
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Erum Shoeb | + 2991 word(s) | 2991 | 2021-08-06 11:41:47 | | | |
| 2 | Conner Chen | Meta information modification | 2991 | 2021-09-16 03:21:32 | | |
Video Upload Options
Naturally occurring viral nanomaterials have gained popularity owing to their biocompatible and biodegradable nature. Plant virus nanoparticles (VNPs) can be used as nanocarriers for a number of biomedical applications. Plant VNPs are inexpensive to produce, safe to administer and efficacious as treatments. Features which distinguish plant viruses from synthetic nanocarriers include stability, flexibility, diversity in shape and size for use in drug delivery and the nontoxic nature of plant viruses in humans. Cancer is one of the most common death causing disease worldwide and it is characterized by uncontrolled rapid cell division and differentiation. VNPs are an ideal choice to apply for cancer treatment owing to the enhanced permeation and retention (EPR) potential of cancer cells for these nanoparticles, whereas VNPs cannot penetrate through healthy tissues due to tightly packed endothelial cells.
1. Plant Virus Architecture
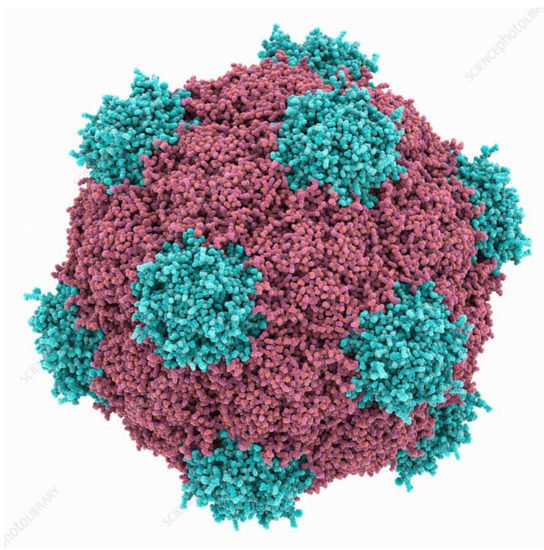
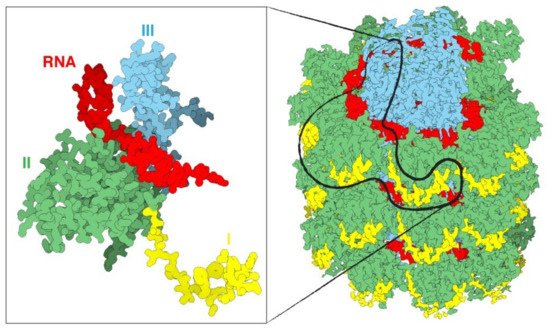
2. Biomedical Applications of Plant Virus Nanoparticles
References
- Nikitin, N.; Trifonova, E.; Evtushenko, E.; Kirpichnikov, M.; Atabekov, J.; Karpova, O. Comparative Study of Non-Enveloped Icosahedral Viruses Size. PLoS ONE 2015, 10, e0142415.
- Aljabali, A.A.; Shah, S.N.; Evans-Gowing, R.; Lomonossoff, G.P.; Evans, D.J. Chemically-coupled-peptide-promoted virus nanoparticle templated mineralization. Integr. Biol. (Camb.) 2011, 3, 119–125.
- Nikitin, N.; Trifonova, E.; Karpova, O.; Atabekov, J. Examination of biologically active nanocomplexes by nanoparticle tracking analysis. Microsc. Microanal. 2013, 19, 808–813.
- Petrova, E.K.; Nikitin, N.A.; Trifonova, E.A.; Protopopova, A.D.; Karpova, O.V.; Atabekov, J.G. The 5′-proximal region of Potato virus X RNA involves the potential cap-dependent “conformational element” for encapsidation. Biochimie 2015, 115, 116–119.
- Zhou, J.C.; Soto, C.M.; Chen, M.S.; Bruckman, M.A.; Moore, M.H.; Barry, E.; Ratna, B.R.; Pehrsson, P.E.; Spies, B.R.; Confer, T.S. Biotemplating rod-like viruses for the synthesis of copper nanorods and nanowires. J. Nanobiotechnology 2012, 10, 18.
- Steinmetz, N.F.; Shah, S.N.; Barclay, J.E.; Rallapalli, G.; Lomonossoff, G.P.; Evans, D.J. Virus-templated silica nanoparticles. Small 2009, 5, 813–816.
- Liu, Z.; Qiao, J.; Niu, Z.; Wang, Q. Natural supramolecular building blocks: From virus coat proteins to viral nanoparticles. Chem. Soc. Rev. 2012, 41, 6178–6194.
- Pokorski, J.K.; Steinmetz, N.F. The art of engineering viral nanoparticles. Mol. Pharm. 2011, 8, 29–43.
- Kaiser, C.R.; Flenniken, M.L.; Gillitzer, E.; Harmsen, A.L.; Harmsen, A.G.; Jutila, M.A.; Douglas, T.; Young, M.J. Biodistribution studies of protein cage nanoparticles demonstrate broad tissue distribution and rapid clearance in vivo. Int. J. Nanomed. 2007, 2, 715–733.
- Flynn, C.E.; Lee, S.-W.; Peelle, B.R.; Belcher, A.M. Viruses as vehicles for growth, organization and assembly of materials11The Golden Jubilee Issue—Selected topics in Materials Science and Engineering: Past, Present and Future, edited by S. Suresh. Acta Mater. 2003, 51, 5867–5880.
- Wen, A.M.; Wang, Y.; Jiang, K.; Hsu, G.C.; Gao, H.; Lee, K.L.; Yang, A.C.; Yu, X.; Simon, D.I.; Steinmetz, N.F. Shaping bio-inspired nanotechnologies to target thrombosis for dual optical-magnetic resonance imaging. J. Mater. Chem. B 2015, 3, 6037–6045.
- Rong, J.; Niu, Z.; Lee, L.A.; Wang, Q. Self-assembly of viral particles. Curr. Opin. Colloid Interface Sci. 2011, 16, 441–450.
- Sokullu, E.; Soleymani Abyaneh, H.; Gauthier, M.A. Plant/Bacterial Virus-Based Drug Discovery, Drug Delivery, and Therapeutics. Pharmaceutics 2019, 11, 211.
- Narayanan, K.; Han, S.S. Icosahedral plant viral nanoparticles—Bioinspired synthesis of nanomaterials/nanostructures. Adv. Colloid Interface Sci. 2017, 248.
- Shukla, S.; Ablack, A.L.; Wen, A.M.; Lee, K.L.; Lewis, J.D.; Steinmetz, N.F. Increased tumor homing and tissue penetration of the filamentous plant viral nanoparticle Potato virus X. Mol. Pharm. 2013, 10, 33–42.
- Zeng, Q.; Wen, H.; Wen, Q.; Chen, X.; Wang, Y.; Xuan, W.; Liang, J.; Wan, S. Cucumber mosaic virus as drug delivery vehicle for doxorubicin. Biomaterials 2013, 34, 4632–4642.
- Pokorski, J.K.; Breitenkamp, K.; Liepold, L.O.; Qazi, S.; Finn, M.G. Functional virus-based polymer-protein nanoparticles by atom transfer radical polymerization. J. Am. Chem. Soc. 2011, 133, 9242–9245.
- Bruckman, M.A.; Hern, S.; Jiang, K.; Flask, C.A.; Yu, X.; Steinmetz, N.F. Tobacco mosaic virus rods and spheres as supramolecular high-relaxivity MRI contrast agents. J. Mater. Chem. B 2013, 1, 1482–1490.
- Wen, J.; Xu, Y.; Li, H.; Lu, A.; Sun, S. Recent applications of carbon nanomaterials in fluorescence biosensing and bioimaging. Chem. Commun. 2015, 51, 11346–11358.
- Niehl, A.; Appaix, F.; Bosca San Jose, S.; van der Sanden, B.; Nicoud, J.-F.; Bolze, F.; Heinlein, M. Fluorescent Tobacco mosaic virus-Derived Bio-Nanoparticles for Intravital Two-Photon Imaging. Front. Plant Sci. 2016, 6.
- Hu, H.; Zhang, Y.; Shukla, S.; Gu, Y.; Yu, X.; Steinmetz, N.F. Dysprosium-Modified Tobacco Mosaic Virus Nanoparticles for Ultra-High-Field Magnetic Resonance and Near-Infrared Fluorescence Imaging of Prostate Cancer. ACS Nano 2017, 11, 9249–9258.
- Li, R.; Wu, W.; Liu, Q.; Wu, P.; Xie, L.; Zhu, Z.; Yang, M.; Qian, X.; Ding, Y.; Yu, L.; et al. Intelligently targeted drug delivery and enhanced antitumor effect by gelatinase-responsive nanoparticles. PLoS ONE 2013, 8, e69643.
- Li, R.; Xie, L.; Zhu, Z.; Liu, Q.; Hu, Y.; Jiang, X.; Yu, L.; Qian, X.; Guo, W.; Ding, Y.; et al. Reversion of pH-induced physiological drug resistance: A novel function of copolymeric nanoparticles. PLoS ONE 2011, 6, e24172.
- Beatty, P.H.; Lewis, J.D. Cowpea mosaic virus nanoparticles for cancer imaging and therapy. Adv. Drug Deliv. Rev. 2019, 145, 130–144.
- Ma, Y.-Y.; Jin, K.-T.; Wang, S.-B.; Wang, H.-J.; Tong, X.-M.; Huang, D.-S.; Mou, X.-Z. Molecular Imaging of Cancer with Nanoparticle-Based Theranostic Probes. Contrast Media Mol. Imaging 2017, 2017, 1026270.
- Key, J.; Leary, J.F. Nanoparticles for multimodal in vivo imaging in nanomedicine. Int. J. Nanomed. 2014, 9, 711–726.
- Hansen, A.E.; Petersen, A.L.; Henriksen, J.R.; Boerresen, B.; Rasmussen, P.; Elema, D.R.; Rosenschöld, P.M.A.; Kristensen, A.T.; Kjær, A.; Andresen, T.L. Positron Emission Tomography Based Elucidation of the Enhanced Permeability and Retention Effect in Dogs with Cancer Using Copper-64 Liposomes. ACS Nano 2015, 9, 6985–6995.
- Shoeb, E.; Hefferon, K. Future of cancer immunotherapy using plant virus-based nanoparticles. Future Sci. OA 2019, 5, FSO401.
- Chroboczek, J.; Szurgot, I.; Szolajska, E. Virus-like particles as vaccine. Acta. Biochim. Pol. 2014, 61, 531–539.
- Chung, H.-J.; Islam, S.; Rahman, M.; Hong, S.-T. Neuroprotective function of Omi to α-synuclein-induced neurotoxicity. Neurobiol. Dis. 2020, 136, 104706.
- Bachmann, M.F.; Jennings, G.T. Vaccine delivery: A matter of size, geometry, kinetics and molecular patterns. Nat. Rev. Immunol. 2010, 10, 787–796.
- Lebel, C.; Treit, S.; Beaulieu, C. Diffusion MRI of typical white matter development from childhood to adulthood. NMR Bio-Medicine 2017, in press.
- Shukla, A.K.; Spurrier, J.; Kuzina, I.; Giniger, E. Hyperactive Innate Immunity Causes Degeneration of Dopamine Neurons upon Altering Activity of Cdk5. Cell Rep. 2019, 26, 131–144.
- Jobsri, J.; Allen, A.; Rajagopal, D.; Shipton, M.; Kanyuka, K.; Lomonossoff, G.P.; Ottensmeier, C.; Diebold, S.S.; Stevenson, F.; Savelyeva, N. Plant Virus Particles Carrying Tumour Antigen Activate TLR7 and Induce High Levels of Protective Antibody. PLoS ONE 2015, 10, e0118096.
- Miermont, A.; Barnhill, H.; Strable, E.; Lu, X.; Wall, K.A.; Wang, Q.; Finn, M.G.; Huang, X. Cowpea Mosaic Virus Capsid: A Promising Carrier for the Development of Carbohydrate Based Antitumor Vaccines. Chem. A Eur. J. 2008, 14, 4939–4947.
- Yin, Z.; Nguyen, H.G.; Chowdhury, S.; Bentley, P.; Bruckman, M.A.; Miermont, A.; Gildersleeve, J.C.; Wang, Q.; Huang, X. Tobacco mosaic virus as a new carrier for tumor associated carbohydrate antigens. Bioconjug. Chem. 2012, 23, 1694–1703.
- Gajewski, T.F.; Schreiber, H.; Fu, Y.-X. Innate and adaptive immune cells in the tumor microenvironment. Nat. Immunol. 2013, 14, 1014–1022.
- Shukla, S.; Wang, C.; Beiss, V.; Cai, H.; Washington, T.; Murray, A.A.; Gong, X.; Zhao, Z.; Masarapu, H.; Zlotnick, A.; et al. The unique potency of Cowpea mosaic virus (CPMV) in situ cancer vaccine. Biomater. Sci. 2020, 8, 5489–5503.
- Lizotte, P.; Wen, A.M.; Sheen, M.R.; Fields, J.; Rojanasopondist, P.; Steinmetz, N.F.; Fiering, S. In situ vaccination with cowpea mosaic virus nanoparticles suppresses metastatic cancer. Nat. Nanotechnol. 2016, 11, 295–303.
- Kerstetter-Fogle, A.; Shukla, S.; Wang, C.; Beiss, V.; Harris, P.L.R.; Sloan, A.E.; Steinmetz, N.F.; Fogle, K. Wang Plant Virus-Like Particle In Situ Vaccine for Intracranial Glioma Immunotherapy. Cancers 2019, 11, 515.
- Czapar, A.E.; Tiu, B.D.B.; Veliz, F.A.; Pokorski, J.K.; Steinmetz, N.F. Slow-Release Formulation of Cowpea Mosaic Virus for In Situ Vaccine Delivery to Treat Ovarian Cancer. Adv. Sci. 2018, 5, 1700991.
- Le, D.H.T.; Lee, K.L.; Shukla, S.; Commandeur, U.; Steinmetz, N.F. Potato virus X, a filamentous plant viral nanoparticle for doxorubicin delivery in cancer therapy. Nanoscale 2017, 9, 2348–2357.
- Wang, C.; Steinmetz, N.F. CD47 Blockade and Cowpea Mosaic Virus Nanoparticle In Situ Vaccination Triggers Phagocytosis and Tumor Killing. Adv. Heal. Mater. 2019, 8, e1801288.
- Masarapu, H.; Patel, B.K.; Chariou, P.L.; Hu, H.; Gulati, N.M.; Carpenter, B.L.; Ghiladi, R.A.; Shukla, S.; Steinmetz, N.F. Physalis Mottle Virus-Like Particles as Nanocarriers for Imaging Reagents and Drugs. Biomacromolecules 2017, 18, 4141–4153.
- Cao, Y.; Ruan, Y.; Shen, T.; Huang, X.; Li, M.; Yu, W.; Zhu, Y.; Man, Y.; Wang, S.; Li, J. AstragalusPolysaccharide Suppresses Doxorubicin-Induced Cardiotoxicity by Regulating the PI3k/Akt and p38MAPK Pathways. Oxidative Med. Cell. Longev. 2014, 2014, 1–12.
- Finbloom, J.A.; Aanei, I.L.; Bernard, J.M.; Klass, S.H.; Elledge, S.K.; Han, K.; Ozawa, T.; Nicolaides, T.P.; Berger, M.S.; Francis, M.B. Evaluation of Three Morphologically Distinct Virus-Like Particles as Nanocarriers for Convection-Enhanced Drug Delivery to Glioblastoma. Nanomaterials 2018, 8, 1007.
- Pitek, A.S.; Jameson, S.A.; Veliz, F.A.; Shukla, S.; Steinmetz, N.F. Serum albumin ‘camouflage’ of plant virus based nanoparticles prevents their antibody recognition and enhances pharmacokinetics. Biomaterials 2016, 89, 89–97.
- Bruckman, M.A.; Czapar, A.E.; VanMeter, A.; Randolph, L.N.; Steinmetz, N.F. Tobacco mosaic virus-based protein nanoparticles and nanorods for chemotherapy delivery targeting breast cancer. J. Control. Release 2016, 231, 103–113.
- Franke, C.E.; Czapar, A.E.; Patel, R.; Steinmetz, N.F. Tobacco Mosaic Virus-Delivered Cisplatin Restores Efficacy in Platinum-Resistant Ovarian Cancer Cells. Mol. Pharm. 2017, 15, 2922–2931.
- Vernekar, A.; Berger, G.; Czapar, A.E.; Veliz, F.A.; Wang, D.I.; Steinmetz, N.F.; Lippard, S.J. Speciation of Phenanthriplatin and Its Analogs in the Core of Tobacco Mosaic Virus. J. Am. Chem. Soc. 2018, 140, 4279–4287.
- Liu, X.; Wu, F.; Tian, Y.; Wu, M.; Zhou, Q.; Jiang, S.; Niu, Z. Size Dependent Cellular Uptake of Rod-like Bionanoparticles with Different Aspect Ratios. Sci. Rep. 2016, 6, 24567.
- Lin, R.D.; Steinmetz, N.F. Tobacco mosaic virus delivery of mitoxantrone for cancer therapy. Nanoscale 2018, 10, 16307–16313.
- Lam, P.; Lin, R.D.; Steinmetz, N.F. Delivery of mitoxantrone using a plant virus-based nanoparticle for the treatment of glioblastomas. J. Mater. Chem. B 2018, 6, 5888–5895.
- Kernan, K.F.; A Carcillo, J. Hyperferritinemia and inflammation. Int. Immunol. 2017, 29, 401–409.
- Tian, Y.; Zhou, M.; Shi, H.; Gao, S.; Xie, G.; Zhu, M.; Wu, M.; Chen, J.; Niu, Z. Integration of Cell-Penetrating Peptides with Rod-like Bionanoparticles: Virus-Inspired Gene-Silencing Technology. Nano Lett. 2018, 18, 5453–5460.
- Medeiros, S.D.O.; Martins, A.N.; Dias, C.G.A.; Tanuri, A.; Brindeiro, R.D.M. Natural transmission of feline immunodeficiency virus from infected queen to kitten. Virol. J. 2012, 9, 99.
- Comellas-Aragonès, M.; Engelkamp, H.; Claessen, V.I.; Sommerdijk, N.A.J.M.; Rowan, A.E.; Christianen, P.C.M.; Maan, J.C.; Verduin, B.J.M.; Cornelissen, J.J.L.M.; Nolte, R.J.M. A virus-based single-enzyme nanoreactor. Nat. Nanotechnol. 2007, 2, 635–639.
- Koyani, C.N.; Kolesnik, E.; Woelkart, G.; Shrestha, N.; Scheruebel, S.; Trummer, C.; Zorn-Pauly, K.; Hammer, A.; Lang, P.; Reicher, H.; et al. Dipeptidyl peptidase-4 independent cardiac dysfunction links saxagliptin to heart failure. Biochem. Pharmacol. 2017, 145, 64–80.
- Sánchez, Y.; Segura, V.; Marín-Béjar, O.; Athie, A.; Marchese, F.; González, J.; Bujanda, L.; Guo, S.; Matheu, A.; Huarte, M. Genome-wide analysis of the human p53 transcriptional network unveils a lncRNA tumour suppressor signature. Nat. Commun. 2014, 5, 5812.
- Strable, E.; Finn, M.G. Chemical Modification of Viruses and Virus-Like Particles. Curr. Top. Microbiol. Immunol. 2009, 327, 1–21.
- Röder, J.; Dickmeis, C.; Fischer, R.; Commandeur, U. Systemic Infection of Nicotiana benthamiana with Potato virus X Nanoparticles Presenting a Fluorescent iLOV Polypeptide Fused Directly to the Coat Protein. BioMed Res. Int. 2018, 2018, 1–12.





