Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Ahmad Gholami | + 1148 word(s) | 1148 | 2021-09-27 05:36:36 | | | |
| 2 | Conner Chen | Meta information modification | 1148 | 2021-10-09 03:34:04 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Gholami, A. Bioactive Agent-Loaded Electrospun Nanofiber Membranes. Encyclopedia. Available online: https://encyclopedia.pub/entry/14900 (accessed on 27 March 2026).
Gholami A. Bioactive Agent-Loaded Electrospun Nanofiber Membranes. Encyclopedia. Available at: https://encyclopedia.pub/entry/14900. Accessed March 27, 2026.
Gholami, Ahmad. "Bioactive Agent-Loaded Electrospun Nanofiber Membranes" Encyclopedia, https://encyclopedia.pub/entry/14900 (accessed March 27, 2026).
Gholami, A. (2021, October 08). Bioactive Agent-Loaded Electrospun Nanofiber Membranes. In Encyclopedia. https://encyclopedia.pub/entry/14900
Gholami, Ahmad. "Bioactive Agent-Loaded Electrospun Nanofiber Membranes." Encyclopedia. Web. 08 October, 2021.
Copy Citation
Despite the advances that have been achieved in developing wound dressings to date, wound healing still remains a challenge in the healthcare system. None of the wound dressings currently used clinically can mimic all the properties of normal and healthy skin. Electrospinning has gained remarkable attention in wound healing applications because of its excellent ability to form nanostructures similar to natural extracellular matrix (ECM). Electrospun dressing accelerates the wound healing process by transferring drugs or active agents to the wound site sooner.
wound dressing
bioactive molecules
wound healing
electrospinning
tissue engineering
1. Introduction
Skin plays an essential role in protecting our body against any physical and chemical injury and water loss, and contributes to the maintenance of bodily homeostasis [1][2][3]. After damage, the skin should restore its integrity to maintain its functions. Wound healing is a vital and complex physiological process consisting of the collaboration of many cell strains and their products [4]. In this process, the growth factors induce cell proliferation, which leads to a combination of several dynamic changes, including soluble mediators, blood cells, the production of the extracellular matrix, and the proliferation of parenchymal cells [5][6]. In healthy skin, the outer layer is the epidermis, which is impermeable and resists the external environment. The dermis, the inner layer of skin, is full of extracellular matrix (ECM), mechanoreceptors, and vasculature, and this layer can also provide the skin with immunity, nutrients, and mechanical strength [6][7].
In general, a wound is a type of injury that involves damage to the epithelial tissue and causes a lack of skin integrity. Wound healing is a natural physiological response to tissue damage. In general, wound healing is performed with four specific stages of hemostasis, inflammation, proliferation, and maturation (Figure 1) [8][9][10]. ECM plays a crucial role in every step of the wound healing process. The ECM acts as a supporting scaffold, which is necessary at all stages of the repair phenomenon [11]. The role of ECM is not only in the activity of cytokines and signaling within cellulitis, but also in mechanical support, which is helpful for activating and regulating the pathways of cell differentiation and proliferation [12][13].
Successful tissue regeneration depends on the complex function and fibrous form of native ECM stimulated by skin substitutes or wound dressing membranes [14]. Various biocompatible and biodegradable scaffolds have been created with three-dimensional cell proliferation as a suitable dressing membrane for wound healing [15][16]. Extensive studies in the field of tissue engineering have been performed to define tissue engineering as described in the workshop of the National Science Foundation, and the role of scaffolds in maintaining and improving tissue function and reconstruction is undeniable [17].
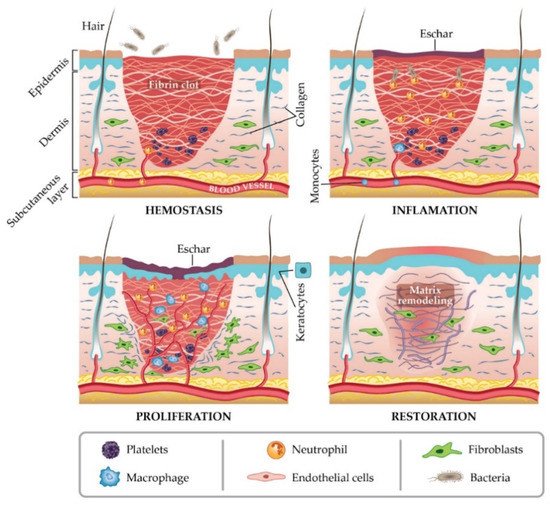
Figure 1. Stages of wound healing. Adapted from I. Negut, V. Grumezescu, A.M. Grumezescu, Treatment strategies for infected wounds, adapted from [18].
Electrospinning is a simple and effective technique that uses electric force to produce nano-/micro-scale continuous fibers made of a polymer solution Figure 2A. The nanofibrous membrane has impressive advantages as skin scaffolds compared to conventional and non-fibrous structures such as hydrogels, due to their similarity to native ECM of skin tissue, so they can be considered as potential scaffolds providing cell adhesion and proliferation, and eventually induce neodermis regeneration [19][20][21].
To date, the electrospinning method is the most practical technique in the field of advanced scaffolds for tissue engineering and delivery of bioactive agents to damaged tissue, which has attracted the attention of researchers [8]. One of the most practical electrospinning methods in tissue engineering is skin regeneration [22][23][24][25][26][27]. Synthetic polymers are not inherently bioactive, and by combining with some active components, the bioactivity of synthetic polymers can be improved [28]. The electrospinning technique allows the production of a suitable nanofibrous membrane to deliver bioactive agents or pharmacological agents to the damaged site of skin to accelerate the skin regeneration process [14][29]. Bioactive incorporation into electrospun nanofiber membrane could be done in four different ways represented in Figure 2B–E.
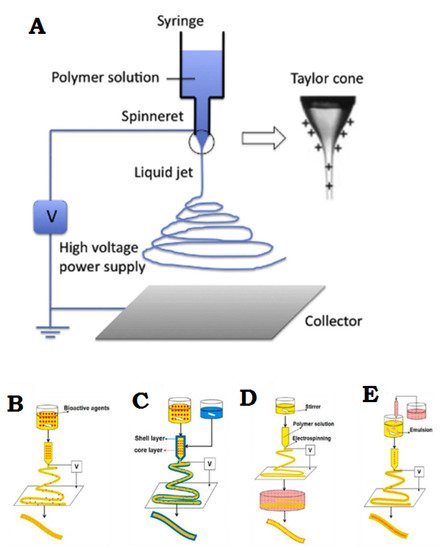
Figure 2. (A) General flowchart of the basic electrospinning process. (B) Adjusting the electrospinning mixture. (C) Set up of shell electrospinning. (D) Surface functional electrospinning settings. (E) Emulsion-based electrospinning operation. (A) is reprinted from [30]; (B–E) are reprinted from [31] Licensed by the American Chemical Society.
2. New Advances in Wound Dressing of Bioactive Nanofibers
A new generation of wound dressings with the ability to quickly accelerate the wound healing process is nanofiber membranes with active materials. The production of this type of membrane can be performed using any kind of electrospun solution. In general, a biologically active factor can be considered to play an active role in the recovery process. Bioactive agents include antibacterial particles, growth factors, stem cells, vitamins, and other factors that are effective in wound healing in combination with nanofiber membranes.
In a new study, for the first time, a hybrid nanofiber membrane was proposed as a bioactive wound dressing by Homaygohar et al. The results of this study showed that biohybrid nanofibers have elastic modulus and elongation (300% and 58%, respectively) comparable to regular poly(ε-caprolactone) (PCL) nanofibers. Survival of WST-8 cells, and NIH3T3 fibroblasts proliferating correctly and interacting with biohybrids were observed in the experimental results. It was also concluded from SEM images that cells can adhere to such nanofibers even better than pure PCL cells. As a result, sufficient traction and mineralization through this BSA/PCL nanofiber biohybrid membrane can accelerate the wound healing process and be considered a suitable option for wound dressing applications [28].
In another study, Cerium-doped bioactive glass (Ce-BG)-loaded chitosan/polyethylene oxide (PEO) nanofibrous scaffolds were fabricated as antibacterial wound dressing. Chitosan/PEO scaffolds had been modified to overcome the instability and mechanical properties problems of these scaffolds in aqueous solutions. Numerous parameters including mechanical properties, antibacterial activity against Gram-positive and Gram-negative bacterial cells, ion release behavior, and swelling properties were studied in this research. This study shows that increasing the Ce-BG/CH (w/w) ratio up to 20% (w/w) can increase the swelling and mechanical properties of scaffolding. By further increasing the ratio, the effects of Ce-BG affinity on the above are reduced. In addition, chitosan/PEO scaffolds containing Ce-BG had mechanical properties very close to those of the skin, and the results showed that they were only 20% shorter than the required length when breaking skin tissue scaffolds, which increased in length by 28.6% when broken [32].
Another interesting study was designed and conducted using Lemon Oil (LMEO) as a natural and highly antibacterial bioactive agent for making electrospun cellulose acetate (CA) nanofiber membranes by Bekzadeh et al. The chemical, antibacterial, morphological, thermal and optical properties of LMEO-loaded CA nanofiber scaffolds were investigated.
The degradation profile of CA with LMEO composition showed no change in the results of the calorimetric analysis. LMEO-loaded CA electrospun nanofibers 100% eliminated Escherichia coli and Staphylococcus aureus in the antibacterial evaluation, even at the lowest LMEO loading concentration. LMEO electrospinning fiber membranes show that nanofibers can retain their antibacterial properties even after two months of storage. Consequently, this nanofiber structure may be a potential candidate as a bioactive wound dressing with long-lasting antibacterial activity [33].
References
- Joodaki, H.; Panzer, M.B. Skin mechanical properties and modeling: A review. Proc. Inst. Mech. Eng. Part. H J. Eng. Med. 2018, 232, 323–343.
- Mousavi, S.; Zarei, M.; Hashemi, S.; Ramakrishna, S.; Chiang, W.-H.; Lai, C.; Gholami, A.; Omidifar, N.; Shokripour, M. Asymmetric Membranes: A Potential Scaffold for Wound Healing Applications. Symmetry 2020, 12, 1100.
- Nejati, R.; Kovacic, D.; Slominski, A. Neuro-immune-endocrine functions of the skin: An overview. Expert Rev. Dermatol. 2013, 8, 581–583.
- Shaw, T.; Martin, P. Wound repair at a glance. J. Cell Sci. 2009, 122, 3209–3213.
- Gonzalez, A.C.D.O.; Costa, T.F.; Andrade, Z.D.A.; Medrado, A.R.A.P. Wound healing-A literature review. An. Bras. Dermatol. 2016, 91, 614–620.
- Rodrigues, M.; Kosaric, N.; Bonham, C.A.; Gurtner, G.C. Wound Healing: A Cellular Perspective. Physiol. Rev. 2019, 99, 665–706.
- Cañedo-Dorantes, L.; Cañedo-Ayala, M. Skin Acute Wound Healing: A Comprehensive Review. Int. J. Inflamm. 2019, 2019, 3706315.
- Afsharian, Y.P.; Rahimnejad, M. Bioactive electrospun scaffolds for wound healing applications: A comprehensive review. Polym. Test. 2020, 93, 106952.
- Alam, W.; Hasson, J.; Reed, M. Clinical approach to chronic wound management in older adults. J. Am. Geriatr. Soc. 2021, 69, 2327–2334.
- Toleubayev, M.; Dmitriyeva, M.; Kozhakhmetov, S.; Igissinov, N.; Turebayev, M.; Omarbekov, A.; Adaibayev, K.; Shakenov, A.; Izimbergenov, M. Regenerative Properties of Recombinant Human Erythropoietin in the Wound Healing. Open Access Maced. J. Med. Sci. 2021, 9, 113–117.
- Olczyk, P.; Mencner, Ł.; Komosińska-Vassev, K. The Role of the Extracellular Matrix Components in Cutaneous Wound Healing. BioMed Res. Int. 2014, 2014, 747584.
- Caldeira, J.; Sousa, A.; Sousa, D.; Barros, D. Extracellular matrix constitution and function for tissue regeneration and repair. In Peptides and Proteins as Biomaterials for Tissue Regeneration and Repair; Elsevier: Amsterdam, The Netherlands, 2018; pp. 29–72.
- Ghorbani, F.; Sahranavard, M.; Nejad, Z.M.; Li, D.; Zamanian, A.; Yu, B. Surface Functionalization of Three Dimensional-Printed Polycaprolactone-Bioactive Glass Scaffolds by Grafting GelMA Under UV Irradiation. Front. Mater. 2020, 7, 348.
- Kalva, S.N.; Augustine, R.; Al Mamun, A.; Dalvi, Y.B.; Vijay, N.; Hasan, A. Active agents loaded extracellular matrix mimetic electrospun membranes for wound healing applications. J. Drug Deliv. Sci. Technol. 2021, 63, 102500.
- EzEldeen, M.; Loos, J.; Nejad, Z.M.; Cristaldi, M.; Murgia, D.; Braem, A.; Jacobs, R. 3D-printing-assisted fabrication of chitosan scaffolds from different sources and cross-linkers for dental tissue engineering. Eur. Cells Mater. 2021, 41, 485–501.
- Norouzi, M.; Boroujeni, S.M.; Omidvarkordshouli, N.; Soleimani, M. Advances in Skin Regeneration: Application of Electrospun Scaffolds. Adv. Healthc. Mater. 2015, 4, 1114–1133.
- Chaudhari, A.A.; Vig, K.; Baganizi, D.R.; Sahu, R.; Dixit, S.; Dennis, V.; Singh, S.R.; Pillai, S.R.; Chaudhari, A.A.; Vig, K.; et al. Future Prospects for Scaffolding Methods and Biomaterials in Skin Tissue Engineering: A Review. Int. J. Mol. Sci. 2016, 17, 1974.
- Negut, I.; Grumezescu, V.; Grumezescu, A.M. Treatment Strategies for Infected Wounds. Molecules 2018, 23, 2392.
- Bacakova, M.; Musilkova, J.; Riedel, T.; Stranska, D.; Brynda, E.; Bacakova, L.; Zaloudkova, M. The potential applications of fibrin-coated electrospun polylactide nanofibers in skin tissue engineering. Int. J. Nanomed. 2016, 11, 771–789.
- Keirouz, A.; Chung, M.; Kwon, J.; Fortunato, G.; Radacsi, N. 2D and 3D electrospinning technologies for the fabrication of nanofibrous scaffolds for skin tissue engineering: A review. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2020, 12, e01626.
- Vig, K.; Chaudhari, A.; Tripathi, S.; Dixit, S.; Sahu, R.; Pillai, S.; Dennis, V.A.; Singh, S.R. Advances in Skin Regeneration Using Tissue Engineering. Int. J. Mol. Sci. 2017, 18, 789.
- Du, P.; Casavitri, C.; Suhaeri, M.; Wang, P.-Y.; Lee, J.H.; Koh, W.-G.; Park, K. A Fibrous Hybrid Patch Couples Cell-Derived Matrix and Poly(l-lactide-co-caprolactone) for Endothelial Cells Delivery and Skin Wound Repair. ACS Biomater. Sci. Eng. 2018, 5, 900–910.
- Grant, J.; Pillai, S.; Perova, T.; Hehir, S.; Hinder, S.; McAfee, M.; Breen, A. Electrospun Fibres of Chitosan/PVP for the Effective Chemotherapeutic Drug Delivery of 5-Fluorouracil. Chemosensors 2021, 9, 70.
- Günday, C.; Anand, S.; Gencer, H.B.; Munafò, S.; Moroni, L.; Fusco, A.; Donnarumma, G.; Ricci, C.; Hatir, P.C.; Türeli, N.G.; et al. Ciprofloxacin-loaded polymeric nanoparticles incorporated electrospun fibers for drug delivery in tissue engineering applications. Drug Deliv. Transl. Res. 2020, 10, 706–720.
- Kharaghani, D.; Gitigard, P.; Ohtani, H.; Kim, K.O.; Ullah, S.; Saito, Y.; Khan, M.Q.; Kim, I.S. Design and characterization of dual drug delivery based on in-situ assembled PVA/PAN core-shell nanofibers for wound dressing application. Sci. Rep. 2019, 9, 12640.
- Rao, G.K.; Kurakula, M.; Yadav, K.S. Application of Electrospun Materials in Gene Delivery. Electrospun Mater. Allied Appl. 2020, 10, 265–306.
- Tuğcu-Demiröz, F.; Saar, S.; Tort, S.; Acartürk, F. Electrospun metronidazole-loaded nanofibers for vaginal drug delivery. Drug Dev. Ind. Pharm. 2020, 46, 1015–1025.
- Homaeigohar, S.; Monavari, M.; Koenen, B.; Boccaccini, A.R. Biomimetic biohybrid nanofibers containing bovine serum albumin as a bioactive moiety for wound dressing. Mater. Sci. Eng. C 2021, 123, 111965.
- Kim, P.-H.; Cho, J.-Y. Myocardial tissue engineering using electrospun nanofiber composites. BMB Rep. 2016, 49, 26–36.
- Ghorani, B.; Tucker, N. Fundamentals of electrospinning as a novel delivery vehicle for bioactive compounds in food nanotechnology. Food Hydrocoll. 2015, 51, 227–240.
- Memic, A.; Abudula, T.; Mohammed, H.S.; Navare, K.J.; Colombani, T.; Bencherif, S.A. Latest Progress in Electrospun Nanofibers for Wound Healing Applications. ACS Appl. Bio Mater. 2019, 2, 952–969.
- Saatchi, A.; Arani, A.R.; Moghanian, A.; Mozafari, M. Cerium-doped bioactive glass-loaded chitosan/polyethylene oxide nanofiber with elevated antibacterial properties as a potential wound dressing. Ceram. Int. 2021, 47, 9447–9461.
- Beikzadeh, S.; Akbarinejad, A.; Swift, S.; Perera, J.; Kilmartin, P.A.; Travas-Sejdic, J. Cellulose acetate electrospun nanofibers encapsulating Lemon Myrtle essential oil as active agent with potent and sustainable antimicrobial activity. React. Funct. Polym. 2020, 157, 104769.
More
Information
Subjects:
Nanoscience & Nanotechnology
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.1K
Revisions:
2 times
(View History)
Update Date:
09 Oct 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





