Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Diego A. Moreno-Fernandez | + 3055 word(s) | 3055 | 2021-11-11 04:32:09 | | | |
| 2 | Peter Tang | Meta information modification | 3055 | 2021-11-12 02:46:58 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Moreno-Fernandez, D. Brassica Bioactives on Human Health. Encyclopedia. Available online: https://encyclopedia.pub/entry/15900 (accessed on 12 May 2026).
Moreno-Fernandez D. Brassica Bioactives on Human Health. Encyclopedia. Available at: https://encyclopedia.pub/entry/15900. Accessed May 12, 2026.
Moreno-Fernandez, Diego. "Brassica Bioactives on Human Health" Encyclopedia, https://encyclopedia.pub/entry/15900 (accessed May 12, 2026).
Moreno-Fernandez, D. (2021, November 11). Brassica Bioactives on Human Health. In Encyclopedia. https://encyclopedia.pub/entry/15900
Moreno-Fernandez, Diego. "Brassica Bioactives on Human Health." Encyclopedia. Web. 11 November, 2021.
Copy Citation
Brassica vegetables and their components, the glucosinolates, have been suggested as good candidates as dietary coadjutants to improve health in non-communicable diseases (NCDs).
Brassica
glucosinolates
clinical studies
human health
1. Introduction
Nowadays, the incidence of non-communicable diseases (NCDs), including obesity, diabetes, cancer, and other chronic conditions, is increasing and showing high mortality indexes worldwide (https://www.who.int/nmh/topics/es/). These diseases are multifactorial, but it has been described that lifestyle, such as smoking or sedentary life, contributes to the prevalence of the NCDs. As a result, different prevention strategies have been developed, mainly the promotion of healthy habits, recommending avoiding a sedentary life, quitting smoking and the consumption of alcoholic drinks, and keeping a healthy diet [1]. To emphasize the importance of these strategies, it has been proven that these habits have an important role in the prevention of cardiovascular diseases and type-II diabetes [2].
Since one of the main points of intervention for a healthy lifestyle is diet, global health recommendations are always encouraging the message of eating a minimum of five portions of fruits and vegetables daily [3][4]. Vegetables are not only a natural source of amino acids and minerals, but also of phytochemicals [5]. In particular, cruciferous vegetables, like broccoli, cabbage, red radish, or Brussels sprouts, are consumed worldwide not only because of their culinary value being popular in many countries with plates being part of the culture legacy, but also because of their diverse content in phytochemicals with high health-promoting benefits [6][7]. Between them vitamin C, polyphenols, and minerals can be found. However, one of the most relevant biomolecules are glucosinolates (GSLs), mainly found in plant species and varieties of the order Brassicales, mainly Brassicaceae as the most well-known representatives of this horticultural, botanical, and socioeconomically relevant botanical family [8]. They consist of a basic structure of a thiohydroximate-O-sulfate group with a glycosylation and, depending on which amino acid they are derived from, a different side chain [8]. GSLs are stable secondary metabolites, but after tissue disruption, they are hydrolyzed by the enzyme myrosinase (EC 3.2.1.147), generating different GSL hydrolysis products (Figure 1) including bioactive isothiocyanates (ITCs) if the pH is 5–8, in the range of neutrality. In some plants, epithiospecifier proteins exist and can modify the outcome of the hydrolysis process, typically to promote other products than ITCs, as it is the case of nitrile specifier protein originating simple nitriles and elemental sulfur [8][9][10]. On the other hand, the epithiospecifier proteins (ESP) with similar function may also carry out a different reaction for the few aliphatic GSLs with a terminal double bond by adding the sulfur to this bond, forming an epitionitrile. The production of thiocyanates is produced at pH > 8, and oxazolidine-2-thiones can be formed if a hydroxyl group is present on carbon 2 (beta position) of the glucosinolate [10][11].
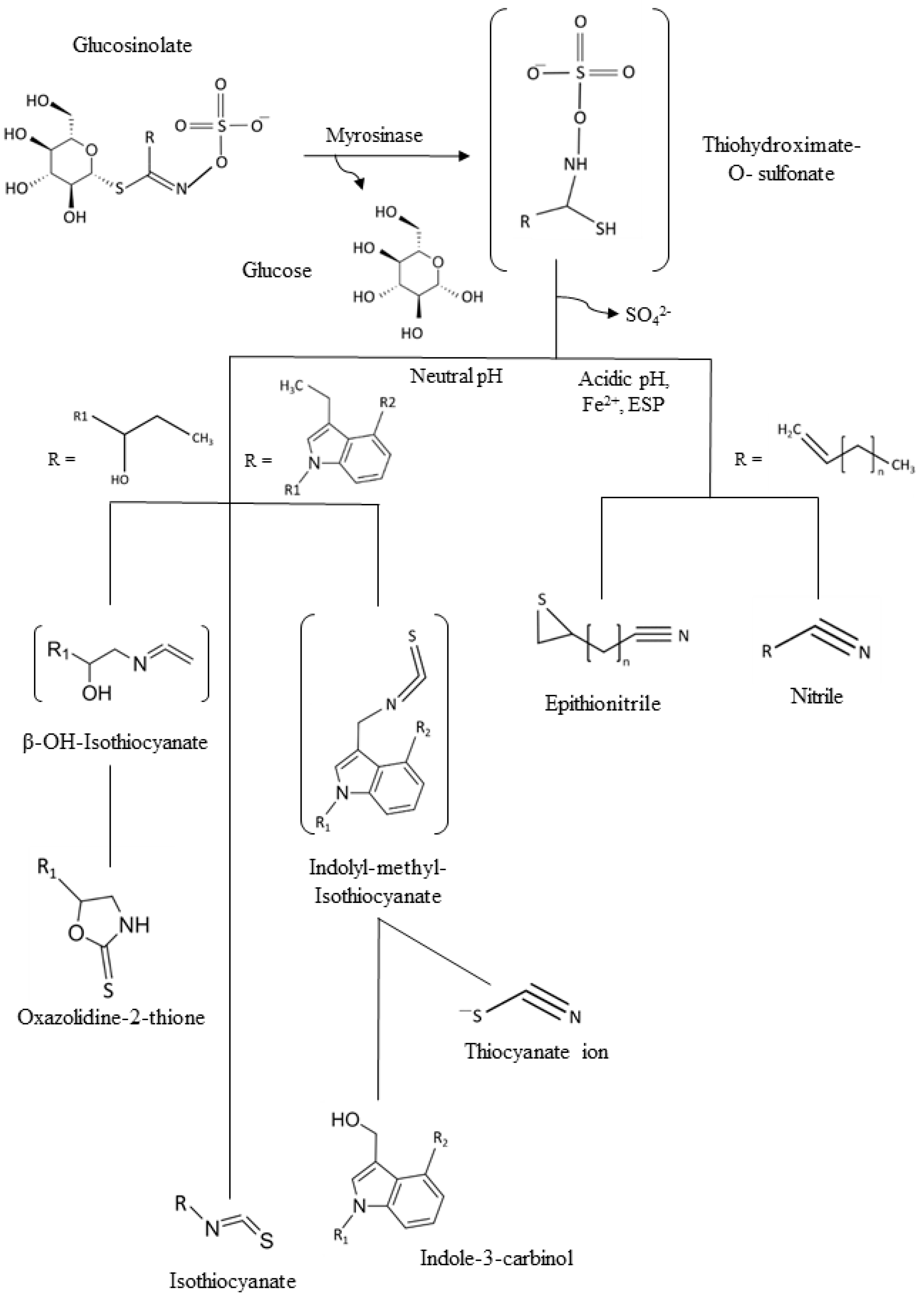
Figure 1. General scheme of the glucosinolates (GSLs) and common hydrolysis products. ESP: Epithiospecifer proteins.
All these studies performed in vitro suggest that cruciferous vegetables mainly from Brassica spp. (either as foods or ingredients rich in bioactive substances) are good candidates as dietary coadjutants to improve human health in NCDs. As a consequence, some clinical and pre-clinical studies have been developed during the last six years. Epidemiological studies have linked the intake of GSLs with the risk of coronary heart disease or type 2 diabetes [12][13]. Nevertheless, this information from large cohorts is usually based in food frequency questionnaires, making it difficult to know the specific effects of the GSLs in the intake.
2. The Clinical Evidences of Health-Promotion with Brassica Bioactives from a Critical Point of View
2.1. Short-Time Studies
Within the short-term studies, we found two documents on prostate [14] and melanoma [15], three papers on respiratory pathologies [16][17][18], and one on type 2 diabetes [19]. The main limitation observed in these studies is the length or duration of the study, of 1 month or less, because the validity of the data in terms of changes in bioavailability of the intake of compounds could be acceptable, but for the evaluation of data from tissues, the time of exposition is too low. In the article of Gee et al. [14], a Phase Ib study, studied the concentration of DIM in prostate tissue after its administration in three groups: 200 mg, 400 mg, or placebo. They found a positive increase of DIM in plasma, but not concomitant or related with the presence of DIM in the prostate tissues, and the initial hypothesis was not corroborated. On the other hand, the urinary 2/16-hydroxysterone increased significantly by 68% (p = 0.030) in the group administrated with 400 mg of DIM. The levels of PSA taken separately, were also non-significant, but comparing between the group of 400 mg versus placebo, the reduction was of 1.8 times (p = 0.1). The gene expression of the CYP1A1 in blood after 15 days in the Group of 200 mg was significantly reduced by 31% (p = 0.032) and more importantly for the reduction for the CYP2B6 was highly significant by 87% (p = 0.040).
In Tahata et al. [15], patients with more than two nevus or cell-proliferation structures of ≥4 mm Ø were selected and showed significant increases (ratio of concentration >1.5) in 14 out of the 92 proteins identified as tumor suppressors during the time of the study. On the other hand, it was not possible to relate the positive data on the significant reduction of proinflammatory cells such as IP-10, MCP-1, MIG, and MIP-1b to the supplementation of broccoli sprout extract (BSE) enriched in sulforaphane (SFN) because of different problems, such as reduced size in terms of number of participants, samples that were too small, and of a different nevus, which made the analysis difficult and induced higher inter-lesion variability. From the size of the nevus, it was not possible to obtain significant differences, only a general increase in size that was less patent in the patients treated with 100 µg BSE–SFN. Against these results it is worth highlighting that the changes in the size of the nevus in a period of one month are unusual and the comparisons between pictures depends in the illumination characteristic of the images, being unreliable data when analyzing the results. One of the strengths of this work actually are the improvements suggested for the future to use a different technique for the biopsies (in patch), to reduce the stress in the area of lesion, and to use a dermatoscope to substitute the pictures because of their great variability according to the illumination conditions.
2.2. Medium-Length Studies
The analysis of the articles of medium-length duration between 2 and 8 months indicated that three were dedicated to studies on cancer in different localizations, two studies were on prostate cancer, and one study was on breast cancer.
In both studies of Alumkal et al. [20] and Cipolla et al. [21], the main factor studied was the change in the PSA (prostate specific antigen) parameter. In their study, Alumkal et al. ambitiously established the objective of a reduction of 50% in the level of PSA after 20 weeks of intervention, an objective that was not achieved. The obtained reductions were significant and in the range of 3% to 20%. In both studies it was also positive to find an increased time for doubling the amount of PSA after the intervention was found (pre- and post-intervention in placebo from 6.1 to 16.6 months [20]; 9.6 to 31.9 months, in the treated group).
Cipolla et al. [21] were focused in the changes in PSA over time, which were not significant when adjusting the data to the months (m0, m1, m3, and m6); but, when eliminating the m1, the reduction was of 0.0180 ng/mL/month. Besides, the median of log PSA was 38.5% lower in the intervention group than in the placebo, and the progression of the PSA was by 71.8% in the placebo versus the 44.4% in the SFN-treated group. Looking into the changes of the phase II enzymes, the GSTM1, and the inhibition of the HDAC in the work of Alumkal et al. [20], non-significant differences were found. In both studies, the time for the intervention was similar, namely 20 and 24 weeks, respectively, but the amount of SFN was much higher in the work of Cipolla et al. [21], and that probably favored the better more encouraging final results in this study. In any case, Atwell et al. [22] planned to evaluate the efficacy of the consumption of the broccoli sprouts extract to modify the activity of the HDAC (histone deacetylase) and the breast tumor marker CA (carcinogenic antigen) in order to improve the diagnosis in women with benign disease and non-invasive CA. They proposed an intervention using six pills with a theoretical content of SFN of 30 mg in three administrations per day. A point in favor of this article is that they carried out an internal analysis of the composition of the capsules, and determined the total daily dosage of 224 mg of SFN instead of the declared 180 mg of SFN as designated by the supplier of the supplement. When evaluating the results, the levels of the HDAC before and after administration between groups were not statistically different. Only when comparing the two groups, treated and untreated, after the intervention was a significant difference observed, with a reduction in the HDAC in the group with the treatment. It should be noted that the authors also carried out a stratified data analysis considering the consumption of NSAIDs, using this variable as independent, and they found a significant reduction in the HDAC in the patients that were non-regular consumers of NSAIDs (p = 0.04), and it was not possible to find significant differences in the group of consumers of NSAIDs. Additionally, the biomarkers H3K18ac, H3K9ac, HDAC3, HDAC6, Ki-67, and P21 did not present statistically significant differences in the pre- or post-analysis. When comparing the levels of pre- and post-treatment within each group, a significant reduction was found in Ki-67 and HDAC3 in benign tissues in the SFN treated group, and a significant reduction of H3K9ac in the tissular DCIS in the placebo group. As a conclusion of this work, the authors did not plan as a hypothesis that the NSAIDs inhibited the synthesis of prostaglandins, and this could suppress the expression of regulator proteins through the involvement of the recruitment of the HDAC. The increased recruitment of the HDAC in the chromatin could prevent the inhibition associated with the consumption of SFN, and that could be a potential explanation for the observed data, and for the future studies using interventions with SFN, longer studies will be required to confirm these findings.
2.3. Long-Term Studies
Finally, we proceeded with the analysis of the articles studying long-term interventions (Table S3). In these works, we found various publications on different types of cancers: prostate [23][24], breast [25], ovarian [26], and pancreas (pilot [27] and development [28]), and one work on type 2 diabetes mellitus [12] and a letter to the editor [29] as a result of this study.
The works of Paltsev et al. [23] and Traka et al. [24] were both dedicated to prostate cancer, for a period of 12 months, but with different interventions and different parameters of the stage of development. In the first one [23], the intervention was carried out using capsules with 900 mg of DIM (3,3-diindolylmethane) and considered data of morphological changes (MI formula to calculate presence of cancer) in the prostate, and registration of the prostatic intraepithelial neoplasias (PIN) of low and high degree as well as the number of foci of cancer and urological dynamic parameters (maximum, average, and residual urinary flow) and erectile function. In the second work [24], with a higher dosage of glucoraphanin (parental glucosinolate of SFN) through the intake of different broccoli soups (based on broccolis with different contents of glucoraphanin), specific parameters included the change in the genetic expression of the RNA, changes in metabolites after biopsy from prostate tissue, and the plasma PSA levels. In the results of Paltsev et al. [23], a significant reduction in the cancerous and precancerous cells was obtained in the treated group (from MI = 50 before treatment to MI = 0.08 after treatment), obtaining a complete regression in 45.5% of the patients (5 out of 11). Despite the results, the extremely reduced sample size in this study made it impossible to establish a significant difference between groups. On the other hand, Traka et al. [24] observed changes in the genetic expression in all the patients, considering as reference the group that took the soup with the unmodified broccoli (not enriched). The changes were more striking in the patients that took the soups with enriched broccoli (with much higher glucoraphanin than regular broccoli, through the presence of one or two alleles of the Myb28 gene), but the study did not reach a significant result, because the number of patients or participants was below the minimum necessary of 78 participants, in order to detect significant changes of p < 0.02. The authors concluded that a diet rich in glucoraphanin may attenuate the transcriptional changes that take place in the prostate, but the data were not conclusive because of that limitation. It became clear, as in other studies with natural products and interventions for different types of diseases, that the size and number of participants should be enough to generate sound results that would help in the elaboration of recommendations for dietary options with food rich in bioactives such as Brassica vegetables, microgreens, and sprouts.
The works of Thomson et al. [25] on breast cancer and Kiselev et al. [26] on ovarian cancer were studies with the highest number of participants and the longest periods of evaluation. Thomson et al. [25] studied the effects of diindolylmethane (DIM) + tamoxifen administration for 12 months. Previously, a tolerance test was carried out with the 10 first participants to establish a 150 mg dosage of DIM. From the aims of the study, the main objective was to analyze the breast density, and it had to be discarded because of the high incidence of participants with bilateral mastectomy, and therefore, they analyzed the effects on 2-OHE/16-α-OHE (alpha hydroxylated estrogen), the levels of strone, stradiol, and SHBG (sex hormone binding globulin). The statistical power of this study was established under 88% because it needed at least 55 participants per group, and in this case, the study was finished by 51 patients in the placebo group and 47 in the intervention group. The results support the hypothesis that the DIM (from Brassica) induced an increase in the 2-OHE/16-α-OHE in urine (p = 0.001) and increased the circulating SHGB in the patients with the combined treatment with tamoxifen. Besides that, the pharmacokinetic of the tamoxifen was changed, reducing the circulating forms of this drug. The results are positive but cannot be used for generalization.
On the other hand, the work of Kiselev et al. [26] provided data on the survival and the survival without progression of ovarian cancer at 5 years in the patients with the combined treatment of EGCG (epigallocatechin gallate of green tea) and indole-3-carbinol (I3C)—the precursor of DIM. The combined I3C + EGCG demonstrated a 1.5 times increase in the survival free of cancer, a significant reduction in recurrences of the ovarian cancer with ascites in the treated group (by 8%–9%) when compared with the untreated group (by 60%–63%). The first results were observed in the pre-surgery stage because the majority of the patients (81%–85%) in the therapeutic branches of maintenance 1–3 could take a successful reduction surgery to eliminate all the visible tumor focus. On the contrary, for the participants in branches 4–5, it was not possible to carry out such procedure. Additionally, the levels of CA-125 were statistically lower in the 1–3 pre-surgery groups and after treatment than in the control group. Based on these data, the authors concluded that these were preliminary results, but they hypothesized that the treatment of I3C + EGCG, administered as part of the maintenance therapy during and after the combined treatment, inhibited the cancer precursor cells at the ovarian level, and therefore, diminished significantly the incidence of recurrence (ascites), and this was also redundant for higher rate of survival, global survival, and free-of-cancer survival.
From the article of Lozanovski et al. [27], a pilot study, we found that one of their inclusion criteria made it difficult to extrapolate the results to all the participants, because it only considered patients without gastrointestinal symptoms, which are common symptoms of this pathology and in the chemotherapy treatments. With respect to the objective of the study, they aimed to analyze the effects of the intake of encapsulated broccoli sprouts as co-adjuvants in the chemotherapy and proposed the intake of 15 capsules of broccoli sprouts at a time, giving 90 mg of SFN and 180 mg of glucoraphanin, a never before tested dosage, which was established because of the poor life expectancy of the patients and the aggressive characteristics of this type of cancer. The results of Lozanovski et al. [28], later on, established that the secondary effects of the chemotherapy, the lack of appetite, nausea, vomits, diarrhea, mouth sores, etc., were factors that made it very difficult for the patients to intake 15 pills at once, as the study initially planned. Besides, the masking of the bioactive agent was not possible because the pills were easily distinguishable. All these raised inconveniences, the progression of the pancreatic cancer and the GI symptomatology, led to a high rate of drop-off of 72% in the treatment group and 55% abandonment in the control group, and therefore, the results were not significant.
In order to finish this critical analysis, the only study of cohorts included in this review, from the group of Ma et al. [12], analyzed data from 200,907 people free of disease (cancer, diabetes mellitus, or cardiovascular disease) at the beginning of the study. To establish this, they used semi-quantitative polls of food frequency: vegetables, fruits, whole grains, sugar drinks, juices, nuts, legumes, red meat, processed foods, trans-fats, omega-3 fatty acid rich foods, polyunsaturated fats, salt, and alcohol consumption. They also included the risk factors for type 2 diabetes mellitus (T2DM), body weight, smoking, pharmaceutical treatments or vitamin complex intake, family history of diabetes, chronic diseases, hypertension, dyslipidemia, and physical activity calculated on a weekly basis. With all this information, the authors presented the results that there was a risk for T2DM with the intake of cruciferous foods and glucosinolates. However, in agreement with the work of Oliviero et al. [29], this study presented big limitations because it did not review or consider the cooking procedures, it did not quantify the amount of vegetables consumed on a daily or weekly basis, and it did not quantify the glucosinolate contents in the different vegetables consumed. Besides, the population sample was homogenous, because all of the participants were health-area workers, and the results are not extrapolatable to the general population. Finally, it should be highlighted that other nutrients and cofounding factors were not considered in the study, and they could affect the development of these pathologies.
References
- Colditz, G.A.; Wollin, K.Y.; Gehlert, S. Applying What We Know to Accelerate Cancer Prevention. Sci. Transl. Med. 2012, 4, 127.
- Alkhatib, A.; Tsang, C.; Tiss, A.; Bahorun, T.; Arefanian, H.; Barake, R.; Khadir, A.; Tuomilehto, J. Functional foods and lifestyle approaches for diabetes prevention and management. Nutrients 2017, 9, 1310.
- Patravale, V.B.; Naik, S.V.; Dhage, S.N. Role of Diet, Functional Foods, and Nutraceuticals in Brain Disorders. In Nutraceutical and Functional Foods in Disease Prevention; Raj, K.K., Anil, K.S., Rajesh, K.K., Eds.; Information Resources Management Association: Pennsylvania, PA, USA, 2018; pp. 256–287.
- Anderson, J.V.; Bybee, D.I.; Brown, R.M.; McLean, D.F.; Garcia, E.M.; Breer, M.L.; Schillo, B.A. 5 A day fruit and vegetable intervention improves consumption in a low income population. J. Am. Diet. Assoc. 2001, 101, 195–202.
- Avato, P.; Argentieri, M.P. Brassicaceae: A rich source of health improving phytochemicals. Phytochem. Rev. 2015, 14, 1019–1033.
- Sita, G.; Hrelia, P.; Tarozzi, A.; Morroni, F. Isothiocyanates are promising compounds against oxidative stress, neuroinflammation and cell death that may benefit neurodegeneration in Parkinson’s disease. Int. J. Mol. Sci. 2016, 17, 1454.
- Royston, K.J.; Tollefsbol, T.O. The Epigenetic Impact of Cruciferous Vegetables on Cancer Prevention. Curr. Pharmacol. Rep. 2015, 1, 46–51.
- Blažević, I.; Montaut, S.; Burčul, F.; Olsen, C.E.; Burow, M.; Rollin, P.; Agerbirk, N. Glucosinolate structural diversity, identification, chemical synthesis and metabolism in plants. Phytochemistry 2020, 169, e112100.
- Grubb, C.D.; Abel, S. Glucosinolate metabolism and its control. Trends Plant Sci. 2006, 11, 89–100.
- Hanschen, F.S.; Schreiner, M. Isothiocyanates, nitriles, and epithionitriles from glucosinolates are affected by genotype and developmental stage in Brassica oleracea varieties. Front. Plant Sci. 2017, 8, 1095.
- Wittstock, U.; Burow, M. Tipping the scales-Specifier proteins in glucosinolate hydrolysis. IUBMB Life 2007, 59, 744–751.
- Ma, L.; Liu, G.; Sampson, L.; Willett, W.C.; Hu, F.B.; Sun, Q. Dietary glucosinolates and risk of type 2 diabetes in 3 prospective cohort studies. Am. J. Clin. Nutr. 2018, 107, 617–625.
- Ma, L.; Liu, G.; Zong, G.; Sampson, L.; Hu, F.B.; Willett, W.C.; Rimm, E.B.; Manson, J.E.; Rexrode, K.M.; Sun, Q. Intake of glucosinolates and risk of coronary heart disease in three large prospective cohorts of US men and women. Clin. Epidemiol. 2018, 10, 749–762.
- Gee, J.R.; Saltzstein, D.R.; Messing, E.; Kim, K.M.; Kolesar, J.; Huang, W.; Havighurst, T.C.; Harris, L.; Wollmer, B.W.; Jarrard, D.; et al. Phase Ib placebo-controlled, tissue biomarker trial of diindolylmethane (BR-DIMNG) in patients with prostate cancer who are undergoing prostatectomy. Eur. J. Cancer Prev. 2016, 25, 312–320.
- Tahata, S.; Singh, S.V.; Lin, Y.; Hahm, E.R.; Beumer, J.H.; Christner, S.M.; Rao, U.N.; Sander, C.; Tarhini, A.A.; Tawbi, H.; et al. Evaluation of biodistribution of sulforaphane after administration of oral broccoli sprout extract in melanoma patients with multiple atypical nevi. Cancer Prev. Res. 2018, 11, 429–437.
- Sudini, K.; Diette, G.B.; Breysse, P.N.; McCormack, M.C.; Bull, D.; Biswal, S.; Zhai, S.; Brereton, N.; Peng, R.D.; Matsui, E.C. A Randomized Controlled Trial of the Effect of Broccoli Sprouts on Antioxidant Gene Expression and Airway Inflammation in Asthmatics. J. Allergy Clin. Immunol. Pract. 2016, 4, 932–940.
- Wise, R.A.; Holbrook, J.T.; Criner, G.; Sethi, S.; Rayapudi, S.; Sudini, K.R.; Sugar, E.A.; Burke, A.; Thimmulappa, R.; Singh, A.; et al. Lack of effect of oral sulforaphane administration on Nrf2 expression in COPD: A randomized, double-blind, placebo controlled trial. PLoS ONE 2016, 11, 1–15.
- Brown, R.H.; Reynolds, C.; Brooker, A.; Talalay, P.; Fahey, J.W. Sulforaphane improves the bronchoprotective response in asthmatics through Nrf2-mediated gene pathways. Respir. Res. 2015, 16, 1–12.
- Bahadoran, Z.; Tohidi, M.; Nazeri, P.; Mehran, M.; Azizi, F.; Mirmiran, P. Effect of broccoli sprouts on insulin resistance in type 2 diabetic patients: A randomized double-blind clinical trial. Int. J. Food Sci. Nutr. 2012, 63, 767–771.
- Alumkal, J.J.; Slottke, R.; Schwartzman, J.; Cherala, G.; Graff, J.N.; Beer, T.M.; Ryan, C.W.; Koop, D.R.; Gao, L.; Flamiatos, J.F.; et al. A phase II study of sulforaphane-rich broccoli sprout extracts in men with recurrent prostate cancer Joshi. Investig. New Drugs 2015, 33, 480–489.
- Cipolla, B.G.; Mandron, E.; Marc Lefort, J.; Coadou, Y.; Della Negra, E.; Corbel, L.; Le Scodan, R.; Rahmene Azzouzi, A.; Mottet, N. Effect of sulforaphane in men with biochemical recurrence after radical prostatectomy. Cancer Prev. Res. 2015, 8, 712–719.
- Atwell, L.L.; Zhang, Z.; Mori, M.; Farris, P.; Vetto, J.T.; Naik, A.M.; Oh, K.Y.; Thuillier, P.; Ho, E.; Shannon, J. Sulforaphane bioavailability and chemopreventive activity in women scheduled for breast biopsy. Cancer Prev. Res. 2015, 8, 1184–1191.
- Paltsev, M.; Kiselev, V.; Drukh, V.; Muyzhnek, E.; Kuznetsov, I.; Andrianova, E.; Baranovskiy, P. First results of the double-blind randomized placebo-controlled multicenter clinical trial of DIM-based therapy designed as personalized approach to reverse prostatic intraepithelial neoplasia (PIN). EPMA J. 2016, 7, 1–8.
- Traka, M.H.; Melchini, A.; Coode-Bate, J.; Al Kadhi, O.; Saha, S.; Defernez, M.; Troncoso-Rey, P.; Kibblewhite, H.; O’Neill, C.M.; Bernuzzi, F.; et al. Transcriptional changes in prostate of men on active surveillance after a 12-mo glucoraphanin-rich broccoli intervention-results from the Effect of Sulforaphane on prostate CAncer PrEvention (ESCAPE) randomized controlled trial. Am. J. Clin. Nutr. 2019, 109, 1133–1144.
- Thomson, C.A.; Chow, H.H.S.; Wertheim, B.C.; Roe, D.J.; Stopeck, A.; Maskarinec, G.; Altbach, M.; Chalasani, P.; Huang, C.; Strom, M.B.; et al. A randomized, placebo-controlled trial of diindolylmethane for breast cancer biomarker modulation in patients taking tamoxifen. Breast Cancer Res. Treat. 2017, 165, 97–107.
- Kiselev, V.I.; Ashrafyan, L.A.; Muyzhnek, E.L.; Gerfanova, E.V.; Antonova, I.B.; Aleshikova, O.I.; Sarkar, F.H. A new promising way of maintenance therapy in advanced ovarian cancer: A comparative clinical study. BMC Cancer 2018, 18, 1–16.
- Lozanovski, V.J.; Houben, P.; Hinz, U.; Hackert, T.; Herr, I.; Schemmer, P. Pilot study evaluating broccoli sprouts in advanced pancreatic cancer (POUDER trial)-study protocol for a randomized controlled trial. Trials 2014, 15, 1–8.
- Lozanovski, V.J.; Polychronidis, G.; Gross, W.; Gharabaghi, N.; Mehrabi, A.; Hackert, T.; Schemmer, P.; Herr, I. Broccoli sprout supplementation in patients with advanced pancreatic cancer is difficult despite positive effects—Results from the POUDER pilot study. Investig. New Drugs 2019.
- Oliviero, T.; Verkerk, R.; Dekker, M. Reply to “Dietary glucosinolates and risk of type 2 diabetes in 3 prospective cohort studies”. Am. J. Clin. Nutr. 2018, 108, 425.
More
Information
Subjects:
Food Science & Technology; Nutrition & Dietetics; Others
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
824
Revisions:
2 times
(View History)
Update Date:
12 Nov 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





