Urothelial carcinoma of the bladder (UCB) is among the top 10 most common cancers in the world, with an estimated 80,000 new cases and 17,000 deaths in the United States each year. Significant advances have been made in the management of bladder cancer since the 1990s. More accurate staging has been achieved with refined tissue imaging, and advances in surgical techniques have been combined with improved chemotherapy regimens. Even more, the 5-year survival rate for patients with non-muscle invasive UCB is over 90% and radical cystectomy is the treatment of choice for patients with surgically resectable disease without evidence of metastatic disease. However, patients with muscle-invasive bladder cancer or disseminated disease have a much lower survival rate, suggesting that the occurrence of metastasis has a significant impact on the prognosis of patients with bladder cancer. Considering the impact of metastatic disease on treatment options and patient prognosis, the importance of timely detection and prevention of metastasis in UCB cannot be overemphasized.
1. Introduction
In cancer progression, S1PR1 is thought to be highly expressed in bladder cancer cells and is associated with poor patient prognosis [1]. S1P can promote cancer cell viability, survival, growth, and transformation by activating S1PR1 [2]. In addition, S1PR1 overexpression is associated with the convening of regulatory T cells (Treg) [3], suggesting S1PR1 as a potential prognostic biomarker and therapeutic target for UCB patients. Clinically, FTY-720 (Fingolimod) is used as an S1PR1 inhibitor and is widely used in multiple sclerosis or as an immunomodulator [4][5]. FTY-720 is reported to promote apoptosis of bladder cancer cells [6].
2. Analysis on Results
2.1. Retrospective Evaluation of the Association between S1PR1 Expression and Bladder Cancer Prognosis Shows Divergent Results in Different Databases
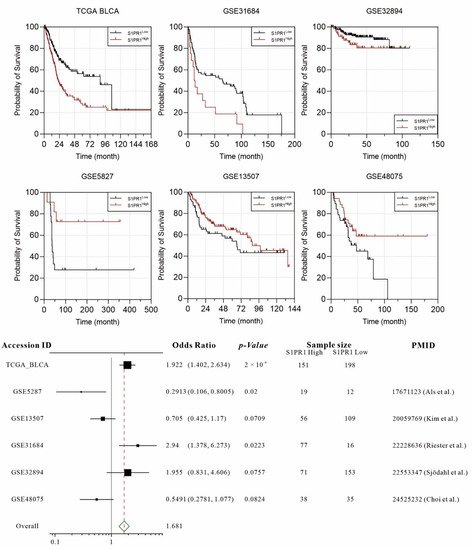
Several publications have demonstrated the association of S1PR1 overexpression with worse prognosis in bladder cancer patients. To comprehensively evaluate the association between S1PR1 expression and prognosis of bladder cancer patients, we collected six available databases including GSE5287, GSE13507, GSE31684, GSE32894, GSE48075, and TCGA BLCA. In order to unbiasedly group the S1PR expression, the “Evaluate Cutpoints” application in R was used to find the best cut-off point for the lowest
p-value in survival of S1PR1 expression
[7]. The results showed that high S1PR1 expression in TCGA BLCA (OR: 1.922,
p-value: 0.0002), GSE 32894 (OR: 1.955,
p-value: 0.0757), and GSE31684 (OR: 2.94,
p-value: 0.0223) was associated with worse prognosis. On the contrary, high S1PR1 expression was associated with better prognosis in GSE5287 (OR: 0.2913,
p-value: 0.02), GSE48075 (OR: 0.5491,
p-value: 0.0824), and GSE13507 (OR: 0.705,
p-value: 0.0709) (
Figure 1). The meta-analysis showed that the overall odds ratio still reached 1.681, indicating that, in general, high S1PR1 expression was associated with a worse prognosis in bladder cancer.
Figure 1. Individual and integrated analysis of the association between S1PR1 mRNA expression and patient prognosis in published bladder cancer clinical databases
[8][9][10][11][12].
2.2. Differences in S1PR1 Expression and Prognosis of Patients with Bladder Cancer May Be Related to the Degree of Neutrophil Infiltration
Considering that S1PR1 is an important receptor related to the regulation of migration by various immune cells
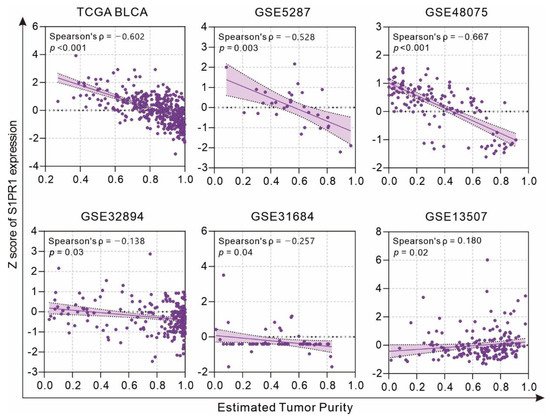
[13], clinical specimens may have different levels of immune cell infiltration affecting the S1PR1 mRNA expression in bulky tumors. Evaluation of the association between tumor purity and S1PR1 expression using the estimate score strategy showed that S1PR1 expression was negatively correlated with tumor purity in all databases except GSE13507 (
Figure 2), suggesting that high S1PR1 expression in bulk tissue may be associated with enriched immune or stromal cell infiltration.
Figure 2. Association of S1PR1 expression with tumor purity.
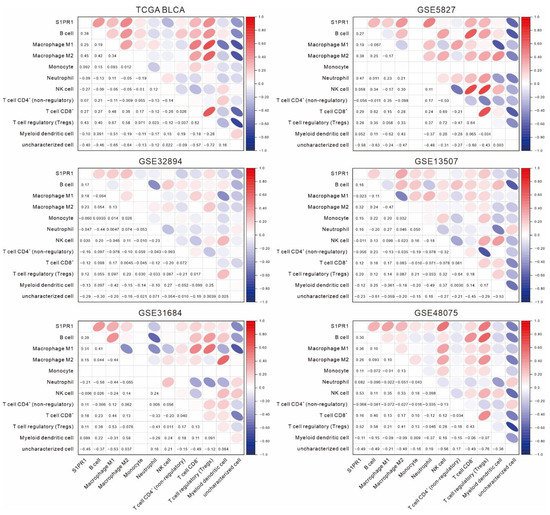
Further evaluation of the correlation between S1PR1 and immune cell populations by QUANTISEQ showed that S1PR1 expression was positively correlated with B cells, macrophage (M1 and M2), and regulatory T cells in all databases (Figure 3). Interestingly, although not all correlations were significant, S1PR1 was positively correlated with neutrophil infiltration in all three databases with better prognosis (GSE5287: ρ = 0.47, GSE13507: ρ = 0.16, GSE48075: ρ = 0.082). In contrast, S1PR1 was negatively correlated with neutrophil (TCGA BLCA: ρ = −0.094, GSE32894: ρ = −0.047, GSE31684: ρ = −0.21) in the three databases where S1PR1 was associated with poorer prognosis. This suggests that the enriched neutrophil infiltration may directly affect the prognosis prediction of bladder cancer patients using S1PR1 expression.
Figure 3. Association of S1PR1 expression with various immune cell infiltrations. The association of S1PR1 expression in bladder cancer tissues with QUANTISEQ simulated multiple immune cell infiltration was assessed using Spearman’s correlation.
2.3. Comprehensive Assessment of S1PR1 Expression Differences between Bladder Cancer Tumors and Normal Tissue
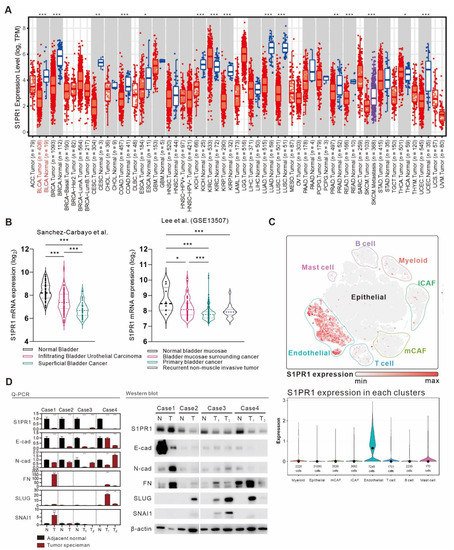
Given the negative correlation between S1PR1 expression and estimated tumor purity, we suggest that the proportion of normal tissue adulterated in tumor samples may also affect S1PR1 expression. To clarify this issue, we attempted to identify the differential expression of S1PR1 in normal tissue and bladder cancer tumor tissue. Gene expression analysis of the TCGA database provided by Timer 2.0
[14] showed that S1PR1 expression was significantly higher in normal tissue than in tumor tissue in most cancers, including BLCA (
Figure 4A). Further, the association of S1PR1 expression differences with tissue types was evaluated in the clinical database of bladder cancer provided by Sanchez-Carbayo et al. and Lee et al. (
Figure 4B)
[15][16], showing that S1PR1 expression was higher in normal bladder tissue and decreased as the tumor histology became more defined. For example, S1PR1 expression was significantly lower in superficial bladder cancer or primary bladder cancer than in infiltrating bladder urothelial carcinoma or bladder mucosae surrounding cancer, suggesting that the actual expression of S1PR1 may be affected when the tumor sample contains normal tissue. To further characterize the expression distribution of S1PR1 in clinical bladder carcinoma samples, we analyzed S1PR1 expression using the bladder urothelial carcinoma single-cell RNA sequencing database (
Figure 4C) published by Chen et al.
[17]. The results showed that S1PR1 was mainly expressed in endothelial cells and to a lesser extent in various immune cells. In addition, endothelial cells were mainly found in normal and high-grade bladder urothelial carcinoma rather than low-grade (refer to
Figure 1b in the publication of Chen et al., 2020), suggesting that high-grade tumors contain a high proportion of endothelial cells, which may be related to the high expression of endothelial and S1PR1 due to tumor invasion of normal tissues.
Figure 4. A comprehensive analysis of the differences in S1PR1 expression in bladder cancer tumors versus normal tissues. (
A) Differences in S1PR1 expression in tumors and normal tissues among the 34 cancer types of TCGA adopted from TIMER 2.0 searching “S1PR1”. (
B) Differences in S1PR1 expression in normal versus clinically defined histopathological bladder cancer tissues from the bladder cancer database published by Sanchez-Carbayo et al. and Lee et al
[9][10]. (GSE13507). (
C) Bladder urothelial carcinoma single-cell RNA sequencing database adopted from Chen et al. showed that S1PR1 was mostly expressed in endothelial cells, followed by immune cells. (
D) mRNA and protein expression of S1PR1 and EMT marker in four bladder cancer tumors and adjacent normal tissues. Student’s t-test or one-way ANOVA was utilized to analyze the statistical significance of the differences in S1PR1 expression between groups. *
p < 0.05, **
p < 0.01, ***
p < 0.001. (N: adjacent normal tissue, T: tumor tissue, T1/2: samples from two separated tumors tissues, E-cad: E-cadherin, N-cad: N-cadherin, FN: Fibronectin).
We evaluated S1PR1 expression in normal or tumor tissues from four clinical cases and analyzed the epithelial–mesenchymal transition (EMT) marker together, which is thought to be regulated by S1PR1 expression, but with different effects in different tissues or spatial and temporal contexts
[18][19]. The results showed that S1PR1 mRNA and protein expression were mostly amplified in normal tissues, similar to E-cadherin expression, while N-cadherin, FN, SLUG, and SNAI1, which are mesenchymal markers, were higher in tumor tissues, suggesting a preliminary association between S1PR1 expression and EMT in bladder cancer (
Figure 4D).
2.4. Comparison of S1PR1 Expression in Bladder Cancer Cell Lines with Clinical Databases Reveals its Potential Function in Cell Adhesion
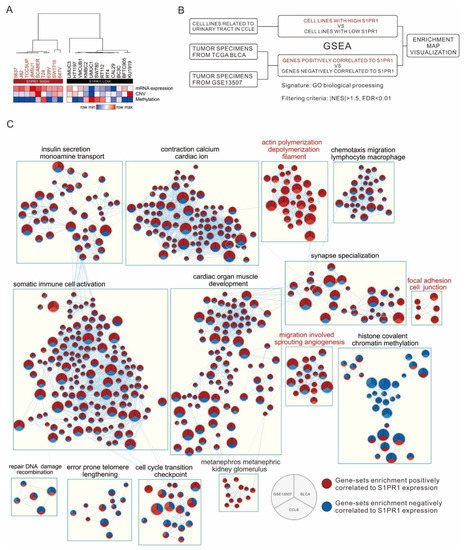
There are many factors in clinical bulk tissue that may affect S1PR1 expression, such as the infiltration of endothelial or immune cells, which may lead to misinterpretation of the biological response of S1PR1 expression in bladder cancer cells. To understand the direct effect of S1PR1 expression on bladder cancer cells, 21 bladder cancer-related cell lines were screened from the CCLE database for S1PR1 expression analysis, showing that the high S1PR1 expression group generally had lower methylation and the low S1PR1 expression group had higher methylation in addition to lower copy number variation (Figure 5A). Further, GSEA was performed after clustering cell lines with high or low S1PR1 expression. On the other hand, GSEA was performed with S1PR1 expression-related genes in TCGA BLCA (high S1PR1 associated with poor prognosis) and GSE13507 (low S1PR1 associated with poor prognosis) (illustrated as Figure 5B), and the results of the three GSEAs were visualized using the Enrichment map in Cytoscape (Figure 5C). The results showed that the node cluster associated with immune cell activation in the clinical database was not enriched in the cell lines, suggesting that the biological response associated with S1PR1 expression in clinical tissues is indeed influenced by the immune microenvironment. On the contrary, we observed that angiogenesis and cell adhesion gene clusters were positively associated with S1PR1 expression in all three, suggesting that the real effect of S1PR1 on bladder cancer is related to these gene clusters.
Figure 5. Investigation of S1PR1 expression in bladder cancer cells for authentic biological response enrichment associations. (A) Heat map demonstrating S1PR1 mRNA expression (fragments per kilobase per million, FPKM), copy number variation (log value), and methylation (β value) of 21 uroepithelial carcinomas in the CCLE database with unsupervised hierarchical clustering. (B) Flow chart presenting comparison of S1PR1 expression and biological response association in CCLE bladder cancer cell lines or clinical bladder cancer tumors (TCGA BLCA, GSE13057). (C) Enrichment map visualization showing the GSEA scores of gene-sets significantly enriched in (B). The color represents the degree of normalized enrichment score (NES); red means the gene-sets are enriched in high S1PR1 samples (NES > 1.5) and blue means the gene-sets are enriched in low S1PR1 samples (NES < −1.5). All presented enriched gene sets have passed the screening criteria of p-value < 0.05, FDR < 0.01. Gene-sets without edge linkage were excluded to increase the ease of visualization of the results.
2.5. S1PR1 Expression Shows an Opposite Association with the Promoting of Epithelial–Mesenchymal Transition
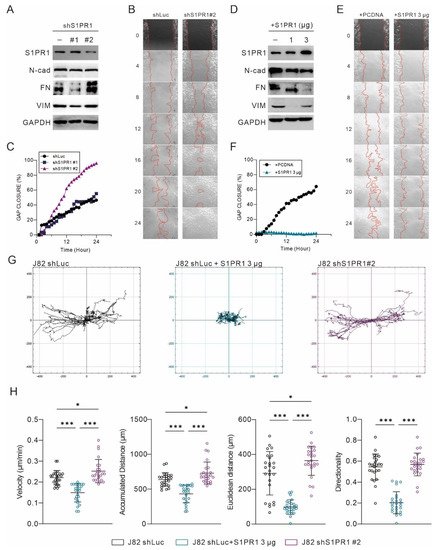
Among the 21 uroepithelial cancer cell lines, only four cell lines, T24, J82, JMSU1, and SCABER, expressed high amounts of S1PR1, and its mRNA expression was not even detected in most cell lines. The expression of S1PR1 was positively correlated with the variation of copy number, especially in JMSU1 and SCABER, which had a high copy number of the S1PR1 gene (Figure 4A). Only the J82 bladder cancer cell line has a near normal copy number and moderate mRNA expression of the S1PR1 gene. Therefore, to avoid potential interference with genetic abnormalities, the J82 bladder cancer cell line was used to establish a stable expression of S1PR1 targeting shRNA clone (Figure 6A–C). In addition, the control cells (J82 shLuc) were transfected with pcDNA3.1-S1PR1 plasmids for overexpression (Figure 6D–F).
Figure 6. Negative correlation between S1PR1 expression and bladder cancer cell motility. (
A,
D) Western blot demonstrating the efficacy of S1PR1 expression manipulation and matching EMT marker expression. (
B,
E) Twenty-four hour live cell image tracking showed the progressions in gap area at multiple time points. (
C,
F) Gap closure (%) presents the effect of manipulating S1PR1 expression on wound healing. (
G) Migration tracking plot for J82 shLuc, shLuc + S1PR1 (3 μg), and J82 shS1PR1#2. Cell migration was tracked for 48 h after the wound healing assay started, with cell positions determined every 30 min. In each panel, the center indicates the starting point. (
H) Statistical analysis of the J82 tracking cell migration rate, distance, and directionality, with lines showing mean and standard deviation. J82 shLuc (n = 29); J82 + S1PR1 3 μg (n = 30); J82 shS1PR1#2 (n = 30). *
p < 0.05, ***
p < 0.001. (VIM: vimentin, GAPDH: glyceraldehyde 3 phosphate dehydrogenase) (for uncropped Western Blot images, please refer to
Supplementary File S1).
Confirming the successful manipulation of S1PR1 expression in J82 cells, it was found that S1PR1 inhibition was associated with the enhancement of EMT (Figure 6A), while S1PR1 over-expression inhibited EMT (Figure 6D). A further 24 h live cell imaging showed that the rate of gap healing was significantly increased when S1PR1 expression was inhibited (Figure 6B). In particular, there was a significant upregulation in the clone that significantly inhibited S1PR1 expression (shS1PR1#2) (Figure 6C). In contrast, the rate of gap healing was significantly reduced upon overexpression of S1PR1 (3 μg) (Figure 6E,F). As assessed by the cell migration trajectory (Figure 6G), overexpression of S1PR1 significantly inhibited the migration distance of J82. Further evaluation of velocity, accumulated distance, Euclidean distance, and directionality showed that the over-expression of S1PR1 significantly inhibited cell mobility, suggesting that S1PR1 may affect cell movement by modulating cell adhesion (Figure 6H).
2.6. The Administration of FTY-720 Promotes EMT in Bladder Carcinoma
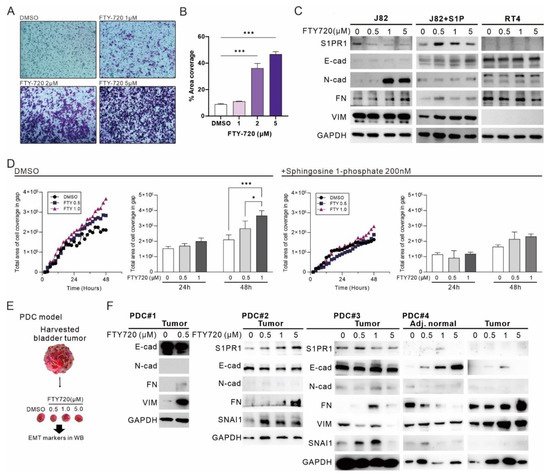
FTY-720 is identified to inhibit cell proliferation and promote apoptosis by regulating S1PR1; however, the effect of FTY-720 on EMT in bladder cancer remains unknown. Transwell migration assay showed that the metastatic capacity of J82 increased with increasing FTY-720 treatment dose (
Figure 7A), and the total cell coverage area was significantly increased with 2 and 5 μM FTY-720 treatment (
Figure 7B). Similarly, FTY720 treatment inhibited E-cadherin expression and promoted mesenchymal marker expression, a phenomenon that was disturbed by the addition of S1P, but not in RT4 cells that did not express S1PR1 (
Figure 7C). In the wound healing assay, treatment with FTY720 promoted gap closure, especially at 48 h, with a significant difference. In contrast, FTY720 treatment in the presence of S1P had no significant effect on gap closure (
Figure 7D), suggesting that inhibition of S1PR1 by FTY720 promoted EMT in bladder cancer cells. Finally, to understand the overall effect of FTY720 treatment on human bladder cancer tumors, we established a patient-derived tumor culture model
[20] (
Figure 7E). A decrease in E-cadherin and increase in mesenchymal marker due to FTY720 treatment was observed in all four cases (
Figure 7F), suggesting that bladder cancer tumors and cell lines respond similarly to FTY-720 treatment. In particular, in the case of PDC#4, the response to FTY720 in adjacent normal tissues and bladder cancer tumors showed an opposite trend, implying that FTY-720 may have divergent responses in different types of tissues or cancers.
Figure 7. The administration of FTY-720 promotes EMT in bladder carcinoma. (
A) Transwell migration assay showing the effect of FTY-720 treatment for 24 h (×100). (
B) Area coverage (%) was calculated by quantifying the ratio of cell coverage per field using ImageJ. (
C) Western blot showed the expression of S1PR1- and EMT-related markers after different dosage treatment of FTY-720 in J82 and RT4. Sphingosine 1-phosphate (200 nM) was added at the same time of FTY-720 treatment. (
D) J82 cell gap coverage curves treated with FTY-720 in the presence and absence of S1P and statistical analysis of the number of cells in the gap at 24 h and 48 h. The curve data are presented as mean values only to reduce reading interference. (
E) Illustration of patient-derived tumor culture (PDC) model. (
F) Western blot showed four sets of clinical bladder cancer tumors with S1PR1- and EMT-related marker expression measured 48 h after FTY-720 addition. Bar charts are presented as the mean ± SD based on three independent experiments. *
p < 0.05, ***
p < 0.001 (for uncropped Western Blot images, please refer to
Supplementary File S1).
3. Current Insights
The TCGA database shows that S1PR1 expression is significantly higher in normal tissues than in tumor tissues for most cancers (
Figure 3A). The work of Sanchez-Carbayo et al. and Lee et al. showed that S1PR1 expression was reduced at low grade tumor (
Figure 3B). Further, the single-cell RNA sequencing results reported by Chen et al. showed that most S1PR1 expression was from endothelial cells and a few from multiple immune cells, both of which were underrepresented in low grade tumor (
Figure 3C). These results suggest that the expression of S1PR1 might be deeply affected by the purity of tumor cell composition in the lesioned tissue. Samples collected from high-grade tumor cells are more likely to be adulterated with normal tissue, which may explain the association of high S1PR1 expression with poor prognosis observed in some databases. Nevertheless, whether S1PR1 expression in tumor cell may affect the generation of tumor-associated endothelial cells requires further investigation. As our analysis shows that S1PR1 expression is associated with angiogenesis (
Figure 3C), it is possible that bladder cancer cells overexpressing S1PR1 may affect tumor progression by altering microenvironmental angiogenesis
[21][22][23]. When S1PR1 expression was manipulated in J82, it was shown that overexpression of S1PR1 had an inhibitory effect on bladder cancer cell migration, possibly associated with enhanced cellular apposition, echoing the analysis in
Figure 3C. Conversely, shRNA interference with S1PR1 expression or inhibition of S1PR1 by FTY-720 accelerated bladder cancer cell migration, and the addition of S1P antagonized the effect of FTY-720. Moreover, a similar phenomenon could be observed in the patient-derived tumor primary culture model, suggesting that clinical inhibition of S1PR1 may cause accelerated metastasis of bladder cancer cells.
High expression of S1PR1 was significantly associated with poor prognosis in multiple cancer databases, raising the possibility of its potential role in promoting tumorigenesis. Based on this inference, FTY-720 has been reported and demonstrated to induce apoptosis in a variety of cancer cells, including bladder cancer
[6][24]. Moreover, the mechanism of EMT inhibition by FTY-720 in cholangiocarcinoma and glioblastoma has also been proposed
[25][26]. However, our study clearly indicates that reduction of S1PR1 expression by human manipulation may cause a promotion in EMT in bladder cancer cells and patient tumor tissue, a phenomenon consistent with FTY-720 treatment. Different responses to FTY-720 in normal or tumor tissues also indicate divergences in response between various types of tissues or cancers; this may be due to the fact that normal tissue is usually rich in endothelium
[27][28]. Although a majority of the literature has confirmed the induction of apoptosis in cancer cells by FTY-720, we still need to pay attention to its potential role in EMT induction. Furthermore, whether the apoptosis induction caused by FTY-720 is related to the EMT-induced anoikis needs to be further clarified. The inhibition of S1PR1 contributes to the reduced interaction with the ECM, thus allowing a higher migration ability. Cell proliferation may be reduced as a result and apoptosis may occur owing to separation from the matrix
[29]. Furthermore, cancer cells may thus develop an anti-anoikis mechanism and become more resistant to chemotherapy or accelerate the progression of metastasis
[30][31][32].