Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Ramar Thangam | + 1744 word(s) | 1744 | 2021-08-31 11:18:14 | | | |
| 2 | Rita Xu | Meta information modification | 1744 | 2021-09-07 08:53:11 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Thangam, R. Engineered Polymer Nanoparticle. Encyclopedia. Available online: https://encyclopedia.pub/entry/13950 (accessed on 10 May 2026).
Thangam R. Engineered Polymer Nanoparticle. Encyclopedia. Available at: https://encyclopedia.pub/entry/13950. Accessed May 10, 2026.
Thangam, Ramar. "Engineered Polymer Nanoparticle" Encyclopedia, https://encyclopedia.pub/entry/13950 (accessed May 10, 2026).
Thangam, R. (2021, September 07). Engineered Polymer Nanoparticle. In Encyclopedia. https://encyclopedia.pub/entry/13950
Thangam, Ramar. "Engineered Polymer Nanoparticle." Encyclopedia. Web. 07 September, 2021.
Copy Citation
Engineering polymeric nanoparticles for their shape, size, surface chemistry, and functionalization using various targeting molecules has shown improved biomedical applications for nanoparticles. Polymeric nanoparticles have created tremendous therapeutic platforms, particularly applications related to chemo- and immunotherapies in cancer.
polymer nanoparticles
nano-vaccines
immune cells
immunotherapy
tumor microenvironments
1. Introduction
In managing human diseases, the biological forms of vaccines play a prominent role in activating the host’s adaptive and innate immune responses. Vaccines are commonly used as a prophylactic treatment for infectious diseases and have recently been used as therapeutic agents for cancer [1][2][3][4]. Recent developments in cancer nano-vaccines and their prime role on host immune systems have generated interest among chemical and biomedical engineers in producing novel targeted nanomedicine-based vaccines for priming the body’s immune defense to elicit anti-tumor responses [5][6][7][8]. For prospective applications, nanoparticulate adjuvants such as polymeric nanoparticles have been shown as promising antigen delivery vehicles for site-specific, targeted vaccine delivery [9]. In combination with therapeutic adjuvants, targeting agents, and immuno-therapeutic molecules, polymeric nanoparticles become potential functional candidates for therapies [10][11][12][13].
Cancer is one of the deadliest diseases and kills millions of people every year across the globe. According to the Global Cancer Observatory (GCO), an interactive web-based platform, approximately 10 million cancer deaths and 19.3 million new cancer cases were expected globally in 2020 [14]. There are a variety of cancer therapeutic approaches that exist. Among these, chemo-, photo- (thermal, dynamic), radio-, and immuno-therapies are the most evolved to target tumor cells at their sites and inducing cell death [15][16][17]. Despite good signs of progress in cancer therapeutic approaches, drug resistance, recurrence, low therapeutic responsiveness, and non-specific toxicity with undesired side effects are major concerns noted in current cancer therapies [18][19][20]. The advent and success of immuno-therapeutic strategies via applying nanomaterials are promising for patients by extending their life span. Immuno-therapeutic approaches have advantages in reducing the overall burden of primary tumors and metastatic lesions [21]. Currently, clinicians consider immunotherapy as a gold-standard treatment choice for many cancer types [22][23]. Hence, the development of various immuno-therapeutic nanoengineered antibodies and therapeutic molecules is the current focus of research for successful treatments [24].
Currently, engineered antibodies are used to induce immune stimulation via blocking the immune checkpoints to show anticancer responses. The use of antibodies in combination with selective delivery approaches using various polymeric nanomaterials with functionalized nanocarrier systems would be a beneficial approach, and this could profoundly increase the success of treatment towards clinical applications (Figure 1). The multifunctional immunomodulatory role of nanomedicine in cancer immunotherapy is subsidizing the field of nanomaterials in immunology. Thus, the innovative developments of nanomedicine and immunotherapy through combined nanomaterial systems can enhance immunomodulatory functions in the subsequent stages of polymeric vaccine manipulations for clinical use. Occasionally, autoimmune diseases can show some unpredicted side effects in cancer immunotherapy. Polymeric nanomaterial-based cancer immuno-therapeutic strategies are shown to be more effective against hematological malignancies than solid tumors. This is mainly due to their low effectiveness in tumor penetration through their abnormal extracellular matrices, endothelial barriers, and distinct tumor microenvironments (TMEs) [25][26]. By developing highly effective polymeric nanomaterials with precise targeting of TMEs across the endothelial border, it is possible to improve the efficacy of cancer immunotherapies in solid tumors.
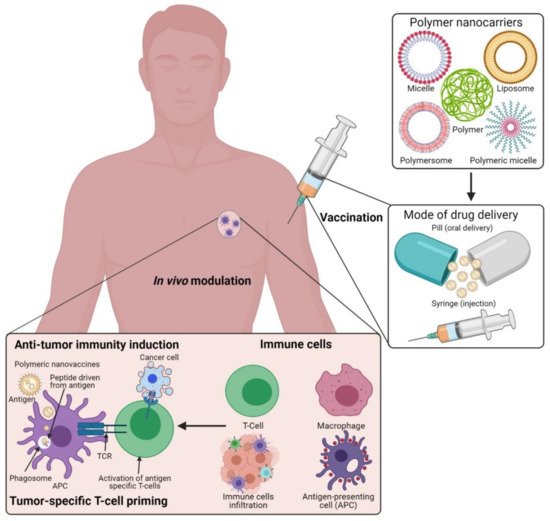
Figure 1. Schematic illustration of the polymeric nanocarrier systems as adjuvants for cancer vaccine deliveries and triggering host-immune-response-mediated cell death in vivo. (Images were created with the help of BioRender.com, accessed on 15 August 2021).
In recent years, engineered polymeric nanoparticles have been extensively used as a novel drug delivery platform for achieving safe and effective cancer immunotherapy [8][23][27]. The tumor immune responses and long-lasting tumor suppression effects can be achieved via vaccines like immuno-therapeutic agents, which have attracted clinicians to utilize them for their beneficial effects [28][29]. The target-specific anti-tumor immune functions of the polymeric nanoparticles can also be improved via introducing aromatic amino acid mutations in genetically encoded regions of the tumor target-specific neoantigens (non-self-peptides) [26][30]. Thus, the polymeric nanocarriers with engineered cancer neoantigen-based (cancer-specific differentially expressed antigens) vaccines have been widely used for immunotherapies via reducing autoimmunity while enhancing cytotoxic T lymphocyte (CTL) responses [27][31]. Similarly, dendritic cells (DCs) that activate antigen targeting and ligand presentation to conquer neoantigen-dependent cancer vaccines have been used to improve therapeutic functions [32][33]. It has been proven that DCs and immune regulators of type-I interferons (e.g., IFN-γ) are predominant molecules that link the innate and adaptive immunity in patients for the uptake and exposure of naive T-cells in the activation of tumor-specific CTL responses [34][35]. Several reports in cancer nanomedicine-based drug delivery approaches have revealed the functions of polymeric nanoparticles in the activation of stimulator of interferon genes (STING) and toll-like receptors (TLRs), thereby enhancing the CD8+ T-cells in preclinical animal models via stimulating the STING mechanistic pathways [36][37][38][39][40].
2. Evolution of Polymeric Nanomaterials and Their Engineering Strategies in Cancer Immunotherapies
Many clinical pathologies have improved immuno-therapeutic applications by the use of polymeric nanomaterials. However, understanding the mechanisms behind cancer immunity is an urgent need for deciding on suitable strategies for cancer immunotherapies [41][42]. In recent years, advancements in polymer science and nanotechnology have provided approaches to improving cancer immunotherapies. Multidisciplinary fields bridging chemistry, physics, biology, engineering, and medicine have helped design, synthesize, and characterize nanomaterials and extend their applications in various immunotherapies. Upon considering this, the biomedical investigations and the rapid developments of polymer-based nanoparticles and their deep understanding in medical fields have resulted in their clinical applications, including in cancer treatment. The potential of polymeric nanoparticles is extensive due to their unique and multifaceted functions, including adjustable surface area, easy tunability for cell-materials, and cell–chemical–biological interactions to accommodate higher drug contents during drug delivery applications [43]. The prominent phenomenon of polymeric nanoparticles is the easy surface functionalization for achieving precise target-tissue biodistribution and blood clearance upon circulation [44]. In addition, polymeric nanoparticles could also exhibit different functionalities via the incorporation of theranostic molecules such as fluorescent probes, drug conjugates, or antibody–drug conjugates. These molecules can be easily coupled onto the surfaces of the polymeric biomaterials [45][46].
The use of polymeric nanoparticles combined with metal nanoparticles to deliver cancer immunotherapies has already been approved for clinical applications and many are currently undergoing clinical trials [47]. The organic and inorganic nano biomaterials possess higher biocompatibility with reduced side effects when modified with bio-functional polymers. In addition, several organic nanoparticles with polymeric modifications have been under investigation for clinical applications as vaccines, as a means to prolong drug delivery systems, and as biocompatible dermal and tropical drugs. Likewise, the intravenous delivery of organic nanomaterials is also efficiently utilized for cancer immuno- and gene therapies [48][49]. Organic nanomaterials with the modifications of polymer materials, especially for immunotherapy, have shown broader and more prolonged success rates in preclinical models because of their increased biocompatibility with targeting efficiency [50]. Despite these, the biologically engineered polymer nanomaterials modified with organic polymers have demonstrated long-term safety with less accumulation in clearance organs such as the kidney, liver, and spleen. Hence, the utilization of polymer-based engineering approaches to make multifunctional nanoparticles has evolved to show promising delivery vehicles with increased drug loading capacity, controlled delivery, antibody targeting, and targeted accumulation at the localized TMEs as switchable candidates during therapeutic applications [51]. Upon considering all these factors, designing a polymer nanoparticle-based vaccine will always require an antigen as a primary vital content that is generally present in a short-peptide form to trigger the adaptive host immune responses [52]. Moreover, a long-lasting and robust immune response could also be achieved through natural killer (NK) cells [53]. Thus, developing the polymeric vaccine as a potential cancer immuno-therapeutic agent to effectively internalize via tumor homing and penetration through cell membranes is of immediate need for expanding this field in cancer immunotherapies.
3. Functional Immunotherapies Delivered to Cancer Cells via Polymeric Nanoparticles
The prerequisite of polymeric nanoparticles in cancer immunotherapy is to induce many signaling events of cancer cells to achieve treatment outcomes [54][55]. The cancer immunity cycle (Figure 2) plays a significant role in activating immunotherapy responses in the tumor to remove cancer cells or alter the TME by releasing tumor antigens. Upon delivery of polymeric nanoparticles to the tumor site, the cancer cells undergo numerous biological changes to induce apoptosis-mediated cell death pathways and release cancer antigens for further immune activation to induce CTL responses. The released cancer cancer-specific antigens are presented by the antigen-presenting cells (APCs) in the context of the major histocompatibility complexes (MHCs) [56]. MHCs play a prime role in the immune system by enhancing the modulatory functions in the infected cells during vaccination. MHCs have a highly pronounced cell surface display of intracellularly derived proteins and peptides for immune activation. Furthermore, the MHCs presenting dendritic cells (DCs) and tumor antigens stimulate the activation of immature T-cells in the lymph node to trigger the tumor-specific CTLs and natural killer (NK) cells which allow them to control cellular events and identify TMEs in tumorigenesis [32][57][58]. CTLs effectively communicate with the T-cell signaling molecules of the receptors and MHCs to attract tumor cells.
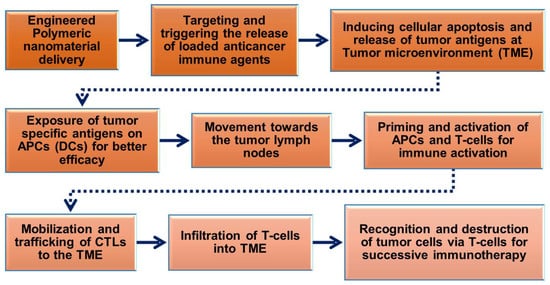
Figure 2. Schematic flow chart showing the roles of engineered polymeric nanomaterials in the activation and regulation of cancer immunotherapy via modulation of tumor microenvironments (TMEs).
Furthermore, polymeric nanoparticles facilitate T-cell-mediated cellular apoptosis, which eventually stimulates the release of cancer antigens to boost further immune responses [43][48][58]. This could maintain the events that promote specific immunity to cancer, which can also be disturbed through multiple barriers that reduce the immuno-therapeutic mechanisms. In addition, the pro-inflammatory responses from the polarized macrophages (M1) [59] are highly involved in regulating and killing tumor cells via the production of immunosuppressive cytokines [60]. As shown in Figure 3, the various forms of delivered polymeric nanomaterials can facilitate the internalization of APCs through modulating migration, uptake, and maturation in the weakly acidic TMEs. This could further enable the production of tumor antigens that enhance NPs’ immunomodulatory potential, such as through reducing regulatory T-cell population, DC activation, and macrophage repolarization (M2 to M1) to induce immunogenic cell death (ICD). These actions are potentiated in the TMEs through the regulation of various cytotoxic T-cell-associated immune modulations and encouraging immunotherapies (Figure 2 and Figure 3). Immunosuppressive candidates such as PD-1, PD-L1, and CTLA-4 are involved in decreasing tumor cell proliferation. These conditions may be involved in reducing the immuno-therapeutic efficiencies that are the cause of the existing problems with current cancer immunotherapies [61][62]. Nanomaterials, including polymeric nanoparticles could intervene in these conditions, to boost cancer immunity towards successive immuno-therapeutics [63][64].
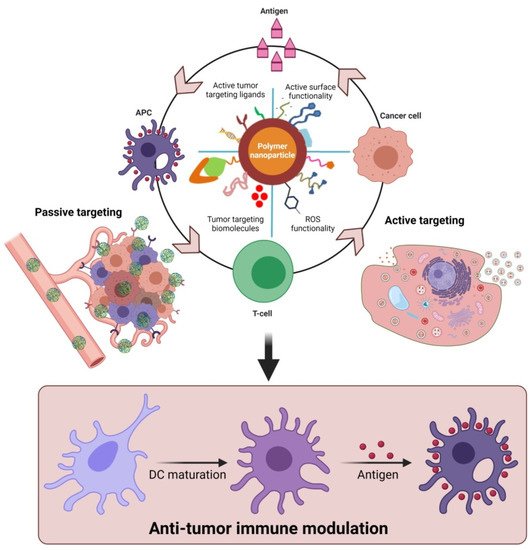
Figure 3. Proposed role of functional polymeric nanomaterials with immunostimulatory molecules in regulating anti-tumor potential upon delivery as cancer-specific vaccine systems. The delivery of polymer nanomaterials could enhance the tumor-specific immune modulation and improve anti-cancer responses via activation of DCs. (Images were created with the help of BioRender.com, accessed on 15 August 2021).
References
- Brubaker, S.W.; Bonham, K.S.; Zanoni, I.; Kagan, J.C. Innate immune pattern recognition: A cell biological perspective. Annu. Rev. Immunol. 2015, 33, 257–290.
- Vetter, V.; Denizer, G.; Friedland, L.R.; Krishnan, J.; Shapiro, M. Understanding modern-day vaccines: What you need to know. Ann. Med. 2018, 50, 110–120.
- Marshall, J.S.; Warrington, R.; Watson, W.; Kim, H.L. An introduction to immunology and immunopathology. Allergy Asthma Clin. Immunol. 2018, 14, 49.
- June, C.H.; Warshauer, J.T.; Bluestone, J.A. Is autoimmunity the Achilles’ heel of cancer immunotherapy? Nat. Med. 2017, 23, 540–547.
- Jo, S.D.; Nam, G.-H.; Kwak, G.; Yang, Y.; Kwon, I.C. Harnessing designed nanoparticles: Current strategies and future perspectives in cancer immunotherapy. Nano Today 2017, 17, 23–37.
- Sun, Q.; Barz, M.; De Geest, B.G.; Diken, M.; Hennink, W.E.; Kiessling, F.; Lammers, T.; Shi, Y. Nanomedicine and macroscale materials in immuno-oncology. Chem. Soc. Rev. 2019, 48, 351–381.
- Hodi, F.S.; O’Day, S.J.; McDermott, D.F.; Weber, R.W.; Sosman, J.A.; Haanen, J.B.; Gonzalez, R.; Robert, C.; Schadendorf, D.; Hassel, J.C. Improved survival with ipilimumab in patients with metastatic melanoma. N. Engl. J. Med. 2010, 363, 711–723.
- Siriwon, N.; Kim, Y.J.; Siegler, E.; Chen, X.; Rohrs, J.A.; Liu, Y.; Wang, P. CAR-T cells surface-engineered with drug-encapsulated nanoparticles can ameliorate intratumoral T-cell hypofunction. Cancer Immunol. Res. 2018, 6, 812–824.
- Gondan, A.I.B.; Ruiz-de-Angulo, A.; Zabaleta, A.; Blanco, N.G.; Cobaleda-Siles, B.M.; García-Granda, M.J.; Padro, D.; Llop, J.; Arnaiz, B.; Gato, M. Effective cancer immunotherapy in mice by polyIC-imiquimod complexes and engineered magnetic nanoparticles. Biomaterials 2018, 170, 95–115.
- Pierini, F.; Nakielski, P.; Urbanek, O.; Pawłowska, S.; Lanzi, M.; De Sio, L.; Kowalewski, T.A. Polymer-based nanomaterials for photothermal therapy: From light-responsive to multifunctional nanoplatforms for synergistically combined technologies. Biomacromolecules 2018, 19, 4147–4167.
- O’Donnell, J.S.; Teng, M.W.; Smyth, M.J. Cancer immunoediting and resistance to T cell-based immunotherapy. Nat. Rev. Clin. Oncol. 2019, 16, 151–167.
- Shao, F.; Zhang, M.; Xu, L.; Yin, D.; Li, M.; Jiang, Q.; Zhang, Q.; Yang, Y. Multiboosting of Cancer Immunotherapy by a Core–Shell Delivery System. Mol. Pharm. 2019, 17, 338–348.
- Alshamsan, A.; Hamdy, S.; Haddadi, A.; Samuel, J.; El-Kadi, A.O.; Uludağ, H.; Lavasanifar, A. STAT3 knockdown in B16 melanoma by siRNA lipopolyplexes induces bystander immune response in vitro and in vivo. Transl. Oncol. 2011, 4, 178–188.
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2021, 71, 209–249.
- Lu, K.; He, C.; Guo, N.; Chan, C.; Ni, K.; Lan, G.; Tang, H.; Pelizzari, C.; Fu, Y.-X.; Spiotto, M.T. Low-dose X-ray radiotherapy–radiodynamic therapy via nanoscale metal–organic frameworks enhances checkpoint blockade immunotherapy. Nat. Biomed. Eng. 2018, 2, 600–610.
- Duan, X.; Chan, C.; Guo, N.; Han, W.; Weichselbaum, R.R.; Lin, W. Photodynamic therapy mediated by nontoxic core–shell nanoparticles synergizes with immune checkpoint blockade to elicit antitumor immunity and antimetastatic effect on breast cancer. J. Am. Chem. Soc. 2016, 138, 16686–16695.
- Lan, G.; Ni, K.; Xu, Z.; Veroneau, S.S.; Song, Y.; Lin, W. Nanoscale metal–organic framework overcomes hypoxia for photodynamic therapy primed cancer immunotherapy. J. Am. Chem. Soc. 2018, 140, 5670–5673.
- Fan, Q.; Chen, Z.; Wang, C.; Liu, Z. Toward biomaterials for enhancing immune checkpoint blockade therapy. Adv. Funct. Mater. 2018, 28, 1802540.
- Chang, M.; Wang, M.; Wang, M.; Shu, M.; Ding, B.; Li, C.; Pang, M.; Cui, S.; Hou, Z.; Lin, J. A multifunctional Cascade bioreactor based on hollow-structured Cu2MoS4 for synergetic cancer chemo-dynamic therapy/starvation therapy/phototherapy/immunotherapy with remarkably enhanced efficacy. Adv. Mater. 2019, 31, 1905271.
- Aikins, M.E.; Xu, C.; Moon, J.J. Engineered Nanoparticles for Cancer Vaccination and Immunotherapy. Acc. Chem. Res. 2020, 53, 2094–2105.
- Zhang, X.; Xia, L.-Y.; Chen, X.; Chen, Z.; Wu, F.-G. Hydrogel-based phototherapy for fighting cancer and bacterial infection. Sci. China Mater. 2017, 60, 487–503.
- Liu, Y.; Bhattarai, P.; Dai, Z.; Chen, X. Photothermal therapy and photoacoustic imaging via nanotheranostics in fighting cancer. Chem. Soc. Rev. 2019, 48, 2053–2108.
- Lu, J.; Liu, X.; Liao, Y.-P.; Salazar, F.; Sun, B.; Jiang, W.; Chang, C.H.; Jiang, J.; Wang, X.; Wu, A.M. Nano-enabled pancreas cancer immunotherapy using immunogenic cell death and reversing immunosuppression. Nat. Commun. 2017, 8, 1811.
- Wang, S.; Sun, Z.; Hou, Y. Engineering Nanoparticles toward the Modulation of Emerging Cancer Immunotherapy. Adv. Healthc. Mater. 2020, 10, 2000845.
- Zhao, Y.-D.; Muhetaerjiang, M.; An, H.-W.; Fang, X.; Zhao, Y.; Wang, H. Nanomedicine Enables Spatiotemporally Regulating Macrophage-based Cancer Immunotherapy. Biomaterials 2020, 268, 120552.
- He, C.; Duan, X.; Guo, N.; Chan, C.; Poon, C.; Weichselbaum, R.R.; Lin, W. Core-shell nanoscale coordination polymers combine chemotherapy and photodynamic therapy to potentiate checkpoint blockade cancer immunotherapy. Nat. Commun. 2016, 7, 12499.
- Cabrita, R.; Lauss, M.; Sanna, A.; Donia, M.; Larsen, M.S.; Mitra, S.; Johansson, I.; Phung, B.; Harbst, K.; Vallon-Christersson, J. Tertiary lymphoid structures improve immunotherapy and survival in melanoma. Nature 2020, 577, 561–565.
- Apetoh, L.; Ladoire, S.; Coukos, G.; Ghiringhelli, F. Combining immunotherapy and anticancer agents: The right path to achieve cancer cure? Ann. Oncol. 2015, 26, 1813–1823.
- Pei, M.; Xu, R.; Zhang, C.; Wang, X.; Li, C.; Hu, Y. Mannose-functionalized antigen nanoparticles for targeted dendritic cells, accelerated endosomal escape and enhanced MHC-I antigen presentation. Colloids Surf. B Biointerfaces 2021, 197, 111378.
- Harari, A.; Graciotti, M.; Bassani-Sternberg, M.; Kandalaft, L.E. Antitumour dendritic cell vaccination in a priming and boosting approach. Nat. Rev. Drug Discov. 2020, 19, 635–652.
- Scheetz, L.; Park, K.S.; Li, Q.; Lowenstein, P.R.; Castro, M.G.; Schwendeman, A.; Moon, J.J. Engineering patient-specific cancer immunotherapies. Nat. Biomed. Eng. 2019, 3, 768–782.
- Cho, N.-H.; Cheong, T.-C.; Min, J.H.; Wu, J.H.; Lee, S.J.; Kim, D.; Yang, J.-S.; Kim, S.; Kim, Y.K.; Seong, S.-Y. A multifunctional core–shell nanoparticle for dendritic cell-based cancer immunotherapy. Nat. Nanotechnol. 2011, 6, 675–682.
- Akita, H.; Kogure, K.; Moriguchi, R.; Nakamura, Y.; Higashi, T.; Nakamura, T.; Serada, S.; Fujimoto, M.; Naka, T.; Futaki, S. Nanoparticles for ex vivo siRNA delivery to dendritic cells for cancer vaccines: Programmed endosomal escape and dissociation. J. Control. Release 2010, 143, 311–317.
- Demaria, O.; Cornen, S.; Daëron, M.; Morel, Y.; Medzhitov, R.; Vivier, E. Harnessing innate immunity in cancer therapy. Nature 2019, 574, 45–56.
- Eyquem, J.; Mansilla-Soto, J.; Giavridis, T.; van der Stegen, S.J.; Hamieh, M.; Cunanan, K.M.; Odak, A.; Gönen, M.; Sadelain, M. Targeting a CAR to the TRAC locus with CRISPR/Cas9 enhances tumour rejection. Nature 2017, 543, 113–117.
- van der Meel, R.; Sulheim, E.; Shi, Y.; Kiessling, F.; Mulder, W.J.; Lammers, T. Smart cancer nanomedicine. Nat. Nanotechnol. 2019, 14, 1007–1017.
- Shang, T.; Yu, X.; Han, S.; Yang, B. Nanomedicine-based tumor photothermal therapy synergized immunotherapy. Biomater. Sci. 2020, 8, 5241–5259.
- Lynn, G.M.; Laga, R.; Jewell, C.M. Induction of anti-cancer T cell immunity by in situ vaccination using systemically administered nanomedicines. Cancer Lett. 2019, 459, 192–203.
- Wilson, D.R.; Sen, R.; Sunshine, J.C.; Pardoll, D.M.; Green, J.J.; Kim, Y.J. Biodegradable STING agonist nanoparticles for enhanced cancer immunotherapy. Nanomed. Nanotechnol. Biol. Med. 2018, 14, 237–246.
- Han, H.D.; Byeon, Y.; Kang, T.H.; Jung, I.D.; Lee, J.-W.; Shin, B.C.; Lee, Y.J.; Sood, A.K.; Park, Y.-M. Toll-like receptor 3-induced immune response by poly(d,l-lactide-co-glycolide) nanoparticles for dendritic cell-based cancer immunotherapy. Int. J. Nanomed. 2016, 11, 5729–5742.
- Sau, S.; Alsaab, H.O.; Bhise, K.; Alzhrani, R.; Nabil, G.; Iyer, A.K. Multifunctional nanoparticles for cancer immunotherapy: A groundbreaking approach for reprogramming malfunctioned tumor environment. J. Control. Release 2018, 274, 24–34.
- Han, S.; Huang, K.; Gu, Z.; Wu, J. Tumor immune microenvironment modulation-based drug delivery strategies for cancer immunotherapy. Nanoscale 2020, 12, 413–436.
- Korangath, P.; Barnett, J.D.; Sharma, A.; Henderson, E.T.; Stewart, J.; Yu, S.-H.; Kandala, S.K.; Yang, C.-T.; Caserto, J.S.; Hedayati, M. Nanoparticle interactions with immune cells dominate tumor retention and induce T cell–mediated tumor suppression in models of breast cancer. Sci. Adv. 2020, 6, eaay1601.
- Prakash, S.; Kumbhojkar, N.; Clegg, J.R.; Mitragotri, S. Cell-bound Nanoparticles for Tissue Targeting and Immunotherapy: Engineering of the Particle-Membrane Interface. Curr. Opin. Colloid Interface Sci. 2020, 52, 101408.
- Chen, F.; Hableel, G.; Zhao, E.R.; Jokerst, J.V. Multifunctional nanomedicine with silica: Role of silica in nanoparticles for theranostic, imaging, and drug monitoring. J. Colloid Interface Sci. 2018, 521, 261–279.
- Ulbrich, K.; Hola, K.; Subr, V.; Bakandritsos, A.; Tucek, J.; Zboril, R. Targeted drug delivery with polymers and magnetic nanoparticles: Covalent and noncovalent approaches, release control, and clinical studies. Chem. Rev. 2016, 116, 5338–5431.
- Xie, Y.Q.; Wei, L.; Tang, L. Immunoengineering with biomaterials for enhanced cancer immunotherapy. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2018, 10, e1506.
- Su, S.; Hu, B.; Shao, J.; Shen, B.; Du, J.; Du, Y.; Zhou, J.; Yu, L.; Zhang, L.; Chen, F. CRISPR-Cas9 mediated efficient PD-1 disruption on human primary T cells from cancer patients. Sci. Rep. 2016, 6, 20070.
- Ren, J.; Liu, X.; Fang, C.; Jiang, S.; June, C.H.; Zhao, Y. Multiplex genome editing to generate universal CAR T cells resistant to PD1 inhibition. Clin. Cancer Res. 2017, 23, 2255–2266.
- Zeng, Z.; Pu, K. Improving Cancer Immunotherapy by Cell Membrane-Camouflaged Nanoparticles. Adv. Funct. Mater. 2020, 30, 2004397.
- Singh, A.P.; Biswas, A.; Shukla, A.; Maiti, P. Targeted therapy in chronic diseases using nanomaterial-based drug delivery vehicles. Signal. Transduct. Target. Ther. 2019, 4, 33.
- Sukumar, U.K.; Rajendran, J.C.B.; Gambhir, S.S.; Massoud, T.F.; Paulmurugan, R. SP94-Targeted Triblock Copolymer Nanoparticle Delivers Thymidine Kinase–p53–Nitroreductase Triple Therapeutic Gene and Restores Anticancer Function against Hepatocellular Carcinoma in Vivo. ACS Appl. Mater. Interfaces 2020, 12, 11307–11319.
- Irvine, D.J.; Hanson, M.C.; Rakhra, K.; Tokatlian, T. Synthetic nanoparticles for vaccines and immunotherapy. Chem. Rev. 2015, 115, 11109–11146.
- Zu, M.; Ma, L.; Zhang, X.; Xie, D.; Kang, Y.; Xiao, B. Chondroitin sulfate-functionalized polymeric nanoparticles for colon cancer-targeted chemotherapy. Colloids Surf. B Biointerfaces 2019, 177, 399–406.
- Zhang, C.; Pu, K. Molecular and nanoengineering approaches towards activatable cancer immunotherapy. Chem. Soc. Rev. 2020, 49, 4234–4253.
- Chen, Q.; Chen, M.; Liu, Z. Local biomaterials-assisted cancer immunotherapy to trigger systemic antitumor responses. Chem. Soc. Rev. 2019, 48, 5506–5526.
- Rajendrakumar, S.K.; Uthaman, S.; Cho, C.-S.; Park, I.-K. Nanoparticle-based phototriggered cancer immunotherapy and its domino effect in the tumor microenvironment. Biomacromolecules 2018, 19, 1869–1887.
- Wu, Y.; Gu, W.; Li, L.; Chen, C.; Xu, Z.P. Enhancing PD-1 gene silence in T lymphocytes by comparing the delivery performance of two inorganic nanoparticle platforms. Nanomaterials 2019, 9, 159.
- Thangam, R.; Kim, M.S.; Bae, G.; Kim, Y.; Kang, N.; Lee, S.; Jung, H.J.; Jang, J.; Choi, H.; Li, N. Remote Switching of Elastic Movement of Decorated Ligand Nanostructures Controls the Adhesion-Regulated Polarization of Host Macrophages. Adv. Funct. Mater. 2021, 31, 2008698.
- Bose, R.J.; Tharmalingam, N.; Garcia Marques, F.J.; Sukumar, U.K.; Natarajan, A.; Zeng, Y.; Robinson, E.; Bermudez, A.; Chang, E.; Habte, F. Reconstructed apoptotic bodies as targeted “nano decoys” to treat intracellular bacterial infections within macrophages and cancer cells. ACS Nano 2020, 14, 5818–5835.
- García-Aranda, M.; Redondo, M. Targeting protein kinases to enhance the response to anti-PD-1/PD-L1 immunotherapy. Int. J. Mol. Sci. 2019, 20, 2296.
- Conde, J.; Bao, C.; Tan, Y.; Cui, D.; Edelman, E.R.; Azevedo, H.S.; Byrne, H.J.; Artzi, N.; Tian, F. Dual targeted immunotherapy via in vivo delivery of biohybrid RNAi-peptide nanoparticles to tumor-associated macrophages and cancer cells. Adv. Funct. Mater. 2015, 25, 4183–4194.
- Luo, Y.-L.; Xu, C.-F.; Li, H.-J.; Cao, Z.-T.; Liu, J.; Wang, J.-L.; Du, X.-J.; Yang, X.-Z.; Gu, Z.; Wang, J. Macrophage-specific in vivo gene editing using cationic lipid-assisted polymeric nanoparticles. ACS Nano 2018, 12, 994–1005.
- Tu, K.; Deng, H.; Kong, L.; Wang, Y.; Yang, T.; Hu, Q.; Hu, M.; Yang, C.; Zhang, Z. Reshaping tumor immune microenvironment through acidity-responsive nanoparticles featured with CRISPR/Cas9-mediated programmed death-ligand 1 attenuation and chemotherapeutics-induced immunogenic cell death. ACS Appl. Mater. Interfaces 2020, 12, 16018–16030.
More
Information
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.1K
Revisions:
2 times
(View History)
Update Date:
08 Sep 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





