Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Xing, Y. EXO70 Gene Family in Cotton. Encyclopedia. Available online: https://encyclopedia.pub/entry/15446 (accessed on 25 May 2026).
Xing Y. EXO70 Gene Family in Cotton. Encyclopedia. Available at: https://encyclopedia.pub/entry/15446. Accessed May 25, 2026.
Xing, Yadi. "EXO70 Gene Family in Cotton" Encyclopedia, https://encyclopedia.pub/entry/15446 (accessed May 25, 2026).
Xing, Y. (2021, October 27). EXO70 Gene Family in Cotton. In Encyclopedia. https://encyclopedia.pub/entry/15446
Xing, Yadi. "EXO70 Gene Family in Cotton." Encyclopedia. Web. 27 October, 2021.
Copy Citation
The EXO70 gene is a vital component of the exocytosis complex and participates in biological processes ranging from plant cell division to polar growth. There are many EXO70 genes in plants and their functions are extensive, but little is known about the EXO70 gene family in cotton. Here, we analyzed four cotton sequence databases, identified 165 EXO70 genes, and divided them into eight subgroups (EXO70A–EXO70H) based on their phylogenetic relationships. EXO70A had the most exons (≥11), whereas the other seven each had only one or two exons.
EXO70
Gossypium
evolution analysis
transcriptome
expression analysis
1. Introduction
Vesicle transport is an extremely important cytological process in eukaryotes. It moves proteins, lipids, and other substances between the inner membrane system and the cells, and establishes cell polarity, secretion, growth, division, and wall formation [1]. Tethering is a key step in vesicle transport. Large multi-subunit tethering complexes were first discovered in yeast [2]. Exocysts tether different vesicles to the exocytosis site required for cellular secretion [3]. They are evolutionarily conserved octameric protein complexes composed of Sec3, Sec5, Sec6, Sec8, Sec10, Sec15, EXO70, and EXO84 [4][5]. EXO70 plays a key role in exocyst assembly [6]. It recruits exocysts on the target membrane and interacts with Rho protein to regulate SNARE complex assembly and activation there via SEC6. In this manner, EXO70 mediates polar exocytosis [7][8].
The exocyst subunits are encoded by a single gene in yeast and just a few genes in metazoans. However, 23 EXO70 subunits encoded by various loci have been identified in Arabidopsis [9][10]. The EXO70s in terrestrial plant genomes have even more copies. This phenomenon is unique to the EXO70 subunit of the exocyst [11]. In the fungal and animal genomes sequenced to date, only one EXO70 coding gene was found. Hence, multiple EXO70 gene copies are unique to higher terrestrial plants [12]. Certain EXO70 functions might have been alienated during evolution and participated in other biological processes besides membrane vesicle transport. Alternatively, various EXO70 functions are specialized and form different exocysts from other subunits that participate in specific membrane vesicle transport processes in the organization, carrier substrate, or transport link [12]. The expression profiles of the 23 members of the Arabidopsis EXO70 family have been analyzed. Expression of this gene family has the following characteristics: spatiotemporal expression specificity at the cell and tissue levels; no constitutive expression; and specific expression in dividing, growing, differentiating, and secreting cells [12]. Plant EXO70 gene family members participate at the transcriptional level in the biological processes of different cell types via cell- and tissue-specific expression patterns.
EXO70 is an important part of the secretory complex mediating exocytosis, and it regulates neurite growth in animal cells, epithelial cell polarity, and cell movement and morphogenesis [13][14][15][16][17]. In plants, EXO70 regulates pollen tube elongation and polarization, root hair growth, cell wall material deposition, cell plate activation and maturation, defense and autophagy, and so on [10][18][19][20][21]. Defects in AtEXO70C2 gene function affect pollen tube growth, which results in significant male-specific transmission defects in Arabidopsis [22]. EXO70H4- and PMR4-dependent corpus callosum deposition in trichomes is necessary for cell wall silicification [23]. EXO70B1 knockdown resulted in impaired light-induced stomatal opening [24]. AtEXO70B1 and AtEXO70B2 regulate FLS2 to participate in plant immune response [25]. AtEXO70D regulates cytokinin sensitivity by mediating the selective autophagy of Type-A ARR protein, thereby maintaining cell homeostasis and normal plant growth and development [26]. OsEXO70A1, OsEXO70L2, and AtEXO70A1 affect tracheary element (TE) development [27][28][29]. Hence, the roles of EXO70 in plant organ development have undergone differentiation.
Cotton is a major global economic crop. It is a source of seed, fiber, oil, and medicine [30][31]. The development of novel high-quality cotton varieties is of great commercial importance. The high copy numbers and tissue-specific functions of the EXO70 gene in plants suggest that targeting EXO70 to construct high-quality cotton is feasible. To date, however, few studies have investigated the cotton EXO70 gene. The only report is that GhEXO70B1 may respond to stressors by mediating cell autophagy [32].
2. Identification and Analysis of the Phylogenetic Relationship of the EXO70 Gene Family in Cotton
The exocytosis complex subunits comprise mostly EXO70 gene family members. There are 23 and 47 EXO70 genes in the model dicotyledon Arabidopsis thaliana and the monocotyledon rice, respectively. Here, we identified 165 EXO70 genes among the four cotton subspecies included in the CottonFGD database, namely, G. hirsutum, G. barbadense, G. arboretum, and G. raimondii. There were 27, 26, 55, and 57 genes in G. arboretum, G. raimondii, G. barbadense, and G. hirsutum, respectively. We also identified 48 EXO70 genes in the tobacco database (Figure 1A). A phylogenetic analysis of the evolutionary relationships of 23 Arabidopsis EXO70s, 41 rice EXO70s, 165 cotton EXO70s, and 48 tobacco EXO70s (Figure 1C) showed that cotton EXO70 resembled Arabidopsis EXO70. Both of these plants are dicotyledons [9][33] and their EXO70s could be divided into eight categories. Relative to the monocotyledon rice, the dicotyledons lacked four EXO70 categories, such as EXO70I–EXO70L [34]. Hence, the EXO70 gene may be markedly differentiated between monocotyledons and dicotyledons. Monocotyledons possess more EXO70 genes than dicotyledons. Based on the phylogenetic tree, the grouping and naming of Arabidopsis, cotton EXO70s can be divided into eight subgroups (EXO70A–EXO70H) containing 12, 6, 29, 12, 27, 12, 22, and 45 genes, respectively (Figure 1B).
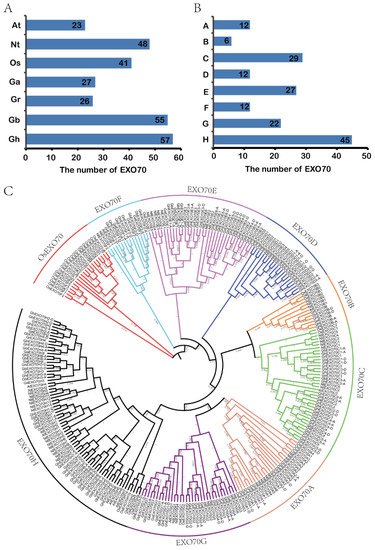
Figure 1. Numbers and phylogenetic relationships of EXO70 family genes in Arabidopsis, rice, upland cotton, sea island cotton, Asian cotton, Raymond cotton, and tobacco. (A) Numbers of EXO70 family genes in Arabidopsis, rice, upland cotton, sea island cotton, Asian cotton, Raymond cotton, and tobacco. At: Arabidopsis thaliana; Nt: Nicotiana tabacum; Os: Oryza sativa; Ga: G. arboretum; Gr: G. raimondii; Gb: G. barbadense; Gh: G. hirsutum. (B) Quantitative statistics for each subgroup of the EXO70 family genes in upland cotton, sea island cotton, Asian cotton, and Raymond cotton. (C) Phylogenetic analysis of EXO70 family genes in Arabidopsis, rice, upland cotton, sea island cotton, Asian cotton, Raymond cotton, and tobacco.
According to the cotton EXO70 gene classification, we named the 57 EXO70 genes in upland cotton as GhEXO70A1–GhEXO70A2, GhEXO70B, GhEXO70C1–GhEXO70C5, GhEXO70D1–GhEXO70D2, GhEXO70E1–GhEXO70E6, GhEXO70F1–GhEXO70F2, GhEXO70G1–GhEXO70G4, and GhEXO70H1–GhEXO70H8. Groups A and D were represented by -A and -D, respectively. We predicted their genome locations, protein lengths, numbers of exons, isoelectric points, protein molecular weights, and subcellular locations. The numbers of exons widely varied among GhEXO70 genes. All four GhEXO70A genes had the most exons (≥11 each) (Table 1). Further analysis of the exons of the EXO70 gene in Arabidopsis and rice showed that only Group A contained more exons in Arabidopsis and rice. The number of EXO70 in Arabidopsis A group was ≥9, and the number of EXO70 in rice A group was ≥12. Therefore, this phenomenon is not unique to cotton EXO70s, but is conserved in plants. The subcellular localization prediction results showed that GhEXO70 was mostly localized in the cell membrane, cytoplasm, or nucleus, which was consistent with the reported subcellular localization results of EXO70 from H. villosa [35] (Table 1).
Table 1. Nomenclature and analysis of physicochemical properties of EXO70 family genes in G. hirsutum.
| Gene ID | Name | Chromosome | Start | End | Exon Number | Protein Length (aa) | Molecular Weight (kDa) | Isoelectric Point | Subcellular Location |
|---|---|---|---|---|---|---|---|---|---|
| Gh_A10G1765 | GhEXO70A1-A | A10 | 92,079,304 | 92,086,988 | 12 | 650 | 73.432 | 8.308 | Cell membrane |
| Gh_D10G2039 | GhEXO70A1-D | D10 | 56,152,841 | 56,160,513 | 12 | 650 | 73.429 | 8.131 | Cell membrane |
| Gh_A09G1270 | GhEXO70A2-A | A09 | 64,875,878 | 64,879,803 | 11 | 640 | 72.863 | 8.901 | Cell membrane |
| Gh_D09G1272 | GhEXO70A2-D | D09 | 39,824,276 | 39,828,245 | 11 | 644 | 73.467 | 9.329 | Cell membrane, cytoplasm |
| Gh_A03G0212 | GhEXO70B-A | A03 | 3,226,810 | 3,228,732 | 1 | 640 | 72.955 | 4.951 | Cell membrane, cytoplasm |
| Gh_D03G1369 | GhEXO70B-D | D03 | 42,280,554 | 42,282,476 | 1 | 640 | 72.962 | 4.914 | Cell membrane, cytoplasm |
| Gh_A01G1064 | GhEXO70C1-A | A01 | 36,760,999 | 36,763,005 | 1 | 668 | 77.345 | 8.781 | Cell membrane, cytoplasm |
| Gh_D01G1124 | GhEXO70C1-D | D01 | 23,935,803 | 23,937,809 | 1 | 668 | 76.941 | 8.482 | Cell membrane, cytoplasm |
| Gh_A10G0625 | GhEXO70C2-A | A10 | 9,989,979 | 9,992,177 | 1 | 732 | 84.71 | 4.555 | Nucleus |
| Gh_D10G0774 | GhEXO70C2-D | D10 | 9,213,316 | 9,215,523 | 1 | 735 | 85.065 | 4.537 | Nucleus |
| Gh_A04G0860 | GhEXO70C3-A | A04 | 55,909,662 | 55,911,515 | 1 | 617 | 70.763 | 4.975 | Cell membrane, cytoplasm |
| Gh_D04G1359 | GhEXO70C3-D | D04 | 44,235,768 | 44,237,621 | 1 | 617 | 70.799 | 5.024 | Cell membrane, cytoplasm |
| Gh_A09G0369 | GhEXO70C4-A | A09 | 20,318,531 | 20,320,444 | 1 | 637 | 73.612 | 5.53 | Cell membrane, cytoplasm |
| Gh_D09G0388 | GhEXO70C4-D | D09 | 14,114,761 | 14,116,674 | 1 | 637 | 73.682 | 5.35 | Cell membrane, cytoplasm |
| Gh_A05G2929 | GhEXO70C5-A | A05 | 70,962,860 | 70,964,782 | 1 | 640 | 73.595 | 6.664 | Cell membrane, cytoplasm |
| Gh_D04G0713 | GhEXO70C5-D | D04 | 14,452,693 | 14,454,615 | 1 | 640 | 73.566 | 6.384 | Cell membrane, cytoplasm |
| Gh_A10G2233 | GhEXO70D1-A | scaffold2452_A10 | 2396 | 4279 | 1 | 627 | 71.136 | 5.36 | Cell membrane, cytoplasm |
| Gh_D10G0529 | GhEXO70D1-D | D10 | 5,107,684 | 5,109,567 | 1 | 627 | 71.084 | 5.278 | Cell membrane, cytoplasm |
| Gh_A05G1157 | GhEXO70D2-A | A05 | 11,706,409 | 11,708,259 | 1 | 616 | 69.599 | 5.35 | Cell membrane |
| Gh_D05G1334 | GhEXO70D2-D | D05 | 11,742,012 | 11,743,850 | 1 | 612 | 69.264 | 4.972 | Cell membrane, cytoplasm |
| Gh_A09G0090 | GhEXO70E1-A | A09 | 2,303,006 | 2,304,805 | 1 | 599 | 69.086 | 4.988 | Cell membrane, cytoplasm |
| Gh_D09G0087 | GhEXO70E1-D | D09 | 2,312,361 | 2,314,160 | 1 | 599 | 69.308 | 5.089 | Cell membrane, cytoplasm |
| Gh_D01G1051 | GhEXO70E2-D | D01 | 19,584,687 | 19,585,379 | 2 | 134 | 15.208 | 6.674 | Cell membrane |
| Gh_A05G3215 | GhEXO70E3-A | A05 | 84,043,718 | 84,045,679 | 1 | 653 | 74.613 | 4.761 | Cell membrane |
| Gh_D04G0392 | GhEXO70E3-D | D04 | 6,206,299 | 6,208,260 | 1 | 653 | 74.511 | 4.731 | Cell membrane |
| Gh_D12G0327 | GhEXO70E4-D | D12 | 4,666,459 | 4,667,427 | 1 | 322 | 36.173 | 4.875 | Cell membrane |
| Gh_A09G2154 | GhEXO70E5-A | A09 | 74,578,905 | 74,580,839 | 1 | 644 | 73.351 | 5.784 | Cell membrane, cytoplasm |
| Gh_D09G2359 | GhEXO70E5-D | D09 | 50,553,799 | 50,555,733 | 1 | 644 | 73.402 | 6.24 | Cell membrane, cytoplasm |
| Gh_A12G2651 | GhEXO70E6-A | scaffold3396_A12 | 4667 | 6610 | 1 | 647 | 73.345 | 5.788 | Cell membrane |
| Gh_D12G1810 | GhEXO70E6-D | D12 | 50,610,382 | 50,612,325 | 1 | 647 | 73.122 | 5.417 | Cell membrane |
| Gh_A03G0449 | GhEXO70F1-A | A03 | 9,703,926 | 9,705,884 | 1 | 652 | 73.754 | 4.614 | Cell membrane, cytoplasm |
| Gh_D03G1089 | GhEXO70F1-D | D03 | 36,373,153 | 36,375,111 | 1 | 652 | 73.79 | 4.587 | Cell membrane |
| Gh_A12G1712 | GhEXO70F2-A | A12 | 78,884,906 | 78,886,861 | 2 | 593 | 67.361 | 4.566 | Cell membrane |
| Gh_D12G1873 | GhEXO70F2-D | D12 | 51,339,965 | 51,341,920 | 2 | 593 | 67.36 | 4.589 | Cell membrane, cytoplasm |
| Gh_A13G1576 | GhEXO70G1-A | A13 | 74,625,245 | 74,627,293 | 1 | 682 | 77.054 | 8.387 | Cell membrane, cytoplasm |
| Gh_D13G1935 | GhEXO70G1-D | D13 | 54,742,538 | 54,744,586 | 1 | 682 | 76.932 | 8.152 | Cell membrane, cytoplasm |
| Gh_A05G0971 | GhEXO70G2-A | A05 | 9,685,126 | 9,687,123 | 1 | 665 | 74.839 | 6.629 | Cell membrane, nucleus |
| Gh_D05G1080 | GhEXO70G2-D | D05 | 9,208,639 | 9,210,636 | 1 | 665 | 74.79 | 6.457 | Cell membrane, nucleus |
| Gh_A13G1577 | GhEXO70G3-A | A13 | 74,629,925 | 74,631,973 | 1 | 682 | 77.006 | 8.013 | Cell membrane, cytoplasm |
| Gh_D13G1936 | GhEXO70G3-D | D13 | 54,747,144 | 54,749,192 | 1 | 682 | 77.129 | 8.008 | Cell membrane, cytoplasm |
| Gh_A05G1829 | GhEXO70G4-A | A05 | 19,148,304 | 19,150,892 | 2 | 705 | 80.991 | 6.269 | Cell membrane, nucleus |
| Gh_D05G2026 | GhEXO70G4-D | D05 | 18,583,197 | 18,585,799 | 2 | 706 | 81.133 | 6.088 | Cell membrane |
| Gh_A05G2577 | GhEXO70H1-A | A05 | 36,616,771 | 36,618,594 | 1 | 607 | 68.055 | 7.626 | Cell membrane |
| Gh_D05G2864 | GhEXO70H1-D | D05 | 32,263,469 | 32,264,524 | 1 | 351 | 39.036 | 8.216 | Cell membrane, nucleus |
| Gh_A04G0671 | GhEXO70H2-A | A04 | 45,359,837 | 45,362,111 | 2 | 621 | 70.248 | 5.721 | Cell membrane, cytoplasm |
| Gh_D04G1136 | GhEXO70H2-D | D04 | 37,216,953 | 37,218,701 | 1 | 582 | 65.703 | 5.697 | Cell membrane, cytoplasm |
| Gh_A11G2905 | GhEXO70H3-A | A11 | 92,971,783 | 92,973,285 | 1 | 500 | 56.067 | 7.521 | Cell membrane, nucleus |
| Gh_D11G3290 | GhEXO70H3-D | D11 | 65,820,199 | 65,822,067 | 1 | 622 | 69.832 | 7.178 | Cell membrane, cytoplasm |
| Gh_A01G1870 | GhEXO70H4-A | A01 | 98,692,379 | 98,694,289 | 1 | 636 | 71.827 | 7.493 | Cell membrane, cytoplasm |
| Gh_D01G2127 | GhEXO70H4-D | D01 | 60,327,492 | 60,329,402 | 1 | 636 | 72.148 | 7.783 | Cell membrane, cytoplasm |
| Gh_A07G0865 | GhEXO70H5-A | A07 | 15,194,291 | 15,196,123 | 1 | 610 | 69.021 | 6.068 | Cell membrane, cytoplasm |
| Gh_D07G0937 | GhEXO70H5-D | D07 | 12,444,178 | 12,446,010 | 1 | 610 | 68.925 | 6.316 | Cell membrane, cytoplasm |
| Gh_A05G0839 | GhEXO70H6-A | A05 | 8,379,960 | 8,381,828 | 1 | 622 | 70.171 | 5.757 | Cell membrane |
| Gh_D05G3898 | GhEXO70H6-D | scaffold4075_D05 | 141,419 | 143,287 | 1 | 622 | 70.049 | 5.361 | Cell membrane, cytoplasm |
| Gh_A03G0316 | GhEXO70H7-A | A03 | 5,680,420 | 5,682,288 | 1 | 622 | 70.772 | 5.209 | Cell membrane, cytoplasm |
| Gh_D03G1262 | GhEXO70H7-D | D03 | 40,093,224 | 40,095,089 | 1 | 621 | 70.603 | 5.889 | Cell membrane, cytoplasm |
| Gh_A11G2904 | GhEXO70H8-A | A11 | 92,966,319 | 92,968,183 | 2 | 568 | 63.859 | 8.556 | Cell membrane, cytoplasm |
3. Chromosome Distribution Analysis of EXO70 in the Cotton Genome
Diploid G. arboretum, G. raimondii, and Arabidopsis have 27, 26, and 23 EXO70 genes, respectively, while diploid rice has 47. Tetraploid G. hirsutum and G. barbadense have 57 and 55 EXO70 genes, respectively. The 27 EXO70 genes of G. arboretum are located on chromosomes 1–2, 4–5, 7, and 9–13, respectively (Figure 2C). The 26 EXO70 genes of G. raimondii are located on chromosomes 1–3 and 6–12, respectively (Figure 2D). The 57 EXO70 genes of G. hirsutum are located on chromosomes 1, 3–5, 7, and 9–13 in groups A and D, respectively (Figure 2A). The 55 EXO70 genes of G. barbadense are located on chromosomes 1, 3–5, 7, and 9–13 in group A and on chromosomes 1, 3–5, 7, 9–10, and 12–13 in group D (Figure 2B).
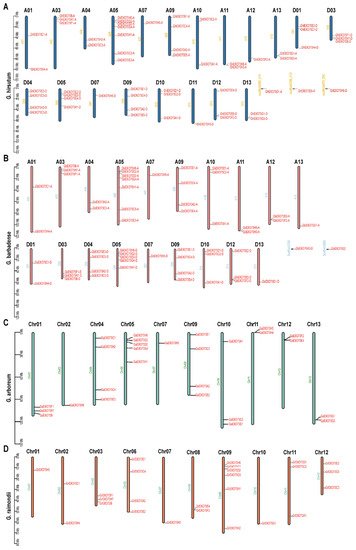
Figure 2. Chromosome distributions of EXO70 in upland cotton, sea island cotton, Asian cotton, and Raymond cotton. (A) Chromosome distribution map of EXO70 in upland cotton. (B) Chromosome distribution map of EXO70 in sea island cotton. (C) Chromosome distribution map of EXO70 in Asian cotton. (D) Chromosome distribution map of EXO70 in Raymond cotton.
Statistical analysis of the EXO70 gene distributions on the chromosomes revealed that there were relatively more EXO70 genes on chromosomes 5 and 9 in G. arboretum, G. barbadense, and G. hirsutum, but no EXO70 genes on chromosome 6 or 8. The EXO70 gene on chromosome 9 was distributed in G. raimondii, but that which was on chromosome 5 was not distributed. The EXO70 gene distributions on chromosomes 6 and 8 of G. raimondii (four and two, respectively) were the opposite of those for the other three cotton species (Figure 2; Table 2).
Table 2. Number of EXO70s in each chromosome of different cotton species.
| Chromosome | Ga (27) | Gr (26) | Gb (55) | Gh (57) | Total | ||
|---|---|---|---|---|---|---|---|
| A | D | A | D | A | D | ||
| Chr.1 | 3 | 1 | 2 | 2 | 2 | 3 | 13 |
| Chr.2 | 1 | 2 | 0 | 0 | 0 | 0 | 3 |
| Chr.3 | 0 | 3 | 3 | 3 | 3 | 3 | 15 |
| Chr.4 | 4 | 0 | 2 | 4 | 2 | 4 | 16 |
| Chr.5 | 5 | 0 | 7 | 5 | 7 | 4 | 28 |
| Chr.6 | 0 | 4 | 0 | 0 | 0 | 0 | 4 |
| Chr.7 | 1 | 1 | 1 | 1 | 1 | 1 | 6 |
| Chr.8 | 0 | 2 | 0 | 0 | 0 | 0 | 2 |
| Chr.9 | 4 | 6 | 4 | 4 | 4 | 4 | 26 |
| Chr.10 | 3 | 1 | 3 | 3 | 2 | 3 | 15 |
| Chr.11 | 2 | 3 | 2 | 0 | 2 | 1 | 10 |
| Chr.12 | 2 | 3 | 2 | 3 | 1 | 3 | 14 |
| Chr.13 | 2 | 0 | 1 | 1 | 2 | 2 | 8 |
| total | 27 | 26 | 27 | 26 | 26 | 28 | 160 |
| unknown | 0 | 0 | 1 | 1 | 2 | 1 | 5 |
The number of EXO70 genes in tetraploid cotton was nearly twice that in diploid cotton. The diploid cotton species (G. arboretum and G. raimondii) contained two EXO70As, one EXO70B, two EXO70Ds, and two EXO70Fs, whereas the tetraploid cotton species had twice these EXO70 gene copy numbers (Table 3). The numbers of EXO70Cs, EXO70Gs, and EXO70Hs in tetraploid cotton were twice those in the autodiploid species and equal to the sum of the number in the allodiploid species (Table 3). In polyploid cotton, then, the number of EXO70 genes increases via genome polyploidization. Most GhEXO70 genes are highly parallel in the At group and Dt subgenome. The exception is that GhEXO70E2-D and GhEXO70E4-D have no homologs in the At subgenome, while GhEXO70H8-A has no homologs in the Dt subgenome, indicating that they may be lost during evolution.
Table 3. Numbers of EXO70s in each subgroup of different cotton varieties.
| Subgroup | G. arboretum | G. raimondii | G. barbadense | G. hirsutum |
|---|---|---|---|---|
| A | 2 | 2 | 4 | 4 |
| B | 1 | 1 | 2 | 2 |
| C | 4 | 5 | 10 | 10 |
| D | 2 | 2 | 4 | 4 |
| E | 4 | 4 | 9 | 10 |
| F | 2 | 2 | 4 | 4 |
| G | 4 | 3 | 7 | 8 |
| H | 8 | 7 | 15 | 15 |
4. Analysis of EXO70 Gene Structure in G. hirsutum
G. hirsutum is the major global cotton variety and was the focus of research attention here. Structural analysis of its 57 GhEXO70 genes showed that all of them had one or two exons except for GhEXO70A, which had 10 or 11 exons. All GhEXO70 genes with similar structures are grouped in the same clade. Moreover, the genes with closely related phylogeny in the same subgroup also had similar structures. Within the same subgroup, however, certain genes exhibited entirely different structures. GhEXO70E2-D contained two exons, while the other genes within the same subgroup had only one. Similarly, GhEXO70G4 contained two exons, whereas GhEXO70G1–GhEXO70G3 each contained a single exon. GhEXO70H2-A and GhEXO70H8-A each contained two exons while the other genes within the same subgroup had only one (Figure 3).
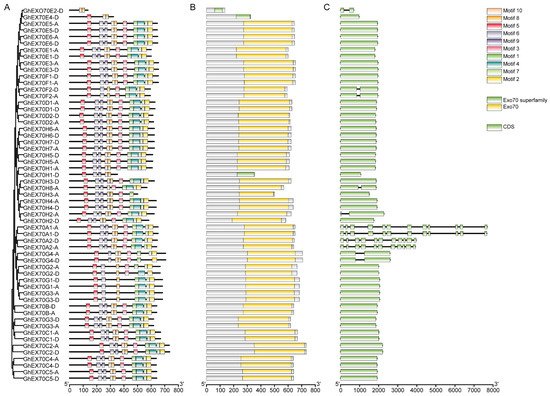
Figure 3. EXO70 family motif, domain, and gene structure in upland cotton. (A) Phylogenetic tree and motif of GhEXO70 proteins. (B) The conserved domains in GhEXO70 proteins. (C) Gene structure of the GhEXO70 family.
We used MEME online software to analyze the conserved motifs in the GhEXO70 protein and study its motif composition diversity and conservation. Figure 3 shows that 10 motifs (1–10) were identified, and each one was localized mainly to the C-terminal of the gene. Therefore, the C-terminal sequence of the GhEXO70 protein is highly conserved. The motif types revealed that the GhEXO70 gene members in subgroups A, B, C, and D were highly conserved and included all motifs. The GhEXO70s gene members in the other subgroups presented with obvious differences in motif type distribution, and some of them were lost. GhEXO70E2-D, GhEXO70E4-D, GhEXO70H1-D, and GhEXO70G2-D contained two, three, five, and six motifs, respectively. The functions of Motifs 1–10 have not been elucidated. Nevertheless, analysis of the conserved domains via the NCBI Conserved Domain Database (CCD) disclosed that they comprise the Exo70 domain (Figure 3).
The PFam03081 domain at the C-terminus of the EXO70 protein is characteristic of the EXO70 superfamily [12], and all 165 predicted homologous clone EXO70 proteins possess it. However, the amino acid sequence lengths differed among EXO70 proteins and were in the range of 134–735 aa (average length = 618.736842105263 aa) (Table 1). It was discovered that most GhEXO70 genes lacked a transmembrane (TM) structure. Only GhEXO70E2-D might possess a transmembrane region. Therefore, it may have evolved along with eukaryote evolution. For the prediction of the transmembrane domain of Arabidopsis EXO70, the results showed that AtEXO70C1, AtEXO70C2, AtEXO70H5, AtEXO70H8, and AtEXO70A3 have transmembrane domains, but they are not obvious, and the other EXO70s have no transmembrane domains. The prediction results of rice EXO70 show that OsEXO70A3, OsEXO70A4, OsEXO70H1a, OsEXO70H1b, OsEXO70H2, OsEXO70H3, OsEXO70H4, OsEXO70I3, OsEXO70I4, OsEXO70L1, OsEXO6K1, OsEXO70L, OsEXO70J1, OsEXO70J1, OsEXO70J2, OsEXO70J6, OsEXO70J8, OsEXO70K1, OsEXO7K2, and OsEXO70L1 have a transmembrane domain. In addition, OsEXO70A4 has a more obvious transmembrane domain at the C-terminus, and none of the other rice EXO70s has a transmembrane domain. Among the prediction results of the transmembrane domain of cotton EXO70, only GhEXO70E2-D has a transmembrane domain, and the others have no transmembrane domain. Both Arabidopsis and cotton contain fewer EXO70s with transmembrane domains. As rice is a monocot, it may be evolving to have more EXO70, and there are more EXO70s with transmembrane domains.
5. Analysis of EXO70 Gene Expression Patterns in G. arboretum and G. hirsutum
Gene expression has spatiotemporal properties. The expression patterns of the various members of the EXO70 gene family may indicate the potential biological effects of these genes. We analyzed expression profile data in the CottonFGD and Cottongen (https://www.cottongen.org/, accessed on 22 March 2021) databases to clarify the spatiotemporal expression characteristics of the EXO70 gene. In G. hirsutum and G. arboretum, the EXO70 gene is commonly expressed in the roots, stems, leaves, flowers, fibers, and ovules and has spatiotemporal properties (Figure 4). GhEXO70A1-A, GhEXO70A1-D, GhEXO70B-A, GhEXO70B-D, GhEXO70D1-A, GhEXO70E1-A, GhEXO70E6-A, GhEXO70F2-D, and other genes in G. hirsutum are generally expressed at high levels and in various tissues. The GhEXO70H3-A gene is expressed mainly in the stamens, whereas the GhEXO70H5-A and GhEXO70H5-D genes are expressed mainly during the early stages of ovule development. In G. arboretum, the GaEXO70A1, GaEXO70E4, GaEXO70B, GaEXO70F1, GaEXO70F2, GaEXO70D1, GaEXO70E1 genes are generally highly expressed in different tissues, while GaEXO70A2 is expressed mainly in the 15D fibers.
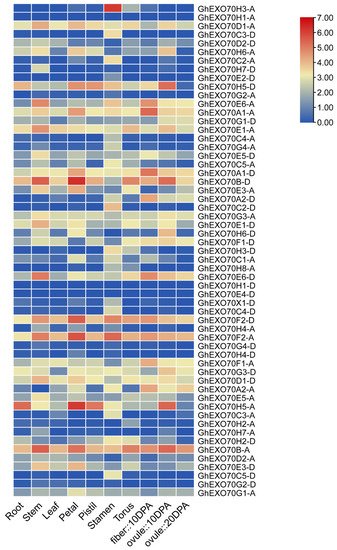
Figure 4. EXO70 family gene expression patterns in different tissues and organs of upland cotton.
Ubiquitous EXO70 expression suggests that this gene is implicated in cotton growth and development. The GaEXO70A2 gene is expressed mainly in the fibers and might participate in cotton fiber development. The GhEXO70H3-A gene is expressed mainly in the stamens and may be associated with cotton fertility. The GhEXO70H5-A and GhEXO70H5-D genes are expressed mainly in the early stages of ovule development and could be involved in cotton seed formation.
6. EXO70 Gene Transcription Regulation Analysis
Spatiotemporal gene expression is regulated mainly by transcription factors (TFs) and epigenetics [36]. The observed differences in spatiotemporal expression of the various EXO70 genes may be related to their promoter specificity. We intercepted the 2-kb sequence upstream of the cotton EXO70 gene start codon and used the PlantCARE database (http://bioinformatics.psb.ugent.be/webtools/plantcare/html/, accessed on 20 April 2021) to analyze the cis-elements in the promoter region. A total of 1081 cis-elements were predicted in the 57 GhEXO70 gene promoter regions. Of these, 10 and 11 categories were related to phytohormones and environmental stressors, respectively. The functions of the cis-elements in phytohormone and environmental stress response are highlighted in Figure 5. Among the predicted phytohormone response elements, the ERE, ABRE, and CGTCA motifs were the most abundant. Hence, the GhEXO70 gene might respond to ethylene, abscisic acid, and methyl jasmonate (MeJA) (Figure 5A). Ten environmental stress-related elements were identified and mainly involved drought stress (MYC), stress response (STRE), and anaerobic induction (ARE) (Figure 5B). Therefore, the EXO70 gene may participate in the response to adversity. To further verify whether the above cis-acting elements are unique to cotton EXO70s, we also analyzed the EXO70s gene promoters in Arabidopsis and rice. The results indicate that the promoters of EXO70 genes in Arabidopsis and rice also contain cis-acting elements that respond to environmental stress and plant hormones. It shows that this phenomenon is not unique to cotton EXO70, but is conserved in plants.
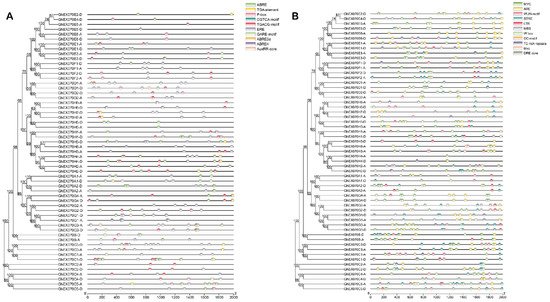
Figure 5. Cis-acting elements in GhEXO70 promoter. (A) Cis-elements involved in phytohormones were predicted. ABRE: cis-acting regulatory element involved in abscisic acid response. AuxRR-core: cis-acting regulatory element involved in auxin response. CGTCA-motif: cis-acting regulatory element involved in methyl jasmonate (MeJA) response. GARE-motif: gibberellin response element. TGACG-motif: cis-acting regulatory element involved in MeJA response. TGA-element: auxin response element. ERE: cis-acting ethylene response element. P-box: gibberellin response element. (B) Predicted cis-elements involved in environmental stress response. GC-motif: enhancer-like elements involved in specific hypoxia induction. LTR: cis-acting elements involved in low temperature response. MBS: MYB binding sites related to drought induction. STRE: stress response elements. TC-rich repetitive sequences: cis-acting elements involved in defense and stress responses. WUN-motif: wound response elements. MYC: cis-acting elements involved in drought stress. W box: cis-acting elements involved in sugar metabolism and plant defense signals. DRE core: dehydration response element. ARE: cis-acting regulatory element for anaerobic induction.
7. Expression Analysis and Subcellular Location of GhEXO70A1-A
The EXO70A1 gene is the most widely studied of all plant EXO70 genes. In Arabidopsis, AtEXO70A1 differentiates tubular molecules and regulates seed coat, root hair, stigma papillae development, and Kjeldahl band formation [37][38][39]. OsEXO70A1 plays important roles in vascular bundle differentiation and mineral nutrient assimilation [28]. In this study, we used GhEXO70A1-A in an experimental study on cotton EXO70 genes. We tested the GhEXO70A1-A gene expression patterns. GhEXO70A1-A was predominantly expressed in the stems, leaves, and flowers but its expression levels were low in the roots, ovules, and cotyledons (Figure 6A).
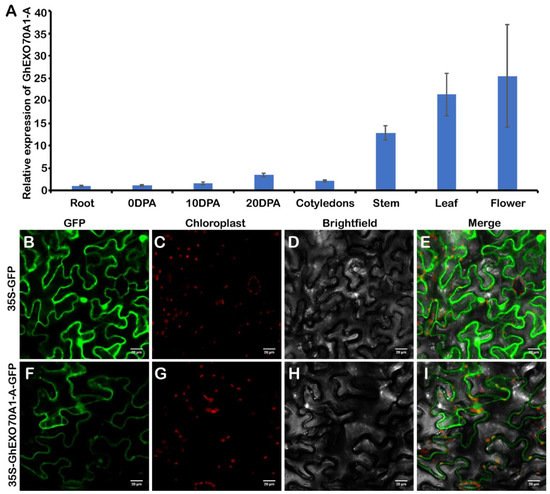
Figure 6. GhEXO70A1-A expression and subcellular localization analyses. (A) GhEXO70A1-A expression analyses in various upland cotton tissues. (B–I) Subcellular GhEXO70A1-A localization in tobacco. GFP: green fluorescence. Chloroplast: chloroplast spontaneous red fluorescence. Merge: green and red fluorescence and bright field fusion. (B–E): 35S-GFP empty vector as control. (F–I): 35S-GhEXO70A1-A-GFP vector located in plasma membrane. Bar: 10 μm. Data are means ± SD for three replicates.
Subcellular GhEXO70A1-A protein localization predicted its roles in biological processes. Transient 35S-GhEXO70A1-A-GFP expression in tobacco produced a fluorescent signal. GhEXO70A1-A induced signals on the plasma membrane (Figure 6B). Thus, GhEXO70A1-A was localized to the endomembrane system. This discovery was consistent with the roles of EXO70s in vesicle transport.
8. GhEXO70A1-A Protein Interaction Analysis
We used a yeast two-hybrid (Y2H) assay to explore the interactions among GhEXO70A1-A and the other subunits of the exocytosis complex. Plasmids containing GhEXO70A1-A and the other subunits of the exocytosis complex were co-transformed into Y2H Gold cells, which can grow on SD/-Leu-Trp. However, the cells were inoculated onto SD/-Ade/-His/-Leu/-Trp medium and only GhEXO70A1-A and GhEXO84A, Gh EXO84B, GhEXO84C co-transformed cells could grow on it and express X-α-Gal activity. GhEXO70A1-A interacted with EXO84A, EXO84B, and EXO84C (Figure 7), which means that it may function as a subunit of the exocytosis complex.
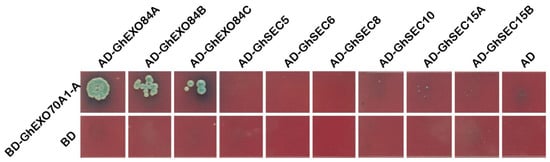
Figure 7. Y2H analysis of interactions among GhEXO70A1-A and other exocyst subunits. GhEXO70A1-A is connected to PGBK-T7 carrier. Other subunits of secretory complex are connected to PGAD-T7 carrier. BD: PGBK-T7 empty vector. AD: PGAD-T7 empty vector.
9. VIGS Silencing of GhEXO70A1-A Causes Changes in Signaling Pathways and Gene Expression
Gene silencing is an effective method of studying gene function. To explore the functions of GhEXO70A1-A in cotton, we constructed GhEXO70A1-A-gene-silenced cotton plants by virus-induced gene silencing (VIGS). qPCR demonstrated that the GhEXO70A1-A gene was successfully knocked down (Figure 8A). We then used next-generation sequencing (NGS) technology to detect any changes in the transcriptome of GhEXO70A1-A-silenced leaves. However, except for GhEXO70C1-A, GhEXO70H6-D, and GhEXO70H6-A, which decreased to 32.1%, 46.9%, and 56.6% of the control, all other genes fell to more than 60% of the control, and the fold increase was also less than 1. Although the three genes GhEXO70C1-A, GhEXO70H6-D, and GhEXO70H6-A declined slightly, their expression abundance was also very low. The above results show that the knockdown of GhEXO70A1-A by VIGS does affect the expression of other EXO70 genes, but the effect is not significant after analysis. The changes in differential genes should be mainly caused by the changes in GhEXO70A1-A.
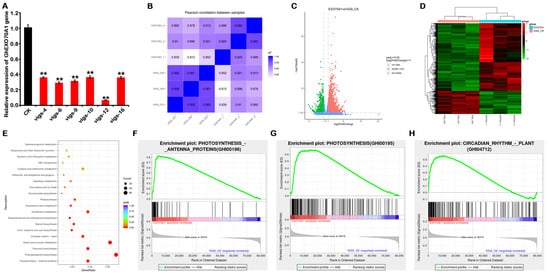
Figure 8. Transcriptome sequencing of differences in cotton transcriptome expression after GhEXO70A1-A gene silencing. (A) GhEXO70A1-A expression levels after virus-induced gene silencing (VIGS). Relative GhEXO70A1-A expression levels in plants numbered 4, 6, 9, 10, 12, and 16 significantly decreased. (B) Correlation analyses of transcriptome samples. (C) Differential gene volcano map in transcriptome. (D) Differential gene heat map in transcriptome. (E) KEGG functional enrichment dot plot of DEGs. (F) GSEA diagram showing changes in photosynthesis antenna proteins after EXO70A1 gene silencing. (G) GSEA diagram showing changes in photosynthetic pathway after EXO70A1 gene silencing. (H) GSEA diagram showing changes in circadian rhythm pathway after EXO70A1 gene silencing. Data are means of three replicates ± SD. ** p < 0.01.
Correlation analyses among samples disclosed significant differences between the GhEXO70A1-A-silenced (EXO70A1) and the control (VIGS-CK) groups (Figure 8B). Thus, GhEXO70A1-A silencing in cotton altered the gene expression profiles. Differentially expressed genes (DEG) were those that met the criteria of |log2(Fold Change)| ≥ 1 and p ≤ 0.05. A total of 3264 upregulated and 1103 downregulated genes were screened, as shown in a volcano graph (Figure 8C) and a heat map (Figure 8D). Kyoto Encyclopedia of Genes and Genomes (KEGG) functional enrichment of the DEGs (Figure 8E) displayed 13 pathways with p < 0.01. These included photosynthesis antenna protein, phenylpropane biosynthesis, flavonoid biosynthesis, starch and sucrose metabolism, circadian rhythm—plant, keratin, cork and wax biosynthesis, steroid biosynthesis, sesquiterpenoid and triterpenoid biosynthesis, glutathione metabolism, cyano-amino acid metabolism, photosynthesis, and glucosinolate biosynthesis (Table 4). GSEA results showed that GhEXO70A1-A was significantly related to photosynthesis antenna protein, photosynthesis, and circadian rhythm—plants (Figure 8F–H). Of the 13 significantly different pathways, all except for circadian rhythm—plants were related to metabolism. Therefore, cotton leaf GhEXO70A1-A may regulate biochemical anabolism and catabolism.
Table 4. DEG function pathway enrichment.
| KEGG ID | Description | Gene Ratio | Bg Ratio | p Value | Up | Down |
|---|---|---|---|---|---|---|
| ghi00196 | Photosynthesis antenna proteins | 33/877 | 53/11,853 | 1.32 × 10−24 | 33 | 0 |
| ghi00940 | Phenylpropanoid biosynthesis | 61/877 | 285/11,853 | 1.91 × 10−14 | 50 | 11 |
| ghi00941 | Flavonoid biosynthesis | 33/877 | 101/11,853 | 9.05 × 10−14 | 32 | 1 |
| ghi00500 | Starch and sucrose metabolism | 55/877 | 324/11,853 | 4.27 × 10−9 | 45 | 10 |
| ghi04712 | Circadian rhythm—plant | 28/877 | 127/11,853 | 1.26 × 10−7 | 25 | 3 |
| ghi00073 | Cutin, suberine, and wax biosynthesis | 17/877 | 64/11,853 | 2.56 × 10−6 | 15 | 2 |
| ghi00100 | Steroid biosynthesis | 19/877 | 84/11,853 | 8.89 × 10−6 | 18 | 1 |
| ghi00909 | Sesquiterpenoid and triterpenoid biosynthesis | 15/877 | 58/11,853 | 1.41 × 10−5 | 11 | 4 |
| ghi00480 | Glutathione metabolism | 33/877 | 249/11,853 | 0.00076 | 20 | 13 |
| ghi00460 | Cyanoamino acid metabolism | 16/877 | 91/11,853 | 0.00095 | 16 | 0 |
| ghi00195 | Photosynthesis | 20/877 | 138/11,853 | 0.002806 | 20 | 0 |
| ghi00966 | Glucosinolate biosynthesis | 7/877 | 29/11,853 | 0.004374 | 1 | 6 |
| ghi00670 | One carbon pool by folate | 10/877 | 56/11,853 | 0.007353 | 10 | 0 |
References
- Bonifacino, J.S.; Glick, B.S. The mechanisms of vesicle budding and fusion. Cell 2004, 116, 153–166.
- Whyte, J.R.; Munro, S. Vesicle tethering complexes in membrane traffic. J. Cell Sci. 2002, 115, 2627–2637.
- Munson, M.; Novick, P. The exocyst defrocked, a framework of rods revealed. Nat. Struct. Mol. Biol. 2006, 13, 577–581.
- Hsu, S.C.; Ting, A.E.; Hazuka, C.D.; Davanger, S.; Kenny, J.W.; Kee, Y.; Scheller, R.H. The mammalian brain rsec6/8 complex. Neuron 1996, 17, 1209–1219.
- TerBush, D.R.; Maurice, T.; Roth, D.; Novick, P. The Exocyst is a multiprotein complex required for exocytosis in Saccharomyces cerevisiae. EMBO J. 1996, 15, 6483–6494.
- He, B.; Guo, W. The exocyst complex in polarized exocytosis. Curr. Opin. Cell Biol. 2009, 21, 537–542.
- Sivaram, M.V.; Saporita, J.A.; Furgason, M.L.; Boettcher, A.J.; Munson, M. Dimerization of the exocyst protein Sec6p and its interaction with the t-SNARE Sec9p. Biochemistry 2005, 44, 6302–6311.
- Wu, H.; Turner, C.; Gardner, J.; Temple, B.; Brennwald, P. The Exo70 subunit of the exocyst is an effector for both Cdc42 and Rho3 function in polarized exocytosis. Mol. Biol. Cell 2010, 21, 430–442.
- Elias, M.; Drdova, E.; Ziak, D.; Bavlnka, B.; Hala, M.; Cvrckova, F.; Soukupova, H.; Zarsky, V. The exocyst complex in plants. Cell. Biol. Int. 2003, 27, 199–201.
- Synek, L.; Schlager, N.; Elias, M.; Quentin, M.; Hauser, M.T.; Zarsky, V. AtEXO70A1, a member of a family of putative exocyst subunits specifically expanded in land plants, is important for polar growth and plant development. Plant J. 2006, 48, 54–72.
- Cvrckova, F.; Grunt, M.; Bezvoda, R.; Hala, M.; Kulich, I.; Rawat, A.; Zarsky, V. Evolution of the land plant exocyst complexes. Front. Plant Sci. 2012, 3, 159.
- Li, S.; van Os, G.M.; Ren, S.; Yu, D.; Ketelaar, T.; Emons, A.M.; Liu, C.M. Expression and functional analyses of EXO70 genes in Arabidopsis implicate their roles in regulating cell type-specific exocytosis. Plant Physiol. 2010, 154, 1819–1830.
- Zuo, X.; Zhang, J.; Zhang, Y.; Hsu, S.C.; Zhou, D.; Guo, W. Exo70 interacts with the Arp2/3 complex and regulates cell migration. Nat. Cell Biol. 2006, 8, 1383–1388.
- Dupraz, S.; Grassi, D.; Bernis, M.E.; Sosa, L.; Bisbal, M.; Gastaldi, L.; Jausoro, I.; Caceres, A.; Pfenninger, K.H.; Quiroga, S. The TC10-Exo70 complex is essential for membrane expansion and axonal specification in developing neurons. J. Neurosci. 2009, 29, 13292–13301.
- Xiong, X.; Xu, Q.; Huang, Y.; Singh, R.D.; Anderson, R.; Leof, E.; Hu, J.; Ling, K. An association between type Igamma PI4P 5-kinase and Exo70 directs E-cadherin clustering and epithelial polarization. Mol. Biol. Cell 2012, 23, 87–98.
- Zhao, Y.; Liu, J.; Yang, C.; Capraro, B.R.; Baumgart, T.; Bradley, R.P.; Ramakrishnan, N.; Xu, X.; Radhakrishnan, R.; Svitkina, T.; et al. Exo70 generates membrane curvature for morphogenesis and cell migration. Dev. Cell 2013, 26, 266–278.
- Fujita, A.; Koinuma, S.; Yasuda, S.; Nagai, H.; Kamiguchi, H.; Wada, N.; Nakamura, T. GTP hydrolysis of TC10 promotes neurite outgrowth through exocytic fusion of Rab11- and L1-containing vesicles by releasing exocyst component Exo70. PLoS ONE 2013, 8, e79689.
- Fendrych, M.; Synek, L.; Pecenkova, T.; Toupalova, H.; Cole, R.; Drdova, E.; Nebesarova, J.; Sedinova, M.; Hala, M.; Fowler, J.E.; et al. The Arabidopsis exocyst complex is involved in cytokinesis and cell plate maturation. Plant Cell 2010, 22, 3053–3065.
- Pecenkova, T.; Hala, M.; Kulich, I.; Kocourkova, D.; Drdova, E.; Fendrych, M.; Toupalova, H.; Zarsky, V. The role for the exocyst complex subunits Exo70B2 and Exo70H1 in the plant-pathogen interaction. J. Exp. Bot. 2011, 62, 2107–2116.
- Kulich, I.; Pecenkova, T.; Sekeres, J.; Smetana, O.; Fendrych, M.; Foissner, I.; Hoftberger, M.; Zarsky, V. Arabidopsis exocyst subcomplex containing subunit EXO70B1 is involved in autophagy-related transport to the vacuole. Traffic 2013, 14, 1155–1165.
- Zarsky, V.; Kulich, I.; Fendrych, M.; Pecenkova, T. Exocyst complexes multiple functions in plant cells secretory pathways. Curr. Opin. Plant Biol. 2013, 16, 726–733.
- Synek, L.; Vukasinovic, N.; Kulich, I.; Hala, M.; Aldorfova, K.; Fendrych, M.; Zarsky, V. EXO70C2 Is a Key Regulatory Factor for Optimal Tip Growth of Pollen. Plant Physiol. 2017, 174, 223–240.
- Kulich, I.; Vojtikova, Z.; Sabol, P.; Ortmannova, J.; Nedela, V.; Tihlarikova, E.; Zarsky, V. Exocyst Subunit EXO70H4 Has a Specific Role in Callose Synthase Secretion and Silica Accumulation. Plant Physiol. 2018, 176, 2040–2051.
- Hong, D.; Jeon, B.W.; Kim, S.Y.; Hwang, J.U.; Lee, Y. The ROP2-RIC7 pathway negatively regulates light-induced stomatal opening by inhibiting exocyst subunit Exo70B1 in Arabidopsis. New Phytol. 2016, 209, 624–635.
- Wang, W.; Liu, N.; Gao, C.; Cai, H.; Romeis, T.; Tang, D. The Arabidopsis exocyst subunits EXO70B1 and EXO70B2 regulate FLS2 homeostasis at the plasma membrane. New Phytol. 2020, 227, 529–544.
- Acheampong, A.K.; Shanks, C.; Cheng, C.; Schaller, G.E.; Dagdas, Y.; Kieber, J.J. EXO70D isoforms mediate selective autophagic degradation of type-A ARR proteins to regulate cytokinin sensitivity. Proc. Natl. Acad. Sci. USA 2020, 117, 27034–27043.
- Li, S.; Chen, M.; Yu, D.; Ren, S.; Sun, S.; Liu, L.; Ketelaar, T.; Emons, A.M.; Liu, C.M. EXO70A1-mediated vesicle trafficking is critical for tracheary element development in Arabidopsis. Plant Cell 2013, 25, 1774–1786.
- Tu, B.; Hu, L.; Chen, W.; Li, T.; Hu, B.; Zheng, L.; Lv, Z.; You, S.; Wang, Y.; Ma, B.; et al. Disruption of OsEXO70A1 Causes Irregular Vascular Bundles and Perturbs Mineral Nutrient Assimilation in Rice. Sci. Rep. 2015, 5, 18609.
- Xing, Y.; Wang, N.; Zhang, T.; Zhang, Q.; Du, D.; Chen, X.; Lu, X.; Zhang, Y.; Zhu, M.; Liu, M.; et al. SHORT-ROOT 1 is critical to cell division and tracheary element development in rice roots. Plant J. 2021, 105, 1179–1191.
- Dowd, K.M.; Pelitire, S.M.; Delhom, C.D. Molecular biology and physiology: Seed-fiber ratio, seed index, and seed tissue and compositional properties of current cotton cultivars. J. Cotton Sci. 2018, 22, 60–74.
- Yu, Q.; Hu, Z.; Shen, Y.; Jiang, Y.; Pan, P.; Hou, T.; Pan, Z.Q.; Huang, J.; Sun, Y. Gossypol inhibits cullin neddylation by targeting SAG-CUL5 and RBX1-CUL1 complexes. Neoplasia 2020, 22, 179–191.
- Ding, Y.P.; Ge, X.Y.; Wang, P.; Wu, J.; Wang, S.F.; Li, F.G. Functional analysis of cotton salt stress response gene GhEXO70B1. J. Cotton Sci. 2018, 30, 4–15.
- Chong, Y.T.; Gidda, S.K.; Sanford, C.; Parkinson, J.; Mullen, R.T.; Goring, D.R. Characterization of the Arabidopsis thaliana exocyst complex gene families by phylogenetic, expression profiling, and subcellular localization studies. New Phytol. 2010, 185, 401–419.
- Yang, K.; Zhang, Y.; Lv, J.; Zhao, Y.B.; Zhang, H.C.; Hang, X.; He, G. EXO70 doubling in Arabidopsis and rice genomes. Chin. Sci. Bull. 2015, 1, 38–51.
- Zhao, J.; Zhang, X.; Wan, W.; Zhang, H.; Liu, J.; Li, M.; Wang, H.; Xiao, J.; Wang, X. Identification and Characterization of the EXO70 Gene Family in Polyploid Wheat and Related Species. Int. J. Mol. Sci. 2019, 20, 60.
- Gaharwar, A.K.; Singh, I.; Khademhosseini, A. Engineered biomaterials for in situ tissue regeneration. Nat. Rev. Mater. 2020, 5, 686–705.
- Kulich, I.; Cole, R.; Drdová, E.; Cvrcková, F.; Soukup, A.; Fowler, J.; Zárský, V. Arabidopsis exocyst subunits SEC8 and EXO70A1 and exocyst interactor ROH1 are involved in the localized deposition of seed coat pectin. New Phytol. 2010, 188, 615–625.
- Kalmbach, L.; Hématy, K.; De Bellis, D.; Barberon, M.; Fujita, S.; Ursache, R.; Daraspe, J.; Geldner, N. Transient cell-specific EXO70A1 activity in the CASP domain and Casparian strip localization. Nat. Plants 2017, 3, 17058.
- Vukašinović, N.; Oda, Y.; Pejchar, P.; Synek, L.; Pečenková, T.; Rawat, A.; Sekereš, J.; Potocký, M.; Žárský, V. Microtubule-dependent targeting of the exocyst complex is necessary for xylem development in Arabidopsis. New Phytol. 2017, 213, 1052–1067.
More
Information
Subjects:
Agriculture, Dairy & Animal Science
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.1K
Revisions:
2 times
(View History)
Update Date:
27 Oct 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





