| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Lynn Htet Htet Aung | + 2755 word(s) | 2755 | 2020-07-22 05:33:47 | | | |
| 2 | Bruce Ren | -2 word(s) | 2753 | 2020-07-27 06:14:53 | | | | |
| 3 | Bruce Ren | Meta information modification | 2753 | 2020-08-04 04:07:48 | | | | |
| 4 | Bruce Ren | -11 word(s) | 2742 | 2020-10-28 04:50:04 | | |
Video Upload Options
Recent studies have shown that RNAs are susceptible to oxidative damage and oxidized RNA is able to break the RNA strand, and affect the protein synthesis, which can lead to cell degradation and cell death. Studies have shown that RNA oxidation is one of the early events in the formation and development of neurodegenerative disorders, including Alzheimer’s disease, Parkinson’s disease, and amyotrophic lateral sclerosis. However, its molecular mechanism, as well as its impact on these diseases, are still unclear. In this article, we review the different types of RNA oxidative damage and the neurodegenerative diseases that are reported to be associated with RNA oxidative damage. In addition, we discuss recent findings on the association between RNA oxidative damage and the development of neurodegenerative diseases, which will have great significance for the development of novel strategies for the prevention and treatment of these diseases.
In the history of nucleic acid research, DNA has always been the main research focus. After the sketch of the human genome was completed in 2000, RNA has been started to gain more attention due to its abundancies in the cell and its essential role in cellular physiology and pathologies. Recent studies have shown that RNAs are susceptible to oxidative damage and oxidized RNA is able to break the RNA strand, and affect the protein synthesis, which can lead to cell degradation and cell death. Studies have shown that RNA oxidation is one of the early events in the formation and development of neurodegenerative disorders, including Alzheimer's disease, Parkinson's disease, and amyotrophic lateral sclerosis. However, its molecular mechanism, as well as its impact on these diseases, are still unclear. In this article, we review the different types of RNA oxidative damage and the neurodegenerative diseases that are reported to be associated with RNA oxidative damage. In addition, we discuss recent findings on the association between RNA oxidative damage and the development of neurodegenerative diseases, which will have great significance for the development of novel strategies for the prevention and treatment of these diseases.
1. Introduction
RNA is known to be essential for all living cells and performs many other functions besides protein synthesis. Contrary to DNA damage studies, RNA damage has only recently been concerned [1]. Although the RNA is only encoded by a small part of the genome in higher organisms, research shows that a great majority of the genome is transcribed, which indicates that the function of a large amount of RNA has not been revealed [2]. In the cell, RNA accounts for 80% to 90% of the total cellular nucleic acid and is more abundant than DNA. Therefore, RNA can be a principal target for nucleic acid damage agents. RNA damage may affect cells as a result of any changes in RNA function. Many factors like ultraviolet light, reactive oxygen species, and nitrogen (ROS and RNS) can induce oxidative damage in RNA. Intracellular RNAs suffer from the same oxidative damage as other biological macromolecules such as DNA. RNA damage may have serious adverse effects on RNA chain, protein synthesis, and cell function. RNA is mainly single-stranded and its bases are not protected by hydrogen bonds or specific proteins, so it is more susceptible to oxidative damage than DNA [3]. Therefore, oxidative damage of protein-coding RNA or non-coding RNA can affect the gene expression. Although RNA oxidation is not as fatal as genomic mutations, RNA oxidative damage is a typical feature of neuronal fragility, suggesting that RNA oxidation may promote the occurrence of chronic degeneration [4][5], including Alzheimer's disease (AD), Parkinson's disease (PD), Lewy body dementia, and prion diseases [6]. Increasing research evidence that preventing damaged nucleotides play a role in translation can significantly reduce the harmful effects of oxidative RNA on protein synthesis. An in-depth study on the molecular mechanisms involved in oxidative RNA damage may provide important implications for the pathogenesis and treatment strategies of neurodegenerative disorders and other diseases [7][8].
2. RNA Oxidation
Because the structure of ribonucleic acid is similar to deoxyribonucleic acid, the nucleotide structures of RNA and DNA are still similar after oxidation modification, especially guanosine. 8-oxidized guanosine is the most studied oxidized RNA damage marker at present. 8-oxidized guanosine exists widely in various tissues and body fluids (such as the brain, spinal cord, liver, artery, urine, cerebrospinal fluid, etc.) [9][10][11]. Reactive oxygen species (ROS), ultraviolet rays (UV), and alkylating agents can cause nucleic acid cross-linking, nucleic acid strand breakage, and base modification damage and other RNA oxidative damage under physiological or pathological conditions, of which the most important factor is ROS [12]. Some factors in the body, such as abnormal mitochondrial function, usually increase the number of ROS, and certain physiological reactions also produce ROS. Studies have already revealed that ROS not only play a cytotoxic role directly but also indirectly regulate the expression of some genes by affecting the cellular signal transduction system and interact with ribose, bases, nucleotides, and oligonucleotides [13]. ROS induce chemical reactions, which lead to the oxidative modification of RNA.
Due to the following four reasons, the brain is more susceptible to oxidative damage. (1) High oxygen consumption of the brain: the oxygen consumption of the human brain accounts for 20–25% of the total oxygen consumption of the human body [14]. (2) The brain is prone to high levels of polyunsaturated fatty acids [14]. (3) High levels of transition metals in the brain can catalyze the reaction that produces ROS [15]. (4) Compared with other organs, the brain is relatively lacking in antioxidant enzymes: for example, the content of catalase in the brain is only 10–20% of the liver and heart [16]. At this time, superoxide (·-O2), hydrogen peroxide (H2O2), and the hydroxyl radical (·OH) produced by the normal mitochondrial electron transport chain through normal cell metabolism will threaten neurons, leaving the neurons of the brain continuously exposed to ROS [14][15][16]. The·OH can only diffuse a few nanometers in the tissue [17] and ·−O2 can hardly penetrate cell membrane [18]. When the level of ROS in the cell exceeds the defense ability of the cell, the RNA widely present in the cytoplasm is continuously in contact with the ROS and is attacked by the ROS, causing oxidative damage to the RNA. In general, we do not pay much attention to the cause of RNA damage because maybe that damaged molecules will not accumulate due to the transient nature of RNA. However, the damage caused by ROS occurs within a few minutes, and the average half-life of most people's mRNA can last for 10 hours [1]. In addition, stable RNA species (mainly rRNA and tRNA) constitute the majority of cellular RNA and will not degrade during exponential growth [19]. Therefore, the damaged RNA will accumulate in the cell and pose a challenging problem for the cell. In the brain of AD patients, dysfunctional mitochondria may produce large amounts of ROS [20], and 8-oxo-guanosine (8-oxoGuo) levels were detected in the brain of AD patients after death [21]. All of these suggest that the formation of ROS that causes RNA oxidation in AD is likely to be the cause of mitochondrial abnormalities. Interestingly, the phenomenon of mitochondrial abnormalities [22] is also found in the substantia nigra of PD, which makes this mechanism a common theme in the cascade of neurodegenerative diseases [23].
3. Mechanism of RNA Oxidation
3.1. Structural Changes in Oxidized RNAs
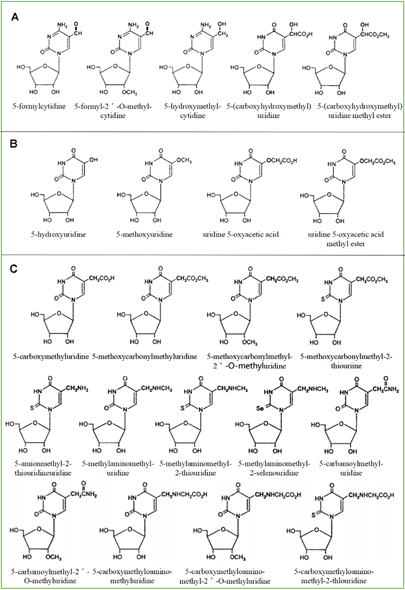
As a product of normal metabolism and obtaining from exogenous sources, living cells produce highly active -OH, which is likely to cause a lot of the oxidative damage of biological macromolecules including RNAs. The most common source of -OH in cells is the Fenton and Haber-Weiss reaction [24]. −OH generated near RNA can readily modify RNA because they are so active that they cannot diffuse from where they are formed. Therefore, the modification caused by the hydroxyl group is the most diverse type of RNA damage. There are more than 20 distinct types of -OH that destroy substrates [25]. Currently, 8-hydroxyguanosine (8-OHG) is the most common biomarker that can indicate RNA oxidation. The highly active hydroxyl radical first reacts with guanine to form C8-OH adducts and then produces 8-OHG, accompanied by the loss of an electron (e−) and proton (H+). According to the chemical structure of all known modified RNA nucleosides, they are divided into four categories base on the following chemical properties [24]: (1) The size of the modification group: small or large, like methyl- versus isopentenyl- groups. (2) The nature of basic substituents: simple or derivatives of other natural ingredients like amino acids and sugars and thio-threonine derivatives. (3) Type of modification: primary or secondary, such as adenine isoprenylation and Q or Y nucleoside side-chain substitution. (4) Synthetic pathway: enzymatic pathway (prenylation and methylation) or random pathway (hydroxylation or peroxidation). Among all known RNAs, tRNA is mainly responsible for carrying amino acids into ribosomes, synthesizing proteins under the guidance of mRNA, and can also participate in DNA synthesis by serving as reverse transcriptase primers. One of the structural characteristics of tRNA is that it contains more modified components, and most of the modified components in nucleic acids are found in tRNA [26]. The modified bases and nucleosides found in tRNA have the following characteristics: (1) modified bases and nucleosides tend to appear in many tRNA at position 34 (Swing position, when the anticodon of tRNA is paired with the codon of mRNA, the first two pairs strictly abide by the rule of base pairing, but the third pair of bases has a certain degree of freedom to "swing"); (2) it can produce pyrimidine residues, most of which are uridine derivatives; (3) it is easy to occur at position five outside the ring; (4) after modification, two uridine derivatives are generated in many cases (Figure 1). Based on their secondary reactions with formylcytosine (Figure 1A), hydroxyuridine (Figure 1B), and carboxyuridine (Figure 1C), these RNA-modified nucleosides can be further divided into three subgroups. In fact, oxidative damage-induced cleavage and fragmentation of tRNA can be observed in yeast and human cell lines [27]. Under oxidizing conditions, tRNA is cleaved at the anticodon loop into half molecules of 30–45 nucleotides in length [28][29], known as tRNA-derived stress-inducing fragments (tiRNAs) [30]. This conformational change of tRNA can be used as a biomarker to identify organ damage or reflect clinical prognosis [31]. In addition, tRNAs are an essential part of the translation mechanism. Oxidative damage to tRNA may lead to defects in codon-anticodon pairing or aminoacylation, which may lead to the production of incorrectly encoded proteins [8][21].
Figure 1. Modified pyrimidine nucleosides found in tRNA. They are divided into three subgroups according to possible factors: formylcytosine (A), hydroxyuridine (B), and carboxyuridine (C).
3.2. Different Forms of RNA Oxidative Damage
We have known for decades that poly (A)+mRNA accounts for only a small portion (about 1–2%) of the total cellular RNAs. RNA inside cells is primarily made up of rRNAs and tRNAs. Additionally, there are many different types of non-coding RNAs, such as microRNAs (miRNAs), small nucleolar RNAs (snoRNAs), and small nuclear RNAs (snRNAs) [32]. These non-coding RNAs play an essential role in mRNA splicing regulation, non-spliced RNA modification, and mRNA translation [33]. In regions affected by AD, which is the cytoplasm of hippocampal neurons, bound redox-active iron oxidizes rRNAs [34]. Oxidation of rRNA in the brains of patients with AD and mild cognitive impairment (MCI) is significantly increased, and oxidative damage also occurs in other types of cytoplasmic RNAs (such as tRNA and miRNA) in diseased tissues [35]. There are many forms of oxidative damage to RNA, which can affect the body in the following ways:
(1) Direct RNA strand breaks: Jacobs et al. found that the most common form of damage to RNA is the direct strand break [36]. The possible mechanism is the production of nucleic acid bases or their peroxyl groups, resulting in the removal of hydrogen atoms from adjacent ribose molecules, which in turn leads to a breakage of the RNA strand.
(2) Translation errors caused by oxidized RNA: Oxidized mRNA can induce translation errors, which can lead to premature termination of translation or degradation of peptide chains, resulting in short-chain polypeptides and protein variation [37]. In addition, oxidative changes of bases on mRNAs cause mismatches with the bases on tRNAs during translation and can result in protein variation.
(3) RNA oxidative damage can cause protein synthesis disorders: Experiments in rat primary nerve cell culture showed that the RNA in nerve cells is selectively oxidized when oxidative damage occurs. Generally, mRNA is more likely to be oxidized. During the transcription process, the presence of oxidized bases on the oxidized mRNA chain will cause transcription errors in the body, which will lead to the next translation error. Once an error occurs during translation, the expression of the corresponding protein will change. Ding et al. [38] demonstrated that once RNA is oxidized, protein synthesis in primary neurons and neural SH-SY5Y cells is significantly reduced. This decrease gradually increases with the extension of the oxidation time.
3.3. RNA Repair and Prevention of Oxidative Damage
In previous studies, it was believed that RNA only served as a messenger in the expression of genetic material, while ignoring the occurrence of oxidative damage to RNA may have important physiological and pathological effects [39]. Cells produce different repair mechanisms when dealing with different forms of RNA damage. Cells have evolved a variety of mRNA monitoring and control mechanisms to eliminate false transcripts, including nonsense-mediated mRNA decay (NMD), mRNA without stop codons, and degradation of translationally blocked mRNAs wait [40]. The oxidative modification of RNA is also affected by the RNA monitoring mechanism. Eukaryotic cells use NMD as the major mechanism to monitor RNA. Transcripts containing premature stop codons can be identified and degraded to avoid the accumulation of truncated protein products and reduce cytotoxicity [41]. Studies show that E. coli enzyme AlkB and human homologous enzyme hABH3 can directly reverse the alkylation damage of RNA by hydroxylating methyl groups on damaged RNA bases, and oxidized bases on RNA may be repaired [42].
4. Discussion and Future Perspectives
With the rapid development of technology, people gradually realize that RNA is more susceptible to oxidative damage, and the oxidative damage of RNA is related to the early development of various neurodegenerative diseases such as AD, PD, and ALS [7]. This phenomenon may represent a common theme of the pathogenesis of neurodegenerative diseases, and it interferes with the normal translation process, promotes the synthesis of variant proteins, and subsequently initiates inappropriate cell fate pathways, eventually leading to various neurodegenerative diseases [4]. In this review, we emphasize that the appearance of RNA oxidative damage can suggest the occurrence of neurodegenerative diseases. Current research has confirmed that 8-OHG can be used as a marker of RNA oxidative damage to indicate the degree of RNA oxidative damage in the body, providing some reference information for understanding the course and treatment effect of neurodegenerative diseases such as AD and PD [24]. Understanding the important role of RNA oxidative damage in the pathogenesis of neurodegenerative diseases and the possible cellular therapeutic mechanisms may provide clues for the treatment strategies of neurodegenerative diseases in the future.
Oxidative damage to RNA can lead to interruption of the translation process and impaired protein synthesis. However, we are still in the preliminary stage of understanding the role of RNA oxidation in protein synthesis inhibition and cell death. We believe that the direction of further research can focus on the following issues. Under normal physiological conditions, whether oxidatively damaged RNA is degraded or repaired by the body and what protein is involved in the process is a very challenging question, and thus further research is needed [43][44]. It may be possible to identify proteins involved in this process by isolating repair/degradation complexes and applying protein microarrays or proteomics analysis. How to prevent oxidative damage to RNA through transgenic or pharmacological methods is also a good research direction. In order to better understand the functional role of RNA oxidation in the pathogenesis of various diseases, it is necessary to further study the above mechanisms.
In addition, recent research on RNA oxidation has a more interesting point: RNA oxidation is a feature of aging brain neurons, and it is more prominently observed in the early fragile neurons of age-related neurodegenerative diseases [45]. This suggests that the aging process may be closely related to RNA oxidation and many researchers have conducted preliminary research on this phenomenon. Comparative experimental studies on the brains of young and old mice [46] and studies on the brains of accelerated aging mice [47] suggest that the increase in 8-OHG in neurons of the brain is related to age. These findings further support RNA oxidation involved in the process of aging and neurodegenerative diseases. However, only a few studies have elaborated on this phenomenon. Continued in-depth understanding of the consequences of RNA oxidative damage and the underlying mechanism of aging will help provide better anti-aging methods in the future.
References
- Li, Z.; Wu, J.; DeLeo, C.J. RNA damage and surveillance under oxidative stress. IUBMB Life 2006, 58, 581–588, doi:10.1080/15216540600946456.
- Wurtmann, E.J.; Wolin, S.L. RNA under attack: Cellular handling of RNA damage. Crit. Rev. Biochem. Mol. Boil. 2009, 44, 34–49, doi:10.1080/10409230802594043.
- Pérez-Cano, L.; Fernandez-Recio, J. Dissection and prediction of RNA-binding sites on proteins. Biomol. Concepts 2010, 1, 345–355, doi:10.1515/bmc.2010.037.
- Nunomura, A.; Moreira, P.; Castellani, R.J.; Lee, H.-G.; Zhu, X.; Smith, M.A.; Perry, G. Oxidative Damage to RNA in Aging and Neurodegenerative Disorders. Neurotox. Res. 2012, 22, 231–248, doi:10.1007/s12640-012-9331-x.
- Kilchert, C.; Sträßer, K.; Kunetsky, V.; Änkö, M.-L. From parts lists to functional significance—RNA–protein interactions in gene regulation. Wiley Interdiscip. Rev. RNA 2020, 11, e1582, doi:10.1002/wrna.1582.
- Essack, M.; Salhi, A.; Van Neste, C.; Bin Raies, A.; Tifratene, F.; Uludag, M.; Hungler, A.; Zaric, B.; Zafirovic, S.; Gojobori, T.; et al. DES-ROD: Exploring Literature to Develop New Links between RNA Oxidation and Human Diseases. Oxidative Med. Cell. Longev. 2020, 2020, 5904315–13, doi:10.1155/2020/5904315.
- Nunomura, A.; Hofer, T.; Moreira, P.; Castellani, R.J.; Smith, M.A.; Perry, G. RNA oxidation in Alzheimer disease and related neurodegenerative disorders. Acta Neuropathol. 2009, 118, 151–166, doi:10.1007/s00401-009-0508-1.
- Nunomura, A.; Lee, H.-G.; Zhu, X.; Perry, G. Consequences of RNA oxidation on protein synthesis rate and fidelity: Implications for the pathophysiology of neuropsychiatric disorders. Biochem. Soc. Trans. 2017, 45, 1053–1066, doi:10.1042/bst20160433.
- Abe, T.; Tohgi, H.; Isobe, C.; Murata, T.; Sato, C. Remarkable increase in the concentration of 8-hydroxyguanosine in cerebrospinal fluid from patients with Alzheimer’s disease. J. Neurosci. Res. 2002, 70, 447–450, doi:10.1002/jnr.10349.
- Hayashi, M.; Araki, S.; Kohyama, J.; Shioda, K.; Fukatsu, R. Oxidative nucleotide damage and superoxide dismutase expression in the brains of xeroderma pigmentosum group A and Cockayne syndrome. Brain Dev. 2005, 27, 34–38, doi:10.1016/j.braindev.2004.04.001.
- Shi, F.; Nie, B.; Gan, W.; Zhou, X.-Y.; Takagi, Y.; Hayakawa, H.; Sekiguchi, M.; Cai, J.-P. Oxidative damage of DNA, RNA and their metabolites in leukocytes, plasma and urine ofMacaca mulatta: 8-oxoguanosine in urine is a useful marker for aging. Free. Radic. Res. 2012, 46, 1093–1098, doi:10.3109/10715762.2012.689428.
- Santana, I.; Sá, A.; Guerreiro, R.; Bras, J.; Hardy, J.; Rogaeva, E.; Oliveira, C. P1-348: Early onset familial Alzheimer’s disease: First Portuguese cases. Alzheimer’s Dement. 2006, 2, S199, doi:10.1016/j.jalz.2006.05.726.
- Cadet, J. Oxidative damage to DNA: Formation, measurement and biochemical features. Mutat. Res./Fundam. Mol. Mech. Mutagen. 2003, 531, 5–23, doi:10.1016/j.mrfmmm.2003.09.001.
- Coyle, J.; Puttfarcken, P. Oxidative stress, glutamate, and neurodegenerative disorders. Science 1993, 262, 689–695, doi:10.1126/science.7901908.
- Mattson, M.P.; Chan, S.L.; Duan, W. Modification of Brain Aging and Neurodegenerative Disorders by Genes, Diet, and Behavior. Physiol. Rev. 2002, 82, 637–672, doi:10.1152/physrev.00004.2002.
- Halliwell, B. Reactive Oxygen Species and the Central Nervous System. J. Neurochem. 1992, 59, 1609–1623, doi:10.1111/j.1471-4159.1992.tb10990.x.
- Joenje, H. Genetic toxicology of oxygen. Mutat. Res. 1989, 219, 193–208, doi:10.1016/0921-8734(89)90001-5.
- Takahashi, M.-A.; Asada, K. Superoxide anion permeability of phospholipid membranes and chloroplast thylakoids. Arch. Biochem. Biophys. 1983, 226, 558–566, doi:10.1016/0003-9861(83)90325-9.
- Deutscher, M.P. Degradation of RNA in bacteria: Comparison of mRNA and stable RNA. Nucleic Acids Res. 2006, 34, 659–666, doi:10.1093/nar/gkj472.
- Hirai, K.; Aliev, G.; Nunomura, A.; Fujioka, H.; Russell, R.L.; Atwood, C.S.; Johnson, A.B.; Kress, Y.; Vinters, H.V.; Tabaton, M.; et al. Mitochondrial Abnormalities in Alzheimer’s Disease. J. Neurosci. 2001, 21, 3017–3023, doi:10.1523/JNEUROSCI.21-09-03017.2001.
- Nunomura, A.; Perry, G.; Pappolla, M.A.; Wade, R.; Hirai, K.; Chiba, S.; Smith, M.A. RNA Oxidation Is a Prominent Feature of Vulnerable Neurons in Alzheimer’s Disease. J. Neurosci. 1999, 19, 1959–1964.
- Schapira, A.H.; Cooper, J.; Dexter, D.; Clark, J.B.; Jenner, P.; Marsden, C.D. Mitochondrial Complex I Deficiency in Parkinson’s Disease. J. Neurochem. 1990, 54, 823–827, doi:10.1111/j.1471-4159.1990.tb02325.x.
- Nunomura, A.; Zhu, X.; Perry, G. Modulation of Parkinson’s Disease Associated Protein Rescues Alzheimer’s Disease Degeneration. J. Alzheimer’s Dis. 2016, 55, 73–75, doi:10.3233/jad-160878.
- Barciszewski, J.; Barciszewska, M.Z.; Siboska, G.; Rattan, S.; Clark, B.F. Some unusual nucleic acid bases are products of hydroxyl radical oxidation of DNA and RNA. Mol. Boil. Rep. 1999, 26, 231–238, doi:10.1023/a:1007058602594.
- Wiseman, H.; Halliwell, B. Damage to DNA by reactive oxygen and nitrogen species: Role in inflammatory disease and progression to cancer. Biochem. J. 1996, 313, 17–29, doi:10.1042/bj3130017.
- McCown, P.J.; Ruszkowska, A.; Kunkler, C.N.; Breger, K.; Hulewicz, J.P.; Wang, M.C.; Springer, N.A.; Brown, J.A. Naturally occurring modified ribonucleosides. Wiley Interdiscip. Rev. RNA 2020, e1595, doi:10.1002/wrna.1595.
- Thompson, D.M.; Lu, C.; Green, P.J.; Parker, R. tRNA cleavage is a conserved response to oxidative stress in eukaryotes. RNA 2008, 14, 2095–2103, doi:10.1261/rna.1232808.
- Yamasaki, S.; Ivanov, P.; Hu, G.-F.; Anderson, P. Angiogenin cleaves tRNA and promotes stress-induced translational repression. J. Cell Boil. 2009, 185, 35–42, doi:10.1083/jcb.200811106.
- Fu, H.; Feng, J.; Liu, Q.; Sun, F.; Tie, Y.; Zhu, J.; Xing, R.; Sun, Z.; Zheng, X. Stress induces tRNA cleavage by angiogenin in mammalian cells. FEBS Lett. 2008, 583, 437–442, doi:10.1016/j.febslet.2008.12.043.
- Mishima, E.; Inoue, C.; Saigusa, D.; Inoue, R.; Ito, K.; Suzuki, Y.; Jinno, D.; Tsukui, Y.; Akamatsu, Y.; Araki, M.; et al. Conformational Change in Transfer RNA Is an Early Indicator of Acute Cellular Damage. J. Am. Soc. Nephrol. 2014, 25, 2316–2326, doi:10.1681/ASN.2013091001.
- Simms, C.; Zaher, H.S. Quality control of chemically damaged RNA. Cell. Mol. Life Sci. 2016, 73, 3639–3653, doi:10.1007/s00018-016-2261-7.
- Dahariya, S.; Paddibhatla, I.; Kumar, S.; Raghuwanshi, S.; Pallepati, A.; Gutti, R.K. Long non-coding RNA: Classification, biogenesis and functions in blood cells. Mol. Immunol. 2019, 112, 82–92, doi:10.1016/j.molimm.2019.04.011.
- Cao, X.; Yeo, G.; Muotri, A.R.; Kuwabara, T.; Gage, F.H. NONCODING RNAS IN THE MAMMALIAN CENTRAL NERVOUS SYSTEM. Annu. Rev. Neurosci. 2006, 29, 77–103, doi:10.1146/annurev.neuro.29.051605.112839.
- Honda, K.; Smith, M.A.; Zhu, X.; Baus, D.; Merrick, W.C.; Tartakoff, A.M.; Hattier, T.; Harris, P.L.; Siedlak, S.L.; Fujioka, H.; et al. Ribosomal RNA in Alzheimer Disease Is Oxidized by Bound Redox-active Iron. J. Boil. Chem. 2005, 280, 20978–20986, doi:10.1074/jbc.m500526200.
- Shan, X.; Chang, Y.; Lin, C.-L.G. Messenger RNA oxidation is an early event preceding cell death and causes reduced protein expression. FASEB J. 2007, 21, 2753–2764, doi:10.1096/fj.07-8200com.
- Jacobs, A.C.; Resendiz, M.J.E.; Greenberg, M.M. Direct Strand Scission from a Nucleobase Radical in RNA. J. Am. Chem. Soc. 2010, 132, 3668–3669, doi:10.1021/ja100281x.
- Tanaka, M.; Chock, P.B.; Stadtman, E.R. Oxidized messenger RNA induces translation errors. Proc. Natl. Acad. Sci. USA 2006, 104, 66–71, doi:10.1073/pnas.0609737104.
- Ding, Q.; Dimayuga, E.; Bruce-Keller, A.J. Oxidative stress alters neuronal RNA- and protein-synthesis: Implications for neural viability. Free. Radic. Res. 2007, 41, 903–910, doi:10.1080/10715760701416996.
- Yan, L.L.; Zaher, H.S. How do cells cope with RNA damage and its consequences? J. Boil. Chem. 2019, 294, 15158–15171, doi:10.1074/jbc.REV119.006513.
- Pawlicka, K.; Kalathiya, U.; Alfaro, J. Nonsense-Mediated mRNA Decay: Pathologies and the Potential for Novel Therapeutics. Cancers 2020, 12, 765, doi:10.3390/cancers12030765.
- Kishor, A.; Fritz, S.E.; Hogg, J.R. Nonsense‐mediated mRNA decay: The challenge of telling right from wrong in a complex transcriptome. Wiley Interdiscip. Rev. RNA 2019, 10, e1548, doi:10.1002/wrna.1548.
- Feyzi, E.; Sundheim, O.; Westbye, M.; Aas, P.; Vågbø, C.B.; Otterlei, M.; Slupphaug, G.; Krokan, H. RNA Base Damage and Repair. Curr. Pharm. Biotechnol. 2007, 8, 326–331, doi:10.2174/138920107783018363.
- Brégeon, D.; Sarasin, A. Hypothetical role of RNA damage avoidance in preventing human disease. Mutat. Res. Mol. Mech. Mutagen. 2005, 577, 293–302, doi:10.1016/j.mrfmmm.2005.04.002.
- Bellacosa, A.; Moss, E.G. RNA Repair: Damage Control. Curr. Boil. 2003, 13, R482–R484, doi:10.1016/s0960-9822(03)00408-1.
- Nunomura, A.; Tamaoki, T.; Motohashi, N.; Nakamura, M.; McKeel, D.W.; Tabaton, M.; Lee, H.-G.; Smith, M.A.; Perry, G.; Zhu, X. The earliest stage of cognitive impairment in transition from normal aging to Alzheimer disease is marked by prominent RNA oxidation in vulnerable neurons. J. Neuropathol. Exp. Neurol. 2012, 71, 233–241, doi:10.1097/NEN.0b013e318248e614.
- Cui, L.; Hofer, T.; Rani, A.; Leeuwenburgh, C.; Foster, T.C. Comparison of lifelong and late life exercise on oxidative stress in the cerebellum. Neurobiol. Aging 2009, 30, 903–909, doi:10.1016/j.neurobiolaging.2007.09.005.
- Song, X.-N.; Zhang, L.-Q.; Liu, D.-G.; Lin, J.; Zheng, J.-D.; Dai, D.-P.; Hei, A.-L.; Hayakawa, H.; Sekiguchi, M.; Cai, J.-P. Oxidative Damage to RNA and Expression Patterns of MTH1 in the Hippocampi of Senescence-Accelerated SAMP8 Mice and Alzheimer’s Disease Patients. Neurochem. Res. 2011, 36, 1558–1565, doi:10.1007/s11064-011-0484-4.