
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Ken Iwatsuki | + 1955 word(s) | 1955 | 2021-08-12 07:52:54 | | | |
| 2 | Conner Chen | Meta information modification | 1955 | 2021-09-22 04:16:17 | | |
Video Upload Options
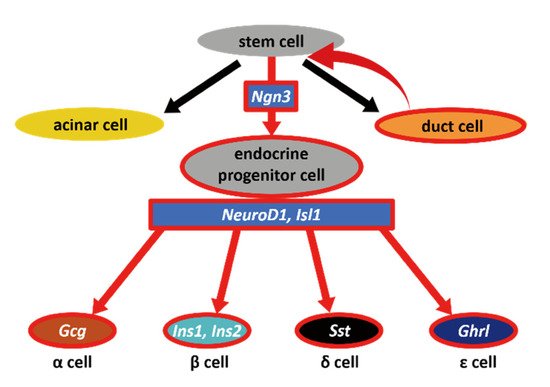
The production of pancreatic β cells is the most challenging step for curing diabetes using next-generation treatments. Adult pancreatic endocrine cells are thought to be maintained by the self-duplication of differentiated cells, and pancreatic endocrine neogenesis can only be observed when the tissue is severely damaged. Experimentally, this can be performed using a method named partial duct ligation (PDL). We performed PDL on neurogenin3 (Ngn3)-GFP transgenic mice to determine the origin of endocrine precursor cells and evaluate their potential to differentiate into multiple cell types. Ngn3-expressing cells, which were marked with GFP, appeared after PDL operation. Because some GFP-positive cells were found proximally to the duct, we hypothesized that Ngn3-positive cells arise from the pancreatic duct. Therefore, we next developed an in vitro pancreatic duct culture system using Ngn3-GFP mice and examined whether Ngn3-positive cells emerge from this duct. Our results demonstrate that in adult mice, Ngn3-positive endocrine precursor cells arise from the pancreatic ducts both in vivo and in vitro experiments indicating that the pancreatic duct could be a potential donor for therapeutic use.
1. Introduction
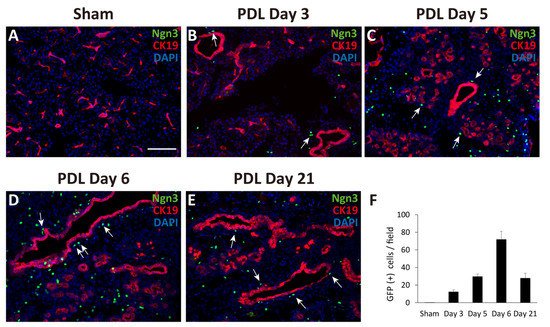
2. Observation of Ngn3-Positive Cells in the Ligated Part of the Pancreas at 3, 5, 6, and 21 Days after Surgery
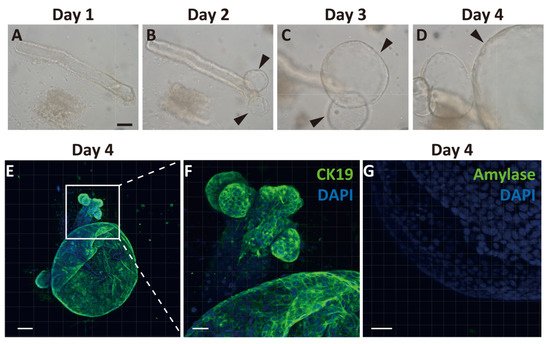
3. Generation of Pancreatic Ductal Organoids from Ngn3-GFP Mice
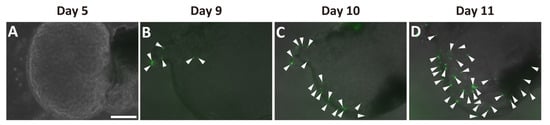
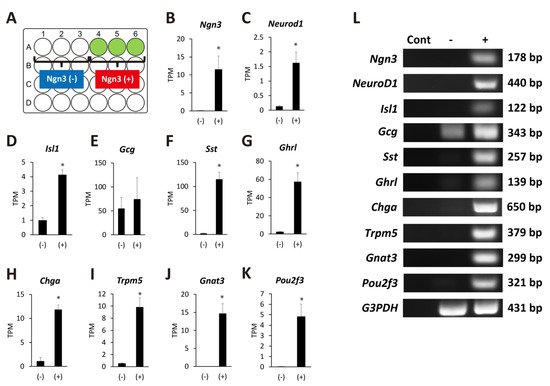
4. Ngn3-Positive Cells Can Differentiate into Endocrine Cells
5. Discussion
References
- Zhou, Q.; Melton, D.A. Pancreas regeneration. Nature 2018, 557, 351–358.
- Loomans, C.J.M.; Williams Giuliani, N.; Balak, J.; Ringnalda, F.; van Gurp, L.; Huch, M.; Boj, S.F.; Sato, T.; Kester, L.; de Sousa Lopes, S.M.C.; et al. Expansion of adult human pancreatic tissue yields organoids harboring progenitor cells with endocrine differentiation potential. Stem Cell Rep. 2018, 10, 712–724.
- Kopp, J.L.; Grompe, M.; Sander, M. Stem cells versus plasticity in liver and pancreas regeneration. Nat. Cell Biol. 2016, 18, 238–245.
- Dor, Y.; Brown, J.; Martinez, O.I.; Melton, D.A. Adult pancreatic β-cells are formed by self-duplication rather than stem-cell differentiation. Nature 2004, 429, 41–46.
- Matveyenko, A.V.; Veldhuis, J.D.; Butler, P.C. Adaptations in pulsatile insulin secretion, hepatic insulin clearance, and β-cell mass to age-related insulin resistance in rats. Am. J. Physiol. Endocrinol. Metab. 2008, 295, E832–E841.
- Sorenson, R.L.; Brelje, T.C. Adaptation of islets of Langerhans to pregnancy: β-cell growth, enhanced insulin secretion and the role of lactogenic hormones. Horm. Metab. Res. 1997, 29, 301–307.
- Kloppel, G.; Lohr, M.; Habich, K.; Oberholzer, M.; Heitz, P.U. Islet pathology and the pathogenesis of type 1 and type 2 diabetes mellitus revisited. Surv. Synth. Pathol. Res. 1985, 4, 110–125.
- Bruning, J.C.; Winnay, J.; Bonner-Weir, S.; Taylor, S.I.; Accili, D.; Kahn, C.R. Development of a novel polygenic model of NIDDM in mice heterozygous for IR and IRS-1 null alleles. Cell 1997, 88, 561–572.
- Teta, M.; Rankin, M.M.; Long, S.Y.; Stein, G.M.; Kushner, J.A. Growth and regeneration of adult beta cells does not involve specialized progenitors. Dev. Cell 2007, 12, 817–826.
- Lipsett, M.; Finegood, D.T. β-cell neogenesis during prolonged hyperglycemia in rats. Diabetes 2002, 51, 1834–1841.
- Xu, X.; D’Hoker, J.; Stange, G.; Bonne, S.; De Leu, N.; Xiao, X.; Van de Casteele, M.; Mellitzer, G.; Ling, Z.; Pipeleers, D.; et al. β cells can be generated from endogenous progenitors in injured adult mouse pancreas. Cell 2008, 132, 197–207.
- Nir, T.; Melton, D.A.; Dor, Y. Recovery from diabetes in mice by. β cell regeneration. J. Clin. Invest. 2007, 117, 2553–2561.
- Szkudelski, T. The mechanism of alloxan and streptozotocin action in B cells of the rat pancreas. Physiol. Res. 2001, 50, 537–546.
- Thorel, F.; Damond, N.; Chera, S.; Wiederkehr, A.; Thorens, B.; Meda, P.; Wollheim, C.B.; Herrera, P.L. Normal glucagon signaling and β-cell function after near-total α-cell ablation in adult mice. Diabetes 2011, 60, 2872–2882.
- Inada, A.; Nienaber, C.; Katsuta, H.; Fujitani, Y.; Levine, J.; Morita, R.; Sharma, A.; Bonner-Weir, S. Carbonic anhydrase II-positive pancreatic cells are progenitors for both endocrine and exocrine pancreas after birth. Proc. Natl. Acad. Sci. USA 2008, 105, 19915–19919.
- Li, W.C.; Rukstalis, J.M.; Nishimura, W.; Tchipashvili, V.; Habener, J.F.; Sharma, A.; Bonner-Weir, S. Activation of pancreatic-duct-derived progenitor cells during pancreas regeneration in adult rats. J. Cell Sci. 2010, 123, 2792–2802.
- Wang, R.N.; Kloppel, G.; Bouwens, L. Duct- to islet-cell differentiation and islet growth in the pancreas of duct-ligated adult rats. Diabetologia 1995, 38, 1405–1411.
- Criscimanna, A.; Speicher, J.A.; Houshmand, G.; Shiota, C.; Prasadan, K.; Ji, B.; Logsdon, C.D.; Gittes, G.K.; Esni, F. Duct cells contribute to regeneration of endocrine and acinar cells following pancreatic damage in adult mice. Gastroenterology 2011, 141, 1451–1462.e6.
- Huch, M.; Bonfanti, P.; Boj, S.F.; Sato, T.; Loomans, C.J.; van de Wetering, M.; Sojoodi, M.; Li, V.S.; Schuijers, J.; Gracanin, A.; et al. Unlimited in vitro expansion of adult bi-potent pancreas progenitors through the Lgr5/R-spondin axis. EMBO J. 2013, 32, 2708–2721.
- Pan, F.C.; Bankaitis, E.D.; Boyer, D.; Xu, X.; Van de Casteele, M.; Magnuson, M.A.; Heimberg, H.; Wright, C.V. Spatiotemporal patterns of multipotentiality in Ptf1a-expressing cells during pancreas organogenesis and injury-induced facultative restoration. Development 2013, 140, 751–764.
- Van de Casteele, M.; Leuckx, G.; Baeyens, L.; Cai, Y.; Yuchi, Y.; Coppens, V.; De Groef, S.; Eriksson, M.; Svensson, C.; Ahlgren, U.; et al. Neurogenin 3+ cells contribute to β-cell neogenesis and proliferation in injured adult mouse pancreas. Cell Death Dis. 2013, 4, e523.
- Chintinne, M.; Stange, G.; Denys, B.; Ling, Z.; ’t Veld, P.; Pipeleers, D. Beta cell count instead of beta cell mass to assess and localize growth in beta cell population following pancreatic duct ligation in mice. PLoS ONE 2012, 7, e43959.
- Kopp, J.L.; Dubois, C.L.; Schaffer, A.E.; Hao, E.; Shih, H.P.; Seymour, P.A.; Ma, J.; Sander, M. Sox9+ ductal cells are multipotent progenitors throughout development but do not produce new endocrine cells in the normal or injured adult pancreas. Development 2011, 138, 653–665.
- Rankin, M.M.; Wilbur, C.J.; Rak, K.; Shields, E.J.; Granger, A.; Kushner, J.A. β-Cells are not generated in pancreatic duct ligation-induced injury in adult mice. Diabetes 2013, 62, 1634–1645.
- Nakajima, C.; Kamimoto, K.; Miyajima, K.; Matsumoto, M.; Okazaki, Y.; Kobayashi-Hattori, K.; Shimizu, M.; Yamane, T.; Oishi, Y.; Iwatsuki, K. A method for identifying mouse pancreatic ducts. Tissue Eng. C Methods 2018, 24, 480–485.
- Jiang, F.X.; Li, K.; Archer, M.; Mehta, M.; Jamieson, E.; Charles, A.; Dickinson, J.E.; Matsumoto, M.; Morahan, G. Differentiation of Islet Progenitors Regulated by Nicotinamide into Transcriptome-Verified beta Cells That Ameliorate Diabetes. Stem Cells 2017, 35, 1341–1354.
- Gradwohl, G.; Dierich, A.; LeMeur, M.; Guillemot, F. Neurogenin3 is required for the development of the four endocrine cell lineages of the pancreas. Proc. Natl. Acad. Sci. USA 2000, 97, 1607–1611.
- Van de Casteele, M.; Leuckx, G.; Cai, Y.; Yuchi, Y.; Coppens, V.; De Groef, S.; Van Gassen, N.; Baeyens, L.; Heremans, Y.; Wright, C.V.; et al. Partial duct ligation: β-Cell proliferation and beyond. Diabetes 2014, 63, 2567–2577.
- Shih, H.P.; Kopp, J.L.; Sandhu, M.; Dubois, C.L.; Seymour, P.A.; Grapin-Botton, A.; Sander, M. A notch-dependent molecular circuitry initiates pancreatic endocrine and ductal cell differentiation. Development 2012, 139, 2488–2499.
- Minami, K.; Okuno, M.; Miyawaki, K.; Okumachi, A.; Ishizaki, K.; Oyama, K.; Kawaguchi, M.; Ishizuka, N.; Iwanaga, T.; Seino, S. Lineage tracing and characterization of insulin-secreting cells generated from adult pancreatic acinar cells. Proc. Natl. Acad. Sci. USA 2005, 102, 15116–15121.
- Miyamoto, Y.; Maitra, A.; Ghosh, B.; Zechner, U.; Argani, P.; Iacobuzio-Donahue, C.A.; Sriuranpong, V.; Iso, T.; Meszoely, I.M.; Wolfe, M.S.; et al. Notch mediates TGFα-induced changes in epithelial differentiation during pancreatic tumorigenesis. Cancer Cell 2003, 3, 565–576.
- Miyatsuka, T.; Kaneto, H.; Shiraiwa, T.; Matsuoka, T.A.; Yamamoto, K.; Kato, K.; Nakamura, Y.; Akira, S.; Takeda, K.; Kajimoto, Y.; et al. Persistent expression of PDX-1 in the pancreas causes acinar-to-ductal metaplasia through Stat3 activation. Genes Dev. 2006, 20, 1435–1440.
- Rooman, I.; Heremans, Y.; Heimberg, H.; Bouwens, L. Modulation of rat pancreatic acinoductal transdifferentiation and expression of PDX-1 in vitro. Diabetologia 2000, 43, 907–914.





