
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | M. R. Mozafari | + 2815 word(s) | 2815 | 2021-11-05 03:40:46 | | | |
| 2 | M. R. Mozafari | Meta information modification | 2815 | 2021-11-11 04:34:13 | | | | |
| 3 | Peter Tang | Meta information modification | 2815 | 2021-11-11 06:34:19 | | |
Video Upload Options
Nanoscale lipid bilayers, or nanoliposomes, are generally spherical vesicles formed by the dispersion of phospholipid molecules in a water-based medium by energy input. Nanoliposomes and tocosomes are able to provide protection and release of sensitive food-grade bioactive materials in a sustained manner. They are being utilized for the encapsulation of different types of bioactive materials (such as drugs, vaccines, antimicrobials, antioxidants, minerals and preservatives), for the enrichment and fortification of different food and nutraceutical formulations and manufacturing of functional products.
1. Introduction
2. Nanoliposome Technology
3. Tocosomes
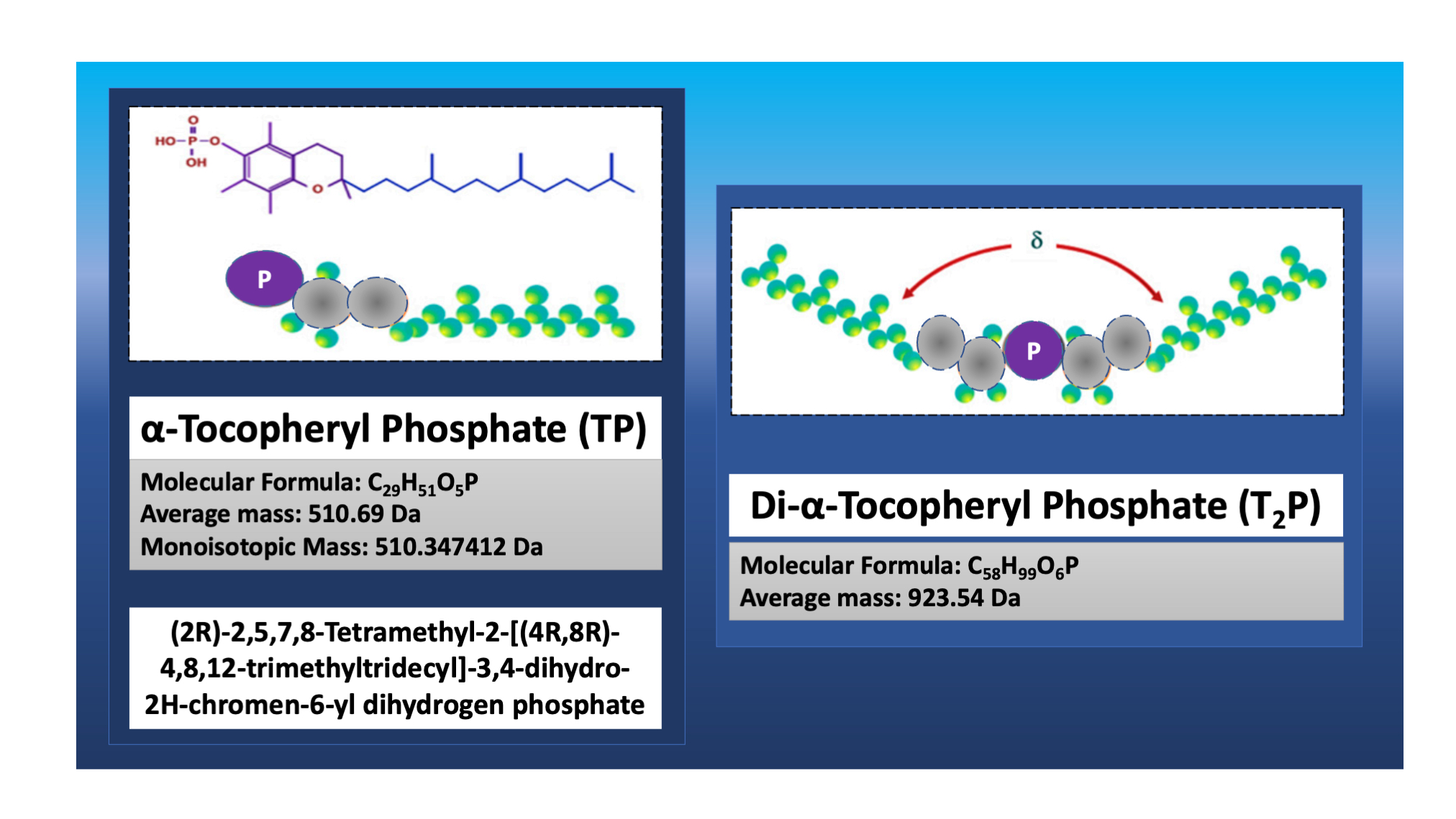
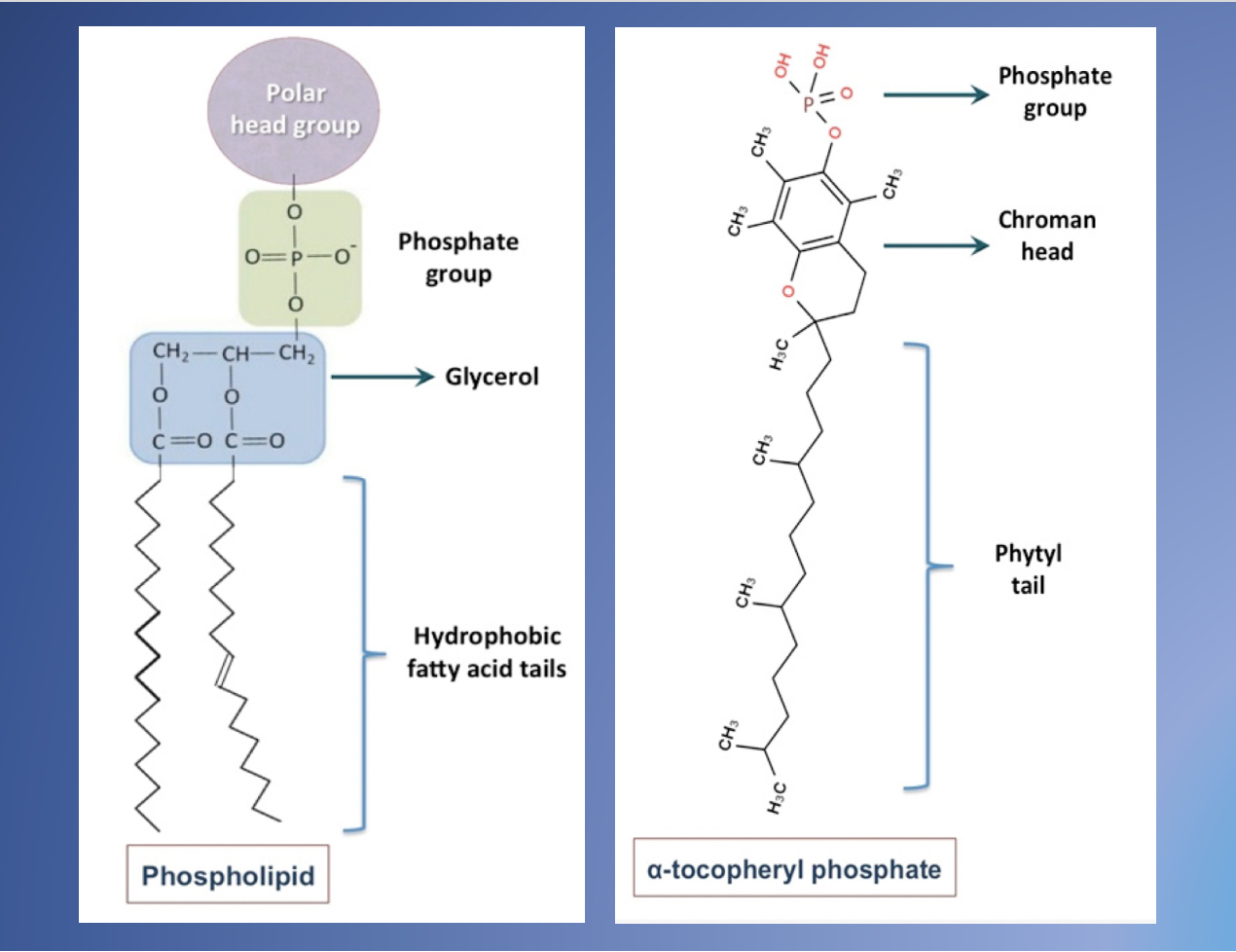
4. Differences between Tocosomes and Nanoliposomes
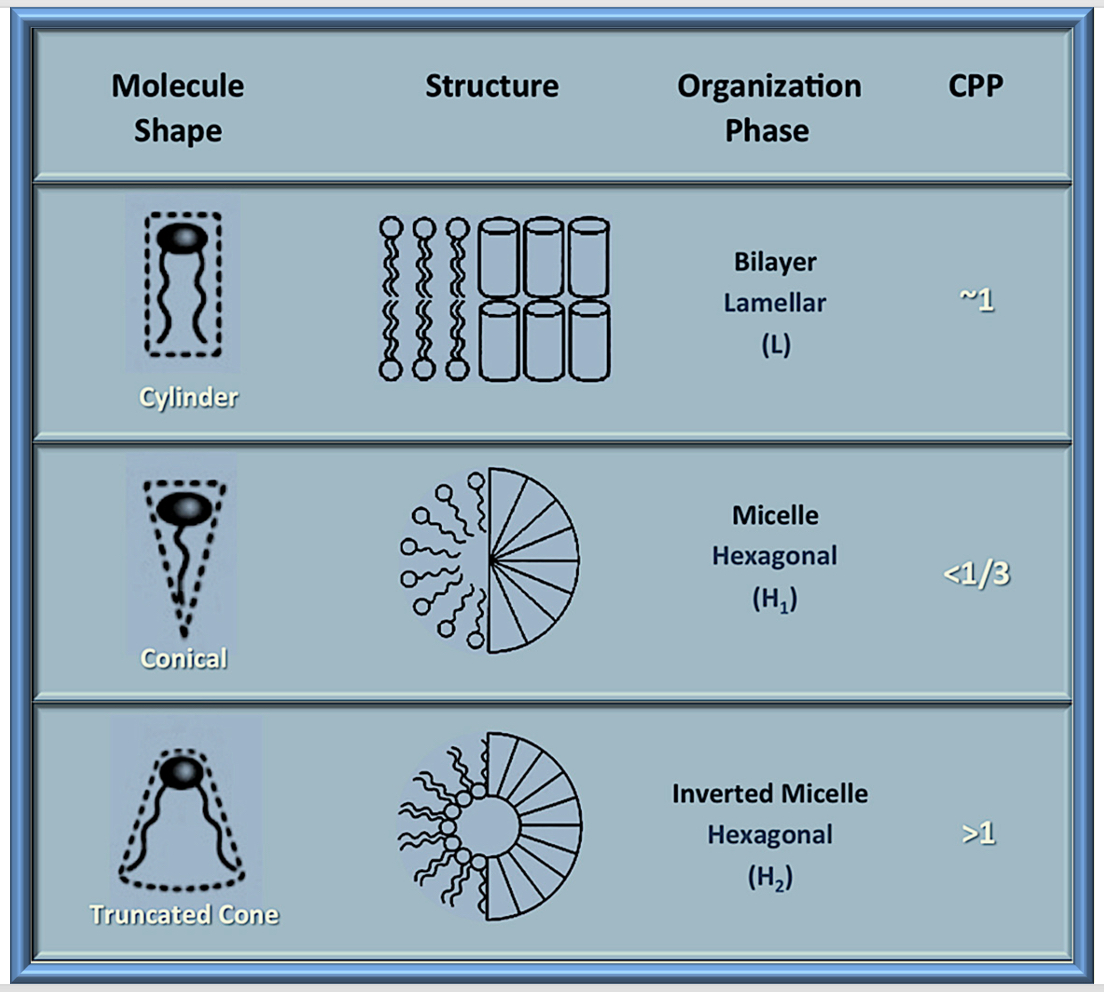
5. Mechanism of Formation of Nanoliposomes and Tocosomes
|
Method |
Advantages |
Disadvantages |
|---|---|---|
|
Thin-film hydration method |
High solubility of ingredients in the initial stage of the process |
Use of potentially toxic solvents, time consuming, difficult to scale-up |
|
Ethanol/ether injection |
Simple procedure |
Organic solvent residue, nozzle blockage in ether system, time consuming, sterilization issue |
|
Reverse phase evaporation |
Simple design, acceptable encapsulation efficiency |
Not suitable for the encapsulation of sensitive material due to large quantity of organic solvent use, time consuming, sterilization issue |
|
Microfluidisation |
Control of particle size, large volume manufacture in a continuous and reproducible manner |
Employment of high pressures (up to l0,000 psi) |
|
Supercritical Fluid Process (SFP) |
Control of particle size, possibility of in situ sterilization, low organic solvent consumption |
High cost, low yield, high pressure up to 350 bar used |
|
Dual asymmetric centrifugation |
Simple method, yields products with narrow size distribution, high encapsulation efficiency |
Not suitable for bulk production, high pressure and high shear force |
|
Sonication |
Simple and fast technique |
Overheating of the sample causing degradation, sonicator tips releases metal particles into the product |
|
Heating Method |
Organic solvent free, scalable |
High temperature requirement |
|
Mozafari Method |
Simple design, safe and mild procedure, organic solvent free, easily scalable |
New method, Reproducibility need to be attested under different conditions |
|
Binary Nanodispersions |
Organic solvent free, not requiring secondary emulsifier |
Requires ultrasonication |
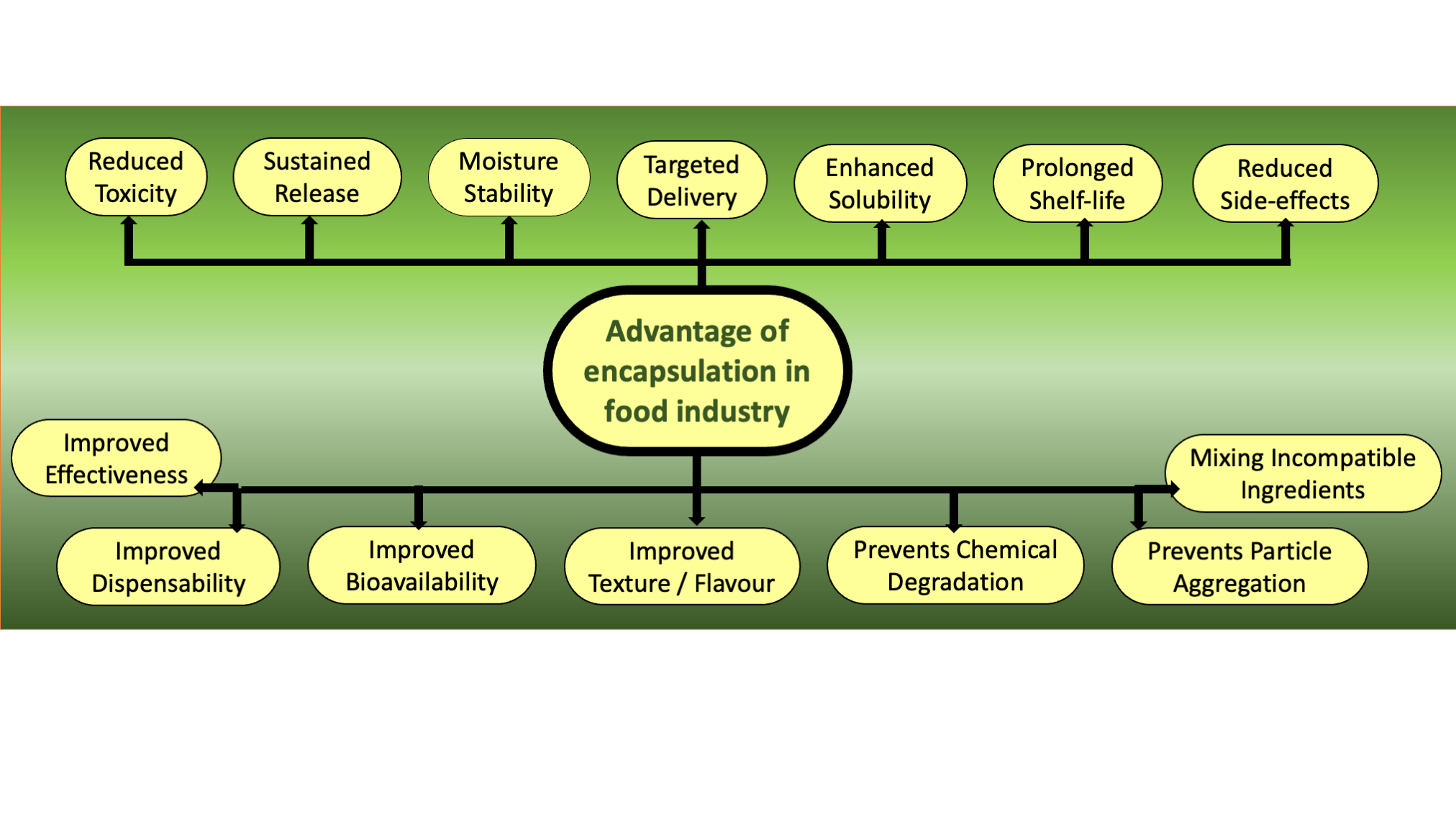
6. Applications in the Food Industry
References
- Bratovcic, A.; Suljagic, J. Micro-and nano-encapsulation in food industry. Croat. J. Food Sci. Technol. 2019, 11, 113–121.
- Jafari, S.M. Lipid-Based Nanostructures for Food Encapsulation Purposes (Vol. 2); Academic Press: Cambridge, MA, USA, 2019.
- Costa, S.S.; Machado, B.A.S.; Martin, A.R.; Bagnara, F.; Ragadalli, S.A.; Alves, A.R.C. Drying by spray drying in the food industry: Micro-encapsulation, process parameters and main carriers used. Afr. J. Food Sci. 2015, 9, 462–470.
- Srivastava, Y.; Semwal, A.D.; Sharma, G.K. Application of various chemical and mechanical microencapsulation techniques in food sector-A. review. Int. J. Food Ferment. Technol. 2013, 3, 1–13.
- Cerqueira, M.A.; Pinheiro, A.C.; Ramos, O.L.; Silva, H.; Bourbon, A.I.; Vicente, A.A. Advances in food nanotechnology. In Emerging Nanotechnologies in Food Science; Busquets, R., Ed.; Elsevier: Oxford, UK, 2017; pp. 11–38.
- Yurdugul, S.; Mozafari, M.R. Recent advances in micro-and nanoencapsulation of food ingredients. Cell. Mol. Biol. Lett. 2004, 9, 64–65.
- Mozafari, M.R.; Khosravi-Darani, K.; Borazan, G.G.; Cui, J.; Pardakhty, A.; Yurdugul, S. Encapsulation of food ingredients using nanoliposome technology. Int. J. Food Prop. 2008, 11, 833–844.
- Khorasani, S.; Danaei, M.; Mozafari, M.R. Nanoliposome technology for the food and nutraceutical industries. Trends Food Sci. Technol. 2018, 79, 106–115.
- Lokhande, S.S. Liposome drug delivery: An update review. Pharma Sci. Monit. 2018, 9, 188–202.
- Mozafari, M.R.; Zareie, M.H.; Piskin, E.; Hasirci, V. Formation of supramolecular structures by negatively charged liposomes in the presence of nucleic acids and divalent cations. Drug Deliv. 1998, 5, 135–141.
- Zareie, M.H.; Mozafari, M.R.; Hasirci, V.; Piskin, E. Scanning tunnelling microscopy investigation of liposome-DNA-Ca2+ complexes. J. Liposome Res. 1997, 7, 491–502.
- Arya, M.; Yale University. Compositions and Methods for Treating Metabolic Disorders. U.S. Patent Application 15/781,606, 2017.
- Mozafari, M.R.; Omri, A. Importance of divalent cations in nanolipoplex gene delivery. J. Pharm. Sci. 2007, 96, 1955–1966.
- Edris, A.E. Formulation and shelf life stability of water-borne lecithin nanoparticles for potential application in dietary supplements field. J. Diet. Suppl. 2012, 9, 211–222.
- Mozafari, M.R.; Johnson, C.; Hatziantoniou, S.; Demetzos, C. Nanoliposomes and their applications in food nanotechnology. J. Liposome Res. 2008, 18, 309–327.
- Martin, F.J. Pharmaceutical manufacturing of liposomes. Drugs Pharm. Sci. 1990, 41, 267–316.
- Mozafari, M.R. Nanoliposomes: Preparation and analysis. In Liposomes; Humana Press: Totowa, NJ, USA, 2010; pp. 29–50.
- Mozafari, M.R.; Javanmard, R.; Raji, M. Tocosome: Novel drug delivery system containing phospholipids and tocopheryl phosphates. Int. J. Pharm. 2017, 528, 381–382.
- Azzi, A. Tocopheryl phosphate, a novel natural form of vitamin E: In vitro and in vivo studies. FASEB J. 2006, 20, LB79–LB80.
- Gianello, R.; Libinaki, R.; Azzi, A.; Gavin, P.D.; Negis, Y.; Zingg, J.M.; West, S.M. α-Tocopheryl phosphate: A novel, natural form of vitamin E. Free Radic. Biol. Med. 2005, 39, 970–976.
- Ogru, E.; Gianello, R.; Libinaki, R.; Smallridge, A.; Bak, R.; Geytenbeek, S.; West, S. Vitamin E phosphate: An endogenous form of vitamin E. In Free Radicals and Oxidative Stress: Chemistry, Biochemistry and Pathophysiological Implications; Galaris, D., Ed.; Medimond Pub. Inc.: Bologna, Italy, 2003; pp. 127–132.
- Munteanu, A.; Zingg, J.M.; Ogru, E.; Libinaki, R.; Gianello, R.; West, S.; Azzi, A. Modulation of cell proliferation and gene expression by α-tocopheryl phosphates: Relevance to atherosclerosis and inflammation. Biochem. Biophys. Res. Commun. 2004, 318, 311–316.
- Libinaki, R.; Tesanovic, S.; Heal, A.; Nikolovski, B.; Vinh, A.; Widdop, R.E.; Ogru, E. Effect of tocopheryl phosphate on key biomarkers of inflammation: Implication in the reduction of atherosclerosis progression in a hypercholesterolaemic rabbit model. Clin. Exp. Pharmacol. Physiol. 2010, 37, 587–592.
- Saitoh, Y.; Yumoto, A.; Miwa, N. α-tocopheryl phosphate suppresses tumor invasion concurrently with dynamic morphological changes and delocalization of cortactin from invadopodia. Int. J. Oncol. 2009, 35, 1277–1288.
- Nishio, K.; Ishida, N.; Saito, Y.; Ogawa-Akazawa, Y.; Shichiri, M.; Yoshida, Y.; Niki, E. α-Tocopheryl phosphate: Uptake, hydrolysis, and antioxidant action in cultured cells and mouse. Free Radic. Biol. Med. 2011, 50, 1794–1800.
- Mukherjee, S.; Lekli, I.; Das, M.; Azzi, A.; Das, D.K. Cardioprotection with α-tocopheryl phosphate: Amelioration of myocardial ischemia reperfusion injury is linked with its ability to generate a survival signal through Akt activation. Biochim. Biophys. Acta (BBA)–Mol. Basis Dis. 2008, 1782, 498–503.
- Kamal-Eldin, A.; Appelqvist, L.A. The chemistry and antioxidant properties of tocopherols and tocotrienols. Lipids 1996, 31, 671–701.
- Mohammadabadi, M.R.; Mozafari, M.R. Enhanced efficacy and bioavailability of thymoquinone using nanoliposomal dosage form. J. Drug Deliv. Sci. Technol. 2018, 47, 445–453.
- Mohammadabadi, M.R.; Mozafari, M.R. Development of nanoliposome-encapsulated thymoquinone: Evaluation of loading efficiency and particle characterization. J. Biopharm. 2019, 11, 39–46.
- Aveling, E.; Zhou, J.; Lim, Y.F.; Mozafari, M.R. Targeting lipidic nanocarriers: Current strategies and problems. Pharmakeftiki 2006, 19, 101–109.
- Maherani, B.; Arab-Tehrany, E.; Mozafari, M.R.; Gaiani, C.; Linder, M. Liposomes: A review of manufacturing techniques and targeting strategies. Curr. Nanosci. 2011, 7, 436–452.
- Kulkarni, C. Lipid self-assemblies and nanostructured emulsions for cosmetic formulations. Cosmetics 2016, 3, 37.
- Lasic, D.D. Mechanisms of liposome formation. J. Liposome Res. 1995, 5, 431–441.
- Lasic, D.D. Novel applications of liposomes. Trends Biotechnol. 1998, 16, 307–321.
- Lasic, D.D.; Walker, S.; Bode, C.J.; Paquet, K.J. Kinetic and thermodynamic effects on the structure and formation of phosphatidylcholine vesicles. Hepatology 1991, 13, 1010–1013.
- Mozafari, M.R.; Mortazavi, S.M. Nanoliposomes: From Fundamentals to Recent Developments; Trafford Pub. Ltd.: Oxford, UK, 2005.
- Katouzian, I.; Esfanjani, A.F.; Jafari, S.M.; Akhavan, S. Formulation and application of a new generation of lipid nano-carriers for the food bioactive ingredients. Trends Food Sci. Technol. 2017, 68, 14–25.
- Wang, T.; Xue, J.; Hu, Q.; Zhou, M.; Luo, Y. Preparation of lipid nanoparticles with high loading capacity and exceptional gastrointestinal stability for potential oral delivery applications. J. Colloid Interface Sci. 2017, 507, 119–130.
- Verma, M.L. Nanobiotechnology advances in enzymatic biosensors for the agri-food industry. Environ. Chem. Lett. 2017, 15, 555–560.
- Colas, J.C.; Shi, W.; Rao, V.M.; Omri, A.; Mozafari, M.R.; Singh, H. Microscopical investigations of nisin-loaded nanoliposomes prepared by Mozafari method and their bacterial targeting. Micron 2007, 38, 841–847.
- Poudel, A.; Gachumi, G.; Wasan, K.M.; Dallal Bashi, Z.; El-Aneed, A.; Badea, I. Development and Characterization of Liposomal Formulations Containing Phytosterols Extracted from Canola Oil Deodorizer Distillate along with Tocopherols as Food Additives. Pharmaceutics 2019, 11, 185.
- Patel, S.; Patel, R.; Dharamsi, A. A review: Proliposomes as a stable novel drug delivery. World J. Pharm. Res. 2019, 8, 638–651.
- Sanguansri, L.; Augustin, M.A. Microencapsulation in functional food product development. Funct. Food Prod. Dev. 2010, 1, 1–23.
- Khanniri, E.; Bagheripoor-Fallah, N.; Sohrabvandi, S.; Mortazavian, A.M.; Khosravi-Darani, K.; Mohammad, R. Application of liposomes in some dairy products. Crit. Rev. Food Sci. Nutr. 2016, 56, 484–493.





