
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Martina Pummerova | + 2952 word(s) | 2952 | 2021-08-27 10:55:10 | | | |
| 2 | Beatrix Zheng | + 96 word(s) | 3048 | 2021-09-04 15:22:34 | | |
Video Upload Options
This study investigated the effect of natural antioxidants inherent to beetroot (Beta vulgaris var. Vulgaris) on the ageing of environmentally friendly plastics. Certain properties were examined in this context, comprising thermal, mechanical, and morphological properties. A visual evaluation of relevant changes in the given polymers (polylactide and polycaprolactone) was conducted during an ageing test in a UV chamber (45 °C, 70% humidity) for 720 h. The films were prepared by a casting process, in which samples with the extract of beetroot were additionally incorporated in a common filler (bentonite), this serving as a carrier for the extract. The results showed the effect of the incorporated antioxidant, which was added to stabilize the biodegradable films. Its efficiency during the ageing test in the polymers tended to exceed or be comparable to that of the reference sample.
[1]1. Introduction
2. Analysis on Research Results
2.1. Characterization of Beetroot Extract (BRE)
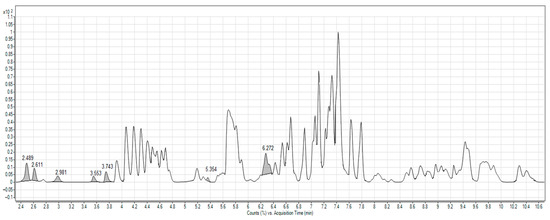
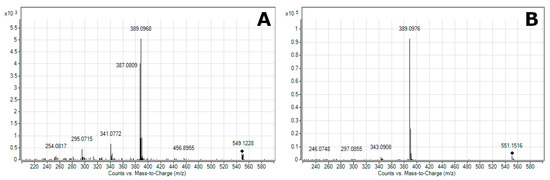
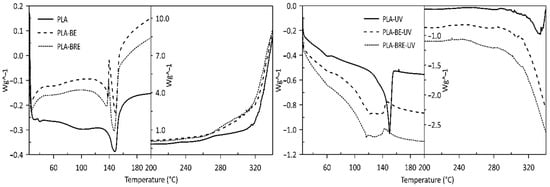
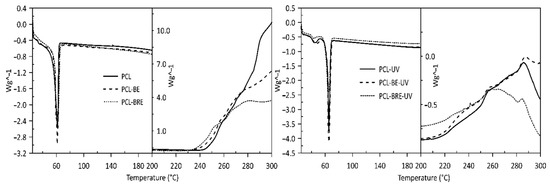
2.2. Thermal Analysis
| Sample | Tg [°C] | Tm [°C] | ∆Hm [J/g] | Tonset [°C] | Tpeak [°C] | ∆H [J/g] |
|---|---|---|---|---|---|---|
| PLA | 53 | 146 | 36 | 254 | 355 | 1805 |
| PLA-BE | 57 | 143 | 39 | 267 | 349 | 1564 |
| PLA-BRE | 57 | 145 | 38 | 265 | 347 | 1746 |
| PLA-UV | 49 | 151 | 42 | 212 | - | - |
| PLA-BE-UV | 53 | 133 | 37 | 216 | - | - |
| PLA-BRE-UV | 57 | 135 | 39 | 218 | - | - |
| Sample | Tm1 [°C] | Tm2 [°C] | Tcc [°C] | ∆Hcc [J/g] | ∆Hm [J/g] | Tonset [°C] | Tpeak [°C] | ∆H [J/g] |
|---|---|---|---|---|---|---|---|---|
| PCL | 65 | - | 34 | 3 | 88 | 245 | 312 | 3168 |
| PCL-BE | 65 | - | 34 | 3 | 86 | 240 | 314 | 2254 |
| PCL-BRE | 66 | - | 35 | 3 | 90 | 236 | 281 | 1157 |
| PCL-UV | 66 | 39 | 26 | 3 | 92 | 215 | 286 | 254 |
| PCL-BE-UV | 64 | 38 | 36 | 3 | 99 | 216 | 288 | 307 |
| PCL-BRE-UV | - | 64 | 30 | 1 | 93 | 208 | 265 | 143 |
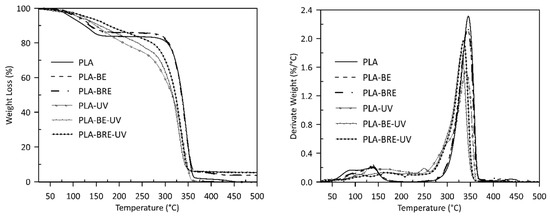
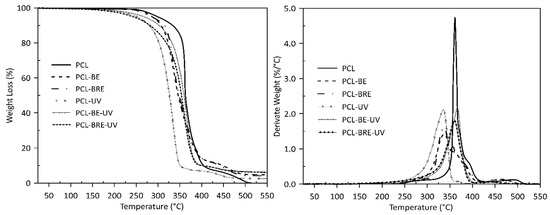
| Samples | Tonset (°C) | T10 (°C) | T50 (°C) | T90 (°C) | Mass Loss (%) |
|---|---|---|---|---|---|
| PLA | 359 | 123 | 337 | 355 | 100 |
| PLA-BE | 358 | 140 | 336 | 357 | 95 |
| PLA-BRE | 357 | 136 | 335 | 356 | 95 |
| PLA-UV | 354 | 149 | 315 | 348 | 100 |
| PLA-BE-UV | 343 | 156 | 316 | 342 | 95 |
| PLA-BRE-UV | 347 | 175 | 322 | 346 | 95 |
| PCL | 368 | 333 | 363 | 350 | 100 |
| PCL-BE | 378 | 307 | 346 | 449 | 95 |
| PCL-BRE | 378 | 315 | 348 | 443 | 95 |
| PCL-UV | 350 | 278 | 326 | 350 | 99 |
| PCL-BE-UV | 381 | 302 | 359 | 397 | 95 |
| PCL-BRE-UV | 379 | 281 | 354 | 398 | 95 |
2.3. Mechanical Properties
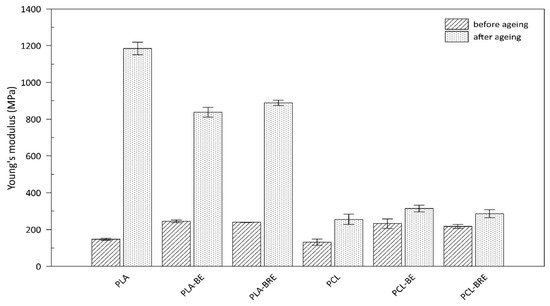
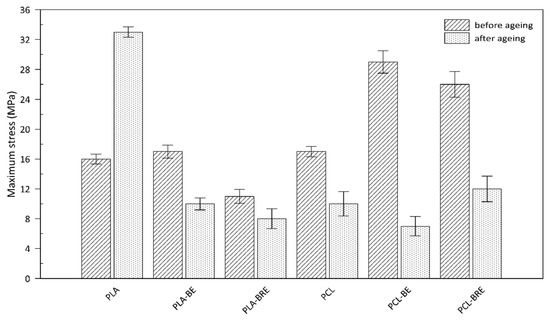
| Samples | Before Solar Ageing | After Solar Ageing | Af (‒) | ||
|---|---|---|---|---|---|
| σ (MPa) | ε (%) | σ (MPa) | ε (%) | ||
| PLA | 15 ± 1 | 354 ± 18 | 16 ± 2 | 24 ± 6 | 0.07 ± 0.01 |
| PLA-BE | 16 ± 2 | 301 ± 25 | 6 ± 2 | 5 ± 1 | 0.01 ± 0.00 |
| PLA-BRE | 10 ± 1 | 363 ± 14 | 1 ± 1 | 9 ± 1 | 0.00 ± 0.00 |
| PCL | 17 ± 1 | 1006 ± 9 | 8 ± 3 | 14 ± 6 | 0.01 ± 0.01 |
| PCL-BE | 22 ± 2 | 1008 ± 6 | 6 ± 3 | 8 ± 3 | 0.00 ± 0.01 |
| PCL-BRE | 22 ± 2 | 1062 ± 8 | 11± 3 | 375 ± 42 | 0.15 ± 0.00 |
2.4. Colour Measurement
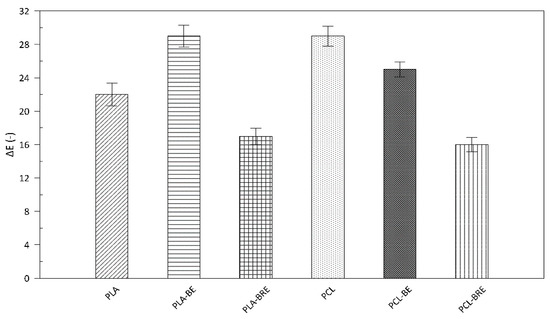
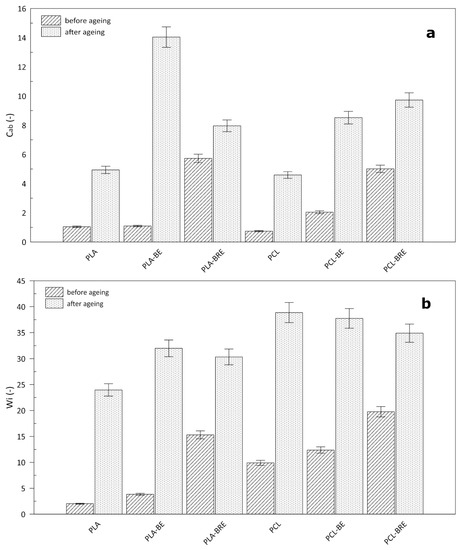
2.5. Fourier-Transform Infrared Spectroscopy (FT-IR)
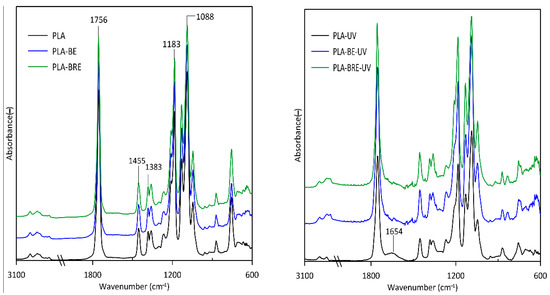
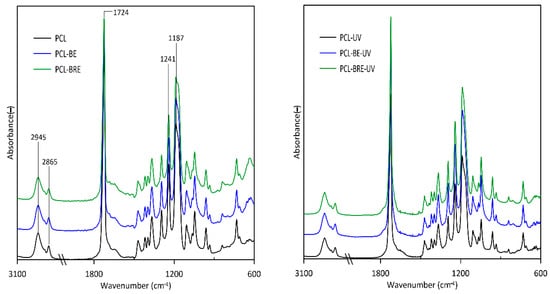
2.6. Scanning Electron Microscopy (SEM)


References
- Petra Drohsler; Jaroslav Cisar; Tomas Sopik; Vladimir Sedlarik; Martina Pummerova; Effect of an Antioxidant Based on Red Beetroot Extract on the Abiotic Stability of Polylactide and Polycaprolactone. Molecules 2021, 26, 5190, 10.3390/molecules26175190.
- Jamshidian, M.; Tehrany, E.A.; Imran, M.; Akhtar, M.J.; Cleymand, F.; Desobry, S. Structural, mechanical and barrier properties of active PLA–antioxidant films. J. Food Eng. 2012, 110, 380–389.
- Zeid, A.; Karabagias, I.K.; Nassif, M.; Kontominas, M.G. Preparation and evaluation of antioxidant packaging films made of polylactic acid containing thyme, rosemary, and oregano essential oils. J. Food Process. Preserv. 2019, 43, e14102.
- Quiles-Carrillo, L.; Montava-Jordà, S.; Boronat, T.; Sammon, C.; Balart, R.; Torres-Giner, S. On the Use of Gallic Acid as a Potential Natural Antioxidant and Ultraviolet Light Stabilizer in Cast-Extruded Bio-Based High-Density Polyethylene Films. Polymers 2020, 12, 31.
- Byun, Y.; Kim, Y.; Whiteside, S. Characterization of an antioxidant polylactic acid (PLA) film prepared with α-tocopherol, BHT and polyethylene glycol using film cast extruder. J. Food Eng. 2010, 100, 239–244.
- Fasihnia, S.H.; Peighambardoust, S.H.; Peighambardoust, S.J.; Oromiehie, A.; Soltanzadeh, M.; Peressini, D. Migration analysis, antioxidant, and mechanical characterization of polypropylene-based active food packaging films loaded with BHA, BHT, and TBHQ. J. Food Sci. 2020, 85, 2317–2328.
- Hanafi, H.; Nurdiani, N.; Sirait, S.; Widyahapsari, D.; Irawan, C. Migration Test of Polylactic Acid Packaging that Modified with (Butyl hydroxy toluene) and (Tert butyl hydroxy quinon) Synthetic Antioxidant in Food Simulant. Orient. J. Chem. 2019, 35, 552–556.
- Veiga-Santos, P.; Silva, L.; Oliveira de souza, C.; Silva, J.; Albuquerque, E.; Druzian, J. Coffee-cocoa additives for bio-based antioxidant packaging. Food Packag. Shelf Life 2018, 18, 37–41.
- Kwak, H.S.; Ji, S.; Jeong, Y. The effect of air flow in coffee roasting for antioxidant activity and total polyphenol content. Food Control 2017, 71, 210–216.
- Bae, I.; Ham, H.; Jeong, M.; Kim, D.; Kim, H. Simultaneous determination of 15 phenolic compounds and caffeine in teas and mate using RP-HPLC/UV detection: Method development and optimization of extraction process. Food Chem. 2015, 172, 469–475.
- Vostrejs, P.; Adamcová, D.; Vaverková, M.; Enev, V.; Kalina, M.; Machovsky, M.; Šourková, M.; Marova, I.; Kovalcik, A. Active biodegradable packaging films modified with grape seeds lignin. RSC Adv. 2020, 10, 29202–29213.
- Rehman, S.; Abbasi, K.; Qayyum, A.; Jahangir, M.; Sohail, A.; Nisa, S.; Tareen, M.; Tareen, M.; Sopade, P. Comparative analysis of citrus fruits for nutraceutical properties. Food Sci. Technol. 2019, 40, 153–157.
- Chan, C.L.; Gan, R.-Y.; Corke, H. The phenolic composition and antioxidant capacity of soluble and bound extracts in selected dietary spices and medicinal herbs. Int. J. Food Sci. Technol. 2016, 51, 565–573.
- Kirschweng, B.; Tátraaljai, D.; Földes, E.; Pukanszky, B. Natural antioxidants as stabilizers for polymers. Polym. Degrad. Stab. 2017, 145, 25–40.
- Fu, Y.; Shi, J.; Xie, S.-Y.; Zhang, T.-Y.; Soladoye, O.; Aluko, R. Red Beetroot Betalains: Perspectives on Extraction, Processing, and Potential Health Benefits. J. Agric. Food Chem. 2020, 68, 11595–11611.
- Ravichandran, K.; Saw, N.M.M.T.; Mohdaly, A.; Gabr, A.; Kastell, A.; Riedel, H.; Cai, Z.; Knorr, D.; Smetanska, I. Impact of processing of red beet on betalain content and antioxidant activity. Food Res. Int. 2013, 50, 670–675.
- Bastos, E.; Schliemann, W. Betalains as Antioxidants. In Plant Antioxidants and Health; Springer: Cham, Switzerland, 2021; pp. 1–44. ISBN 978-3-030-45299-5.
- Belhadj Slimen, I.; Najar, T.; Abderrabba, M. Chemical and Antioxidant Properties of Betalains. J. Agric. Food Chem. 2017, 65, 675–689.
- Gokhale, S.; Lele, S. Betalain Content and Antioxidant Activity of Beta vulgaris: Effect of Hot Air Convective Drying and Storage. J. Food Process. Preserv. 2014, 38, 585–590.
- Vargas-Rubóczki, T.; Raczkó, V.; Takácsné Hájos, M. Evaluation of morphological parameters and bioactive compounds in different varieties of beetroot (Beta vulgaris L. ssp. esculenta GURKE var. rubra L.). Int. J. Hortic. Sci. 2015, 21, 31–35.
- Shyamala, B.; Prakash, J. Nutritional Content and Antioxidant Properties of Pulp Waste from Daucus carota and Beta vulgaris. Malays. J. Nutr. 2010, 16, 397–408.
- Tsao, R. Chemistry and biochemistry of dietary polyphenols. Nutrients 2010, 2, 1231–1246.
- Chiorcea-Paquim, A.-M.; Enache, T.A.; De Souza Gil, E.; Oliveira-Brett, A.M. Natural phenolic antioxidants electrochemistry: Towards a new food science methodology. Compr. Rev. Food Sci. Food Saf. 2020, 19, 1680–1726.
- Rosecler, M.; Rossetto, M.R.; Vianello, F.; Rocha, S.; Pace, G.; Lima, G. Antioxidant substances and pesticide in parts of beet organic and conventional manure. Afr. J. Plant Sci. 2009, 3, 245–253.
- Lembong, E.; Utama, G.L.; Saputra, R. Phytochemical Test, Vitamin C Content and Antioxidant Activities Beet Root (Beta vulgaris Linn.) Extracts as Food Coloring Agent from Some Areas in Java Island. In IOP Conference Series: Materials Science and Engineering; IOP Publishing: Bristol, UK, 2019; Volume 306, p. 012010.
- Jiratanan, T.; Liu, R. Antioxidant Activity of Processed Table Beets (Beta vulgaris var, conditiva) and Green Beans (Phaseolus vulgaris L.). J. Agric. Food Chem. 2004, 52, 2659–2670.
- Kujala, T.; Vienola, M.; Klika, K.; Loponen, J.; Pihlaja, K. Betalain and phenolic compositions of four beetroot (Beta vulgaris) cultivars. Eur. Food Res. Technol. 2002, 214, 505–510.
- Slatnar, A.; Stampar, F.; Veberic, R.; Jakopič, J. HPLC-MSn Identification of Betalain Profile of Different Beetroot (Beta vulgaris L. ssp. vulgaris) Parts and Cultivars. J. Food Sci. 2015, 80, C1952–C1958.
- Nestora, S.; Merlier, F.; Prost, E.; Haupt, K.; Rossi, C.; Bui, B.T.S. Solid-phase extraction of betanin and isobetanin from beetroot extracts using a dipicolinic acid molecularly imprinted polymer. J. Chromatogr. A 2016, 1465, 47–54.
- Dintcheva, N.T.; Al-Malaika, S.; Morici, E.; Arrigo, R. Thermo-oxidative stabilization of poly(lactic acid)-based nanocomposites through the incorporation of clay with in-built antioxidant activity. J. Appl. Polym. Sci. 2017, 134, 44974.
- Syarofi, R.; Wirjosentono, B.; Tamrin; Rihayat, T. Mechanical Properties, Morphology and Thermal Degradation of PCL (Poly ε-Caprolactone) Biodegradable Polymer Blended Nanocomposites with Aceh Bentonite as Filler. In IOP Conference Series: Materials Science and Engineering; IOP Publishing: Bristol, UK, 2019; Volume 536, p. 12040.
- Suryani; Agusnar, H.; Wirjosentono, B.; Rihayat, T.; Aidy, N. Improving the quality of biopolymer(poly lactic acid) with the addition of bentonite as filler. In IOP Conference Series: Materials Science and Engineering; IOP Publishing: Bristol, UK, 2017; Volume 222, p. 012008.
- Arrigo, R.; Dintcheva, N. Natural Anti-oxidants for Bio-polymeric Materials. Arch. Chem. Res. 2017, 1, 2.
- Salević, A.; Prieto, C.; Cabedo, L.; Nedović, V.; Lagaron, J. Physicochemical, Antioxidant and Antimicrobial Properties of Electrospun Poly(ε-caprolactone) Films Containing a Solid Dispersion of Sage (Salvia officinalis L.) Extract. Nanomaterials 2019, 9, 270.
- Marra, A.; Cimmino, S.; Silvestre, C. Effect of TiO2 and ZnO on PLA degradation in various media. Adv. Mater. Sci. 2017, 2, 1–8.
- Kosowska, K.; Szatkowski, P. Influence of ZnO, SiO2 and TiO2 on the aging process of PLA fibers produced by electrospinning method. J. Therm. Anal. Calorim. 2019, 140, 1769–1778.
- Darain, F.; Chan, W.Y.; Chian, K. Performance of Surface-Modified Polycaprolactone on Growth Factor Binding, Release, and Proliferation of Smooth Muscle Cells. Soft Mater. 2010, 9, 64–78.
- França, D.; Morais, D.; Bezerra, E.; Araujo, E.; Wellen, R. Photodegradation Mechanisms on Poly(ε-caprolactone) (PCL). Mater. Res. 2018, 21.





