
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Carmine Cpacchione | + 1239 word(s) | 1239 | 2021-08-18 10:58:57 | | | |
| 2 | Beatrix Zheng | + 164 word(s) | 1403 | 2021-08-27 10:41:38 | | |
Video Upload Options
The terpolymerization of CO2 with epoxides and organic anhydrides or cyclic esters offers the possibility, combining the ROCOP with ring-opening polymerization (ROP), to access a wide range of materials containing polycarbonate and polyester segments along the polymer chain, showing enhanced properties with respect to the simple APC. This review will cover the last advancements in the field, evidencing the crucial role of the catalytic system in determining the microstructural features of the final polymer.
1. Introduction
In particular, the alternating ring-opening copolymerization (ROCOP) of CO 2 with epoxides has offered a conceptually simple route for the synthesis of aliphatic polycarbonates (APC), which show a clear advantage in terms of biodegradability with respect to polyolefins [1][2][3]. APC, however, often display poor chemical and mechanical properties compared to aromatic polycarbonates, and the incorporation of epoxides with various structural features does not always result in an improvement in the final properties of APC [4][5][6]. Aliphatic polyesters are another important class of biopolymers that can be conveniently obtained by the ring-opening polymerization (ROP) of cyclic esters [7][8][9][10] or by the ROCOP of epoxides with cyclic anhydrides [11][12][13][14]. Transition metal complexes generally catalyze all these polymerization processes through a coordination–insertion mechanism. Actually, taking a closer look at the polymerization mechanism of the ROCOP of epoxides with CO 2 or cyclic anhydrides depicted in Scheme 1 , it is evident that the formation of the metal-alkoxo bond (a’ in Scheme 1 ) is a key intermediate in the propagation process. In analogy, the ROP of cyclic esters proceeds via the formation of a metal-alkoxo bond (a in Scheme 1 ) that allows the ring-opening of the following monomer unit.
Given this mechanistic scenario, it is easy to imagine that it is possible to design a metal complex able to promote both types of polymerization, allowing us to obtain various block-copolymers. In principle, it is possible to obtain copolymers with polycarbonate and polyester segments and modulate the nature of the polycarbonate and polyester blocks, permitting the synthesis of new materials with tailored properties.
Notwithstanding the potential of such an approach, the efforts to develop efficient catalytic systems that cannot only incorporate CO 2 but also give rise to unprecedented new materials have raised good results only recently.
This entry will cover the last advancements (since 2003) in the metal-catalyzed and metal-free terpolymerization of CO 2 with epoxides and cyclic esters or cyclic organic anhydrides for the obtaining of polycarbonate–polyester copolymers.
2. Terpolymerization of CO2 with Epoxides and Cyclic Anhydrides
The same catalytic system 4 was also used for the synthesis of pseudo-interpenetrating poly(propylenecarbonate) by the terpolymerization ( p CO 2 = 5.4 MPa, T = 70 °C, t = 36 h) of CO 2 with PO and pyromellitic dianhydride (PMDA) up to 4% (in this case, M n increased up to 862 kg mol −1 ), resulting in a noticeable improvement in the mechanical and thermal properties with respect to the corresponding polycarbonate [15].
In analogy to the polymerization process observed by Coates in the case of the (bdi)Zn complexes, the formation of the polyester is favored over the formation of the polycarbonate, resulting in the formation of a poly(ester- block -carbonate). The authors also showed that DSC of poly(ester- block -carbonate) is inconclusive in giving information about the blocky microstructure of the copolymer because the polyester and polycarbonate phases are completely miscible, giving a single value for the T g . Furthermore, in the case of complex 10, the authors noticed that the presence of CO 2 in the polymerization feed completely suppresses the formation of polyether linkages. In particular, by copolymerizing the equimolar amount of CHO and CPrA in the presence of CO 2, the pure polyester was obtained, while without CO 2 an amount of 15–30% of polyether linkages was observed. For all terpolymerizations, M n (up to 19.2 kg mol −1 ) showed a linear correlation with conversion and the Ð was ≤1.6, indicating controlled behavior.
Since the discovery by Feng and coworkers that triethyl borane (TEB) in combination with onium halides or alkoxides promotes the formation of polycarbonates by coupling CO 2 with PO or CHO, the efforts to extend the use of this metal-free system to the terpolymerization of CO 2 with epoxides and anhydrides resulted in the synthesis of terpolymers having various microstructural features [16].
In 2020, Meng reported the quadripolymerization of CO 2 with PA, PO and CHO in the presence of TEB and PPNCl, resulting in the formation of the copolymer ( p CO 2 = 1 MPa, T = 70 °C, t = 24–96 h) with good selectivity (94%) with respect to the cyclic product [17][18]. The microstructure of the resulting quadripolymer was clarified by 1H and 13C NMR showing the presence of four main blocks, i.e., poly(PA- alt -CHO), poly(PA- alt -PO), poly(propylene carbonate) (PPC), and poly(cyclohexene carbonate) (PCHC), and a very low amount of polyether linkages (<1%). In addition, in this case the formation of the polycarbonate segments only starts after the complete PA conversion and thus after the formation of the polyester segments. The resulting polymers display narrow dispersity ( Ð = 1.14–1.21) and a high molecular weight ( M n up to 77 kg mol −1 ). It is worth reporting that the T g can be easily tuned by regulating the feed ratio with a wide temperature range ( T g = 82–116 °C).
3. Terpolymerization of CO2 with Epoxides and Cyclic Esters
The same catalytic system 24 was also used to obtain pentablock copolymers by alternating ROCOP (anhydrides/epoxide), ROP (lactone) and ROCOP (CO 2/epoxide) by using various epoxides (CHO and VCHO), anhydrides (PA, NA), and DL (ε-decalactone). The resulting pentablock copolymers show a single T g (from −35 to 20 °C), low molecular weight (10–16 kg mol −1 ) [19] and Ð = 1.06–1.16.
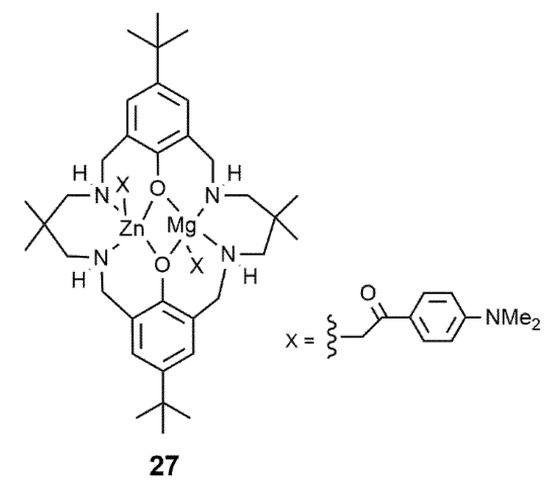
Lately, a heterodinuclear Zn/Mg catalyst 27 ( Figure 1 ) with the same ligand framework promoted the formation of ABA triblock copolymers by using DL with high activity [20].
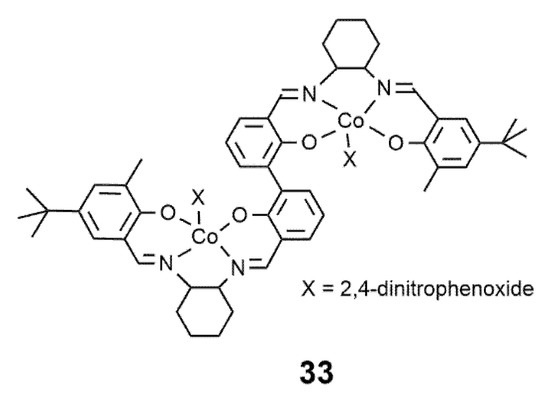
Finally, in the presence of an enantiopure chiral salenCo(III) complex 33 ( Figure 2 ) in combination with PPN-DNP (PPN = bis(triphenylphosphine)iminium, DNP = 2,4-dinitrophenoxide), Lu and coworkers also succeeded in producing CO 2/CHO/BBL terpolymers with isotactic -PCHC blocks [21].
In Table 1 , the main data relating to the terpolymerizations of CO 2 with epoxides and cyclic esters discussed in this second part are summarized.
| Catalyst | Monomers | Polymerization Conditions pCO2, T, t |
Mn * (Ð) kg mol−1 |
Polymer Microstructure | Ref. |
|---|---|---|---|---|---|
| 1 | CO2, PO, CL | 4.0 MPa, 50–90 °C, 16 h | - | random | [22] |
| 4 | CO2, PO, CL | 2.8 MPa, 60 °C, 40 h | 27.5 (1.50–2.97) | block | [23] |
| 4 | CO2, PO, CL | 1.0 MPa, 60 °C, 6 h | 10.8 (1.3–1.6) | grafted | [24] |
| 15–22 | CO2, CHO, LA | 4.0 MPa, 90 °C, 16 h | 41.6 (1.09–1.96) | alternated/random | [25] |
| 23 | CO2, PO, L-LA | 4.0 MPa, 70 °C, 10 h | 15.4 (4.2–9.9) | tapered/random | [26] |
| 24 | CO2, CHO, CL | 0.1 MPa, 80 °C, 2–21 h | 4.8 (1.38–1.49) | block | [27] |
| 24 | CO2, CHO, CL | 0.1 MPa, 80 °C, 16–25 h | 13.8 (1.29–1.49) | block | [28] |
| 24 | CO2, CHO, VCHO, PA/NA, CL | 0.1 MPa, 100 °C | 16 (1.06–1.16) | block | [19] |
| 25–26 | CO2, CHD, DL | 2 MPa, 40–100 °C, 2–24 h | 114 (1.38–1.49) | block | [29] |
| 27 | CO2, CHO, DL | 2 MPa, 80 °C, 21 h | 71.9 (1.07–1.16) | block | [20] |
| 28 | CO2, BBL, CHO/CPO | 0.3–4 MPa, 60 °C, 0.1–7 h | 166 (1.2–1.8) | random/block | [30] |
| 28 | CO2, BBL, LO | 0.9–4 MPa, 40–60 °C, 8–22 h | 233 (1.23–1.39) | random/block | [31] |
| 29 | CO2, SO, LA | 1.5 MPa, 25 °C, 2–6 h | 17.2 (1.04–1.12) | block | [32] |
| 29 | CO2, PO, LA | 1.5 MPa, 25 °C, 1–4 h | 20.1 (1.02–1.04) | block | [33] |
| 30–32 | CO2, PO/CHO/SO, LA | 2 MPa, 60 °C, 4–48 h | 13.6 (1.19–3.15) | block | [34] |
| 33 | CO2, CHO, BBL | 2.0 MPa, 40 °C, 2–4 h | 14.6 (1.19–1.44) | block | [35] |
4. Conclusions
The possibility to terpolymerize CO 2 with epoxides and other cyclic monomers (cyclic esters, organic anhydrides) offers not only a simple way to obtain a wide range of materials with unprecedented properties, but also the possibility to have such material in a completely sustainable way, combining CO 2 with monomers originating from biomasses. The last decade has witnessed tremendous efforts in the development of efficient catalytic systems able to combine the ROP of cyclic esters and the ROCOP of CO 2 or cyclic organic anhydrides with epoxides, allowing us to obtain polymers with various microstructural features spanning from statistical, to AB, ABA, and even more complex architectures. Notwithstanding these endeavors, however, fine control of the microstructure and the molecular weight is still a major challenge in the field. Furthermore, the number of metal centers active in the terpolymerization of CO 2 with epoxides and cyclic esters of anhydrides is still limited, offering active catalysts only in the case of Zn, Cr and Co, and, only in the case of the terpolymerization with cyclic anhydrides, in the presence of metal-free borane-based catalysts.
Therefore, this entry is not only an overview on the progress in the field, but also shows that there is a large space for further developments. More precisely, higher control over the polymer microstructure, an extension to a wider range of monomers and the development of new catalytic systems based on other metal centers to improve the activity and the control of the polymerization process will be highly desirable targets in future developments.
References
- Kernbichl, S.; Rieger, B. Aliphatic Polycarbonates Derived from Epoxides and CO2. In Engineering Solutions for CO2 Conversion; Reina, T.R., Arellano-Garcia, H., Odriozola, J.A., Eds.; Wiley: Weinheim, Germany, 2021; pp. 385–406. ISBN 978-3-527-34652-3.
- Yu, W.; Maynard, E.; Chiaradia, V.; Arno, M.C.; Dove, A.P. Aliphatic Polycarbonates from Cyclic Carbonate Monomers and Their Application as Biomaterials. Chem. Rev. 2021.
- Janeta, M.; Lis, T.; Szafert, S. Zinc Imine Polyhedral Oligomeric Silsesquioxane as a Quattro-Site Catalyst for the Synthesis of Cyclic Carbonates from Epoxides and Low-Pressure CO2. Chem. Eur. J. 2020, 26, 13686–13697.
- Della Monica, F.; Paradiso, V.; Grassi, A.; Milione, S.; Cavallo, L.; Capacchione, C. A Novel [OSSO]-Type Chromium(III) Complex as a Versatile Catalyst for Copolymerization of Carbon Dioxide with Epoxides. Chem. Eur. J. 2020, 26, 5347–5353.
- Wang, Y.; Darensbourg, D.J. Carbon Dioxide-Based Functional Polycarbonates: Metal Catalyzed Copolymerization of CO2 and Epoxides. Coord. Chem. Rev. 2018, 372, 85–100.
- Della Monica, F.; Kleij, A.W. From Terpenes to Sustainable and Functional Polymers. Polym. Chem. 2020, 11, 5109–5127.
- Lecomte, P.; Jérôme, C. Recent Developments in Ring-Opening Polymerization of Lactones. In Synthetic Biodegradable Polymers; Rieger, B., Künkel, A., Coates, G.W., Reichardt, R., Dinjus, E., Zevaco, T.A., Eds.; Advances in Polymer Science; Springer: Berlin/Heidelberg, Germany, 2011; Volume 245, pp. 173–217. ISBN 978-3-642-27153-3.
- Santoro, O.; Zhang, X.; Redshaw, C. Synthesis of Biodegradable Polymers: A Review on the Use of Schiff-Base Metal Complexes as Catalysts for the Ring Opening Polymerization (ROP) of Cyclic Esters. Catalysts 2020, 10, 800.
- Gao, J.; Zhu, D.; Zhang, W.; Solan, G.A.; Ma, Y.; Sun, W.-H. Recent Progress in the Application of Group 1, 2 & 13 Metal Complexes as Catalysts for the Ring Opening Polymerization of Cyclic Esters. Inorg. Chem. Front. 2019, 6, 2619–2652.
- Di Iulio, C.; Jones, M.D.; Mahon, M.F.; Apperley, D.C. Zinc(II) Silsesquioxane Complexes and Their Application for the Ring-Opening Polymerization of Rac-Lactide. Inorg. Chem. 2010, 49, 10232–10234.
- Paul, S.; Zhu, Y.; Romain, C.; Brooks, R.; Saini, P.K.; Williams, C.K. Ring-Opening Copolymerization (ROCOP): Synthesis and Properties of Polyesters and Polycarbonates. Chem. Commun. 2015, 51, 6459–6479.
- Longo, J.M.; Sanford, M.J.; Coates, G.W. Ring-Opening Copolymerization of Epoxides and Cyclic Anhydrides with Discrete Metal Complexes: Structure–Property Relationships. Chem. Rev. 2016, 116, 15167–15197.
- Liang, J.; Ye, S.; Wang, S.; Xiao, M.; Meng, Y. Design and Structure of Catalysts: Syntheses of Carbon Dioxide-Based Copolymers with Cyclic Anhydrides and/or Cyclic Esters. Polym. J. 2021, 53, 3–27.
- Ryzhakov, D.; Printz, G.; Jacques, B.; Messaoudi, S.; Dumas, F.; Dagorne, S.; Le Bideau, F. Organo-Catalyzed/Initiated Ring Opening Co-Polymerization of Cyclic Anhydrides and Epoxides: An Emerging Story. Polym. Chem. 2021, 12, 2932–2946.
- Gao, L.; Feng, J. A One-Step Strategy for Thermally and Mechanically Reinforced Pseudo-Interpenetrating Poly(Propylene Carbonate) Networks by Terpolymerization of CO2, Propylene Oxide and Pyromellitic Dianhydride. J. Mater. Chem. A 2013, 1, 3556.
- Zhang, D.; Boopathi, S.K.; Hadjichristidis, N.; Gnanou, Y.; Feng, X. Metal-Free Alternating Copolymerization of CO2 with Epoxides: Fulfilling “Green” Synthesis and Activity. J. Am. Chem. Soc. 2016, 138, 11117–11120.
- Ye, S.; Wang, W.; Liang, J.; Wang, S.; Xiao, M.; Meng, Y. Metal-Free Approach for a One-Pot Construction of Biodegradable Block Copolymers from Epoxides, Phthalic Anhydride, and CO2. ACS Sustain. Chem. Eng. 2020, 8, 17860–17867.
- Liang, J.; Ye, S.; Wang, W.; Fan, C.; Wang, S.; Han, D.; Liu, W.; Cui, Y.; Hao, L.; Xiao, M.; et al. Performance Tailorable Terpolymers Synthesized from Carbon Dioxide, Phthalic Anhydride and Propylene Oxide Using Lewis Acid-Base Dual Catalysts. J. CO2 Util. 2021, 49, 101558.
- Chen, T.T.D.; Zhu, Y.; Williams, C.K. Pentablock Copolymer from Tetracomponent Monomer Mixture Using a Switchable Dizinc Catalyst. Macromolecules 2018, 51, 5346–5351.
- Sulley, G.S.; Gregory, G.L.; Chen, T.T.D.; Peña Carrodeguas, L.; Trott, G.; Santmarti, A.; Lee, K.-Y.; Terrill, N.J.; Williams, C.K. Switchable Catalysis Improves the Properties of CO2-Derived Polymers: Poly(Cyclohexene Carbonate-b-ε-Decalactone-b-Cyclohexene Carbonate) Adhesives, Elastomers, and Toughened Plastics. J. Am. Chem. Soc. 2020, 142, 4367–4378.
- Li, W.-B.; Liu, Y.; Lu, X.-B. Bimetallic Cobalt Complex-Mediated Enantioselective Terpolymerizations of Carbon Dioxide, Cyclohexene Oxide, and β-Butyrolactone. Organometallics 2020, 39, 1628–1633.
- Liu, S.; Xiao, H.; Huang, K.; Lu, L.; Huang, Q. Terpolymerization of Carbon Dioxide with Propylene Oxide and ε-Caprolactone: Synthesis, Characterization and Biodegradability. Polym. Bull. 2006, 56, 53–62.
- Hwang, Y.; Jung, J.; Ree, M.; Kim, H. Terpolymerization of CO2 with Propylene Oxide and ε-Caprolactone Using Zinc Glutarate Catalyst. Macromolecules 2003, 36, 8210–8212.
- Zhou, J.; Wang, W.; Villarroya, S.; Thurecht, K.J.; Howdle, S.M. Epoxy Functionalised Poly (ε-Caprolactone): Synthesis and Application. Chem. Commun. 2008, 44, 5806–5808.
- Kröger, M.; Folli, C.; Walter, O.; Döring, M. Alternating Copolymerization of Carbon Dioxide and Cyclohexene Oxide and Their Terpolymerization with Lactide Catalyzed by Zinc Complexes of N,N Ligands. Adv. Synth. Catal. 2006, 348, 1908–1918.
- Gu, L.; Qin, Y.; Gao, Y.; Wang, X.; Wang, F. One-Pot Terpolymerization of CO2, Propylene Oxide and Lactide Using Rare-Earth Ternary Catalyst. Chin. J. Chem. 2012, 30, 2121–2125.
- Romain, C.; Williams, C.K. Chemoselective Polymerization Control: From Mixed-Monomer Feedstock to Copolymers. Angew. Chem. Int. Ed. 2014, 53, 1607–1610.
- Paul, S.; Romain, C.; Shaw, J.; Williams, C.K. Sequence Selective Polymerization Catalysis: A New Route to ABA Block Copoly (Ester-b-Carbonate-b-Ester). Macromolecules 2015, 48, 6047–6056.
- Carrodeguas, L.P.; Chen, T.T.D.; Gregory, G.L.; Sulley, G.S.; Williams, C.K. High Elasticity, Chemically Recyclable, Thermoplastics from Bio-Based Monomers: Carbon Dioxide, Limonene Oxide and ε-Decalactone. Green Chem. 2020, 22, 8298–8307.
- Kernbichl, S.; Reiter, M.; Adams, F.; Vagin, S.; Rieger, B. CO2-Controlled One-Pot Synthesis of AB, ABA Block, and Statistical Terpolymers from β-Butyrolactone, Epoxides, and CO2. J. Am. Chem. Soc. 2017, 139, 6787–6790.
- Kernbichl, S.; Reiter, M.; Mock, J.; Rieger, B. Terpolymerization of β-Butyrolactone, Epoxides, and CO2: Chemoselective CO2-Switch and Its Impact on Kinetics and Material Properties. Macromolecules 2019, 52, 8476–8483.
- Wu, G.-P.; Darensbourg, D.J.; Lu, X.-B. Tandem Metal-Coordination Copolymerization and Organocatalytic Ring-Opening Polymerization via Water to Synthesize Diblock Copolymers of Styrene Oxide/CO2 and Lactide. J. Am. Chem. Soc. 2012, 134, 17739–17745.
- Darensbourg, D.J.; Wu, G.-P. A One-Pot Synthesis of a Triblock Copolymer from Propylene Oxide/Carbon Dioxide and Lactide: Intermediacy of Polyol Initiators. Angew. Chem. Int. Ed. 2013, 52, 10602–10606.
- Li, X.; Hu, C.; Pang, X.; Duan, R.; Chen, X. One-Pot Copolymerization of Epoxides/Carbon Dioxide and Lactide Using a Ternary Catalyst System. Catal. Sci. Technol. 2018, 8, 6452–6457.
- Li, X.; Duan, R.; Hu, C.; Pang, X.; Deng, M. Copolymerization of Lactide, Epoxides and Carbon Dioxide: A Highly Efficient Heterogeneous Ternary Catalyst System. Polym. Chem. 2021, 12, 1700–1706.





