
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Yoon-Yen Yow | + 3070 word(s) | 3070 | 2021-09-07 11:55:54 | | | |
| 2 | Lindsay Dong | Meta information modification | 3070 | 2021-09-14 04:37:04 | | |
Video Upload Options
Despite the progressive advances, current standards of treatments for peripheral nerve injury do not guarantee complete recovery. Thus, alternative therapeutic interventions should be considered. Complementary and alternative medicines (CAMs) are widely explored for their therapeutic value, but their potential use in peripheral nerve regeneration is underappreciated. Various CAMs enhanced proliferation and migration of Schwann cells in vitro, primarily through activation of MAPK pathway and FGF-2 signaling, respectively. Animal studies demonstrated the ability of CAMs to promote peripheral nerve regeneration and functional recovery, which are partially associated with modulations of neurotrophic factors, pro-inflammatory cytokines, and anti-apoptotic signaling.
1. Introduction
2. Complementary and alternative medicines (CAMs) and Peripheral Nerve Regeneration
2.1. Current Therapeutic Approaches against Peripheral Nerve Injuries
2.2. Mechanisms of Peripheral Nerve Injury and Regeneration
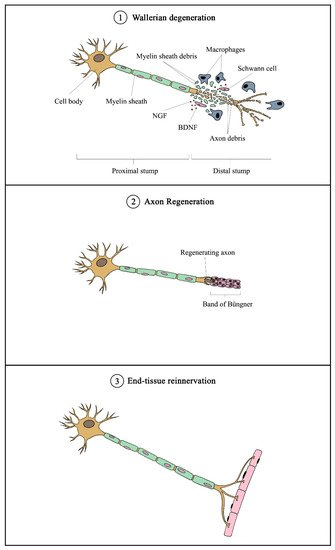
2.3. Role of Schwann Cells in Nerve Regeneration
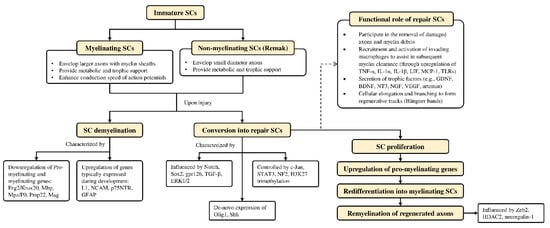
Schwann cells are supportive glial cells that are known to play a pivotal role in the proper functioning and maintenance of peripheral nerves. They are responsible for producing the basal lamina that determines the polarity of SCs and myelinating axons [35]. The myelin sheaths on axons allow the conduction of action potentials at high velocity via the formation of specialized nodes of Ranvier [36]. The high plasticity of SCs allows them to further develop into repair phenotypes in response to nerve injury (Figure 2). Following nerve injury, SCs can re-differentiate into repair SCs that align themselves to form bands of Büngner. This in turn allows axons to emerge from growth cones proximal to the injured site, which then elongate along the bands until the target organ is reinnervated. The repair SCs also participate in the removal of axon and myelin debris, and they can recruit macrophages to assist in the process [37]. In addition, repair SCs can also secrete neurotrophic factors that help promote cellular survival, proliferation, and differentiation, which are all essential for peripheral nerve repair [38]. Due to the importance of SCs in promoting peripheral nerve regeneration, it is expected that any disruption in SC proliferation, such as that caused by impairment in cyclin D1, will affect nerve regeneration following injury [39]. However, findings from past studies suggest that axonal regeneration is independent of SC proliferation [40][41]. Nevertheless, considering the association of SCs with axonal elongation and myelination, it is reasonable to hypothesize that enhanced SC proliferation may lead to greater regenerative potential.
2.4. Experimental Strategies and Neuroprotective Effects of Complementary and Alternative Medicines (CAMs) against Peripheral Nerve Injury
2.4.1. CAMs with Neuroregenerative Potential
| Source | Natural product | Reference |
| Achyranthes bidentata | - | [43][44][45][46][47][48] |
| Alpinate Oxyphyllae Fructus (Alpinia oxyphylla Miq) | Protocatechuic acid | [72][73] |
| Astragalus membranaceus | Astragaloside IV | [49][50][51][52] |
| Centella asiatica | - | [74] |
| Citrus medica var. sarcodactylis | - | [75] |
| Codonopsis pilosula | - | [76] |
| Crocus sativus | Crocin | [77] |
| Curcuma longa | Curcumin | [77][53][54][55][56][57][58] |
| Honeybee | Propolis | [58] |
| Dioscoreae rhizoma | - | [78] |
| Epimedium | Icariin | [79][80] |
| Gardenia jasminoides Ellis |
Genipin | [81] |
| Gastrodia elata Blume | Gastrodin | [82] |
| Ginkgo biloba |
Ginkgo biloba extract (EGb 761) | [83][84] |
| Ginseng | Ginsenoside Rg1, ginsenoside Re | [59][60][61] |
| Green tea | (-)-Epigallocatechin-3-gallate (EGCG) | [85][86] |
| Can be found in a variety of plant species | Syringic acid | [65] |
| Can be found in a variety of plant species | Ursolic acid | [66] |
| Lycium barbarum | - | [87] |
| Can be found in a variety of plant species | Quercetin | [67] |
| Morus sp. | - | [88] |
| Pueraria lobata | Puerarin | [89][90] |
| Radix Hedysari | - | [91][92] |
| Rhodiola rosea L. | Salidroside | [93] |
| Scutellaria baicalensis Georgi |
Baicalin | [94] |
| Trigonella foenum-graecum |
- | [95] |
| Tripterygium wilfordii Hook. F. |
Triptolide | [96] |
| Amanita muscaria |
Muscimol | [97] |
| Hericium erinaceus |
- | [62][63][64] |
| Bogijetong | - | [71] |
| Buyang Huanwu | - | [98] |
| Jiaweibugan | - | [99] |
| Qian-Zheng-San | - | [100] |
2.4.2. In Vitro Studies on Neuroregenerative Potential of CAMs
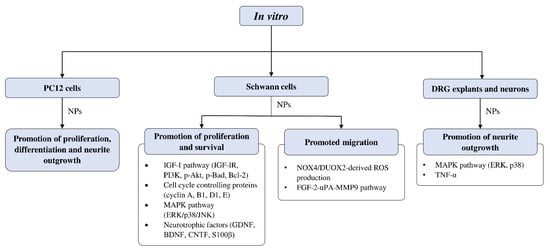
Effects of CAMs on Schwann Cell Activity In Vitro
The studies examining the effects of complementary and alternative medicines and their related natural products on Schwann cells are primarily focused on promoting their proliferation and survival. The molecular mechanisms that were investigated in these studies include signaling pathways such as IGF-I and MAPK, as well as cell cycle controlling proteins and various neurotrophic factors (Figure 3). Past studies have demonstrated that ERK is required for proper myelination of SCs during development [101][102], and ERK signaling was rapidly activated following nerve injury, contributing to SC differentiation [103]. Moreover, evidence suggests that nerve regeneration following injury is closely associated with ERK [104][105], and ERK inhibition leads to impaired regenerative capability [104][106]. On the other hand, inhibition of p38 MAPK prevented SC demyelination and dedifferentiation, indicating its role in promoting the breakdown of myelin following nerve injury [107]. It is not unexpected that cyclins are associated with SC proliferation, as these proteins control cell cycle progression through the interaction of cyclin-dependent kinases. For instance, cyclin D is associated with Cdk4 or Cdk6 in the G1 phase, cyclin A participates with Cdk1 or Cdk2 in the S phase, cyclin E is involved with Cdk2 in G1 and S phases, cyclin B and Cdk1 regulates M phase [108][109].
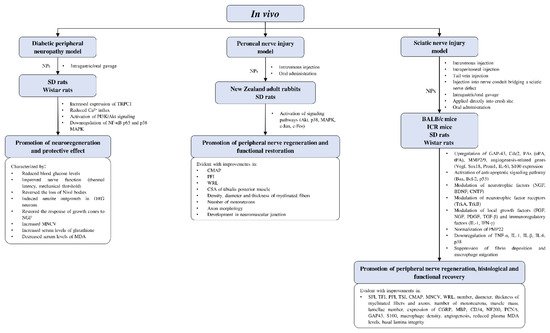
4.4.3. In Vivo Studies on Neuroregenerative Potential of CAMs
2.4.4. Involvement of CAMs in Combinatorial Approaches for the Treatment of PNI
There is increasing evidence that the successful repair and regeneration of nerves will require not just a single treatment strategy, but a multifaceted strategy involving different disciplines. Studies adopting combinatorial approaches have yielded interesting findings. For example, Lycium barbarum polysaccharide incorporated into core-shell structured nanofibrous scaffolds by coaxial electrospinning showed proliferative effects in PC12, SCs, and DRG neurons [87]. In two separate studies, puerarin, the active component extracted from Pueraria lobata roots, as well as rat serum metabolites of P. lobata enhanced the neuroregenerative effects of silicone rubber nerve chambers. Increase in myelinated axons and structurally mature regenerated axons were observed, while muscle reinnervation led to functional recovery, as indicated by an increase in action potential and nerve conduction [89][90]. Similar results were obtained with Buyang Huanwu decoction being administered as a co-treatment alongside silicone rubber nerve chambers, which led to more prominent axonal regeneration [98]. In an SNI model, a magnetic nanocomposite scaffold produced from using magnetic nanoparticles and biodegradable chitosan-glycerophosphate polymer enhanced SC viability, nerve regeneration, and functional recovery when paired with an applied magnetic field [110]. The use of nerve guiding conduits gained popularity over the years. They have been used to isolate regenerating axons from fibrotic tissues, to protect them from mechanical forces, and to guide new-forming tissue as well as condensing growth factors secreted by SCs [111]. The concept was initiated with a simple hollow design but has since advanced to innovative ways of redesigning nerve conduits to further extend their original capabilities 11. The attractive characteristics of modern nerve conduits offer tremendous potentials. These nerve conduits are occasionally paired with other strategies for improving nerve outcomes. For instance, Chang et al. [112] developed a natural biodegradable multi-channeled scaffold with aligned electrospun nanofibers and a neurotrophic gradient, which resulted in superior nerve recovery and less muscle atrophy compared with nerve autografts. Hussin et al. [74] used Centella asiatica (L.) to neurodifferentiate mesenchymal stem cells. This was subsequently developed with decellularized artery as a nerve conduit, which demonstrated functional restoration in an SNI model similar to that of reversed autograft.
3. Conclusions
References
- Navarro, X. Functional evaluation of peripheral nerve regeneration and target reinnervation in animal models: A critical overview. Eur. J. Neurosci. 2016, 43, 271–286.
- Wojtkiewicz, D.M.; Saunders, J.; Domeshek, L.; Novak, C.B.; Kaskutas, V.; Mackinnon, S.E. Social impact of peripheral nerve injuries. Hand 2015, 10, 161–167.
- Li, N.Y.; Onor, G.I.; Lemme, N.J.; Gil, J.A. Epidemiology of peripheral nerve injuries in sports, exercise, and recreation in the United States, 2009–2018. Phys. Sportsmed. 2020, 49, 1–8.
- Scholz, T.; Krichevsky, A.; Sumarto, A.; Jaffurs, D.; Wirth, G.A.; Paydar, K.; Evans, G.R.D. Peripheral nerve injuries: An international survey of current treatments and future perspectives. J. Reconstr. Microsurg. 2009, 25, 339–344.
- Antoniadis, G.; Kretschmer, T.; Pedro, M.T.; König, R.W.; Heinen, C.P.G.; Richter, H.P. Iatrogenic nerve injuries - prevalence, diagnosis and treatment. Dtsch. Arztebl. Int. 2014, 111, 273–279.
- Ciaramitaro, P.; Mondelli, M.; Logullo, F.; Grimaldi, S.; Battiston, B.; Sard, A.; Scarinzi, C.; Migliaretti, G.; Faccani, G.; Cocito, D. Traumatic peripheral nerve injuries: Epidemiological findings, neuropathic pain and quality of life in 158 patients. J. Peripher. Nerv. Syst. 2010, 15, 120–127.
- Grinsell, D.; Keating, C.P. Peripheral nerve reconstruction after injury: A review of clinical and experimental therapies. BioMed Res. Int. 2014, 2014, 698256.
- Ray, W.Z.; Mackinnon, S.E. Management of nerve gaps: Autografts, allografts, nerve transfers, and end-to-side neurorrhaphy. Exp. Neurol. 2010, 223, 77–85.
- Wang, E.W.; Zhang, J.; Huang, J.H. Repairing peripheral nerve injury using tissue engineering techniques. Neural Regen. Res. 2015, 10, 1393–1394.
- Houschyar, K.S.; Momeni, A.; Pyles, M.N.; Cha, J.Y.; Maan, Z.N.; Duscher, D.; Jew, O.S.; Siemers, F.; van Schoonhoven, J. The role of current techniques and concepts in peripheral nerve repair. Plast. Surg. Int. 2016, 2016, 4175293.
- Carvalho, C.R.; Oliveira, J.M.; Reis, R.L. Modern trends for peripheral nerve repair and regeneration: Beyond the hollow nerve guidance conduit. Front. Bioeng. Biotechnol. 2019, 7, 337.
- Brunelli, B.; Gorson, K.C. The use of complementary and alternative medicines by patients with peripheral neuropathy. J. Neurol. Sci. 2004, 218, 59–66.
- Muthuraman, A.; Singh, N.; Jaggi, A.S. Effect of hydroalcoholic extract of Acorus calamus on tibial and sural nerve transection-induced painful neuropathy in rats. J. Nat. Med. 2011, 65, 282–292.
- Zhao, X.; Xu, Y.; Zhao, Q.; Chen, C.R.; Liu, A.M.; Huang, Z.L. Curcumin exerts antinociceptive effects in a mouse model of neuropathic pain: Descending monoamine system and opioid receptors are differentially involved. Neuropharmacology 2012, 62, 843–854.
- Kim, Y.S.; Park, H.J.; Kim, T.K.; Moon, D.E.; Lee, H.J. The effects of Ginkgo biloba extract EGB 761 on mechanical and cold allodynia in a rat model of neuropathic pain. Anesth. Analg. 2009, 108, 1958–1963.
- Calixto, J.B. Efficacy, safety, quality control, marketing and regulatory guidelines for herbal medicines (phytotherapeutic agents). Braz. J. Med. Biol. Res. 2000, 33, 179–189.
- Karimi, A.; Majlesi, M.; Rafieian-Kopaei, M. Herbal versus synthetic drugs; beliefs and facts. J. Nephropharmacology 2015, 4, 27–30.
- Lahlou, M. The success of natural products in drug discovery. Pharmacol. Pharm. 2013, 04, 17–31.
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs over the nearly four decades from 01/1981 to 09/2019. J. Nat. Prod. 2020, 83, 770–803.
- Weaver, B.A. How taxol/paclitaxel kills cancer cells. Mol. Biol. Cell 2014, 25, 2677–2681.
- Noble, R.L. The discovery of the vinca alkaloids—Chemotherapeutic agents against cancer. Biochem. Cell Biol. 1990, 68, 1344–1351.
- Hussain, G.; Wang, J.; Rasul, A.; Anwar, H.; Qasim, M.; Zafar, S.; Aziz, N.; Razzaq, A.; Hussain, R.; de Aguilar, J.L.G.; et al. Current status of therapeutic approaches against peripheral nerve injuries: A detailed story from injury to recovery. Int. J. Biol. Sci. 2020, 16, 116–134.
- Millesi, H. Bridging defects: Autologous nerve grafts. Acta Neurochir. Suppl. 2007, 100, 37–38.
- Griffin, J.W.; Hogan, M.C.V.; Chhabra, A.B.; Deal, D.N. Peripheral nerve repair and reconstruction. J. Bone Joint Surg. Am. 2013, 95, 2144–2151.
- Li, R.; Li, D.H.; Zhang, H.Y.; Wang, J.; Li, X.K.; Xiao, J. Growth factors-based therapeutic strategies and their underlying signaling mechanisms for peripheral nerve regeneration. Acta Pharmacol. Sin. 2020, 41, 1289–1300.
- Kubiak, C.A.; Grochmal, J.; Kung, T.A.; Cederna, P.S.; Midha, R.; Kemp, S.W.P. Stem-cell–based therapies to enhance peripheral nerve regeneration. Muscle Nerve 2020, 61, 449–459.
- Gordon, T.; English, A.W. Strategies to promote peripheral nerve regeneration: Electrical stimulation and/or exercise. Eur. J. Neurosci. 2016, 43, 336–350.
- Choudhari, A.S.; Mandave, P.C.; Deshpande, M.; Ranjekar, P.; Prakash, O. Phytochemicals in cancer treatment: From preclinical studies to clinical practice. Front. Pharmacol. 2020, 10, 1614.
- Kumar, G.P.; Khanum, F. Neuroprotective potential of phytochemicals. Pharmacogn. Rev. 2012, 6, 81–90.
- Rotshenker, S. Wallerian degeneration: The innate-immune response to traumatic nerve injury. J. Neuroinflammation 2011, 8, 1–14.
- Menorca, R.M.G.; Fussell, T.S.; Elfar, J.C. Peripheral nerve trauma: Mechanisms of injury and recovery. Hand Clin. 2013, 29, 317–330.
- Perry, V.H.; Brown, M.C.; Gordon, S. The macrophage response to central and peripheral nerve injury: A possible role for macrophages in regeneration. J. Exp. Med. 1987, 165, 1218–1223.
- Tetzlaff, W. Tight junction contact events and temporary gap junctions in the sciatic nerve fibres of the chicken during Wallerian degeneration and subsequent regeneration. J. Neurocytol. 1982, 11, 839–858.
- Sulaiman, W.; Gordon, T. Neurobiology of peripheral nerve injury, regeneration, and functional recovery: From bench top research to bedside application. Ochsner J. 2013, 13, 100–108.
- Simons, M.; Trotter, J. Wrapping it up: The cell biology of myelination. Curr. Opin. Neurobiol. 2007, 17, 533–540.
- Arancibia-Carcamo, I.L.; Attwell, D. The node of Ranvier in CNS pathology. Acta Neuropathol. 2014, 128, 161–175.
- Jessen, K.R.; Mirsky, R. The success and failure of the Schwann cell response to nerve injury. Front. Cell. Neurosci. 2019, 13, 1–33.
- Arthur-Farraj, P.J.; Latouche, M.; Wilton, D.K.; Quintes, S.; Chabrol, E.; Banerjee, A.; Woodhoo, A.; Jenkins, B.; Rahman, M.; Turmaine, M.; et al. c-Jun reprograms Schwann cells of injured nerves to generate a repair cell essential for regeneration. Neuron 2012, 75, 633–647.
- Atanasoski, S.; Shumas, S.; Dickson, C.; Scherer, S.S.; Suter, U. Differential cyclin D1 requirements of proliferating Schwann cells during development and after injury. Mol. Cell. Neurosci. 2001, 18, 581–592.
- Kim, H.A.; Pomeroy, S.L.; Whoriskey, W.; Pawlitzky, I.; Benowitz, L.I.; Sicinski, P.; Stiles, C.D.; Roberts, T.M. A developmentally regulated switch regenerative growth of Schwann cells through cyclin D1. Neuron 2000, 26, 405–416.
- Yang, D.P.; Zhang, D.P.; Mak, K.S.; Bonder, D.E.; Scott, L.; Kim, H.A. Schwann cell proliferation during Wallerian degeneration is not necessary for regeneration and remyelination of the peripheral nerves: Axon-dependent removal of newly generated Schwann cells by apoptosis. Mol. Cell. Neurosci. 2008, 38, 80–88.
- Nocera, G.; Jacob, C. Mechanisms of Schwann cell plasticity involved in peripheral nerve repair after injury. Cell Mol. Life Sci. 2020, 77, 3977–3989.
- Song, H.; Zhao, H.; Yang, L.; Li, L.; Zhang, T.; Pan, J.; Meng, Y.; Shen, W.; Yuan, Y. Achyranthes bidentata polypeptides promotes migration of Schwann cells via NOX4/DUOX2-dependent ROS production in rats. Neurosci. Lett. 2019, 696, 99–107.
- Cheng, Q.; Yuan, Y.; Sun, C.; Gu, X.; Cao, Z.; Ding, F. Neurotrophic and neuroprotective actions of Achyranthes bidentata polypeptides on cultured dorsal root ganglia of rats and on crushed common peroneal nerve of rabbits. Neurosci. Lett. 2014, 562, 7–12.
- Cheng, Q.; Jiang, C.; Wang, C.; Yu, S.; Zhang, Q.; Gu, X.; Ding, F. The Achyranthes bidentata polypeptide k fraction enhances neuronal growth in vitro and promotes peripheral nerve regeneration after crush injury in vivo. Neural Regen. Res. 2014, 9, 2142–2150.
- Wang, Y.; Shen, W.; Yang, L.; Zhao, H.; Gu, W.; Yuan, Y. The protective effects of Achyranthes bidentata polypeptides on rat sciatic nerve crush injury causes modulation of neurotrophic factors. Neurochem. Res. 2012, 38, 538–546.
- Yuan, Y.; Shen, H.; Yao, J.; Hu, N.; Ding, F.; Gu, X. The protective effects of Achyranthes bidentata polypeptides in an experimental model of mouse sciatic nerve crush injury. Brain Res. Bull. 2010, 81, 25–32.
- Ding, F.; Cheng, Q.; Gu, X. The repair effects of Achyranthes bidentata extract on the crushed common peroneal nerve of rabbits. Fitoterapia 2008, 79, 161–167.
- Zhang, X.H.; Chen, J.J. The mechanism of astragaloside IV promoting sciatic nerve regeneration. Neural Regen. Res. 2013, 8, 2256–2265.
- Cheng, C.Y.; Yao, C.H.; Liu, B.S.; Liu, C.J.; Chen, G.W.; Chen, Y.S. The role of astragaloside in regeneration of the peripheral nerve system. J. Biomed. Mater. Res. A 2006, 76, 463–469.
- Chen, Y.S.; Chen, C.C.; Chang, L.C.; Yao, C.H.; Hsu, Y.M.; Lin, J.H.; Yang, T.Y.; Chen, Y.H. Increased calcitonin gene-related peptide and macrophages are involved in Astragalus membranaceus-mediated peripheral nerve regeneration in rats. Am. J. Chin. Med. 2018, 46, 69–86.
- Fang, W.K.; Ko, F.Y.; Wang, H.L.; Kuo, C.H.; Chen, L.M.; Tsai, F.J.; Tsai, C.H.; Chen, Y.S.; Kuo, W.W.; Huang, C.Y. The proliferation and migration effects of huangqi on RSC96 Schwann cells. Am. J. Chin. Med. 2009, 37, 945–959.
- Tehranipour, M.; Javaheri, R. Neuroprotective effect of Curcuma longa alcoholic extract on peripheral nerves degeneration after sciatic nerve compression in rats. J. Biol. Sci. 2009, 9, 889–893.
- Noorafshan, A.; Omidi, A.; Karbalay-Doust, S. Curcumin protects the dorsal root ganglion and sciatic nerve after crush in rat. Pathol. Res. Pract. 2011, 207, 577–582.
- Noorafshan, A.; Omidi, A.; Karbalay-Doust, S.; Aliabadi, E.; Dehghani, F. Effects of curcumin on the dorsal root ganglion structure and functional recovery after sciatic nerve crush in rat. Micron 2011, 42, 449–455.
- Ma, J.; Liu, J.; Yu, H.; Wang, Q.; Chen, Y.; Xiang, L. Curcumin promotes nerve regeneration and functional recovery in rat model of nerve crush injury. Neurosci. Lett. 2013, 547, 26–31.
- Tello Velasquez, J.; Nazareth, L.; Quinn, R.J.; Ekberg, J.A.K.; St John, J.A. Stimulating the proliferation, migration and lamellipodia of Schwann cells using low-dose curcumin. Neuroscience 2016, 324, 140–150.
- Yüce, S.; Cemal Gökçe, E.; Işkdemir, A.; Koç, E.R.; Cemil, D.B.; Gökçe, A.; Sargon, M.F. An experimental comparison of the effects of propolis, curcumin, and methylprednisolone on crush injuries of the sciatic nerve. Ann. Plast. Surg. 2015, 74, 684–692.
- Lu, M.C.; Lai, T.Y.; Hwang, J.M.; Chen, H.T.; Chang, S.H.; Tsai, F.J.; Wang, H.L.; Lin, C.C.; Kuo, W.W.; Huang, C.Y. Proliferation- and migration-enhancing effects of ginseng and ginsenoside Rg1 through IGF-I- and FGF-2-signaling pathways on RSC96 Schwann cells. Cell Biochem. Funct. 2009, 27, 186–192.
- Ma, J.; Li, W.; Tian, R.; Lei, W. Ginsenoside Rg1 promotes peripheral nerve regeneration in rat model of nerve crush injury. Neurosci. Lett. 2010, 478, 66–71.
- Wang, L.; Yuan, D.; Zhang, D.; Zhang, W.; Liu, C.; Cheng, H.; Song, Y.; Tan, Q. Ginsenoside Re promotes nerve regeneration by facilitating the proliferation, differentiation and migration of Schwann cells via the ERK- and JNK-dependent pathway in rat model of sciatic nerve crush injury. Cell. Mol. Neurobiol. 2015, 35, 827–840.
- Wong, K.H.; Kanagasabapathy, G.; Naidu, M.; David, P.; Sabaratnam, V. Hericium erinaceus (Bull.: Fr.) Pers., a medicinal mushroom, activates peripheral nerve regeneration. Chin. J. Integr. Med. 2016, 22, 759–767.
- Wong, K.H.; Kanagasabapathy, G.; Bakar, R.; Phan, C.W.; Sabaratnam, V. Restoration of sensory dysfunction following peripheral nerve injury by the polysaccharide from culinary and medicinal mushroom, Hericium erinaceus (Bull.: Fr.) Pers. through its neuroregenerative action. Food Sci. Technol. 2015, 35, 712–721.
- Wong, K.H.; Naidu, M.; David, P.; Abdulla, M.A.; Abdullah, N.; Kuppusamy, U.R.; Sabaratnam, V. Peripheral nerve regeneration following crush injury to rat peroneal nerve by aqueous extract of medicinal mushroom Hericium erinaceus (Bull.: Fr) Pers. (Aphyllophoromycetideae). Evid. Based Complement. Altern. Med. 2011, 2011, 580752.
- Lin, Y.; Jiang, X.; Yin, G.; Lin, H. Syringic acid promotes proliferation and migration of Schwann cells via down-regulating MiR-451-5p. Acta Biochim. Biophys. Sin. (Shanghai) 2019, 51, 1198–1207.
- Liu, B.; Liu, Y.; Yang, G.; Xu, Z.; Chen, J. Ursolic acid induces neural regeneration after sciatic nerve injury. Neural Regen. Res. 2013, 8, 2510–2519.
- Wang, W.; Huang, C.Y.; Tsai, F.J.; Tsai, C.C.; Yao, C.H.; Chen, Y.S. Growth-promoting effects of quercetin on peripheral nerves in rats. Int. J. Artif. Organs 2011, 34, 1095–1105.
- Srinivasulu, C.; Ramgopal, M.; Ramanjaneyulu, G.; Anuradha, C.M.; Suresh Kumar, C. Syringic acid (SA) ‒ A review of its occurrence, biosynthesis, pharmacological and industrial importance. Biomed. Pharmacother. 2018, 108, 547–557.
- Baliga, M.S.; Shivashankara, A.R.; Venkatesh, S.; Bhat, H.P.; Palatty, P.L.; Bhandari, G.; Rao, S. Phytochemicals in the prevention of ethanol-induced hepatotoxicity: A revisit. In Dietary Interventions in Liver Disease: Foods, Nutrients, and Dietary Supplements; Watson, R.R., Preedy, V.R., Eds.; Academic Press: Cambridge, MA, USA, 2019; pp. 79–89. ISBN 978-0-12-814466-4.
- Li, Y.; Yao, J.; Han, C.; Yang, J.; Chaudhry, M.T.; Wang, S.; Liu, H.; Yin, Y. Quercetin, inflammation and immunity. Nutrients 2016, 8, 167.
- Kim, K.J.; Namgung, U.; Cho, C.S. Protective effects of Bogijetong decoction and its selected formula on neuropathic insults in streptozotocin-induced diabetic animals. Evid. Based Complement. Altern. Med. 2017, 2017, 4296318.
- Ju, D.T.; Liao, H.E.; Shibu, M.A.; Ho, T.J.; Padma, V.V.; Tsai, F.J.; Chung, L.C.; Day, C.H.; Lin, C.C.; Huang, C.Y. Nerve Regeneration potential of protocatechuic acid in RSC96 Schwann cells by induction of cellular proliferation and migration through IGF-IR-PI3K-Akt signaling. Chin. J. Physiol. 2015, 58, 412–419.
- Chang, Y.M.; Ye, C.X.; Ho, T.J.; Tsai, T.N.; Chiu, P.L.; Tsai, C.C.; Lin, Y.M.; Kuo, C.H.; Tsai, F.J.; Tsai, C.H.; et al. Alpinia oxyphylla Miquel fruit extract activates MAPK-mediated signaling of PAs and MMP2/9 to induce Schwann cell migration and nerve regeneration. Int. J. Artif. Organs 2014, 37, 402–413.
- Hussin, H.M.; Lawi, M.M.; Haflah, N.H.M.; Kassim, A.Y.M.; Idrus, R.B.H.; Lokanathan, Y. Centella asiatica (L.)-neurodifferentiated mesenchymal stem cells promote the regeneration of peripheral nerve. Tissue Eng. Regen. Med. 2020, 17, 237–251.
- Huang, C.Y.; Kuo, W.W.; Shibu, M.A.; Hsueh, M.F.; Chen, Y.S.; Tsai, F.J.; Yao, C.H.; Lin, C.C.; Pan, L.F.; Ju, D.T. Citrus medica var. sarcodactylis (foshou) activates fibroblast growth factor-2 signaling to induce migration of RSC96 Schwann cells. Am. J. Chin. Med. 2014, 42, 443–452.
- Chen, H.T.; Tsai, Y.L.; Chen, Y.S.; Jong, G.P.; Chen, W.K.; Wang, H.L.; Tsai, F.J.; Tsai, C.H.; Lai, T.Y.; Tzang, B.S.; et al. Dangshen (Codonopsis pilosula) activates IGF-I and FGF-2 pathways to induce proliferation and migration effects in RSC96 Schwann cells. Am. J. Chin. Med. 2010, 38, 359–372.
- Tamaddonfard, E.; Farshid, A.A.; Ahmadian, E.; Hamidhoseyni, A. Crocin enhanced functional recovery after sciatic nerve crush injury in rats. Iran. J. Basic Med. Sci. 2013, 16, 83–90.
- Lee, J.M.; Namgung, U.K.; Hong, K.E. Growth-promoting activity of sanyak (Dioscoreae rhizoma) extract on injured sciatic nerve in rats. JAMS J. Acupunct. Meridian Stud. 2009, 2, 228–235.
- Chen, B.; Niu, S.P.; Wang, Z.Y.; Wang, Z.W.; Deng, J.X.; Zhang, P.X.; Yin, X.F.; Han, N.; Kou, Y.H.; Jiang, B.G. Local administration of icariin contributes to peripheral nerve regeneration and functional recovery. Neural Regen. Res. 2015, 10, 84–89.
- Kou, Y.; Wang, Z.; Wu, Z.; Zhang, P.; Zhang, Y.; Yin, X.; Wong, X.; Qiu, G.; Jiang, B. Epimedium extract promotes peripheral nerve regeneration in rats. Evid. Based Complement. Altern. Med. 2013, 2013, 954798.
- Liu, B.S.; Yao, C.H.; Hsu, S.H.; Yeh, T.S.; Chen, Y.S.; Kao, S.T. A novel use of genipin-fixed gelatin as extracellular matrix for peripheral nerve regeneration. J. Biomater. Appl. 2004, 19, 21–34.
- Zuo, W.; Xu, F.; Zhang, K.; Zheng, L.; Zhao, J. Proliferation-enhancing effects of gastrodin on RSC96 Schwann cells by regulating ERK1/2 and PI3K signaling pathways. Biomed. Pharmacother. 2016, 84, 747–753.
- Zhu, Z.; Zhou, X.; He, B.; Dai, T.; Zheng, C.; Yang, C.; Zhu, S.; Zhu, J.; Zhu, Q.; Liu, X. Ginkgo biloba extract (EGb 761) promotes peripheral nerve regeneration and neovascularization after acellular nerve allografts in a rat model. Cell. Mol. Neurobiol. 2015, 35, 273–282.
- Hsu, S.H.; Chang, C.J.; Tang, C.M.; Lin, F.T. In vitro and in vivo effects of Ginkgo biloba extract EGb 761 on seeded Schwann cells within poly (DL-lactic acid-co-glycolic acid) conduits for peripheral nerve regeneration. J. Biomater. Appl. 2004, 19, 163–182.
- Renno, W.M.; Al-Maghrebi, M.; Al-Banaw, A. (-)-Epigallocatechin-3-gallate (EGCG) attenuates functional deficits and morphological alterations by diminishing apoptotic gene overexpression in skeletal muscles after sciatic nerve crush injury. Naunyn. Schmiedebergs. Arch. Pharmacol. 2012, 385, 807–822.
- Renno, W.M.; Al-Maghrebi, M.; Alshammari, A.; George, P. (-)-Epigallocatechin-3-gallate (EGCG) attenuates peripheral nerve degeneration in rat sciatic nerve crush injury. Neurochem. Int. 2013, 62, 221–231.
- Wang, J.; Tian, L.; He, L.; Chen, N.; Ramakrishna, S.; So, K.F.; Mo, X. Lycium barbarum polysaccharide encapsulated poly lactic-co-glycolic acid nanofibers: Cost effective herbal medicine for potential application in peripheral nerve tissue engineering. Sci. Rep. 2018, 8, 8669.
- Lu, M.; Yi, T.; Xiong, Y.; Wang, Q.; Yin, N. Cortex Mori Radicis extract promotes neurite outgrowth in diabetic rats by activating PI3K/AKT signaling and inhibiting Ca2+ influx associated with the upregulation of transient receptor potential canonical channel 1. Mol. Med. Rep. 2020, 21, 320–328.
- Hsiang, S.W.; Lee, H.C.; Tsai, F.J.; Tsai, C.C.; Yao, C.H.; Chen, Y.S. Puerarin accelerates peripheral nerve regeneration. Am. J. Chin. Med. 2011, 39, 1207–1217.
- Chen, H.-T.; Yao, C.-H.; Chao, P.-D.L.; Hou, Y.-C.; Chiang, H.-M.; Hsieh, C.-C.; Ke, C.-J.; Chen, Y.-S. Effect of serum metabolites of Pueraria lobata in rats on peripheral nerve regeneration: In vitro and in vivo studies. J. Biomed. Mater. Res. Part B Appl. Biomater. 2007, 84B, 256–262.
- Wang, Z.; Zhang, P.; Kou, Y.; Yin, X.; Han, N.; Jiang, B. Hedysari extract improves regeneration after peripheral nerve injury by enhancing the amplification effect. PLoS ONE 2013, 8, e67921.
- Wei, S.Y.; Zhang, P.X.; Han, N.; Dang, Y.; Zhang, H.B.; Zhang, D.Y.; Fu, Z.G.; Jiang, B.G. Effects of Hedysari polysaccharides on regeneration and function recovery following peripheral nerve injury in rats. Am. J. Chin. Med. 2009, 37, 57–67.
- Sheng, Q.S.; Wang, Z.J.; Zhang, J.; Zhang, Y.G. Salidroside promotes peripheral nerve regeneration following crush injury to the sciatic nerve in rats. Neuroreport 2013, 24, 217–223.
- Zuo, W.; Wu, H.; Zhang, K.; Lv, P.; Xu, F.; Jiang, W.; Zheng, L.; Zhao, J. Baicalin promotes the viability of Schwann cells in vitro by regulating neurotrophic factors. Exp. Ther. Med. 2017, 14, 507–514.
- Morani, A.S.; Bodhankar, S.L.; Mohan, V.; Thakurdesai, P.A. Ameliorative effects of standardized extract from Trigonella foenum-graecum L. seeds on painful peripheral neuropathy in rats. Asian Pac. J. Trop. Med. 2012, 5, 385–390.
- Zhang, Y.G.; Sheng, Q.S.; Wang, H.K.; Lv, L.; Zhang, J.; Chen, J.M.; Xu, H. Triptolide improves nerve regeneration and functional recovery following crush injury to rat sciatic nerve. Neurosci. Lett. 2014, 561, 198–202.
- Naik, A.K.; Latham, J.R.; Obradovic, A.; Jevtovic-Todorovic, V. Dorsal root ganglion application of muscimol prevents hyperalgesia and stimulates myelin protein expression after sciatic nerve injury in rats. Anesth. Analg. 2012, 114, 674–682.
- Chen, Y.S.; Yao, C.H.; Chen, T.H.; Hsieh, C.L.; Lao, C.J.; Tsai, C.C. Effect of Buyang Huanwu decoction on peripheral nerve regeneration using silicone rubber chambers. Am. J. Chin. Med. 2001, 29, 423–432.
- Wang, Y.; Chen, Z.; Ye, R.; He, Y.; Li, Y.; Qiu, X. Protective effect of Jiaweibugan decoction against diabetic peripheral neuropathy. Neural Regen. Res. 2013, 8, 1113–1121.
- Wang, Z.Y.; Qin, L.H.; Zhang, W.G.; Zhang, P.X.; Jiang, B.G. Qian-Zheng-San promotes regeneration after sciatic nerve crush injury in rats. Neural Regen. Res. 2019, 14, 683–691.
- Newbern, J.M.; Li, X.; Shoemaker, S.E.; Zhou, J.; Zhong, J.; Wu, Y.; Bonder, D.; Hollenback, S.; Coppola, G.; Geschwind, D.H.; et al. Specific functions for ERK/MAPK signaling during PNS development. Neuron 2011, 69, 91–105.
- Ishii, A.; Furusho, M.; Bansal, R. Sustained activation of ERK1/2 MAPK in oligodendrocytes and Schwann cells enhances myelin growth and stimulates oligodendrocyte progenitor expansion. J. Neurosci. 2013, 33, 175–186.
- Harrisingh, M.C.; Perez-Nadales, E.; Parkinson, D.B.; Malcolm, D.S.; Mudge, A.W.; Lloyd, A.C. The Ras/Raf/ERK signalling pathway drives Schwann cell dedifferentiation. EMBO J. 2004, 23, 3061–3071.
- Agthong, S.; Kaewsema, A.; Tanomsridejchai, N.; Chentanez, V. Activation of MAPK ERK in peripheral nerve after injury. BMC Neurosci. 2006, 7, 45.
- Yamazaki, T.; Sabit, H.; Oya, T.; Ishii, Y.; Hamashima, T.; Tokunaga, A.; Ishizawa, S.; Jie, S.; Kurashige, Y.; Matsushima, T.; et al. Activation of MAP kinases, Akt and PDGF receptors in injured peripheral nerves. J. Peripher. Nerv. Syst. 2009, 14, 165–176.
- Huang, H.; Sun, Z.; Liu, H.; Ma, J.; Hu, M. ERK/MAPK and PI3K/AKT signal channels simultaneously activated in nerve cell and axon after facial nerve injury. Saudi J. Biol. Sci. 2017, 24, 1853–1858.
- Yang, D.P.; Kim, J.; Syed, N.; Tung, Y.J.; Bhaskaran, A.; Mindos, T.; Mirsky, R.; Jessen, K.R.; Maurel, P.; Parkinson, D.B.; et al. P38 MAPK activation promotes denervated Schwann cell phenotype and functions as a negative regulator of Schwann cell differentiation and myelination. J. Neurosci. 2012, 32, 7158–7168.
- Pines, J. Four-dimensional control of the cell cycle. Nat. Cell Biol. 1999, 153, E73.
- Lim, S.; Kaldis, P. Cdks, cyclins and CKIs: Roles beyond cell cycle regulation. Development 2013, 140, 3079–3093.
- Liu, Z.; Zhu, S.; Liu, L.; Ge, J.; Huang, L.; Sun, Z.; Zeng, W.; Huang, J.; Luo, Z. A magnetically responsive nanocomposite scaffold combined with Schwann cells promotes sciatic nerve regeneration upon exposure to magnetic field. Int. J. Nanomedicine 2017, 12, 7815–7832.
- Lundborg, G.; Dahlin, L.B.; Danielsen, N.; Gelberman, R.H.; Longo, F.M.; Powell, H.C.; Varon, S. Nerve regeneration in silicone chambers: Influence of gap length and of distal stump components. Exp. Neurol. 1982, 76, 361–375.
- Chang, Y.C.; Chen, M.H.; Liao, S.Y.; Wu, H.C.; Kuan, C.H.; Sun, J.S.; Wang, T.W. Multichanneled nerve guidance conduit with spatial gradients of neurotrophic factors and oriented nanotopography for repairing the peripheral nervous system. ACS Appl. Mater. Interfaces 2017, 9, 37623–37636.





