
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Marc St-Arnaud | + 2097 word(s) | 2097 | 2021-08-17 10:35:22 | | | |
| 2 | Dean Liu | Meta information modification | 2097 | 2021-08-23 02:45:12 | | | | |
| 3 | Mohamed Hijri | + 1 word(s) | 2098 | 2021-08-23 12:37:59 | | |
Video Upload Options
Rhizoremediation of PHCs is facilitated through a process known as the ‘rhizosphere effect’, in which plants exude a myriad of organic compounds into their root-surrounding zone (the rhizosphere), resulting in an increase in abundance and activity of certain rhizospheric microbes, which in turn can degrade or metabolize hydrocarbon contaminants.
1. Introduction
Industrial activities, including mining and extraction of oil and gas, as well as chemical inputs into agricultural production systems, have led to different degrees of environmental contamination worldwide. Petroleum hydrocarbons (PHCs) are among the major pollutants that can pose a serious environmental threat. PHC products have adversely affected various ecosystems, causing disturbing damage to natural habitats with serious economic consequences [1].
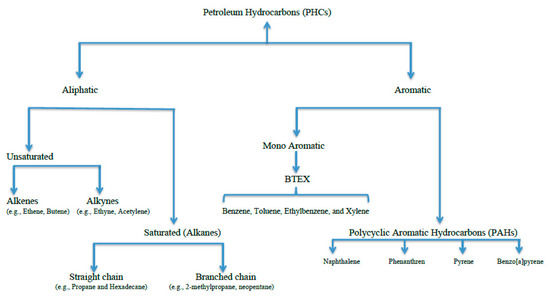
Petroleum hydrocarbons are heterogeneous organic mixtures composed of carbon and hydrogen atoms arranged in varying structural configurations and have different physical and chemical properties [2]. These compounds consist mainly of hydrocarbons and fewer numbers of other non-hydrocarbon constituents, such as nitrogen, oxygen, and sulfur [3][4]. They are broadly classified into two major fractions: aliphatic hydrocarbons and aromatic hydrocarbons ( Figure 1 ). Prior to processing, PHCs are composed, on average, of ~57% aliphatic hydrocarbons, ~29% aromatic hydrocarbons, and ~14% asphaltenes and other polar compounds containing nitrogen, oxygen, and sulfur [5]. Aliphatic hydrocarbons include both linear or branched-chain hydrocarbons, which may be unsaturated (alkenes and alkynes) or saturated (alkanes) [6]. Aromatic hydrocarbons include monocyclic (i.e., benzene, toluene, phenol, etc.) and polycyclic aromatic hydrocarbons (PAHs) ( Figure 1 ). PHCs are the most common pollutants in soil and ground water worldwide. The ever- increasing dependency of modern society on fuel for energy generation in many vital sectors, such as electricity , heat , industry, and transportation has resulted in the extensive exploitation of PHCs [2]. Although environmental transition actions have been taken in many countries, dependency on petroleum will last for some decades, contributing to organic pollution risks.
Soil contamination with PHCs is an international issue, and the magnitude of soil pollution is hard to quantify. For example, in Australia, around 80,000 sites are estimated to be contaminated by PHCs [7], whereas in Canada around 22,000 federal-owned sites are identified as being contaminated by PHCs [8]. In Europe, PHC contamination was observed in at least 342,000 sites [9]. These organic contaminants also pose serious health risks to humans and other organisms in addition to their adverse impact on the soil microflora, leading to environmental quality degradation. For instance, some aromatic substances, such as BTEX and PAHs, are notorious mutagens and carcinogens that can enter our food chain together with lipophilic compounds [10], and they have been linked with probable causes of bladder, kidney, liver, lung, and skin cancers. This explains the growing concern with these contaminants and the urgent need to use all possible means to protect the environment and to find the appropriate techniques to remediate polluted soils.
Various chemical, physical, and thermal conventional techniques have been used to remediate soils contaminated with PHCs. These conventional methods, which can contain, destroy, or separate the pollutants, include a wide range of both in situ and ex situ cleanup technologies, such as asphalt batching, biopiles, chemical oxidation, excavation, hydrolysis, incineration, photolysis, pump and treat, multi-phased slurry reactors, soil vapor extraction, soil washing, and thermal desorption. However, these methods have particular limitations. First, their cost is often prohibitive; for example, it can cost between USD 480 and 813 per m 3 for extraction [11]. Second, chemical procedures only work for specific organic compounds, and they most often destroy soil microbial communities. Third, these methods do not often result in a complete degradation of the pollutants [2][12]. Finally, PHC-contaminated soil contains numerous classes and types of toxic organic compounds, which make the choice of the proper method a challenging task. Hence, phytoremediation is a more recent and promising green-biotechnology that is perceived as an environmentally friendly, more cost-effective, and less destructive approach to cleanup contaminants in the environment.
2. Phytoremediation
Phytoremediation is a remediation technique that relies on the ability of plants and their associated microbiota to accumulate, degrade, sequester, or stabilize harmful environmental contaminants [13][14]. Over the past two decades, the deployment of plants (and their associated microbiota) to remediate a wide spectrum of inorganic and organic pollutants in soil and water environments has been carried out. This technique has been applied to remediate various types of pollutants such as chlorinated solvents [15], explosives [16], heavy metals [17], landfill leachates [18], pesticides [19] , PHC [20], radionuclides [21], and salts [22]. Although phytoremediation is still very much in its infancy, its application has been adopted by a growing number of companies. For example, the phytoremediation market has grown continuously at a rapid rate, with an estimated value of USD 32.2 billion in 2016 and is expected to reach USD 65.7 billion by 2025 [23].
Phytoremediation is an innovative technique that has gained broad public acceptance, not only because it is an environmentally friendly approach but also as it requires less maintenance efforts, minimize site disturbance, and cost-effective process, which is powered by solar energy. However, phytoremediation still remains a marginal option for in situ soil remediation [24]. As any other technique, phytoremediation has some limitations that affect its efficiency, performance, and time consuming. For example, phytoremediation efficiency varies with environmental conditions, such as soil physiochemical properties, contaminant level, and seasonal temperature fluctuations [13][25][26].
Phytoremediation efficiency is dependent on many factors, including plant selection [27]; environmental parameters such as nutrient status, contaminant concentration, and bioavailability; soil pH, etc. [25], in addition to the composition and activity of plant-associated microbiomes. Plants and their associated microbiomes facilitate pollutant uptake from the environment via different processes, including degradation, extraction, stabilization, transformation, and volatilization [13][14]. The type of plant and pollutants plus the environmental conditions are key factors for determining the way in which phytoremediation techniques can be applied. Generally, phytoremediation technologies are divided into five different categories ( Table 1 ). The phytoremediation method suitable for petroleum hydrocarbon-contaminated soil is called rhizoremediation [28], which is defined as the breakdown of organic pollutants by using plants and their root-associated microbiota.
| Category | Mechanism | Target Pollutants | Region of Activity | Reference |
|---|---|---|---|---|
| Phytoextraction | Uptake and concentrate contaminants | Metals (e.g., Cd,Ni), radionuclides (e.g., Pu) | Shoot tissue | [21][29] |
| Phytostabilization | Immobilization and sequestration of contaminants | Primarily metals (e.g., Cu, Zn, Pb) | Root tissue | [30] |
| Phytotransformation | Enzymatic actions | Chlorinated solvents, ammunition waste, herbicides, monoaromatic hydrocarbons | Plant tissue | [15][30] |
| Phytovolatilization | Uptake and evapotranspiration | Volatile organics (e.g., TCE, toluene, MTBE), | Shoot tissue | [14] |
| Rhizoremediation | Breakdown of organic pollutants by using plants and root-associated microbiomes | PHC (e.g., diesel), pesticides (e.g., dimethomorph) | Root | [19][28] |
Rhizoremediation of PHCs is facilitated through a process known as the ‘rhizosphere effect’ [31], in which plants exude a variety of organic compounds into their root-surrounding zone (the rhizosphere), resulting in an increase in abundance and activity of certain rhizospheric microbes, which in turn can degrade or metabolize hydrocarbon contaminants [32]. Understanding the plant–microbiome partnerships, and the underlying processes that govern and control PHC degradation, is a priority challenge in rhizoremediation research nowadays [2][33][34].
3. The Rhizosphere Microbiota and root exudates
In addition to shaping the microbial communities in the rhizosphere, root exudates have other functions that benefit the plant itself. Through root exudation, plants can change the soil physicochemical properties, contributing to nutrient assimilation, reducing the growth of competitor plant species, increasing the abundance of certain beneficial microbes, and regulating the microbiome composition in the rhizosphere [35][36].
Since the beginning of phytoremediation research, many plant species have been tested for their potential to enhance rhizoremediation of PHCs [37]. Plants enhance the degradation of PHCs principally by the unique properties of the plant itself and by providing optimal conditions for microbial proliferation in the rhizosphere [38]. In general, selection of plants suitable for rhizoremediation of PHCs should be based on the following criteria: tolerance to a broad range of PHCs, speed of growth, root morphology, ability to grow in many soil types, and the root exudate profile [38][39][40]. Additionally, plants should not be selected based solely on the contaminant uptake efficiency; their ability to stimulate microbial activity and abundance also should be considered [41][42].
Sequestration and transportation of contaminants inside plant tissues enable plants to remediate PHC-polluted soil [43][44]. Plants can also degrade or transform organic pollutants into less toxic forms via their enzymatic machinery, or synthesizing a variety of defensive proteins and metabolites [13][45]. Therefore, plants can adapt and confront many unfavorable stressful conditions, such as PHC contamination. However, plant growth has been retarded under highly stressed conditions, e.g., PHC pollutants are expected to be lower than those under optimal conditions [46]. Therefore, plant growth may be positively enhanced by the presence of plant growth-promoting rhizobacteria (PGPR) that are able to alleviate stresses in plants via many mechanisms, such as reducing soil nutrient deficiencies (fixing nitrogen, solubilizing phosphorus, and enhancing iron uptake), synthesizing plant hormones, suppressing ethylene production via 1-aminocyclopropane-1-carboxylate (ACC) deaminase activity [46][47], and degrading a broad range of PHCs [2].
Ethylene production by plants at low concentrations can be beneficial. However, when produced at high concentrations, it can stunt plant growth and development by inhibiting root growth [48]. In response to various biotic and abiotic stressor conditions, plants synthesize different enzymes, metabolites, and stress proteins to alleviate the adverse effects of stress [49]; of particular interest is ethylene. Once plants encounter stress, such as flooding, drought, or presence of toxic compounds, plant growth is inhibited because the ethylene precursor, 1-aminocyclopropane-1-carboxylate, is induced [50]. However, certain PGPR can hinder ethylene biosynthesis via production of 1-aminocyclopropane-1-carboxylate deaminase (ACCD) that cleaves the ethylene precursor ACC into alpha-ketobutyrate and ammonia [51], thus balancing ethylene levels and reducing its adverse impact on plant growth [51].
4. Enhancing the Understanding of Mechanisms through Which Host Plants Assemble a Beneficial Microbiota, and How It Functions, under Pollutant Stress
Studies of the rhizosphere microbiota in natural and agricultural settings have generated most of our knowledge about host plant selection processes and plant–microbiome interactions taking place in the rhizosphere and how plants recruit different microbiota from surrounding environments [34][52]. For example, previous studies using 16S rRNA amplicon sequencing revealed that microbial communities in the rhizosphere and adjunct bulk soils are different; the recruitment of rhizosphere microbiota by plants is strongly dependent on the structure and composition of the bulk soil microbiome [53], and different plant genotypes were found to select for different rhizosphere microbiomes [54].
Over the past few years, several experiments have been conducted to optimize phytoremediation systems and improve their efficacy using high-throughput sequencing approaches. For example, Bell et al. [55] used high-throughput 454-pyrosequencing of bacterial 16S rRNA genes and the fungal internal transcribed spacer (ITS) region to compare the community structure and composition of the rhizosphere microbiome of native and non-native Salix cultivars across uncontaminated and PHC-contaminated soil. Their results indicated higher fungal sensitivity to PHC contamination than that found for bacterial communities. Additionally, certain fungal class ( Pezizomycetes ) reacted differently following plant introduction to soils [55], implying the importance of plant species selection in phytoremediation with regard to their impact on plant-associated microbiomes [34]. Similarly, Hassan et al. [56] used 454-pyrosequencing of the AMF 18S rDNA gene to examine how rhizospheric AMF communities are shaped within the rhizosphere of 11 Salix cultivars introduced across non-contaminated and PHC-contaminated soil. While PHC contamination levels had a strong impact on AMF community structure, Salix planting increased the abundance of several AMF families [56], inferring that AMF, possibly due to opportunistic associations with the plant, are involved in plant adaptation to PHC contamination [34].
Tardif et al. [57] amplified the bacterial 16S rRNA gene and fungal ITS regions using Ion Torrent sequencing in order to characterize the variations between plant compartments (bulk soil, rhizosphere soil, roots, and stems) in the microbiome of two Salix cultivars growing under three PHC contamination levels at a former petrochemical site. PHC contamination was found to be the main factor, shaping not only the rhizosphere but also the root and stem microbiome structure [57]. Additionally, the presence of the plant offered a protective buffer zone against PHC pollution in the rhizosphere and other plant tissues, subsequently minimizing the severe effects of PHC contamination on the microbiome composition, as compared with adjunct bulk soil [57]. Finally, increasing PHC contamination caused a shift in the microbial community composition, favoring beneficial microbiome communities such as putative PHC-degraders and PGPR [57].
In a recent study, Mitter et al. [58] used high-throughput Illumina MiSeq amplicon sequencing of the 16S rRNA gene to characterize the bacterial root microbiome associated with annual barley and sweet clover growing in an oil sands reclamation site. Results confirmed that, consistent with previous reports, the rhizosphere compartment produced the strongest differentiation of the root microbiome community structure [53][58][59]; for example, Proteobacteria was the predominant phyla in the endosphere microbiome, whereas phyla such as Acidobacteria and Gemmatimonadetes were restricted only to the rhizosphere microbiome [58]. Additionally, host plants play a major role in shaping the root microbiome community structure [58], implying plants have the ability to select for specific soil microbiota [58].
References
- Brzeszcz, J.; Kaszycki, P. Aerobic bacteria degrading both n-alkanes and aromatic hydrocarbons: An undervalued strategy for metabolic diversity and flexibility. Biodegradation 2018, 29, 359–407.
- Gkorezis, P.; Daghio, M.; Franzetti, A.; Van Hamme, J.; Sillen, W.; Vangronsveld, J. The Interaction between Plants and Bacteria in the Remediation of Petroleum Hydrocarbons: An Environmental Perspective. Front. Microbiol. 2016, 7, 1836.
- Wang, Z.; Fingas, M.; Yang, C.; Christensen, J.H. Crude Oil and Refined Product Fingerprinting: Principles. In Environmental Forensics: Contaminant Specific Guide; Morrison, R.D., Murphy, B.L., Eds.; Academic Press: New York, NY, USA, 2006; pp. 339–407.
- Farrell-Jones, J. Petroleum Hydrocarbons and Polyaromatic Hydrocarbons; Blackwell Publishing CRC Press: New York, NY, USA, 2003.
- Tissot, B.P.; Welte, D.H. Petroleum Formation and Occurrence; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2013.
- Pandey, P.; Pathak, H.; Dave, S. Microbial Ecology of Hydrocarbon Degradation in the Soil: A Review. Res. J. Environ. Toxicol. 2016, 10, 1–15.
- Hoang, S.A.; Lamb, D.; Seshadri, B.; Sarkar, B.; Choppala, G.; Kirkham, M.; Bolan, N.S. Rhizoremediation as a green technology for the remediation of petroleum hydrocarbon-contaminated soils. J. Hazard. Mater. 2020, 401, 123282.
- Secretariat. Federal Contaminated Sites Inventory. Available online: https://www.tbs-sct.gc.ca/fcsi-rscf/home-accueil-eng.aspx (accessed on 18 August 2020).
- Panagos, P.; Van Liedekerke, M.; Yigini, Y.; Montanarella, L. Contaminated Sites in Europe: Review of the Current Situation Based on Data Collected through a European Network. J. Environ. Public Health 2013, 2013, 1–11.
- Henner, P.; Schiavon, M.; Morel, J.-L.; Lichtfouse, E. Polycyclic aromatic hydrocarbon (PAH) occurrence and remediation methods. Analusis 1997, 25, M56–M59.
- Inoue, Y.; Katayama, A. Two-scale evaluation of remediation technologies for a contaminated site by applying economic input–output life cycle assessment: Risk–cost, risk–energy consumption and risk–CO2 emission. J. Hazard. Mater. 2011, 192, 1234–1242.
- Yerushalmi, L.; Rocheleau, S.; Cimpoia, R.; Sarrazin, M.; Sunahara, G.; Peisajovich, A.; Leclair, G.; Guiot, S.R. Enhanced biodegradation of petroleum hydrocarbons in contaminated soil. J. Soil Contam. 1998, 7, 37–51.
- Pilon-Smits, E. Phytoremediation. Annu. Rev. Plant Biol. 2005, 56, 15–39.
- Salt, D.E.; Smith, R.D.; Raskin, I. Phytoremediation. Annu. Rev. Plant Biol. 1998, 49, 643–668.
- Aken, B.V.; Doty, S.L. Transgenic plants and associated bacteria for phytoremediation of chlorinated compounds. Biotechnol. Genet. Eng. Rev. 2009, 26, 43–64.
- Panz, K.; Miksch, K. Phytoremediation of explosives (TNT, RDX, HMX) by wild-type and transgenic plants. J. Environ. Manag. 2012, 113, 85–92.
- Leguizamo, M.A.O.; Gómez, W.D.F.; Sarmiento, M.C.G. Native herbaceous plant species with potential use in phytoremediation of heavy metals, spotlight on wetlands—A review. Chemosphere 2017, 168, 1230–1247.
- Ch, J.A.J.; Romero, R.M. Evaluation of Cajanus cajan (pigeon pea) for phytoremediation of landfill leachate containing chromium and lead. Int. J. Phytoremediat. 2016, 18, 1122–1127.
- Olette, R.; Couderchet, M.; Biagianti, S.; Eullaffroy, P. Toxicity and removal of pesticides by selected aquatic plants. Chemosphere 2008, 70, 1414–1421.
- Newman, L.A.; Reynolds, C.M. Phytodegradation of organic compounds. Curr. Opin. Biotechnol. 2004, 15, 225–230.
- Sharma, S.; Singh, B.; Manchanda, V.K. Phytoremediation: Role of terrestrial plants and aquatic macrophytes in the remediation of radionuclides and heavy metal contaminated soil and water. Environ. Sci. Pollut. Res. 2014, 22, 946–962.
- Devi, S.; Nandwal, A.; Angrish, R.; Arya, S.; Kumar, N.; Sharma, S. Phytoremediation potential of some halophytic species for soil salinity. Int. J. Phytoremediat. 2015, 18, 693–696.
- Transparency Market Research. BioremediationI Technology & Services Market to Reach Valuation gf ~US$ 20 BN by 2030. Available online: https://www.transparencymarketresearch.com/pressrelease/bioremediation-technology-services-market.htm (accessed on 18 September 2020).
- Mench, M.; Lepp, N.; Bert, V.; Schwitzguébel, J.-P.; Gawroński, S.; Schröder, P.; Vangronsveld, J. Successes and limitations of phytotechnologies at field scale: Outcomes, assessment and outlook from COST Action 859. J. Soils Sediments 2010, 10, 1039–1070.
- Vangronsveld, J.; Herzig, R.; Weyens, N.; Boulet, J.; Adriaensen, K.; Ruttens, A.; Thewys, T.; Vassilev, A.; Meers, E.; Nehnevajova, E.; et al. Phytoremediation of contaminated soils and groundwater: Lessons from the field. Environ. Sci. Pollut. Res. 2009, 16, 765–794.
- Arthur, E.L.; Rice, P.J.; Rice, P.J.; Anderson, T.A.; Baladi, S.M.; Henderson, K.L.; Coats, J.R. Phytoremediation—An overview. Crit. Rev. Plant Sci. 2005, 24, 109–122.
- Wenzel, W.W. Rhizosphere processes and management in plant-assisted bioremediation (phytoremediation) of soils. Plant Soil 2008, 321, 385–408.
- Kuiper, I.; Lagendijk, E.L.; Bloemberg, G.V.; Lugtenberg, B.J.J. Rhizoremediation: A Beneficial Plant-Microbe Interaction. Mol. Plant-Microbe Interact. 2004, 17, 6–15.
- Ali, H.; Khan, E.; Sajad, M.A. Phytoremediation of heavy metals—Concepts and applications. Chemosphere 2013, 91, 869–881.
- Behera, K.K. Phytoremediation, transgenic plants and microbes. In Sustainable Agriculture Reviews; Springer: Berlin/Heidelberg, Germany, 2014; pp. 65–85.
- Anderson, T.A.; Guthrie, E.A.; Walton, B.T. Bioremediation in the rhizosphere. Environ. Sci. Technol. 1993, 27, 2630–2636.
- Martin, B.; George, S.J.; Price, C.A.; Ryan, M.; Tibbett, M. The role of root exuded low molecular weight organic anions in facilitating petroleum hydrocarbon degradation: Current knowledge and future directions. Sci. Total Environ. 2014, 472, 642–653.
- Correa-García, S.; Pande, P.; Séguin, A.; St-Arnaud, M.; Yergeau, E. Rhizoremediation of petroleum hydrocarbons: A model system for plant microbiome manipulation. Microb. Biotechnol. 2018, 11, 819–832.
- Ethijs, S.; Sillen, W.; Erineau, F.; Eweyens, N.; Vangronsveld, J. Towards an Enhanced Understanding of Plant–Microbiome Interactions to Improve Phytoremediation: Engineering the Metaorganism. Front. Microbiol. 2016, 7, 341.
- Vieira, S.; Sikorski, J.; Dietz, S.; Herz, K.; Schrumpf, M.; Bruelheide, H.; Scheel, D.; Friedrich, M.W.; Overmann, J. Drivers of the composition of active rhizosphere bacterial communities in temperate grasslands. ISME J. 2019, 14, 463–475.
- Vives-Peris, V.; de Ollas, C.; Gómez-Cadenas, A.; Pérez-Clemente, R.M. Root exudates: From plant to rhizosphere and beyond. Plant Cell Rep. 2019, 39, 3–17.
- Khan, S.; Afzal, M.; Iqbal, S.; Khan, Q.M. Plant–bacteria partnerships for the remediation of hydrocarbon contaminated soils. Chemosphere 2013, 90, 1317–1332.
- Chaudhry, Q.; Blom-Zandstra, M.; Gupta, S.K.; Joner, E.J. Utilising the Synergy between Plants and Rhizosphere Microorganisms to Enhance Breakdown of Organic Pollutants in the Environment (15 pp). Environ. Sci. Pollut. Res. 2004, 12, 34–48.
- Aprill, W.; Sims, R.C. Evaluation of the use of prairie grasses for stimulating polycyclic aromatic hydrocarbon treatment in soil. Chemosphere 1990, 20, 253–265.
- Gaskin, S.E.; Bentham, R.H. Rhizoremediation of hydrocarbon contaminated soil using Australian native grasses. Sci. Total Environ. 2010, 408, 3683–3688.
- Hall, J.; Soole, K.; Bentham, R. Hydrocarbon Phytoremediation in the FamilyFabacea—A Review. Int. J. Phytoremediat. 2011, 13, 317–332.
- Reynolds, C.M.; Wolf, D.C.; Gentry, T.J.; Perry, L.B.; Pidgeon, C.S.; Koenen, B.A.; Rogers, H.B.; Beyrouty, C.A. Plant enhancement of indigenous soil micro-organisms: A low-cost treatment of contaminated soils. Polar Rec. 1999, 35, 33–40.
- Reichenauer, T.G.; Germida, J. Phytoremediation of Organic Contaminants in Soil and Groundwater. ChemSusChem 2008, 1, 708–717.
- Sandermann, H. Plant metabolism of xenobiotics. Trends Biochem. Sci. 1992, 17, 82–84.
- Singer, A.C. Advances in development of transgenic plants for remediation of xenobiotic pollutants. In Phytoremediation and Rhizoremediation: Theoretical Background; Mackova, M., Dowling, D., Macek, T., Eds.; Springer: Dordrecht, UK, 2006; pp. 5–21.
- Glick, B.R.; Stearns, J.C. Making Phytoremediation Work Better: Maximizing a Plant’s Growth Potential in the Midst of Adversity. Int. J. Phytoremediat. 2011, 13, 4–16.
- Hardoim, P.; van Overbeek, L.S.; van Elsas, J.D. Properties of bacterial endophytes and their proposed role in plant growth. Trends Microbiol. 2008, 16, 463–471.
- Vacheron, J.; Desbrosses, G.; Bouffaud, M.-L.; Touraine, B.; Moënne-Loccoz, Y.; Muller, D.; Legendre, L.; Wisniewski-Dyé, F.; Prigent-Combaret, C. Plant growth-promoting rhizobacteria and root system functioning. Front. Plant Sci. 2013, 4, 356.
- Li, J.; Sun, J.; Yang, Y.; Guo, S.; Glick, B.R. Identification of hypoxic-responsive proteins in cucumber roots using a proteomic approach. Plant Physiol. Biochem. 2012, 51, 74–80.
- Li, Q.; Saleh-Lakha, S.; Glick, B.R. The effect of native and ACC deaminase-containing Azospirillum brasilense Cd1843 on the rooting of carnation cuttings. Can. J. Microbiol. 2005, 51, 511–514.
- Glick, B.R. Modulation of plant ethylene levels by the bacterial enzyme ACC deaminase. FEMS Microbiol. Lett. 2005, 251, 1–7.
- Equiza, L.; St-Arnaud, M.; Eyergeau, E. Harnessing phytomicrobiome signaling for rhizosphere microbiome engineering. Front. Plant Sci. 2015, 6, 507.
- Bulgarelli, D.; Rott, M.; Schlaeppi, K.; Van Themaat, E.V.L.; Ahmadinejad, N.; Assenza, F.; Rauf, P.; Huettel, B.; Reinhardt, R.; Schmelzer, E.; et al. Revealing structure and assembly cues for Arabidopsis root-inhabiting bacterial microbiota. Nat. Cell Biol. 2012, 488, 91–95.
- Lundberg, D.S.; Lebeis, S.L.; Paredes, S.H.; Yourstone, S.; Gehring, J.; Malfatti, S.; Tremblay, J.; Engelbrektson, A.; Kunin, V.; Del Rio, T.G.; et al. Defining the core Arabidopsis thaliana root microbiome. Nat. Cell Biol. 2012, 488, 86–90.
- Bell, T.; Hassan, S.E.-D.; Lauron-Moreau, A.; Al-Otaibi, F.; Hijri, M.; Yergeau, E.; St-Arnaud, M. Linkage between bacterial and fungal rhizosphere communities in hydrocarbon-contaminated soils is related to plant phylogeny. ISME J. 2013, 8, 331–343.
- Hassan, S.E.-D.; Bell, T.H.; Stefani, F.O.P.; Denis, D.; Hijri, M.; St-Arnaud, M. Contrasting the Community Structure of Arbuscular Mycorrhizal Fungi from Hydrocarbon-Contaminated and Uncontaminated Soils following Willow (Salix spp. L.) Planting. PLoS ONE 2014, 9, e102838.
- Tardif, S.; Yergeau, E.; Tremblay, J.; Legendre, P.; Whyte, L.G.; Greer, C.W. The Willow Microbiome Is Influenced by Soil Petroleum-Hydrocarbon Concentration with Plant Compartment-Specific Effects. Front. Microbiol. 2016, 7, 1363.
- Mitter, E.K.; De Freitas, J.R.; Germida, J.J. Bacterial Root Microbiome of Plants Growing in Oil Sands Reclamation Covers. Front. Microbiol. 2017, 8, 849.
- Ofek-Lalzar, M.; Sela, N.; Goldman-Voronov, M.; Green, S.; Hadar, Y.; Minz, D. Niche and host-associated functional signatures of the root surface microbiome. Nat. Commun. 2014, 5, 4950.





