inhibit tumor proliferation, Suppress primary tumor growth and lung metastasis |
[50] | |
| Genome engineered mesenchymal stem cells | PEI-coated MSNs | Increase tumor homing ability, reduce undesired side effects of anticancer treatment | A suicide fusion gene and uracil phosphoribosyl transferase | murine NMU mammary tumor | Induce NMU cancer cells death | [51] |
| Reconstituted high-density lipoprotein | Increase therapeutic efficiency and tumor targetability | pDNA encoding TRAIL | Murine melanoma | Induce cancer cell apoptosis, inhibit pulmonary metastasis tumor growth | [52] | |
| Adipocyte | N/A | Achieve local and sustained release of chemotherapeutics within the TME | Rumenic acid and doxorubicin prodrug | Murine melanoma | Promote antitumor efficacy, downregulate of PD-L1 expression | [53] |
| Adipose-derived stem cell | Superparamagnetic iron oxide NPs | Improve selective delivery | Paclitaxel | Murine brain tumor | Enhance therapeutic efficacy and prolong survival time | [54] |
| PLGA | Achieve sustained drug release and increase tumor targeting ability | Pirarubicin | Human pancreatic cancer | Inhibit tumor growth, induce the apoptosis of tumor cells, cause minimal side effects | [55] | |
| Lipid droplet | N/A | Promote anticancer therapy through metabolic intervention | Pyrolipid | Human ovarian cancer | Inhibit tumor growth | [56] |
2. Leukocyte-Based Drug Delivery Systems
Leukocytes, also called white blood cells (WBCs), are a group of colorless cells with a nucleus, in circulation, which are associated with innate and adaptive immune responses [57]. Circulating leukocytes can be mainly divided into five categories based on their morphological and bio-functional features, including neutrophils, monocytes, lymphocytes, eosinophils, and basophils [58]. Among the leukocytes groups, neutrophils, eosinophils, and basophils are collectively known as granulocytes, in which numerous granules are stored and can be released from the cytosol into extracellular space [59]. Chemotaxis is a fundamental ability of leukocytes, which means that leukocytes can migrate towards chemoattractants like formyl peptides, leukotrienes and complement fragments, or migrate away from chemorepellents [60]. When the infection, inflammation or cancer occurs, the recruitment of leukocytes can be triggered, leading to an increasing number of leukocytes in circulation and their follow-up transmigration [61]. Therefore, leukocytes are regarded as promising candidates for the implementation of the treatment of inflammatory diseases and cancers. During the tumor initiation and progression, numerous leukocytes are recruited towards tumor sites, which is known as leukocyte infiltration and can contribute to an inflammatory tumor microenvironment (TME) [62]. In addition to tumor-infiltration leukocytes that play a crucial role in TME, leukocytes in circulation also have great potential for cancer treatment via serving as vehicles. Leukocytes circulating in blood vessels are more likely to locate near the vessel wall instead of the vessel center, causing adhesive interactions between leukocytes in circulation and endothelial cells within the inner blood vessel through intercellular adhesion molecule-1 (ICAM-1) on endothelial cells [21][63]. Interestingly, circulating tumor cells that are essential to tumor progression and metastasis share similar migration and adhesion characteristics in blood vessels as leukocytes, in circulation [64]. Therefore, leukocytes can be potentially exploited as an ideal DDS for cancer treatment (Figure 2).
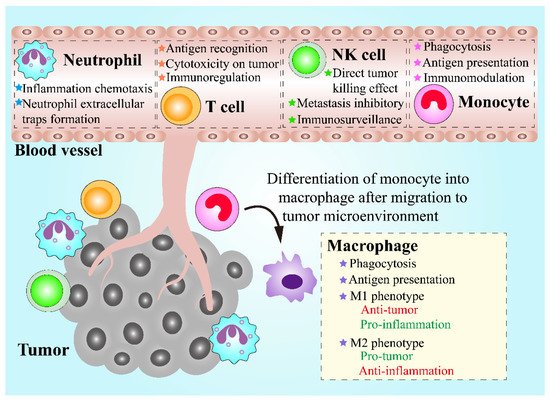
Figure 2. Schematic demonstration of the properties of various leukocytes and their functions in antitumor therapies.
2.1. Neutrophils
Neutrophils are the most abundant leukocytes with unique nuclear morphology and defined granule content, which are a kind of myeloid cells that are sourced from immature neutrophil precursors in the bone marrow [65]. In addition, neutrophils play a crucial role in the immune response against infection and inflammation. Under infectious and inflammatory conditions, the life span of neutrophils is significantly extended, which allows neutrophils to persist for a sufficient period to express cytokines, release bioactive molecules such as granules, and mediate the recruitment of other immune cells [66]. More importantly, not only the release process of neutrophils from the bone marrow to circulation, but also the mobilization of neutrophils to inflammatory and tumor niches, are involved in neutrophil chemotaxis, that is mediated by chemokine receptors and integrin adhesion receptors expressed on neutrophils [67]. In addition to chemotaxis capacity, another important characteristic is neutrophil extracellular traps (NET) formation, whose process is also described as NETosis. NETosis is, in fact, a type of cell death of neutrophils which differs from neutrophil necrosis and apoptosis. In infection, inflammatory disorder niches and TME, NETosis occurs companied by release of intracellular contents, such as chromatin, granules of neutrophils, and carried cargoes [68]. Taken together, neutrophils have great potential to be an excellent drug delivery carrier with tumor targeting nature and drug release ability within TME for cancer therapy.
A variety of nanomaterials have been utilized in DDS in cooperation with neutrophils for cancer treatment. One approach to apply neutrophils as a drug delivery vector is to construct biomimetic drug loaded nanoparticles (NPs) with neutrophils membrane coating. PLGA, fully named as poly (lactic-co-glycolic acid), is a widely used polymer due to its high biocompatibility and biodegradability [69]. Moreover, formulation based on PLGA can achieve a sustained drug release behavior for periods up to 6 months, and up to 19 formulations involved in PLGA material have been approved by FDA [70]. Wang et al. developed PLGA-based NPs that were coated with a neutrophil membrane to carry the chemotherapeutic compound paclitaxel (PTX) for ovarian adenocarcinoma treatment [24]. PTX was preloaded in the PLGA core, then the core was encapsulated by the membrane of neutrophils which was further modified with a tumor necrosis factor-related apoptosis-inducing ligand (TRAIL). The developed system, named TNM-PN, displayed great serum stability and could release PTX in a sustained manner in vitro. Moreover, TRAIL on TNM-PN surface promoted the uptake of TNM-PN by the SKOV3 cells while the neutrophil membrane coating enabled TNM-PN to escape immune elimination as well as prolong their circulation in vivo. In vitro experiments showed that TNM-PN could selectively bind with inflamed human umbilical vein endothelial cells (HUVECs) and in vivo results demonstrated the specific biodistribution and high accumulation of TNM-PN at tumor sites, both indicating the excellent targeting capacity of the constructed nanocomplexes that is a product of the neutrophil membrane clocking. More crucially, SKOV3 tumor-bearing nude mice in TNM-PN treatment group showed significant tumor inhibition compared with other groups, confirming the excellent targeting capacity, successful drug release and high antitumor efficiency of TNM-PN.
Apart from the neutrophil membrane cloaking strategy, another approach to employ neutrophil in drug delivery is the hitchhiking strategy, which means that living neutrophils can pick up NPs and transport them. Based on this concept, Li et al. established a pathogen-mimicking NP, called nano-pathogenoids (NPNs), which could be recognized and internalized by neutrophils, due to the immune response to fight pathogen of neutrophils [25]. NPNs were in a core–shell structure and prepared from cisplatin and photothermal transducer polymer PBIBDF-BT, loaded PEG-b-PLGA mi-cellar NPs, encrusted with outer membrane vesicles (OMVs), which were secreted from Escherichia coli bacteria. Hence, NPNs with pathogenic features could be recognized and internalized by neutrophils in circulation. Additionally, neutrophils could retain the chemotaxis ability after the uptake of NPNs, for which NPNs were capable of hitchhiking neutrophils in circulation to arrive at inflamed issue. Upon photothermal therapy (PTT), numerous tumor cells were killed, accompanied by TME inflammation and follow-up neutrophils recruitment and inflation. Therefore, NPNs could be transferred and accumulated at inflamed tumor tissue as the in vivo results showed, displaying great tumor targeting ability after PTT. In vitro results also revealed that neutrophils could achieve NET formation to release carried contents when they were treated with phorbol myristate acetate mimicking inflammatory condition, and the re-leased NPNs and cisplatin were still active to cause EMT6 cell death. More crucially, in vivo results showed that, once the neutrophil containing NPNs reached the tumor niche, NPNs were sufficiently released and were taken up by EMT6 tumor cells. Finally, this PTT, in combination with neutrophil-mediated chemotherapy, contributed to a 60% tumor-free rate and a 97% tumor growth inhibition, which completely eradicated all tumors after repeated treatment in EMT6-bearing mice.
2.2. Lymphocytes
Lymphocytes are the second largest subtype of leukocytes in human, taking up about 30% of the population [71]. The main subsets of lymphocytes, including T lymphocytes (T cells), B lymphocytes (B cells), and natural killer cells (NK cells), have an essential effect on host immunity and perform their individual function against virus-es, bacteria, and tumor cells, due to their different roles in the immune system. For cancer treatment, lymphocytes or engineered lymphocytes have gained increasing concerns because of the rapid development of cancer immunotherapy [72]. T cell is a critical participant of the adaptive immune system in response to a broad array of antigens mediated by T cell receptors [73]. CD8+ T cells, known as cytotoxic T lymphocyte (CTL), and CD4+ T cells, also named helper T cells, are two important subsets of T cells, which are considered as the principal weapons of immunity against cancer [74]. T cells are capable of homing to the TME where tumor-associated antigens exist [75]. Unlike T cells, NK cells can directly kill cancer cells by secreting cytokines and perform their cytotoxic effect with no demand of the previous encounter with the antigen [76]. Moreover, NK cells are members of the innate immunity system and can recognize the target antigen, independently from major histocompatibility complex I (MHC-I). Based on NK cell receptors regulation, NK cells are capable of realizing the identification and targeting of cancer cells [77]. Moreover, both T cells and NK cells have been engineered to express chimeric antigen receptors (CARs) with tumor specificity, which promotes their efficacy and empower their functions [78]. Therefore, T cells and NK cells show great potential to mediate drug delivery for cancer therapy.
2.2.1. T Lymphocytes
Drug transportation based on T cells have been investigated and several types of drug delivery platform have been proposed. One of the developing approaches of the T cell-based drug delivery strategy is to load drugs into T cells. Steinfeld et al. compared two normal loading methods of T cells’ incorporation with doxorubicin (DOX)-laden magnetite NPs, including electroporation and endocytosis. It was concluded that endocytosis by a smooth coincubation process was an effective method of drug uptake into cells, along with a reduced death rate of T cells [79]. In light of this, Kennedy et al. applied an endocytosis method by colocalization of gold nanospheres (AuNPs) to be loaded into T cells, which demonstrated a high loading efficiency as well as no influence on viability and function of T cells. Both in vitro and in vivo studies confirmed that AuNP-loaded T cells retained their homing ability to tumor sites and achieved specific tumor AuNPs accumulation, displaying the potential to promote the effect of AuNPs-based phototherapy [26].
Apart from loading therapeutic agents or NPs inside cells, attachment of NPs on the surface of T cells is an alternative approach to develop T cell-based drug delivery vehicles. Liposome is an ideal nanomaterial incorporated with T cells due to convenient functionalization with thiol-reactive group for covalent attachment to exofacial thiols on T cell surfaces, based on disulfide bond formation. Wayteck et al. constructed a siRNA-loaded liposome coupled to the surface of CTL [80]. The pyridyldithiopropionate (PDP)-functionalized lipids were incorporated in liposomes bilayer to form PDP-liposome that could couple with free thiol groups highly expressed on CTL mem-brane via a disulfide bond formation. The formed disulfide bond could respond to re-duction stress such as glutathione, a common reducing agent that is abundant within the TME, allowing the detachment of liposome from CTL surface. In vitro results demonstrated that the coupling of liposome to CTL surface was reversible, and the coupling ability could be improved by T cell activation correlated with CD25 receptor expression. More importantly, liposome coupled CTLs could proliferate and cause cytotoxic effect against targeted tumor cells, indicating that the surface attachment did not affect cell proliferation and function. On account of this, another study by Huang et al. showed that polyclonal T cells expressed lymph node-homing receptors incorporated with lipid nanocapsules (NCs) could act as efficient carriers with active targeting ability to lymphoma cells [27]. The lymph node-active targeting ability is crucial because lymphoma is unlike solid tumors, which lack the EPR effect, making the potent therapeutic agent SN-38 display poor pharmacokinetics. In this study, SN-38 was entrapped into NCs containing multilamellar lipid which could covalently bind to free thiols at T cell surface by the formation of a thioether bond. The NCs-functionalized polyclonal T cells could migrate to the lymphoid organs where lymphoma cells resided, which was achieved by organ-specific targeting rather than tumor-specific targeting. In a disseminated lymphoma mice model, this T cell-based DDS reduced tumor burden significantly owing to the enhanced delivery of SN-38 to tumor cells.
In addition to intracellular loading and surface coupling, T cell membrane camouflaged NPs is the third approach for T cell-based vector development. Ma et al. exploited a novel biomimetic nanomaterial based on CAR-T cell membrane-cloaked mesoporous silica NPs (MSNs), aiming to deliver IR780 dye for hepatocellular carcinoma (HCC) treatment [28]. IR780, a photothermal agent, were encapsulated into MSNs to form a biodegradable core. IR780-loaded MSNs were further coated by membrane derived from lentivirus transfected CAR-T cells that could express glypican-3 (GPC3)-specific CARs which enabled CAR-T cells to target HCC tumors by recognizing GPC3 on HCC cells. Both in vitro and in vivo results revealed that GPC3-specific CAR-T cell mem-brane camouflaged IR780-loaded MSNs (CIMs) could selectively accumulate in HCC cells, causing a significant PTT effect for tumor ablation upon 808 nm laser irradiation.
2.2.2. Natural Killer Cells
Similar as T cells, NK cells have also been utilized to create novel cell-based drug delivery platforms in several ways [81]. NK92 cell line is the most extensively characterized type of NK cells and has been approved by FDA for cancer immunotherapy [82]. Siegler et al. constructed drug carriers based on CAR-engineered NK92 cells (CAR.NK cells), whose surface was attached by cross-linked multilamellar liposomal vesicles (cMLVs) containing PTX [29]. CAR.NK cells were generated via retroviral transduction, aiming to increase their homing ability, selectivity, and cytotoxicity towards tumor cells. The cMLVs were functionalized with thiol-reactive maleimide head groups that could covalently conjugate with free thiols highly expressed on NK cell surface, allowing stable attachment of cMLVs to CAR.NK cells surface. An appropriate dose of PTX was encapsulated into cMLVs, which could kill tumor cells and showed no toxicity to CAR.NK cells. According to in vitro experiments, cMLVs were not internalized by NK cells after attachment, and did not cause damages on NK cells, which retained their migratory ability to chemoattractant. CAR.NK cells also released interferon (IFN)-γ; thus, they caused specific cytotoxicity to antigen-expressed cancer cells. In SKOV.CD19 ovarian tumor-bearing mice, the developed DDS CAR.NK.CLV could home to tumor sites and promote PTX accumulation within tumor niches, leading to significant tumor growth inhibition.
The NK cell membrane is also regarded as candidate for NK-cell based drug de-livery. Pitchaimani et al. combined liposomes and NK cells to newly design a kind of liposomal drug vector called NKsome through the infusion of NK cell membrane into liposomes driven by electrostatic interactions [30]. NK92 cells were chosen in this study also, due to their outstanding characteristics, including easy-expansion ex vivo, broad-spectrum targeting capacity, and promoted cytotoxicity. These fusogenic NKsomes could retain the surface receptor proteins of NK92 cells, which allowed NKsomes to mimic properties similar to NK cells, such as tumor homing capacity and immunosurveillance of cancer cells. Furthermore, NK cell membrane-fused NKsomes had positive charged surfaces, which enabled them to escape from lysosomal degradation in target cells due, to the fusion property of NKsomes with the target cell mem-brane. Moreover, this fusion property could realize accurate release of encapsulated drug inside tumor cells. In vitro investigation demonstrated that NKsomes were stable in serum and accumulated significantly in MCF-7 breast cancer cells with wonderful targeting efficiency; therefore, when loaded DOX into NKsomes, the constructed nanocomplexes (DOX@NKsomes) showed superior cytotoxicity to MCF-7 cells without significant immunogenicity. In addition, in vivo results revealed that DOX@NKsomes had longer circulation time and distributed mainly within the TME, which induced a significant inhibitory effect on MCF-7 tumor growth.
2.3. Macrophages and Monocytes
Macrophages are a type of phagocyte and can be roughly classified into M1 macrophages (classically activated macrophages) and M2 macrophages (alternatively activated macrophages) [83]. M1 macrophages promote immune response against tumor cells by secretion of inflammatory cytokines with potent antitumor activity such as tumor necrosis factor, while M2 macrophages show an anti-inflammatory function [84]. On this account, a shift of M2 macrophages towards M1 is in favor of tumor sup-pression. Monocytes are mononuclear phagocytes that have an important impact on tumor growth and progression. In addition, monocytes can differentiate into a macrophages or dendritic cells (DCs) under certain conditions [85]. Tumor cells can generate monocyte chemoattractant protein-1 (CCL-2), also known as MCP-1, which is able to recruit monocytes and macrophages [86]. For this reason, monocytes and macrophages have a tumor targeting ability that can be employed for drug delivery.
Macrophages, as a kind of inflammatory cell, have an innate chemotaxis capacity, which means that they can be driven to inflammation by inflammatory factors such as tumor necrosis factor (TNF)-α, interleukin (IL)-10. C-C chemokine receptor type 2 (CCR2) is expressed on macrophages in response to CCL-2, allowing the specific migration of macrophages towards TME. In addition, an essential requirement that enables the feasibility of cell-based drug delivery is that the loaded drug does not induce damage to carrier cells and would not be degraded inside the cell. Hence, in view of these facts, Qiu et al. exploited a “dual-guide” drug delivery vector based on macro-phages for triple-negative breast cancer treatment [31]. It is essential to give chemo-therapy after surgery for triple-negative breast cancer treatment due to the particularly high risk of recurrence. Given that a sustain inflammation occurs in tumor sites after tumor resection, drug-containing liposomes loaded macrophages were developed for tumor targeting achieved by dual guide of tumor and inflammation, aiming to ensure the distinguished targeting ability. Liposomes were PEGylated and modified with octaarginine (R8), a cell penetrating peptide for promoting tumor cell penetration, to load resveratrol (Res) and PTX simultaneously. The obtained liposomes were co-incubated with macrophages to construct the final system PTX/Res-R8-Lip@MP without damage and polarization on macrophages. In vitro results revealed that PTX/Res-R8-Lip@MP could migrate by nanotube formation in response to both inflammation and tumor attraction and enter into tumor cells accompanied by the re-lease of liposomes. The released R8-modified PEGylated liposomes were taken up by tumor cells through a “two-way delivery” mechanism—cell membrane fusion and cell penetrating—which could achieve sustained release of therapeutic agent Res and PTX. Res could block the pro-inflammation pathway and combination of Res and PTX could enhance inhibition of tumor initiating cells, which showed the significant anti-recurrence and anti-stemness effect. More importantly, PTX/Res-R8-Lip@MP efficiently inhibited tumor regrowth in a 4T1 orthotopic mouse model, avoided inflammation, and promoted tumor cell apoptosis. Besides, Guo et al. developed engineered macrophages by anchoring lipopolysaccharides (LPS) on their surfaces to deliver DOX for lung cancer treatment [32]. LPS, a biomolecule presented on outer membrane of bacterial, could induce tumor-associated macrophages towards the M1 phenotype, which was beneficial to improve the anticancer efficiency of the proposed LM-DOX formulation. In orthotopic lung cancer mice model, LM-DOX could migrate to tumor efficiently in response to CCL2, which facilitated the specific delivery of the encapsulated DOX to tumor cells and the production of TNF-α through activating TAMs by LPS anchored on macrophages. Hence, LM-DOX could achieve the release of TNF-α release from TAMs and DOX accumulation, leading to a synergetic inhibitory effect against A549 lung cancer cells.
In addition to the examples mentioned above, Molinaro et al. described an innovative approach for the development of proteolipid vesicles, which were derived from leukosomes for inflammation targeting [87]. This novel leukosome platform was constructed via reconstituting proteins derived from the membrane of J774 macrophages into phospholipids bilayer of lipid NPs, which was based on a newly approach to com-bine traditional top–down and bottom–up methods. In brief, membrane proteins of macrophages were isolated firstly, which then incorporated into the preparation process of thin layer evaporation approach. Following the formation of phospholipids thin film, PBS containing proteins were used to hydrate for constructing leukosomes. In vitro characterizations, such as fluorescence microscopy and flow cytometry, con-firmed that critical proteins of leukocytes membrane were successfully transferred on-to the leukosomes, which enabled leukosomes to possess wonderful targeting ability to inflamed endothelia. More importantly, in an LPS-induced ear inflammation mouse model, leukosomes showed enhanced accumulation in the inflamed sites, and the targeting capacity was demonstrated, related to lymphocyte function-associated antigen 1 (LFA-1) and CD45 molecules. In addition, leukosomes could load hydrophilic, amphiphilic, and hydrophobic therapeutical compounds without affecting their structure, shape, and size. Hence, due to the fact that TME was inflammatory, this exploited biomimetic leukocyte proteolipid vesicles based on macrophage proteins and lipid NPs have great potential in anti-cancer targeted treatment.
In a similar manner, monocytes can also be utilized as drug delivery vectors. Ibarra et al. investigated human monocyte cells (THP-1) and murine monocytes employed for drug delivery to carry conjugated polymer NPs (CPNs) that could be ap-plied as an outstanding photosensitizer (PS) for photodynamic therapy (PDT) [33]. CPNs could be taken up by both human monocyte cells and murine monocytes and showed no toxicity to monocytes without irradiation. Moreover, ex vivo monocyte activation by LPS could improve the uptake efficiency, as well as the tumor penetration ability. In vitro results indicated that CPN-loaded monocytes could home into and penetrate glioblastoma (GBM), spheroids, and unloaded intracellular CPNs when monocytes differentiated into macrophages. CPN-loaded murine monocytes could cross blood brain barrier and sufficiently accumulate in tumor sites with no cargo re-lease in blood circulation due to the monocyte tropism introduced by GBM cells in orthotopic GL261 cells xenograft GBM mice model. In addition, Wang et al. also used the monocytes-based vector to deliver chemotherapeutic drug DOX for GBM treatment [34]. The obtained nano-DOX-loaded monocytes (Nano-DOX-MC) could migrate to tumor sites due to the tropism of monocyte. GBM cells stimulated lysosomal exocytosis of monocytes that led to the DOX unloading, followed by internalization of DOX by GBM cells. In an orthotopic GBMU87 MG cell xenograft mice model, a high tumor targeting delivery and cancer cells damage were both realized.
Since the aforementioned monocyte-based drug delivery platforms need ex vivo preparation, which is labor-intensive and time-consuming, an alternative in vivo internalization strategy has been proposed for monocyte-based drug delivery. Yang et al. demonstrated that chitosan polymeric micelles (COSA) could almost be internalized by monocytes in circulation after being injected to mice and then delivered to tumor sites [35]. The proposed micelles were comprised of chitosan (CO) and stearic acid (SA) that could produce an amidation reaction between amine groups on CO and carboxyl groups on SA. COSA micelles were able to be selectively taken up by monocytes, especially the Ly-6Chi subset monocytes, mainly via mannose receptor-mediated mechanism and secondly by a Dectin 1 receptor-mediated mechanism. Internalization of COSA had very low effect on both the COSA micelles and monocytes, for which the COSA could be stable, and monocytes could preserve their intrinsic tumor-homing ability. Subsequently, COSA-loaded monocytes were recruited to tumor sites and differentiated into macrophages accompanied by the release of cargoes, resulting in CO-SA accumulation within tumor sites. Due to the great potential of micelles for drug loading, this monocyte-based COSA platform showed momentous significance for cancer targeted therapy. In a similar way, Zheng et al. constructed gold-silver nano-rods (AuNRs), incorporating CpG ligands onto their surfaces via Au–S bonds, followed by the wrap of apoptotic bodies (ABs) that were generated by tumor cells under ultra-violet (UV) light irradiation [36]. AuNRs were prepared and functionalized easily through a simple process, while they could achieve significant tumor ablation directly, due to their high photo-to-heat conversion efficiency. The resultant formulations, AuNR-CpG/ABs, were phagocytized by inflammatory Ly-6C+ monocytes with high selectivity since ABs as waste materials of cells could be recognized and engulfed by circulating monocytes. After injected into EL4 tumor-bearing C57/BL6 mice, AuNR-CpG/Abs were driven by monocytes to accumulate in tumor based on inherent tendency toward rapid tumor recruitment of monocytes. CpG could elicit a significant immune response by DC maturation, proinflammatory cytokine secretion and effector T cell activation. Under irradiation, AuNR-CpG/Abs performed a synergistic therapy that consisted of a strong photothermal effect by AuNRs and enhanced immunostimulation by CpG, leading to tumor ablation and metastasis inhibition.
References
- Afzal, M.; Ameeduzzafar; Alharbi, K.S.; Alruwaili, N.K.; Al-Abassi, F.A.; Al-Malki, A.A.L.; Kazmi, I.; Kumar, V.; Kamal, M.A.; Nadeem, M.S.; et al. Nanomedicine in treatment of breast cancer—A challenge to conventional therapy. Semin. Cancer Biol. 2021, 69, 279–292.
- Tang, L.; Mei, Y.; Shen, Y.; He, S.; Xiao, Q.; Yin, Y.; Xu, Y.; Shao, J.; Wang, W.; Cai, Z. Nanoparticle-Mediated Targeted Drug Delivery to Remodel Tumor Microenvironment for Cancer Therapy. Int. J. Nanomed. 2021, 16, 5811–5829.
- Guo, P.; Huang, J.; Moses, M.A. Cancer Nanomedicines in an Evolving Oncology Landscape. Trends Pharm. Sci. 2020, 41, 730–742.
- Tang, L.; Li, J.; Zhao, Q.; Pan, T.; Zhong, H.; Wang, W. Advanced and Innovative Nano-Systems for Anticancer Targeted Drug Delivery. Pharmaceutics 2021, 13, 1151.
- Mei, Y.; Tang, L.; Xiao, Q.; Zhang, Z.; Zhang, Z.; Zang, J.; Zhou, J.; Wang, Y.; Wang, W.; Ren, M. Reconstituted high density lipoprotein (rHDL), a versatile drug delivery nanoplatform for tumor targeted therapy. J. Mater. Chem. B 2021, 9, 612–633.
- Liu, H.; Mei, Y.; Zhao, Q.; Zhang, A.; Tang, L.; Gao, H.; Wang, W. Black Phosphorus, an Emerging Versatile Nanoplatform for Cancer Immunotherapy. Pharmaceutics 2021, 13, 1344.
- Su, Y.; Liu, Y.; Xu, X.; Zhou, J.; Xu, L.; Xu, X.; Wang, D.; Li, M.; Chen, K.; Wang, W. On-Demand Versatile Prodrug Nanomicelle for Tumor-Specific Bioimaging and Photothermal-Chemo Synergistic Cancer Therapy. ACS Appl. Mater. Interfaces 2018, 10, 38700–38714.
- Ding, Y.; Wang, Y.; Zhou, J.; Gu, X.; Wang, W.; Liu, C.; Bao, X.; Wang, C.; Li, Y.; Zhang, Q. Direct cytosolic siRNA delivery by reconstituted high density lipoprotein for target-specific therapy of tumor angiogenesis. Biomaterials 2014, 35, 7214–7227.
- Celia, C.; Cristiano, M.C.; Froiio, F.; Francesco, M.D.; d’Avanzo, N.; Marzio, L.D.; Fresta, M. Nanoliposomes as Multidrug Carrier of Gemcitabine/Paclitaxel for the Effective Treatment of Metastatic Breast Cancer Disease: A Comparison with Gemzar and Taxol. Adv. Therap. 2020, 4, 2000121.
- Xiao, K.; Luo, J.; Fowler, W.L.; Li, Y.; Lee, J.S.; Xing, L.; Cheng, R.H.; Wang, L.; Lam, K.S. A self-assembling nanoparticle for paclitaxel delivery in ovarian cancer. Biomaterials 2009, 30, 6006–6016.
- Madamsetty, V.S.; Pal, K.; Keshavan, S.; Caulfield, T.R.; Dutta, S.K.; Wang, E.; Fadeel, B.; Mukhopadhyay, D. Development of multi-drug loaded PEGylated nanodiamonds to inhibit tumor growth and metastasis in genetically engineered mouse models of pancreatic cancer. Nanoscale 2019, 11, 22006–22018.
- Mukherjee, S.; Kotcherlakota, R.; Haque, S.; Bhattacharya, D.; Kumar, J.M.; Chakravarty, S.; Patra, C.R. Improved delivery of doxorubicin using rationally designed PEGylated platinum nanoparticles for the treatment of melanoma. Mater. Sci. Eng. C Mater. Biol. Appl. 2020, 108, 110375.
- Paolino, D.; Cosco, D.; Gaspari, M.; Celano, M.; Wolfram, J.; Voce, P.; Puxeddu, E.; Filetti, S.; Celia, C.; Ferrari, M.; et al. Targeting the thyroid gland with thyroid-stimulating hormone (TSH)-nanoliposomes. Biomaterials 2014, 35, 7101–7109.
- Saalik, P.; Lingasamy, P.; Toome, K.; Mastandrea, I.; Rousso-Noori, L.; Tobi, A.; Simon-Gracia, L.; Hunt, H.; Paiste, P.; Kotamraju, V.R.; et al. Peptide-guided nanoparticles for glioblastoma targeting. J. Control. Release 2019, 308, 109–118.
- Imperlini, E.; Celia, C.; Cevenini, A.; Mandola, A.; Raia, M.; Fresta, M.; Orru, S.; Di Marzio, L.; Salvatore, F. Nano-bio interface between human plasma and niosomes with different formulations indicates protein corona patterns for nanoparticle cell targeting and uptake. Nanoscale 2021, 13, 5251–5269.
- Zhang, T.; Li, G.; Miao, Y.; Lu, J.; Gong, N.; Zhang, Y.; Sun, Y.; He, Y.; Peng, M.; Liu, X.; et al. Magnetothermal regulation of in vivo protein corona formation on magnetic nanoparticles for improved cancer nanotherapy. Biomaterials 2021, 276, 121021.
- Giulimondi, F.; Digiacomo, L.; Pozzi, D.; Palchetti, S.; Vulpis, E.; Capriotti, A.L.; Chiozzi, R.Z.; Lagana, A.; Amenitsch, H.; Masuelli, L.; et al. Interplay of protein corona and immune cells controls blood residency of liposomes. Nat. Commun. 2019, 10, 3686.
- Tang, Y.; Wang, X.; Li, J.; Nie, Y.; Liao, G.; Yu, Y.; Li, C. Overcoming the Reticuloendothelial System Barrier to Drug Delivery with a “Don’t-Eat-Us” Strategy. ACS Nano 2019, 13, 13015–13026.
- Ayer, M.; Klok, H.A. Cell-mediated delivery of synthetic nano- and microparticles. J. Control. Release 2017, 259, 92–104.
- Cheng, Z.; Al Zaki, A.; Hui, J.Z.; Muzykantov, V.R.; Tsourkas, A. Multifunctional nanoparticles: Cost versus benefit of adding targeting and imaging capabilities. Science 2012, 338, 903–910.
- Yu, H.; Yang, Z.; Li, F.; Xu, L.; Sun, Y. Cell-mediated targeting drugs delivery systems. Drug Deliv 2020, 27, 1425–1437.
- d’Avanzo, N.; Torrieri, G.; Figueiredo, P.; Celia, C.; Paolino, D.; Correia, A.; Moslova, K.; Teesalu, T.; Fresta, M.; Santos, H.A. LinTT1 peptide-functionalized liposomes for targeted breast cancer therapy. Int. J. Pharm. 2021, 597, 120346.
- Pang, L.; Zhang, C.; Qin, J.; Han, L.; Li, R.; Hong, C.; He, H.; Wang, J. A novel strategy to achieve effective drug delivery: Exploit cells as carrier combined with nanoparticles. Drug Deliv. 2017, 24, 83–91.
- Wang, J.; Gu, X.; Ouyang, Y.; Chu, L.; Xu, M.; Wang, K.; Tong, X. Engineering of Neutrophil Membrane Camouflaging Nanoparticles Realizes Targeted Drug Delivery for Amplified Antitumor Therapy. Int. J. Nanomed. 2021, 16, 1175–1187.
- Li, M.; Li, S.; Zhou, H.; Tang, X.; Wu, Y.; Jiang, W.; Tian, Z.; Zhou, X.; Yang, X.; Wang, Y. Chemotaxis-driven delivery of nano-pathogenoids for complete eradication of tumors post-phototherapy. Nat. Commun. 2020, 11, 1126.
- Kennedy, L.C.; Bear, A.S.; Young, J.K.; Lewinski, N.A.; Kim, J.; Foster, A.E.; Drezek, R.A. T cells enhance gold nanoparticle delivery to tumors in vivo. Nanoscale Res. Lett. 2011, 6, 283.
- Huang, B.N.; Abraham, W.D.; Zheng, Y.R.; Bustamante Lopez, S.C.; Luo, S.S.; Irvine, D.J. Active targeting of chemotherapy to disseminated tumors using nanoparticle-carrying T cells. Sci. Transl. Med. 2015, 7, 291ra94.
- Ma, W.; Zhu, D.; Li, J.; Chen, X.; Xie, W.; Jiang, X.; Wu, L.; Wang, G.; Xiao, Y.; Liu, Z.; et al. Coating biomimetic nanoparticles with chimeric antigen receptor T cell-membrane provides high specificity for hepatocellular carcinoma photothermal therapy treatment. Theranostics 2020, 10, 1281–1295.
- Siegler, E.L.; Kim, Y.J.; Chen, X.; Siriwon, N.; Mac, J.; Rohrs, J.A.; Bryson, P.D.; Wang, P. Combination Cancer Therapy Using Chimeric Antigen Receptor-Engineered Natural Killer Cells as Drug Carriers. Mol. Ther. 2017, 25, 2607–2619.
- Pitchaimani, A.; Nguyen, T.D.T.; Aryal, S. Natural killer cell membrane infused biomimetic liposomes for targeted tumor therapy. Biomaterials 2018, 160, 124–137.
- Qiu, Y.; Ren, K.; Zhao, W.; Yu, Q.; Guo, R.; He, J.; Mei, L.; Liu, Y.; Tang, J.; Xu, S.; et al. A “dual-guide” bioinspired drug delivery strategy of a macrophage-based carrier against postoperative triple-negative breast cancer recurrence. J. Control. Release 2021, 329, 191–204.
- Guo, L.; Zhang, Y.; Wei, R.; Wang, C.; Feng, M. Lipopolysaccharide-anchored macrophages hijack tumor microtube networks for selective drug transport and augmentation of antitumor effects in orthotopic lung cancer. Theranostics 2019, 9, 6936–6948.
- Ibarra, L.E.; Beauge, L.; Arias-Ramos, N.; Rivarola, V.A.; Chesta, C.A.; Lopez-Larrubia, P.; Palacios, R.E. Trojan horse monocyte-mediated delivery of conjugated polymer nanoparticles for improved photodynamic therapy of glioblastoma. Nanomedicine 2020, 15, 1687–1707.
- Wang, C.; Li, K.; Li, T.F.; Chen, Z.; Wen, Y.; Liu, X.; Jia, X.M.; Zhang, Y.C.; Xu, Y.H.; Han, M.; et al. Monocyte-mediated chemotherapy drug delivery in glioblastoma. Nanomedicine 2018, 13, 157–178.
- Yang, X.Q.; Lian, K.K.; Tan, Y.A.; Zhu, Y.; Liu, X.; Zeng, Y.P.; Yu, T.; Meng, T.T.; Yuan, H.; Hu, F.Q. Selective uptake of chitosan polymeric micelles by circulating monocytes for enhanced tumor targeting. Carbohyd. Polym. 2020, 229, 115435.
- Zheng, L.Y.; Hu, X.X.; Wu, H.; Mo, L.T.; Xie, S.T.; Li, J.; Peng, C.; Xu, S.J.; Qiu, L.P.; Tan, W.H. In Vivo Monocyte/Macrophage-Hitchhiked Intratumoral Accumulation of Nanomedicines for Enhanced Tumor Therapy. J. Am. Chem. Soc. 2020, 142, 382–391.
- Zhao, Z.; Ukidve, A.; Gao, Y.; Kim, J.; Mitragotri, S. Erythrocyte leveraged chemotherapy (ELeCt): Nanoparticle assembly on erythrocyte surface to combat lung metastasis. Sci. Adv. 2019, 5, eaax9250.
- Grifantini, R.; Taranta, M.; Gherardini, L.; Naldi, I.; Parri, M.; Grandi, A.; Giannetti, A.; Tombelli, S.; Lucarini, G.; Ricotti, L.; et al. Magnetically driven drug delivery systems improving targeted immunotherapy for colon-rectal cancer. J. Control. Release 2018, 280, 76–86.
- Shi, W.; Qiu, Q.; Feng, Z.; Tong, Z.; Guo, W.; Zou, F.; Yue, N.; Huang, W.; Qian, H. Design, synthesis and immunological evaluation of self-assembled antigenic peptides from dual-antigen targets: A broad-spectrum candidate for an effective antibreast cancer therapy. J. Immunother. Cancer 2021, 9, e002523.
- Gao, M.; Liang, C.; Song, X.; Chen, Q.; Jin, Q.; Wang, C.; Liu, Z. Erythrocyte-Membrane-Enveloped Perfluorocarbon as Nanoscale Artificial Red Blood Cells to Relieve Tumor Hypoxia and Enhance Cancer Radiotherapy. Adv. Mater. 2017, 29.
- Jiang, Q.; Liu, Y.; Guo, R.; Yao, X.; Sung, S.; Pang, Z.; Yang, W. Erythrocyte-cancer hybrid membrane-camouflaged melanin nanoparticles for enhancing photothermal therapy efficacy in tumors. Biomaterials 2019, 192, 292–308.
- Xu, P.; Zuo, H.; Zhou, R.; Wang, F.; Liu, X.; Ouyang, J.; Chen, B. Doxorubicin-loaded platelets conjugated with anti-CD22 mAbs: A novel targeted delivery system for lymphoma treatment with cardiopulmonary avoidance. Oncotarget 2017, 8, 58322–58337.
- Han, X.; Chen, J.; Chu, J.; Liang, C.; Ma, Q.; Fan, Q.; Liu, Z.; Wang, C. Platelets as platforms for inhibition of tumor recurrence post-physical therapy by delivery of anti-PD-L1 checkpoint antibody. J. Control. Release 2019, 304, 233–241.
- Wang, X.; Liang, G.F.; Hao, X.Q.; Feng, S.Y.; Dai, L.; An, J.L.; Li, J.H.; Shi, H.; Feng, W.P.; Zhang, X. Bioinspired Drug Delivery Carrier for Enhanced Tumor-Targeting in Melanoma Mice Model. J. Biomed. Nanotechnol. 2019, 15, 1482–1491.
- Liu, G.; Zhao, X.; Zhang, Y.; Xu, J.; Xu, J.; Li, Y.; Min, H.; Shi, J.; Zhao, Y.; Wei, J.; et al. Engineering Biomimetic Platesomes for pH-Responsive Drug Delivery and Enhanced Antitumor Activity. Adv. Mater. 2019, 31, e1900795.
- Li, L.; Fu, J.; Wang, X.; Chen, Q.; Zhang, W.; Cao, Y.; Ran, H. Biomimetic “Nanoplatelets” as a Targeted Drug Delivery Platform for Breast Cancer Theranostics. ACS Appl. Mater. Interfaces 2021, 13, 3605–3621.
- Jiang, Q.; Wang, K.; Zhang, X.; Ouyang, B.; Liu, H.; Pang, Z.; Yang, W. Platelet Membrane-Camouflaged Magnetic Nanoparticles for Ferroptosis-Enhanced Cancer Immunotherapy. Small 2020, 16, e2001704.
- Shang, Y.; Wang, Q.; Wu, B.; Zhao, Q.; Li, J.; Huang, X.; Chen, W.; Gui, R. Platelet-Membrane-Camouflaged Black Phosphorus Quantum Dots Enhance Anticancer Effect Mediated by Apoptosis and Autophagy. ACS Appl. Mater. Interfaces 2019, 11, 28254–28266.
- Kim, S.W.; Lee, Y.K.; Hong, J.H.; Park, J.Y.; Choi, Y.A.; Lee, D.U.; Choi, J.; Sym, S.J.; Kim, S.H.; Khang, D. Mutual Destruction of Deep Lung Tumor Tissues by Nanodrug-Conjugated Stealth Mesenchymal Stem Cells. Adv. Sci. 2018, 5, 1700860.
- Takayama, Y.; Kusamori, K.; Tsukimori, C.; Shimizu, Y.; Hayashi, M.; Kiyama, I.; Katsumi, H.; Sakane, T.; Yamamoto, A.; Nishikawa, M. Anticancer drug-loaded mesenchymal stem cells for targeted cancer therapy. J. Control. Release 2021, 329, 1090–1101.
- Paris, J.L.; de la Torre, P.; Cabañas, M.V.; Manzano, M.; Flores, A.I.; Vallet-Regí, M. Suicide-gene transfection of tumor-tropic placental stem cells employing ultrasound-responsive nanoparticles. Acta Biomater. 2019, 83, 372–378.
- Chen, K.; Cao, X.; Li, M.; Su, Y.; Li, H.; Xie, M.; Zhang, Z.; Gao, H.; Xu, X.; Han, Y.; et al. A TRAIL-Delivered Lipoprotein-Bioinspired Nanovector Engineering Stem Cell-Based Platform for Inhibition of Lung Metastasis of Melanoma. Theranostics 2019, 9, 2984–2998.
- Wen, D.; Wang, J.; Van Den Driessche, G.; Chen, Q.; Zhang, Y.; Chen, G.; Li, H.; Soto, J.; Liu, M.; Ohashi, M.; et al. Adipocytes as Anticancer Drug Delivery Depot. Matter 2019, 1, 1203–1214.
- Huang, W.C.; Lu, I.L.; Chiang, W.H.; Lin, Y.W.; Tsai, Y.C.; Chen, H.H.; Chang, C.W.; Chiang, C.S.; Chiu, H.C. Tumortropic adipose-derived stem cells carrying smart nanotherapeutics for targeted delivery and dual-modality therapy of orthotopic glioblastoma. J. Control. Release 2017, 254, 119–130.
- Aoki, M.; Kakimoto, K.; Goto, M.; Higuchi, K. Novel Therapeutic Approach Using Drug-loaded Adipose-derived Stem Cells for Pancreatic Cancer. Sci. Rep. 2019, 9, 17971.
- Liang, T.; Wen, D.; Chen, G.; Chan, A.; Chen, Z.; Li, H.; Wang, Z.; Han, X.; Jiang, L.; Zhu, J.J.; et al. Adipocyte-Derived Anticancer Lipid Droplets. Adv. Mater. 2021, 33, e2100629.
- Kameritsch, P.; Renkawitz, J. Principles of Leukocyte Migration Strategies. Trends Cell Biol. 2020, 30, 818–832.
- Umehara, T.; Oka, H.; Nakahara, A.; Matsuno, H.; Murakami, H. Differential leukocyte count is associated with clinical phenotype in Parkinson’s disease. J. Neurol. Sci. 2020, 409, 116638.
- Geering, B.; Stoeckle, C.; Conus, S.; Simon, H.U. Living and dying for inflammation: Neutrophils, eosinophils, basophils. Trends Immunol. 2013, 34, 398–409.
- Montano-Rendon, F.; Grinstein, S.; Walpole, G.F.W. Monitoring Phosphoinositide Fluxes and Effectors During Leukocyte Chemotaxis and Phagocytosis. Front. Cell Dev. Biol. 2021, 9, 626136.
- Do, H.T.T.; Lee, C.H.; Cho, J. Chemokines and their Receptors: Multifaceted Roles in Cancer Progression and Potential Value as Cancer Prognostic Markers. Cancers 2020, 12, 287.
- Zhang, J.; Shi, Z.; Xu, X.; Yu, Z.; Mi, J. The influence of microenvironment on tumor immunotherapy. FEBS J. 2019, 286, 4160–4175.
- Mitchell, M.J.; King, M.R. Leukocytes as carriers for targeted cancer drug delivery. Expert. Opin. Drug Deliv. 2015, 12, 375–392.
- Ganesh, K.; Massague, J. Targeting metastatic cancer. Nat. Med. 2021, 27, 34–44.
- Ng, L.G.; Ostuni, R.; Hidalgo, A. Heterogeneity of neutrophils. Nat. Rev. Immunol. 2019, 19, 255–265.
- Mukaida, N.; Sasaki, S.I.; Baba, T. Two-Faced Roles of Tumor-Associated Neutrophils in Cancer Development and Progression. Int. J. Mol. Sci. 2020, 21, 3457.
- Ley, K.; Hoffman, H.M.; Kubes, P.; Cassatella, M.A.; Zychlinsky, A.; Hedrick, C.C.; Catz, S.D. Neutrophils: New insights and open questions. Sci. Immunol. 2018, 3, eaat4579.
- Masucci, M.T.; Minopoli, M.; Del Vecchio, S.; Carriero, M.V. The Emerging Role of Neutrophil Extracellular Traps (NETs) in Tumor Progression and Metastasis. Front. Immunol. 2020, 11, 1749.
- Monge, M.; Fornaguera, C.; Quero, C.; Dols-Perez, A.; Caldero, G.; Grijalvo, S.; Garcia-Celma, M.J.; Rodriguez-Abreu, C.; Solans, C. Functionalized PLGA nanoparticles prepared by nano-emulsion templating interact selectively with proteins involved in the transport through the blood-brain barrier. Eur. J. Pharm. Biopharm. 2020, 156, 155–164.
- Allen, C.; Evans, J.C. ‘Hip to be square’: Designing PLGA formulations for the future. J. Control. Release 2020, 319, 487–488.
- Huang, Y.; Gao, X.; Chen, J. Leukocyte-derived biomimetic nanoparticulate drug delivery systems for cancer therapy. Acta Pharm. Sin. B 2018, 8, 4–13.
- Sarivalasis, A.; Morotti, M.; Mulvey, A.; Imbimbo, M.; Coukos, G. Cell therapies in ovarian cancer. Adv. Med. Oncol. 2021, 13, 17588359211008399.
- Rao, S.; Gharib, K.; Han, A. Cancer Immunosurveillance by T Cells. Int. Rev. Cell Mol. Biol. 2019, 342, 149–173.
- Speiser, D.E.; Ho, P.C.; Verdeil, G. Regulatory circuits of T cell function in cancer. Nat. Rev. Immunol. 2016, 16, 599–611.
- Farhood, B.; Najafi, M.; Mortezaee, K. CD8(+) cytotoxic T lymphocytes in cancer immunotherapy: A review. J. Cell. Physiol. 2019, 234, 8509–8521.
- Marofi, F.; Abdul-Rasheed, O.F.; Rahman, H.S.; Budi, H.S.; Jalil, A.T.; Yumashev, A.V.; Hassanzadeh, A.; Yazdanifar, M.; Motavalli, R.; Chartrand, M.S.; et al. CAR-NK cell in cancer immunotherapy; A promising frontier. Cancer Sci. 2021, 112, 3427–3436.
- Elahi, R.; Heidary, A.H.; Hadiloo, K.; Esmaeilzadeh, A. Chimeric Antigen Receptor-Engineered Natural Killer (CAR NK) Cells in Cancer Treatment; Recent Advances and Future Prospects. Stem. Cell Rev. Rep. 2021, 1–26.
- Zhang, C.; Hu, Y.; Xiao, W.; Tian, Z. Chimeric antigen receptor- and natural killer cell receptor-engineered innate killer cells in cancer immunotherapy. Cell. Mol. Immunol. 2021, 18, 2083–2100.
- Steinfeld, U.; Pauli, C.; Kaltz, N.; Bergemann, C.; Lee, H.H. T lymphocytes as potential therapeutic drug carrier for cancer treatment. Int. J. Pharm. 2006, 311, 229–236.
- Wayteck, L.; Dewitte, H.; De Backer, L.; Breckpot, K.; Demeester, J.; De Smedt, S.C.; Raemdonck, K. Hitchhiking nanoparticles: Reversible coupling of lipid-based nanoparticles to cytotoxic T lymphocytes. Biomaterials 2016, 77, 243–254.
- Gardiner, C.M. NK cell metabolism. J. Leukoc. Biol. 2019, 105, 1235–1242.
- Enomoto, Y.; Li, P.; Jenkins, L.M.; Anastasakis, D.; Lyons, G.C.; Hafner, M.; Leonard, W.J. Cytokine-enhanced cytolytic activity of exosomes from NK Cells. Cancer Gene Ther. 2021.
- Anderson, N.R.; Minutolo, N.G.; Gill, S.; Klichinsky, M. Macrophage-Based Approaches for Cancer Immunotherapy. Cancer Res. 2021, 81, 1201–1208.
- Tariq, M.; Zhang, J.; Liang, G.; Ding, L.; He, Q.; Yang, B. Macrophage Polarization: Anti-Cancer Strategies to Target Tumor-Associated Macrophage in Breast Cancer. J. Cell. Biochem. 2017, 118, 2484–2501.
- Engblom, C.; Pfirschke, C.; Pittet, M.J. The role of myeloid cells in cancer therapies. Nat. Rev. Cancer 2016, 16, 447–462.
- Nakatsumi, H.; Matsumoto, M.; Nakayama, K.I. Noncanonical Pathway for Regulation of CCL2 Expression by an mTORC1-FOXK1 Axis Promotes Recruitment of Tumor-Associated Macrophages. Cell Rep. 2017, 21, 2471–2486.
- Molinaro, R.; Corbo, C.; Martinez, J.O.; Taraballi, F.; Evangelopoulos, M.; Minardi, S.; Yazdi, I.K.; Zhao, P.; De Rosa, E.; Sherman, M.B.; et al. Biomimetic proteolipid vesicles for targeting inflamed tissues. Nat. Mater. 2016, 15, 1037–1046.
More
Information
Subjects:
Materials Science, Biomaterials
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.4K
Revisions:
3 times
(View History)
Update Date:
29 Mar 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





