Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Fabio Aricò | + 2316 word(s) | 2316 | 2021-08-23 10:32:05 | | | |
| 2 | Dean Liu | Meta information modification | 2316 | 2021-10-08 02:33:51 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Aricò, F. Alkyl Levulinates. Encyclopedia. Available online: https://encyclopedia.pub/entry/14780 (accessed on 02 June 2026).
Aricò F. Alkyl Levulinates. Encyclopedia. Available at: https://encyclopedia.pub/entry/14780. Accessed June 02, 2026.
Aricò, Fabio. "Alkyl Levulinates" Encyclopedia, https://encyclopedia.pub/entry/14780 (accessed June 02, 2026).
Aricò, F. (2021, September 30). Alkyl Levulinates. In Encyclopedia. https://encyclopedia.pub/entry/14780
Aricò, Fabio. "Alkyl Levulinates." Encyclopedia. Web. 30 September, 2021.
Copy Citation
Alkyl levulinates (AlkLevs) are biobased chemicals that occupy a strategic role in biomass valorization as potential substitutes of several oil-based compounds (fuels, solvents, chemical intermediates).
alkyl levulinate
furfuryl alcohol
alcoholysis
purolite resin
heterogenous catalyst
biobased platform chemicals
biorefinery
1. Introduction
Alkyl levulinates (AlkLevs) are biobased chemicals that occupy a strategic role in biomass valorization as potential substitutes of several oil-based compounds [1][2]. Their applications in the fields of biofuels (additives), plasticizers, lubricants, flavoring agents, fragrances, and green solvents have been extensively investigated [3][4].
In particular, short-chain (C1–C4) AlkLevs, i.e., methyl (MetLev), ethyl (EtLev) and butyl levulinate (BuLev), showed great potential as oxygenated fuel additives for gasoline blends [5][6][7]. AlkLevs were also employed as green solvents either as such or as precursors of γ-valerolactone (GVL), a cyclic ester that has found interesting applications in perfume and food industries in addition to being exploited as solvent and fuel additive [8][9].
As depicted in Figure 1, several synthetic routes to AlkLevs are reported in the literature; raw biomass or monomers derived from biomass can be both used as starting materials. The related synthetic procedures are based on an alcoholysis reaction promoted by (Brønsted) acid catalyst where the product selectivity depends on the substrate used [4]. In fact, direct conversion of biomass such as cellulose leads to AlkLevs with low or moderate yields due to a combination of solubility issues and drastic conditions required for the depolymerization step [10]. In this view, starting from simple C6 carbohydrates such as D-Glucose or D-Fructose is more convenient, although once again this acid-catalyzed methodology suffers from some solubility issues and the need for relative high temperature (120–200 °C) [11][12][13][14][15][16][17][18][19][20][21]. It should also be mentioned that, when D-Glucose is employed as substrate, an isomerization step is required, and this negatively affects the AlkLev yield. Furthermore, using simple carbohydrates as starting materials often resulted in the formation of a certain amount of humins due to the presence of 5-(hydroxymethyl) furfural (HMF) as reaction intermediate, a biobased platform chemical well known for its unstable nature.
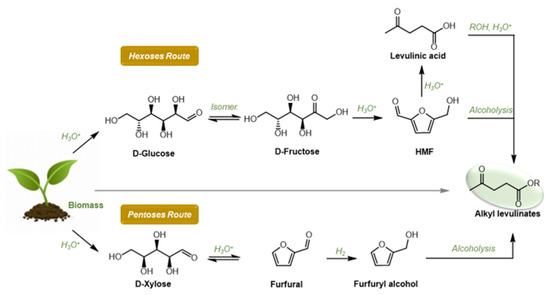
Figure 1. Hexoses and pentoses routed to alkyl levulinates.
2. Ethyl Levulinate as Model Study: Optimization of the Reaction Conditions
Ethyl levulinate (EtLev) was selected as a model compound to test the efficiency of the heterogenous and homogeneous catalysts for the alcoholysis of furfuryl alcohol (Scheme 1, Table 1).
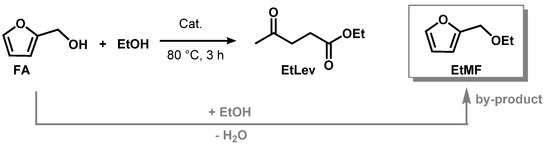
Scheme 1. Synthesis of ethyl levulinate in presence of an acid catalyst.
In a typical experiment, FA was reacted with an excess of ethanol, used as solvent and reagent, in the presence of an acid catalyst. The reaction was conducted under magnetic stirring at reflux temperature (80 °C) for 3 h. Thus, prior to solvent evaporation, the mixture was filtered on a thin layer of basic alumina to remove acid residues that in some cases led to the formation of insoluble black plastic material (Supplementary Figure S1). The product yield was calculated via proton NMR (see Materials and Methods), as this technique allowed for certain identification of the signals related to furfuryl alcohol, ethyl levulinate, and 2-(ethoxymethyl)furan (EtMF) that forms as reaction intermediate.
The first test was conducted in the absence of a catalyst; FA was recovered unaltered from the reaction mixture. Amberlyst-15 was used as referring catalyst as it has been extensively investigated for the alcoholysis of furfuryl alcohol [22][23]. In our reaction conditions, Amberlyst-15 led to a quantitative conversion of FA; however, EtLev formed only in 22% yield.
Purolite exchange resins, available in our laboratory, were then tested. Purolites are polystyrenic strong acid cation resins with different potential applications. Three different Purolite types were investigated, all incorporating sulfonic groups (Supplementary Table S1):
-
Purolite CT275, a macroporous catalyst showing excellent accessibility of active sites;
-
Purolite CT269, a macroporous catalyst with very good mechanical resistance;
-
Purolite CT151, a macroporous polystyrene crosslinked with divinylbenzene.
The Purolites selected were employed both wet and after being oven dried at 100 °C overnight. The presence of interstitial water in these sulfonated resins resulted in a lower efficiency with discrete values of FA conversion and product yield; in contrast, dried Purolites led to more promising results as EtLev yields ranged from 25 to 34%.
Acid-alumina (Al-H) and zeolite were also tested, after calcination at 400 °C overnight; in both cases FA conversion was negligible.
Some homogeneous acid catalysts were investigated. Experiments conducted in the presence of AlCl3 and H2SO4 allowed quantitative conversion of FA and discrete EtLev yield. However, these catalysts were discarded in consideration of their well-known toxicity/corrosivity, and foremost due to the formation of an insoluble black plastic material at the end of the reactions as consequence of the abovementioned concurrent polymerization reactions taking place in strong acidic conditions.
Despite the higher acidity of trifluoroacetic acid compared to acetic acid, both these homogeneous catalysts showed a negligible efficiency in promoting EtLev formation. Similar discouraging results were achieved for the experiments conducted in the presence of oxalic acid and BF3-O(Et)2; the latter showing quantitative conversion but only limited yield.
In conclusion, although all Purolites displayed similar catalytic performances, the oven dried Purolite CT151 showed the best result and was selected for further optimization studies. Its observed enhanced efficiency might be ascribed to the smaller surface area and pore volume of Purolite CT151 compared to the other heterogenous catalysts employed (Supplementary Table S1); these characteristics might allow an optimal interaction with FA.
The effect of the amount of catalyst was next investigated; experiments were conducted by varying the amount of Purolite CT151. Amberlyst-15 was also tested in the same conditions as referring catalyst.
In all experiments, Purolite CT151 confirmed its superior catalytic activity compared to Amberlyst-15 and the latter was no further investigated in the present study.
Although the best results for the Purolite catalyst were obtained when used in 100% wt., the difference in EtLev yield between the reaction conducted with 50% wt. catalyst was too small to justify doubling the amount of catalyst. Thus, further optimization reactions were conducted in the presence of 50% wt. Purolite CT151.
Several experiments were carried out varying the reaction time; FA ethanolysis was carried out for 1, 2, 3, 5, and 7 h after the reaction mixture reached the operating temperature of 80 °C; the results achieved are depicted in Figure 2.
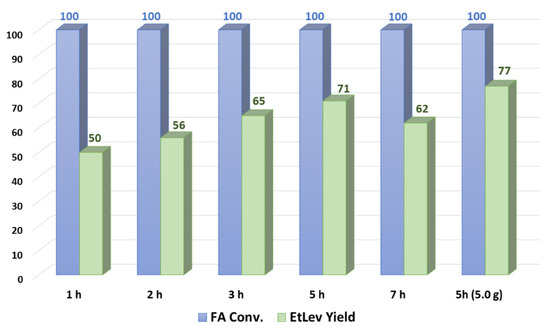
Figure 2. Synthesis of ethyl levulinate over time.
FA conversion reached quantitative value already after 2 h at reflux temperature; however, EtLev yield continued to increase, reaching a maximum at 5 h (71%). This discrepancy might be attributed to the formation of at least one reaction intermediate different from EtMF that was clearly visible in the proton NMR spectrum, although we were not able to identify or isolate it. When the reaction was prolonged to 7 h, a lower yield of EtLev was observed (62%), possibly due to concurrent side reactions and/or product degradation.
To validate the data so far achieved, a larger scale reaction was conducted on 5.0 g of FA; the experiment led to a slightly higher EtLev yield (77% yield) compared to the one achieved on a 1.0 g scale. This result is a good indication that higher scale reactions might perform even better than smaller scale reactions.
Figure 3 reports a possible reaction mechanism for the formation of EtLev from FA. A comprehensive investigation on the alcoholysis of furfuryl alcohol is still lacking, most likely due to the difficulties in identifying all the concurrent synthetic pathways and the related reaction intermediates; thus, further investigations are needed to clarify these aspects.
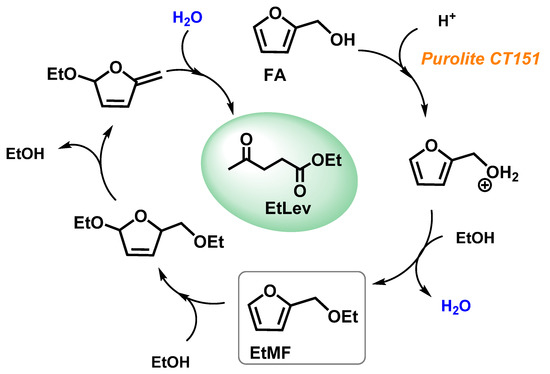
Figure 3. Proposed reaction mechanism for the alcoholysis of FA.
Additional optimization attempts were conducted by varying the operating temperature and the FA concentration, which was previously set arbitrarily at 0.50 M (1.0 gr in 20 mL of EtOH). The reaction carried out at lower temperatures, i.e., from room temperature up to 60 °C, led to reduced substrate conversion and lower EtLev yield (Supplementary Table S2). Similarly, when the FA ethanolysis was conducted on more diluted (0.25 M FA) or more concentrated (1.0 and 2.0 M) solutions, EtLev formed in lower yields (Supplementary Table S3). It can be assumed that at high dilution there is a reduced interaction between FA and ethanol. Meanwhile, working at high concentration, although faster interactions between the reagents took place, unwanted secondary reactions such as polymerization and degradation might also occur more rapidly, affecting product yield.
In conclusion, optimization investigations indicated that the best reaction condition encompassed a 0.5 M solution of FA in ethanol heated at reflux temperature (80 °C) for 5 h; analysis of the reaction mixture indicated an EtLev yield of 71% (77% in larger scale).
3. Alkyl Levulinates Syntheses, Products Isolation, and Catalyst Recycling
With the best-found reaction conditions known, the scope of this procedure was next investigated to develop a library of alkyl levulinates. Thus, FA alcoholysis was conducted in the presence of primary (methanol, 1-propanol, 1-butanol), secondary (2-propanol, 2-butanol), and unsaturated (allyl alcohol) alcohol (Scheme 2, Table 3).
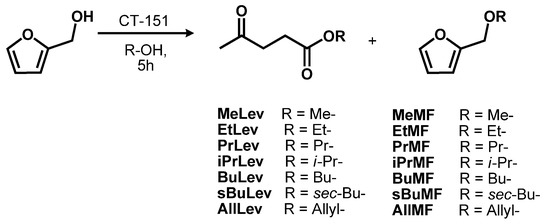
Scheme 2. Synthesis of alkyl levulinates in presence of acid catalyst Purolite CT151.
AlkLevs were achieved in yields ranging from 30% up to 71% according to 1H NMR data. Among them, methyl levulinate (MeLev) was the only product formed in a rather low yield. Thus, an additional experiment was carried out in an autoclave apparatus at 80 °C, doubling the amount of the reagents and catalyst. In these conditions an autogenous pressure of 2 bar was observed, which eventually favored the FA methanolysis reaction, leading to 50% yield of MeLev. A further autoclave test was conducted at 160 °C; in this case the autogenous pressure increased constantly during the reaction up to 44 bar due to the unwanted formation of dimethyl ether. MeLev yield was calculated to be 32%; most likely the reduced yield observed is imputable to degradation of the substrate at this high temperature.
FA alcoholysis in the presence of the other selected alcohols was tested both at 80 °C, and at their reflux temperature. The latter experiments resulted in the formation of the related AlkLevs in higher yields (40–63%).
Note also that to be best of our knowledge, this is the first time that allyl levulinate was prepared via FA alcoholysis. Previous published reports focused mainly on levulinic acid esterification [24].
AllLev is an interesting derivative as the presence of the allyl group within the levulinate backbone can allow further chemical modification of this compound.
Having established the robustness of our procedure, the best experiments were repeated in larger scale starting from 5.0 g of FA to address isolation of the products from the reaction mixture.
After several attempts, we found that the best purification procedure encompassed the filtration of reaction mixture through a thin layer of basic alumina followed by a two-step distillation: (i) a first at atmospheric pressure to recover the residual alcohol, followed by (ii) a second distillation at reduced pressure to recover the pure AlkLev.
The isolated yields of MeLev, EtLev, iPrLev, BuLev, sBuLev, and AllLev are fully consistent with the related values calculated by proton NMR analysis, confirming the validity of the analytical method used. It is interesting to point out that when the methanolysis of FA was conducted in larger scale, MeLev was achieved in higher yield (65%) compared to the 2.0 g scale procedure (50%), similar to that previously observed for the preparation of EtLev (Figure 2).
All AlkLevs were isolated as transparent or yellowish liquid and completely characterized to confirm the proposed structures (see Supplemental Materials).
Furthermore, as part of this investigation, catalyst recycling was also studied, focusing on the case of EtLev preparation.
Purolite CT151, a heterogeneous catalyst, can be recovered from the reaction mixture by simple filtration; however, due to magnetic stirring employed to ensure the reaction mixture homogeneity, the tiny catalyst spheres were reduced to powder. To solve this issue, the ethanolysis of FA was performed without stirring, although the EtLev yield decreased to 23%. Further attempts were carried out inserting nitrogen flow via a glass pipette directly into the reaction mixture to ensure the mixing of the solution. Although in this case EtLev yield improved to 57%, the continuous nitrogen inlet caused a rapid ethanol evaporation that had to be constantly added to the reaction mixture. In contrast, when a 100% wt. of Purolite CT151 (compared to FA) was employed without stirring and/or nitrogen flow, EtLev was achieved with a yield comparable to our previous results, i.e., 66%.
Thus, the experiments on catalyst recycling were conducted employing FA:Purolite CT1515 in 1:1 weight ratio. At the end of each run, the catalyst was recovered, washed with the minimal amount of acetone, and dried overnight at 100 °C. According to the data collected (Figure 4a) the catalyst can be reused for a maximum of four consecutive reactions; in fact, in the fifth run an evident decrease of EtLev yield was observed (44%).
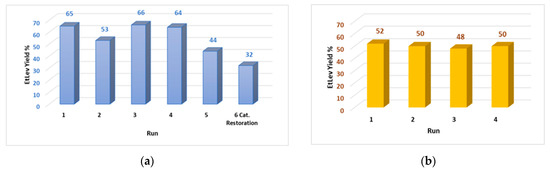
Figure 4. (a) Recycling of the catalyst Purolite CT151; FA (1.0 g, 10.19 mmol), EtOH (20 mL), Purolite CT1515 (100% wt.) at 80 °C for 5 h. (b) Recycling of the catalyst Purolite CT151 and of the exceeding ethanol; FA (5.0 g, 50.9 mmol), EtOH (100 mL), Purolite CT1515 (100% wt.) at 80 °C for 5 h.
Restoration of the catalyst acid sites was attempted by immersion of the spheres in an acid solution of water/methanol; the Purolite was then washed with acetone and dried overnight before being used in a new reaction. However, analysis of the reaction mixture showed that catalyst activity was not reestablished as EtLev yield was even lower than the fifth run (32%). This result was ascribed to the surface poisoning of the catalyst due to deposition of carbonaceous material and polymers that formed during the degradation of furfuryl alcohol. In fact, as the recycling runs proceeded, the Purolite spheres assumed a dark black color consistent with the one of the degradation products (polymers and humins) previously observed.
At an industrial level, catalyst recycling is as relevant as the recovery and reuse of the reagents/solvents used in stoichiometric excess. Thus, some recycling tests were conducted on the five-gram-scale FA ethanolysis; both the catalyst and the exceeding ethanol were recycled for four consecutive runs (Figure 4b). For each recycling test the catalyst was recovered by filtration; the ethanol was recovered by distillation at atmospheric pressure and the product and EtLev purified via distillation at reduced pressure, as previously described. Ethanol distillation was operated in the presence of a mild flow of nitrogen to speed the procedure, although this caused a loss of about 30% (30 mL) of the reagent that, in the following run, had to be replenished with new reagent.
As a result, the large-scale reaction for the ethanolysis of FA allowed isolation of pure EtLev with a constant yield of ca. 50% in four consecutive runs where both ethanol and Purolite were recovered and reused (Figure 4b).
References
- Climent, M.J.; Corma, A.; Iborra, S. Conversion of biomass platform molecules into fuel additives and liquid hydrocarbon fuels. Green Chem. 2014, 16, 516–547.
- Zhang, J.; Wu, S.; Li, B.; Zhang, H. Advances in the catalytic production of valuable levulinic acid derivatives. ChemCatChem 2012, 4, 1230–1237.
- Liu, X.; Yang, W.; Zhang, Q.; Li, C.; Wu, H. Current approaches to alkyl levulinates via efficient valorization of biomass derivatives. Front. Chem. 2020, 8, 794.
- Démolis, A.; Essayem, N.; Rataboul, F. Synthesis and applications of alkyl levulinates. ACS Sustain. Chem. Eng. 2014, 2, 1338–1352.
- Christensen, E.; Williams, A.; Paul, S.; Burton, S.; McCormick, R.L. Properties and performance of levulinate esters as diesel blend components. Energy Fuels 2011, 25, 5422–5428.
- Jia, S.; Ma, J.; Wang, D.; Wang, K.; Zheng, Q.; Song, C.; Guo, X. Fast and efficient upgrading of levulinic acid into long-chain alkyl levulinate fuel additives with a tungsten salt catalyst at low temperature. Sustain. Energy Fuels 2020, 4, 2018–2025.
- Shrivastav, G.; Khan, T.S.; Agarwal, M.; Haider, M.A. Reformulation of gasoline to replace aromatics by biomass-derived alkyl levulinates. ACS Sustain. Chem. Eng. 2017, 5, 7118–7127.
- Shao, Y.; Sun, K.; Li, Q.; Liu, Q.; Zhang, S.; Liu, Q.; Hu, G.; Hu, X. Copper-based catalysts with tunable acidic and basic sites for the selective conversion of levulinic acid/ester to gamma-valerolactone or 1,4-pentanediol. Green Chem. 2019, 21, 4499–4511.
- Vaccaro, L.; Santoro, S.; Curini, M.; Lanari, D. The emerging use of γ-valerolactone as a green solvent. Chem. Today 2017, 35, 46–48.
- Raspolli Galletti, A.M.; Antonetti, C.; Fulignati, S.; Licursi, D. Direct alcoholysis of carbohydrate precursors and real cellulosic biomasses to alkyl levulinates: A critical review. Catalysts 2020, 10, 1221.
- Ramírez, E.; Bringué, R.; Fitè, C.; Iborra, M.; Tejero, J.; Cunill, F. Assessment of ion exchange resins as catalysts for the direct transformation of fructose into butyl levulinate. Appl. Catal. A 2021, 612, 117988.
- Liu, J.; Yang, B.-B.; Wang, X.-Q.; Liu, C.-L.; Yang, R.-Z.; Dong, W.-S. Glucose conversion to methyl levulinate catalyzed by metal ion-exchanged montmorillonites. Appl. Clay Sci. 2017, 141, 118–124.
- Srinivasa Rao, B.; Krishna Kumari, P.; Dhana Lakshmi, D.; Lingaiah, N. One pot selective transformation of biomass derived chemicals towards alkyl levulinates over titanium exchanged heteropoly tungstate catalysts. Cat. Today 2018, 309, 269–275.
- Jiang, L.; Zhou, L.; Chao, J.; Zhao, H.; Lu, T.; Su, Y.; Yang, X.; Xu, J. Direct catalytic conversion of carbohydrates to methyl levulinate: Synergy of solid Brønsted acid and Lewis acid. Appl. Catal. B 2018, 220, 589–596.
- Liu, J.; Wang, X.-Q.; Yang, B.-B.; Liu, C.-L.; Xu, C.-L.; Dong, W.-S. Highly efficient conversion of glucose into methyl levulinate catalyzed by tin-exchanged montmorillonite. Renew. Energy 2018, 120, 231–240.
- Xu, X.; Zhang, X.; Zou, W.; Yue, H.; Tian, G.; Feng, S. Conversion of carbohydrates to methyl levulinate catalyzed by sulfated montmorillonite. Cat. Commun. 2015, 62, 67–70.
- Karnjanakom, S.; Maneechakr, P.; Samart, C.; Kongparakul, S.; Guan, G.; Bayu, S. Direct conversion of sugar into ethyl levulinate catalyzed by selective heterogeneous acid under co-solvent system. Cat. Commun. 2020, 143, 106058.
- Heda, J.; Niphadkar, P.; Bokade, V. Efficient synergetic combination of H-USY and SnO2 for direct conversion of glucose into ethyl levulinate (biofuel additive). Energy Fuels 2019, 33, 2319–2327.
- Tiong, Y.W.; Yap, C.L.; Gan, S.; Yap, W.S.P. Conversion of biomass and its derivatives to levulinic acid and levulinate esters via ionic liquids. Ind. Eng. Chem. Res. 2018, 57, 4749–4766.
- Liu, R.; Chen, J.; Huang, X.; Chen, L.; Ma, L.; Li, X. Conversion of fructose into 5-hydroxymethylfurfural and alkyl levulinates catalyzed by sulfonic acid-functionalized carbon materials. Green Chem. 2013, 15, 2895–2903.
- Song, D.; Zhang, Q.; Sun, Y.; Zhang, P.; Guo, Y.-H.; Hu, J.-L. Design of ordered mesoporous sulfonic acid functionalized ZrO2/organosilica bifunctional catalysts for direct catalytic conversion of glucose to ethyl levulinate. ChemCatChem 2018, 10, 4953–4965.
- Lange, J.P.; van de Graaf, W.D.; Haan, R.J. Conversion of furfuryl alcohol into ethyl levulinate using solid acid catalysts. ChemSusChem 2009, 2, 437–441.
- Jorge, E.Y.C.; Lima, T.D.M.; Lima, C.G.S.; Marchini, L.; Castelblanco, W.N.; Rivera, D.G.; Urquieta-González, E.A.; Varma, R.S.; Paixão, M.W. Metal-exchanged magnetic β-zeolites: Valorization of lignocellulosic biomass-derived compounds to platform chemicals. Green Chem. 2017, 19, 3856–3868.
- Bartoli, G.; Bosco, M.; Carlone, A.; Dalpozzo, R.; Marcantoni, E.; Melchiorre, P.; Sambria, L. Reaction of dicarbonates with carboxylic acids catalyzed by weak lewis acids: General method for the synthesis of anhydrides and esters. Synthesis 2007, 22, 3489–3496.
More
Information
Subjects:
Materials Science, Composites
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.6K
Revisions:
2 times
(View History)
Update Date:
08 Oct 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





