
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Myung-Geol Pang | + 2290 word(s) | 2290 | 2020-07-28 11:00:30 | | | |
| 2 | Myung-Geol Pang | + 10 word(s) | 2300 | 2020-07-30 08:18:59 | | | | |
| 3 | Rita Xu | -963 word(s) | 1337 | 2020-07-31 06:02:44 | | | | |
| 4 | Rita Xu | -3 word(s) | 1334 | 2020-10-26 09:35:08 | | |
Video Upload Options
Bisphenol A (BPA) is an oestrogenic endocrine disruptor widely used in the production of certain plastics, e.g., polycarbonate, hard and clear plastics, and epoxy resins that act as a protective coating for food and beverage cans. Human exposure to this chemical is thought to be ubiquitous. BPA alters endocrine function, thereby causing many diseases in human and animals. In the last few decades, studies exploring the mechanism of BPA activity revealed a direct link between BPA induced oxidative stress and disease pathogenesis. Antioxidants, reducing agents that prevent cellular oxidation reactions, can protect BPA toxicity. Although the important role of antioxidants in minimizing BPA stress has been demonstrated in many studies, a clear consensus on the associated mechanisms is needed, as well as the directives on their efficacy and safety. Herein, considering the distinct biochemical properties of BPA and antioxidants, we provide a framework for understanding how antioxidants alleviate BPA‐associated stress. We summarize the current knowledge on the biological function of enzymatic and non‐enzymatic antioxidants, and discuss their practical potential as BPA‐detoxifying agents.
1. Definition
Bisphenol A (BPA) is an oestrogenic endocrine disruptor widely used in the production of certain plastics, e.g., polycarbonate, hard and clear plastics, and epoxy resins that act as protective coating for food and beverage cans. Human exposure to this chemical is thought to be ubiquitous. BPA alters endocrine function, thereby causing many diseases in human and animals. In the last few decades, studies exploring the mechanism of BPA activity revealed a direct link between BPA-induced oxidative stress and disease pathogenesis. Antioxidants, reducing agents that prevent cellular oxidation reactions, can protect BPA toxicity. Although the important role of antioxidants in minimizing BPA stress has been demonstrated in many studies, a clear consensus on the associated mechanisms is needed, as well as the directives on their efficacy and safety.
2. Introduction
Bisphenol A [BPA; (CH3)2C(C6H4OH)2] is an organic synthetic compound and a major environmental pollutant. It is widely used to manufacture numerous consumer products, including food packaging materials, industrial supplies, dental sealant, and others. BPA is an endocrine disruptor. It interferes with hormone function via oestrogenic, anti-androgenic, and anti-thyroid activity [1][2][3][4]. Exposure to this chemical is ubiquitous, and occurs mostly via the oral (approximately 90%), respiratory, and dermal routes in human and animals [5][6]. In addition, BPA is reportedly passed from the mother to offspring by intrauterine transmission during prenatal embryonic development and via breastfeeding during the early neonatal period [7].
Based on the available evidence, BPA has a very weak binding affinity for certain hormonal receptors. As an example, the binding affinity of BPA to oestrogen receptor is 1000–10,000 times weaker than that of natural oestrogen (diethylstilbestrol) [8]. Normal endocrine signalling does not affect the overall hormone levels to a great extent. However, even small alterations of hormone function can potentially greatly affect biological activity [9]. Consequently, scientists believe that BPA may impact the delicate endocrine balance, leading to diverse pathological outcomes. Interestingly, BPA was initially used as a growth promoter in the cattle and poultry industries, but was later proved to be highly toxic [10]. Specifically, exposure to BPA is linked to cardiovascular disease, brain development abnormalities, obesity, hypertension, thyroid dysfunction, diabetes, breast cancer, infertility, etc., in human, and terrestrial and aquatic animals, as summarized by Rochester [11].
Harmful effects of BPA in cells and tissues (both in vitro and in vivo) are mostly mediated by increased oxidative stress associated with an elevated production of toxic free radicals, in addition to the classical genomic and non-genomic mechanisms of activity [4][5]. Although intracellular free radicals, most importantly, reactive oxygen species (ROS), are critical regulators of cellular physiology, their increased levels can directly affect DNA, RNA, and proteins, subsequently predisposing the cell to pathology [12][13].
By contrast, an antioxidant is a reducing agent that scavenges and neutralizes free radicals, thereby preventing oxidation reactions. Indeed, the important role of enzymatic antioxidants, e.g., superoxide dismutase (SOD), catalase (CAT), or glutathione (GSH) system, in overcoming the harmful effects of BPA has been highlighted in many studies. Simultaneously, the significance of non-enzymatic antioxidants in the biochemistry of living organism has been uncovered in the recent decades. For instance, Lobo et al. [14] reported that vitamin E prevents peroxidation of lipids by scavenging ROS before they damage the cell. Similar, the function of germ cells, neuronal, and kidney cells is preserved by antioxidants during BPA co-exposure and/or administration both in vitro and in vivo [13][15][16][17][18]. Importantly, antioxidants safely interact with free radicals generated upon BPA exposure and terminate the oxidation chain reaction before vital molecules, such as DNA, RNA, and proteins, are damaged [14]. It is tempting to speculate that antioxidants could be used as a potential defence and/or treatment regime against BPA toxicity.
3. Overview of BPA Activity
As an endocrine disrupting chemical, BPA acts as selective modulator of oestrogen receptor (ER), activating oestrogen-related receptor gamma and growth factor receptors [1][3]. Further, it is an antagonist of the thyroid hormone receptor and possesses anti-androgenic properties [1].
BPA has a structural similarity with oestrogen; therefore, capable of binding with both ERα and ERβ [4][18][19]. As an ER modulator, BPA acting via genomic and non-genomic pathways (Figure 1). In the genomic pathway, it binds to ER located in the cytoplasm (cER) or the nucleus (nER). The ER-BPA dimer binds to chromatin and activates transcription factors (TF), thereby leading to the transcription of target genes and affecting cell function [3][20]. In the non-genomic pathway, BPA binds to G protein-coupled receptors (GPCR) and cell membrane-bound ER (mER). Activation of both receptors triggers rapid oestrogen signalling via TF phosphorylation and activation of several kinase systems. In human ovarian cancer cells, such TF phosphorylation is mediated via the activation of mitogen-activated protein kinase (MAPK) and phosphatidylinositol 3-kinase, and changes in cAMP, protein kinase C, and protein kinase A levels that follow BPA binding to GPCR and mER [21]. It is important to note that the levels of circulating oestrogen greatly vary in males and females, which leading to the differential expression of ERs in different cells/tissues between both sexes. This is probably the major reason why BPA affect male and female differently [22]. On the other hand, BPA directly manipulates the cellular microenvironment. Plasma proteins that bind to BPA and protect the target cells from BPA can be changed upon BPA exposure. Detoxification of xenobiotics, including BPA, results in the formation of electrophiles, free radicals, nucleophiles, and redox-active reactants can destroy the DNA, RNA, and proteins [23]. Further, although the majority of BPA is metabolized to generate relatively less toxic BPA glucuronide and BPA sulphate, the remaining free BPA facilitates ROS formation via enzymatic and non-enzymatic reactions [24]. ROS reacting with nicotinamide adenine dinucleotide phosphate species concomitant with enzymatic processing facilitates the formation of diverse oxidative species, such as superoxides, peroxides, and hydroxyl radicals [24]. These free radicals lead to irreversible alteration of gene expression, apoptosis, and cell death. As such BPA interrupts cellular oxidative homeostasis by altering a dynamic balance between oxidative mediators and the activities of antioxidant enzymes, subsequently predispose to apoptosis [25][26].
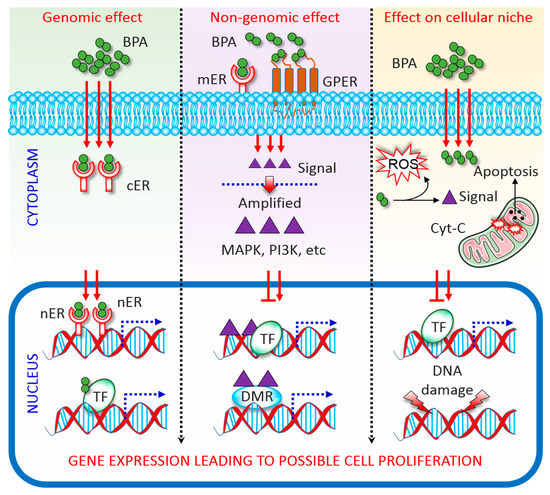
Figure 1. Mechanisms of Bisphenol A (BPA) activity. Genomic, non-genomic, and direct effects exerted by BPA are depicted. See the main text for details. BPA, bisphenol A; DMR, differentially methylated regions; cER, cytoplasmic oestrogen receptor; Cyt-C, cytochrome c; GPCR, G protein-coupled receptor; MAPK, mitogen-activated protein kinase; mER, membrane-bound oestrogen receptor; nER, nuclear oestrogen receptor; PI3K, phosphatidylinositol 3-kinase; ROS, reactive oxygen species; TF, transcription factor.
Meli et al. [18] reported that BPA increases oxidative stress in the rat liver and spermatozoa by lowering the levels of antioxidant enzymes and increasing H2O2 and lipid peroxidation. These harmful effects were also capable of affecting the normal development of the kidney, brain, and testis as shown in mice model [18].
As an androgen receptor antagonist, BPA inhibits N- and C-terminal regions of the androgen receptor. This facilitates the interaction with a silencing mediator for thyroid hormone receptor and nuclear receptor co-repressor, subsequently suppressing the proliferation of Sertoli cells [27]. Although BPA-mediated effects on ER and androgenic receptor are relatively well studied, the mechanisms of BPA activity in different cell types/tissues, need to be investigated.
References
- Vandenberg, L.N.; Maffini, M.V.; Sonnenschein, C.; Rubin, B.S.; Soto, A.M. Bisphenol-A and the great divide: a review of controversies in the field of endocrine disruption. Endocr. Rev. 2008, 30, 75–95, doi:10.1210/er.2008-0021.
- Rahman, S.; Kwon, W.-S.; Lee, J.-S.; Yoon, S.-J.; Ryu, B.-Y.; Pang, M.-G. Bisphenol-A Affects Male Fertility via Fertility-related Proteins in Spermatozoa. Sci. Rep. 2015, 5, 9169, doi:10.1038/srep09169.
- Rahman, S.; Kwon, W.-S.; Yoon, S.-J.; Park, Y.-J.; Ryu, B.-Y.; Pang, M.-G. A novel approach to assessing bisphenol-A hazards using an in vitro model system. BMC Genom. 2016, 17, 577, doi:10.1186/s12864-016-2979-5.
- Rahman, M.S.; Pang, W.K.; Ryu, D.Y.; Park, Y.J.; Pang, M.G. Multigenerational and transgenerational impact of paternal bisphenol A exposure on male fertility in a mouse model. Hum. Reprod. 2020, deaa13, doi: 10.1093/humrep/deaa139.
- Rahman, S.; Kwon, W.-S.; Karmakar, P.C.; Yoon, S.-J.; Ryu, B.-Y.; Pang, M.-G. Gestational Exposure to Bisphenol A Affects the Function and Proteome Profile of F1 Spermatozoa in Adult Mice. Environ. Heal. Perspect. 2016, 125, 238–245, doi:10.1289/EHP378.
- Rahman, S.; Kwon, W.-S.; Ryu, D.-Y.; Khatun, A.; Karmakar, P.C.; Ryu, B.-Y.; Pang, M.-G. Functional and Proteomic Alterations of F1 Capacitated Spermatozoa of Adult Mice Following Gestational Exposure to Bisphenol A. J. Proteome Res. 2017, 17, 524–535, doi:10.1021/acs.jproteome.7b00668.
- Reddivari, L.; Veeramachaneni, D.N.R.; Walters, W.A.; Lozupone, C.; Palmer, J.; Hewage, M.K.K.; Bhatnagar, R.; Amir, A.; Kennett, M.J.; Knight, R.; et al. Perinatal Bisphenol A Exposure Induces Chronic Inflammation in Rabbit Offspring via Modulation of Gut Bacteria and Their Metabolites. mSystems 2017, 2, e00093-17, doi:10.1128/msystems.00093-17.
- Lemmen, J.G.; Arends, R.J.; Van Boxtel, A.L.; Van Der Saag, P.T.; Van Der Burg, B. Tissue- and time-dependent estrogen receptor activation in estrogen reporter mice. J. Mol. Endocrinol. 2004, 32, 689–701, doi:10.1677/jme.0.0320689.
- Saltzman, W.; Ziegler, T.E. Functional Significance of Hormonal Changes in Mammalian Fathers. J. Neuroendocr. 2014, 26, 685–696, doi:10.1111/jne.12176.
- Erler, C.; Novak, J. Bisphenol A Exposure: Human Risk and Health Policy. J. Pediatr. Nurs. 2010, 25, 400–407, doi:10.1016/j.pedn.2009.05.006.
- Rochester, J.R. Bisphenol A and human health: A review of the literature. Reprod. Toxicol. 2013, 42, 132–155, doi:10.1016/j.reprotox.2013.08.008.
- Aitken, R.J.; Paterson, M.; Fisher, H.; Buckingham, D.W.; Van Duin, M. Redox regulation of tyrosine phosphorylation in human spermatozoa and its role in the control of human sperm function. J. Cell Sci. 1995, 108, 2017–25.
- Rahman, S.; Kang, K.-H.; Arifuzzaman, S.; Pang, W.-K.; Ryu, D.-Y.; Song, W.-H.; Park, Y.-J.; Pang, M.-G. Effect of antioxidants on BPA-induced stress on sperm function in a mouse model. Sci. Rep. 2019, 9, 10584–10, doi:10.1038/s41598-019-47158-9.
- Lobo, V.; Patil, A.; Phatak, A.; Chandra, N. Free radicals, antioxidants and functional foods: Impact on human health. Pharmacogn. Rev. 2010, 4, 118–126, doi:10.4103/0973-7847.70902.
- Del Prete, C.; Tafuri, S.; Ciani, F.; Pasolini, M.P.; Ciotola, F.; Albarella, S.; Carotenuto, D.; Peretti, V.; Cocchia, N. Influences of dietary supplementation withLepidium meyenii(Maca) on stallion sperm production and on preservation of sperm quality during storage at 5 °C. Androl. 2018, 6, 351–361, doi:10.1111/andr.12463.
- Losano, J.D.D.A.; Angrimani, D.S.R.; Rui, B.; Bicudo, L.C.; Dalmazzo, A.; Silva, B.C.; Viana, C.H.; Mendes, C.M.; Assumpcao, M.E.; Barnabe, V.H.; et al. The addition of docosahexaenoic acid (DHA) and antioxidants (glutathione peroxidase and superoxide dismutase) in extenders to epididymal sperm cryopreservation in bulls. Zygote 2018, 26, 199–206, doi:10.1017/s0967199418000096.
- Babu, S.; Uppu, S.; Claville, M.O.; Uppu, R.M. Prooxidant actions of bisphenol A (BPA) phenoxyl radicals: implications to BPA-related oxidative stress and toxicity. Toxicol. Mech. Methods 2013, 23, 273–280, doi:10.3109/15376516.2012.753969.
- Kobroob, A.; Peerapanyasut, W.; Chattipakorn, N.; Wongmekiat, O. Damaging Effects of Bisphenol A on the Kidney and the Protection by Melatonin: Emerging Evidences from In Vivo and In Vitro Studies. Oxidative Med. Cell. Longev. 2018, 2018, 1–15, doi:10.1155/2018/3082438.
- Meli, R.; Monnolo, A.; Annunziata, C.; Pirozzi, C.; Ferrante, M.C. Oxidative Stress and BPA Toxicity: An Antioxidant Approach for Male and Female Reproductive Dysfunction. Antioxidants 2020, 9, 405, doi:10.3390/antiox9050405.
- Heldring, N.; Pike, A.; Andersson, S.; Matthews, J.B.; Cheng, G.; Hartman, J.; Tujague, M.; Ström, A.; Treuter, E.; Warner, M.; et al. Estrogen Receptors: How Do They Signal and What Are Their Targets. Physiol. Rev. 2007, 87, 905–931, doi:10.1152/physrev.00026.2006.
- Nadal, A.; Ropero, A.B.; Laribi, O.; Maillet, M.; Fuentes, E.; Soria, B. Nongenomic actions of estrogens and xenoestrogens by binding at a plasma membrane receptor unrelated to estrogen receptor alpha and estrogen receptor beta. Proc. Natl. Acad. Sci. 2000, 97, 11603–11608, doi:10.1073/pnas.97.21.11603.
- Ishunina, T.; Kruijver, F.P.; Balesar, R.; Swaab, D.F. Differential Expression of Estrogen Receptor ? and ? Immunoreactivity in the Human Supraoptic Nucleus in Relation to Sex and Aging 1. J. Clin. Endocrinol. Metab. 2000, 85, 3283–3291, doi:10.1210/jcem.85.9.6826.
- Xin, F.; Susiarjo, M.; Bartolomei, M.S. Multigenerational and transgenerational effects of endocrine disrupting chemicals: A role for altered epigenetic regulation? Semin. Cell Dev. Boil. 2015, 43, 66–75, doi:10.1016/j.semcdb.2015.05.008.
- Gassman, N.R. Induction of oxidative stress by bisphenol A and its pleiotropic effects. Environ. Mol. Mutagen. 2017, 58, 60–71, doi:10.1002/em.22072.
- Wang, K.; Zhao, Z.; Ji, W. Bisphenol A induces apoptosis, oxidative stress and inflammatory response in colon and liver of mice in a mitochondria-dependent manner. Biomed. Pharmacother. 2019, 117, 109182, doi:10.1016/j.biopha.2019.109182.
- Tavakkoli, A.; Abnous, K.; Hassani, F.V.; Hosseinzadeh, H.; Birner-Gruenberger, R.; Mehri, S. Alteration of protein profile in cerebral cortex of rats exposed to bisphenol a: a proteomics study. NeuroToxicology 2020, 78, 1–10, doi:10.1016/j.neuro.2020.01.013.
- Wang, H.; Ding, Z.; Shi, Q.-M.; Ge, X.; Wang, H.-X.; Li, M.-X.; Chen, G.; Wang, Q.; Ju, Q.; Zhang, J.-P.; et al. Anti-androgenic mechanisms of Bisphenol A involve androgen receptor signaling pathway. Toxicol. 2017, 387, 10–16, doi:10.1016/j.tox.2017.06.007.





