
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Senji Okuno | + 2133 word(s) | 2133 | 2021-07-27 08:30:45 | | | |
| 2 | Lindsay Dong | Meta information modification | 2133 | 2021-08-26 07:46:33 | | |
Video Upload Options
In the current aging society of Japan, malnutrition and resultant sarcopenia have been widely identified as important symptomatic indicators of ill health and can cause impairments of longevity and quality of life in older individuals. Elderly individuals are recommended to have sufficient calorie and protein intake so as to enjoy a satisfactory quality of life, including maintaining activities of daily living in order to avoid emaciation and sarcopenia. The prevalence of emaciation and sarcopenia in elderly hemodialysis (HD) patients in Japan is higher than in non-HD elderly subjects due to the presence of malnutrition and sarcopenia associated with chronic kidney disease (CKD). Furthermore, comorbidities, such as diabetes and osteoporosis, induce malnutrition and sarcopenia in HD patients.
1. Introduction
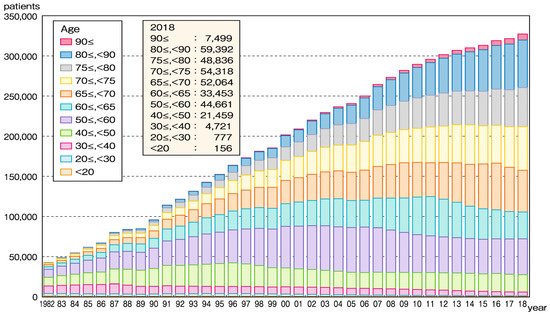
The society of Japan is aging, and the percentage of predialysis-chronic kidney disease (CKD) patients is greater in older populations; thus, it is not surprising that the number of elderly CKD patients who require renal replacement therapy (RRT) has been increasing. Since as few as 3% of dialysis patients can be maintained on peritoneal dialysis [1] and kidney transplantation is uncommon [2], nearly all end-stage CKD patients undergo hemodialysis (HD) as RRT. In fact, the proportion of HD patients in Japan ≥65 years old has increased to 71%, and that of those ≥75 years old has increased to 43% (Figure 1) [3]. An analysis of the annual dialysis data report for the 2018 JSDT renal data registry [3] shows that the increasing age of HD patients in Japan can be accounted for by both elongation of HD duration due to the sophistication of dialysis techniques, and increasing age at the time of HD initiation. The DOPPS study demonstrated that the mortality rate in Japan is the lowest among the DOPPS-participating countries, and has continued to decline given the increasing age of Japanese HD patients. Along with the aging of the HD patient population, the number of co-morbidities that may impair a satisfactory quality of life (QOL), and thus cause emaciation and sarcopenia, is increasing. Impaired QOL in elderly HD patients changes patients’ condition from independent living to the requirement of physical support or nursing care [4], thus impairing the quality of a potential 100-year life.
2. Preferential Occurrence of Malnutrition and Its Mechanism in Pre-Dialysis CKD Patients
Old age is known to be associated with poor nutritional status, while CKD itself is closely associated with malnutrition because of several different mechanisms [5]. First, CKD may be associated with dietary inadequacy in relation to suboptimal energy and protein intake due to poor appetite status, taste perception [6], low diet quality, and/or psychosocial or financial barriers. Furthermore, a reduction in metabolic rate resulting from reduced physical activity and muscle mass can contribute to poor appetite in HD patients [7]. Second, a protein-restricted diet has been recommended for pre-dialysis CKD patients to prevent exacerbation of renal dysfunction, though that might be a risk factor for malnutrition and sarcopenia [8], which is supported by the findings showing the beneficial effects of a high-protein diet or amino acid supplementation on nutritional state, as assessed by improved serum albumin and various nutritional markers [9]. Third, CKD is complicated by a metabolic syndrome termed malnutrition–inflammation complex syndrome (MICS) [10], or protein energy wasting (PEW) syndrome [11], which consist of catabolic inflammatory reactions and cachexia leading to malnutrition. Fourth, it is possible that multiple comorbidities associated with malnutrition and frailty/sarcopenia, such as diabetes mellitus (DM), cardiovascular disease (CVD), cerebrovascular disease, immobility, and insomnia, contribute to malnutrition. Interestingly, a study of common malnutrition in pre-dialysis CKD patients found that more than 50% of nephrologists initiate dialysis for end-stage CKD patients after their nutritional status is impaired [12].
3. Significance of Malnutrition for Various Clinical Outcomes in HD Patients
Along with the common occurrence of malnutrition in end-stage CKD cases, a malnutritional state can continue even when the patient reaches a stable condition on maintenance HD. Although HD initiation allows for a more liberal intake of protein and food so as to improve nutritional state [13][14], dialysis-specific factors still exist that cause malnutrition.
At the time of HD initiation, a patient presenting with malnutrition has a high mortality risk on the basis of low nutrition markers, such as geriatric nutritional risk index (GNRI) [15], subjective global assessment [16], low body mass index (BMI) [17], low serum levels of albumin [15] and cholesterol [16], and low food intake [18].
4. Significance of Sarcopenia in Relation to Harmful Effects of Malnutrition in HD Patients
Sarcopenia was defined in 1988 as an age-related reduction in skeletal muscle mass and function [19], after which the Asia Working Group for Sarcopenia provided a definition for the evaluation of sarcopenia in Asian individuals [20]. Serum albumin, which is reported to be elevated by an increased intake of food, protein, and branched-chain amino acids, also rises with an increase in muscle content. Furthermore, GNRI, a relevant marker for nutrition and mortality, is defined via serum albumin in addition to body weight, which is mainly determined by muscle content. We examined the importance of the creatinine index, another nutritional marker in HD patients without residual renal function, as a predictor of mortality risk [21]. The creatinine index is calculated using the following formula: Cr index = 16.21 ( +1.12 if male) − 0.06 × [age (years] − 0.08 × (single pool Kt/V) + 0.009 × [serum creatinine (μmol/L]. Thus, the creatinine index is a nutritional marker that is mainly determined in HD patients by muscle content, given the lack of apparent residual renal function. We found that lower GNRI and Cr index values were both independently and equally associated with an increased risk of all-cause mortality in a multivariable-adjusted model [21]. Taken together, these findings demonstrate that the mechanism by which malnutrition increases mortality risk in Japanese HD patients can be mostly explained by reduced muscle mass.
5. Preferential Occurrence of Sarcopenia, and the Significance of Muscle Strength Rather Than Muscle Mass in HD Patients
Based on our report [22], with the increasing age of Japanese HD patients, the prevalence of sarcopenia among them was found to have increased to as much as 40% (37% in males and 45% in females). Although the definition of sarcopenia is based on muscle mass measurements by the Asian Working Group for Sarcopenia [20], it remains to be determined whether muscle mass or muscle strength is more important in determining the clinical outcome of sarcopenia in humans. Our study found that serum creatinine has a significantly positive correlation with not only muscle mass, determined via dual-energy X-ray absorptiometry (DXA), but also muscle strength measured by handgrip strength [23]. Therefore, we examined whether muscle mass or muscle strength might be a more important determinant of serum creatinine level in HD patients. Multivariate analysis demonstrated that poor arm muscle quality, calculated using the handgrip strength/DXA-determined arm lean mass ratio, rather than reduced DXA-determined arm lean mass, is responsible for the reduction in serum creatinine in HD patients [24]. This indicates that muscle strength is a more important factor than muscle mass as a determinant for serum creatinine level in HD patients. To confirm the harmful effects of reduced muscle strength on mortality in HD patients in Japan, we also examined the effects of impaired muscle quality, assessed by the reduced muscle strength/muscle mass ratio [23].
6. DM and Sarcopenia in HD Patients
In addition to aging and malnutrition, sarcopenia is known to preferentially occur in HD patients with osteoporosis and DM [25]. Additionally, the rates for the co-existence of sarcopenia, osteoporosis, and DM are known to be higher in HD patients and increase with aging. Although each disease is known to independently affect physical activity and mortality in HD patients, it is possible that DM and osteoporosis, both independently and together with sarcopenia, might reduce longevity and survival rates in these patients. Furthermore, the interaction between these three diseases is important to mention.
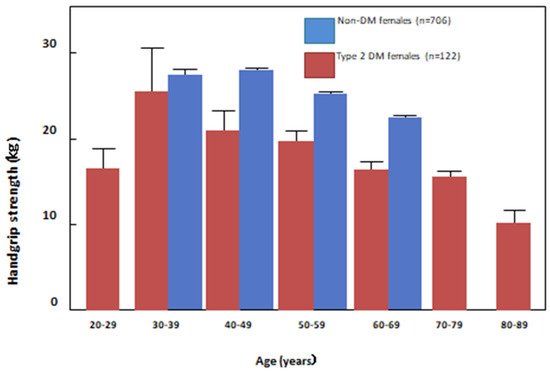
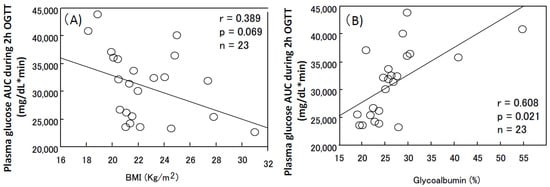
7. Osteoporosis and Sarcopenia in HD Patients
We recently reported that pre-dialysis CKD patients with a fracture exhibited a greater creatinine-based eGFR/cystatin C-based eGFR ratio than those without a fracture [29]. Creatinine-based eGFR is known to overestimate true GFR in aged CKD patients, apparently because of the lower levels of serum creatinine resulting from reduced muscle mass, as observed in HD patients, and it has been shown that cystatin C-based eGFR reflects true GFR more effectively than creatinine-based eGFR in aged CKD patients [30]. Additionally, our results indicate that the fracture rate in pre-dialysis CKD patients is greater in those with than without sarcopenia [29]. Due to the high prevalence of sarcopenia in HD patients in Japan [22], it is reasonable to consider the importance of sarcopenia in the development of osteoporosis and osteoporosis-based fragility fractures. Fall trauma and fracture are amongst the main causes of changes in the conditions of elderly HD patients, from independent living to the requirement of physical support or nursing care.
8. Importance of Sarcopenia as a Treatment Target to Elongate Longevity of HD Patients in the Era of 100-Year Life
References
- Masakane, I.; Hasegawa, T.; Ogata, S.; Kimata, N.; Nakai, S.; Hanafusa, N.; Hamano, T.; Wakai, K.; Wada, A.; Nitta, K. Annual peritoneal dialysis report 2014, the peritoneal dialysis registry. Ren. Replace. Ther. 2017, 21, 119.
- Aikawa, A. Current status and future aspects of kidney transplantation in Japan. Ren. Replace. Ther. 2018, 36, 50.
- Nitta, K.; Goto, S.; Masakane, I.; Hanafusa, N.; Taniguchi, M.; Hasegawa, T.; Nakai, S.; Wada, A.; Hamano, T.; Hoshino, J.; et al. Annual dialysis data report for 2018, JSDT Renal Data Registry: Survey methods, facility data, incidence, prevalence, and mortality. Ren. Replace. Ther. 2020, 6, 41.
- Uy, M.C.; Hospital, C.G.; Lim-Uy, R.; Chua, E. Association of Dialysis Malnutrition Score with Hypoglycemia and Quality of Life Among Patients with Diabetes on Maintenance Hemodialysis. J. ASEAN Fed. Endocr. Soc. 2018, 33, 137–145.
- Dai, L.; Mukai, H.; Lindholm, B.; Heimbürger, O.; Barany, P.; Stenvinkel, P.; Qureshi, A.R. Clinical global assessment of nutritional status as predictor of mortality in chronic kidney disease patients. PLoS ONE 2017, 12, e0186659.
- Carrero, J.J.; Aguilera, A.; Stenvinkel, P.; Gil, F.; Selgas, R.; Lindholm, B. Appetite Disorders in Uremia. J. Ren. Nutr. 2008, 18, 107–113.
- Sahathevan, S.; Khor, B.H.; Ng, H.M.; Gafor, A.H.A.; Mat Daud, Z.A.; Mafra, D.; Karupaiah, T. Understanding Development of Mal-nutrition in Hemodialysis Patients: A Narrative Review. Nutrients 2020, 12, 3147.
- Darmon, P.; Kaiser, M.J.; Bauer, J.M.; Sieber, C.C.; Pichard, C. Restrictive diets in the elderly: Never say never again? Clin. Nutr. 2010, 29, 170–174.
- Eustace, J.A.; Coresh, J.; Kutchey, C.; Te, P.L.; Gimenez, L.F.; Scheel, P.J.; Walser, M. Randomized double-blind trial of oral essential amino acids for dialysis-associated hypoalbuminemia. Kidney Int. 2000, 57, 2527–2538.
- Kalantar-Zadeh, K.; Kopple, J.D.; Block, G.; Humphreys, M.H. A malnutrition-inflammation score is correlated with morbidity and mortality in maintenance hemodialysis patients. Am. J. Kidney Dis. 2001, 38, 1251–1263.
- Carrero, J.J.; Stenvinkel, P.; Cuppari, L.; Ikizler, T.A.; Kalantar-Zadeh, K.; Kaysen, G.; Mitch, W.E.; Price, S.R.; Wanner, C.; Wang, A.Y.; et al. Etiology of the Protein-Energy Wasting Syndrome in Chronic Kidney Disease: A Consensus Statement from the International Society of Renal Nutrition and Metabolism (ISRNM). J. Ren. Nutr. 2013, 23, 77–90.
- Van de Luijtgaarden, M.W.M.; Noordzij, M.; Wanner, C.; Jager, K.J. Renal replacement therapy in Europe-a summary of the 2009 ERA-EDTA Registry Annual Report. Clin. Kidney J. 2012, 5, 109–119.
- Therrien, M.; Byham-Gray, L.; Beto, J. A Review of Dietary Intake Studies in Maintenance Dialysis Patients. J. Ren. Nutr. 2015, 25, 329–338.
- Fujino, Y.; Ishimura, E.; Okuno, S.; Tsuboniwa, N.; Maekawa, K.; Izumotani, T.; Yamakawa, T.; Inaba, M.; Nishizawa, Y. Annual fat mass change is a significant predictor of mortality in female hemodialysis patients. Biomed. Pharmacother. 2006, 60, 253–257.
- Yamada, K.; Furuya, R.; Takita, T.; Maruyama, Y.; Yamaguchi, Y.; Ohkawa, S.; Kumagai, H. Simplifed nutritional screening tools for patients on maintenance hemodialysis. Am. J. Clin. Nutr. 2008, 87, 106–113.
- Kwon, Y.E.; Yoon, C.Y.; Han, I.M.; Han, S.G.; Park, K.S.; Lee, M.J.; Park, J.T.; Han, S.H.; Yoo, T.H.; Kim, Y.L.; et al. Change of Nutritional Status Assessed Using Subjective Global Assessment Is Associated with All-Cause Mortality in Incident Dialysis Patients. Medicine 2016, 95, e2714.
- Bradbury, B.D.; Fissell, R.B.; Albert, J.M.; Anthony, M.S.; Critchlow, C.W.; Pisoni, R.L.; Port, F.K.; Gillespie, B.W. Predictors of Early Mortality among Incident US Hemodialysis Patients in the Dialysis Outcomes and Practice Patterns Study (DOPPS). Clin. J. Am. Soc. Nephrol. 2007, 2, 89–99.
- Murray, D.P.; Young, L.; Waller, J.; Wright, S.; Colombo, R.; Baer, S.; Spearman, V.; Garcia-Torres, R.; Williams, K.; Kheda, M.; et al. Is Dietary Protein Intake Predictive of One-Year Mortality in Dialysis Patients? Am. J. Med. Sci. 2018, 356, 234–243.
- Rosenberg, I.H. Sarcopenia: Origins and Clinical Relevance. J. Nutr. 1997, 127, 990S–991S.
- Chen, L.-K.; Liu, L.-K.; Woo, J.; Assantachai, P.; Auyeung, T.-W.; Bahyah, K.S.; Chou, M.-Y.; Chen, L.-Y.; Hsu, P.-S.; Krairit, O.; et al. Sarcopenia in Asia: Consensus Report of the Asian Working Group for Sarcopenia. J. Am. Med. Dir. Assoc. 2014, 15, 95–101.
- Yamada, S.; Yamamoto, S.; Fukuma, S.; Nakano, T.; Tsuruya, K.; Inaba, M. Geriatric Nutritional Risk Index (GNRI) and Creatinine Index Equally Predict the Risk of Mortality in Hemodialysis Patients: J-DOPPS. Sci. Rep. 2020, 10, 5756.
- Mori, K.; Nishide, K.; Okuno, S.; Shoji, T.; Emoto, M.; Tsuda, A.; Nakatani, S.; Imanishi, Y.; Ishimura, E.; Yamakawa, T.; et al. Impact of diabetes on sarcopenia and mortality in patients undergoing hemodialysis. BMC Nephrol. 2019, 20, 105.
- Yoda, M.; Inaba, M.; Okuno, S.; Yoda, K.; Yamada, S.; Imanishi, Y.; Mori, K.; Shoji, T.; Ishimura, E.; Yamakawa, T.; et al. Poor muscle quality as a predictor of high mortality independent of diabetes in hemodialysis patients. Biomed. Pharmacother. 2012, 66, 266–270.
- Inaba, M.; Kurajoh, M.; Okuno, S.; Imanishi, Y.; Yamada, S.; Mori, K.; Ishimura, E.; Yamakawa, T.; Nishizawa, Y. Poor muscle quality rather than reduced lean body mass is responsible for the lower serum creatinine level in hemodialysis patients with diabetes mellitus. Clin. Nephrol. 2010, 74, 266–272.
- Nakamura, M.; Inaba, M.; Yamada, S.; Ozaki, E.; Maruo, S.; Okuno, S.; Imanishi, Y.; Kuriyama, N.; Watanabe, Y.; Emoto, M.; et al. Association of Decreased Handgrip Strength with Reduced Cortical Thickness in Japanese Female Patients with Type 2 Diabetes Mellitus. Sci. Rep. 2018, 8, 10767.
- Rolland, Y.M.; Perry, H.M., 3rd; Patrick, P.; Banks, W.A.; Morley, J.E. Loss of appendicular muscle mass and loss of muscle strength in young postmenopausal women. J. Gerontol. A Biol. Sci. Med. Sci. 2007, 62, 330–335.
- Saisho, Y.; Tanaka, K.; Abe, T.; Shimada, A.; Kawai, T.; Itoh, H. Glycated albumin to glycated hemoglobin ratio reflects post-prandial glucose excursion and relates to beta cell function in both type 1 and type 2 diabetes. Diabetol. Int. 2011, 2, 146–153.
- Abbatecola, A.M.; for the Health ABC Study; Chiodini, P.; Gallo, C.; Lakatta, E.; Sutton-Tyrrell, K.; Tylavsky, F.A.; Goodpaster, B.; De Rekeneire, N.; Schwartz, A.V.; et al. Pulse wave velocity is associated with muscle mass decline: Health ABC study. AGE 2011, 34, 469–478.
- Kurajoh, M.; Inaba, M.; Nagata, Y.; Yamada, S.; Imanishi, Y.; Emoto, M. Association of cystatin C- and creatinine-based eGFR with osteoporotic fracture in Japanese postmenopausal women with osteoporosis: Sarcopenia as risk for fracture. J. Bone Miner. Metab. 2019, 37, 282–291.
- Dharnidharka, V.R.; Kwon, C.; Stevens, G. Serum cystatin C is superior to serum creatinine as a marker of kidney function: A meta-analysis. Am. J. Kidney Dis. 2002, 40, 221–226.
- Pourhassan, M.; Babel, N.; Sieske, L.; Westhoff, T.H.; Wirth, R. Inflammatory cytokines and appetite in old-er hospitalized patients. Appetite 2021, 166, 105470.





