
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | shubhangi shukla | + 1871 word(s) | 1871 | 2021-08-19 06:20:44 | | | |
| 2 | Lindsay Dong | Meta information modification | 1871 | 2021-08-20 05:01:51 | | |
Video Upload Options
The use of novel photoinitiators (PIs) for free-radical polymerization has attracted significant attention from the scientific community. Quantum PIs, quantum-confined nanoscale crystals with semiconductor properties, have received interest for use in photopolymerization, due to their precisely tunable properties as a function of structural and surface engineering.
1. Introduction
Current advancements in synthetic procedures have led to the development of a variety of quantum PIs with characteristic photoinitiating properties [1][2][3][4][5][6][7]. The surface moieties introduced in the course of nanocrystal synthesis facilitate control over skeletal arrangements, dispersibility, and reactivity at the molecular level [8][9][10][1][2][3][4][5][6][7]. Postsynthetic surface modifications enable the dispersal and stabilization of nanocrystals PIs in formulations for many applications [2][3][4][5][6][7][11][12][13][14][15][16][17][18][19][20].
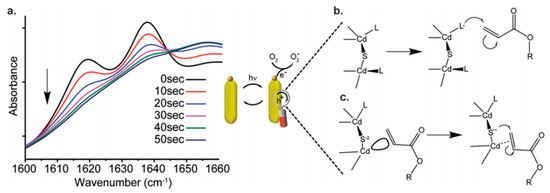
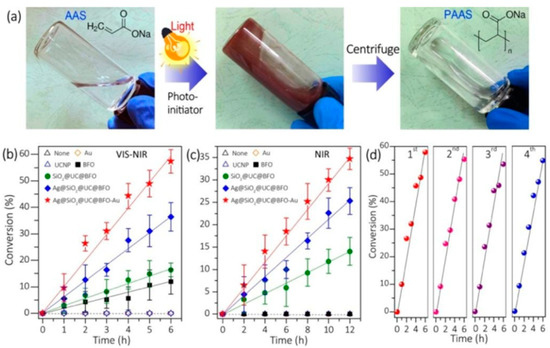
Several classes of quantum PIs have been developed and evaluated for polymerization, with various absorption and excitation wavelength windows [15][16][17][18][19][20]. The mode of action of most of the quantum PIs is generally similar to type I radical initiators, with highly conjugated aromatic moieties; others mimic the activity of type II initiators [2][3][4][5][6][7]. Loir et al. indicated that two pathways exist for initiation in quantum PIs, which involve surface mediated hole transfer (Figure 1) [7]. Typical quantum PIs can be classified as semiconductor nanoparticles (NPs), such as TiO2, ZnO, and CdS NPs [10][1][2][3][4][5][6][7][11][12][13][14][15][16]; hybrid photoinitiators, including composites of metal nanoparticles (MNPs)/silanized metal (organic PIs, MNPs), fluorescent dyes, oligomeric silsesquioxane, and parent PIs, [17] semiconductor NPs (metal/graphene oxide) [18][19][20][21][22][23], and organometallic nanoparticles; panchromatic photoinitiators, namely upconverting nanoparticles (UCNPs) [24][25][26][27][28][29][30][31][32][33][34] and plasmonic nanoparticle composites (e.g., Ag@SiO2@UC@BFO–Au core@triple-shell); near-infrared photoinitiators, such as luminescent lanthanides (e.g., Ln3+, Yb3+, Er3+, and Ho3+) and doped nanomaterials in a crystalline host lattice (NaYF4); [31] magnetic nanoparticles, such as Fe2O3; and metal core-shell nanoparticles (e.g., Ag@AgCl nanocubes) [35][36]. Figure 2 describes the polymerization of acrylic acid sodium (AAS) using the Ag@SiO2@UC@BFO-Au photoinitiator.
2. Categories of Quantum Photoinitiators
2.1. Semiconductor QPIs
Quantum photoinitiators based on semiconductor nanocrystals are considered a viable alternative to traditional organic photoinitiators with low molecular weights. Semiconductor nanocrystals have attracted attention due to their capacity to function as photocatalysts for many types of chemical reactions; these materials offer unique advantages, such as efficient light-harvesting activity, tunable properties, and large surface area-to-volume ratios [9]. These nanocrystals exhibit quantum confinement effects; the properties of these materials may be modified by synthetic control over nanocrystal size, shape, and composition [9].
Semiconductor quantum dots (QD) are solution-dispersible nanocrystals, which have found use as photocatalysts for light-induced polymerization. QDs exhibit strong absorption in the UV-visible range, with large extinction coefficients (ε > 105 M−1 cm−1) [14], large specific surface area values that allow for the interaction with multiple substrates, and higher photostability than organic-based photocatalysts and transition metal complexes [14], Semiconductor suspensions were first used for photopolymerization by Kuriacose et al.; ZnO powders were used for photopolymerization of methyl methacrylate (MMA) in water [8]. The effect of the oxygen level on polymerization was investigated; higher amounts of oxygen were associated with a lower molecular weight and a larger number of chains. On the other hand, too much oxygen in the solution suppressed the propagation step; this phenomenon was reported to be a common limitation for polymerization initiated by oxidative anionic species-releasing photoinitiators.
2.2. Carbon-Based QPIs
2.3. Graphene-Based QPIs
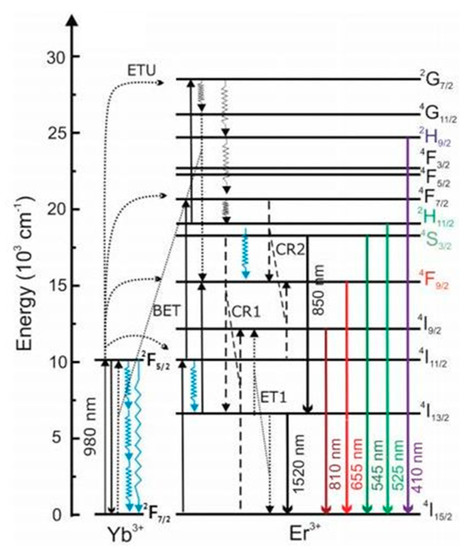
2.4. UCNPs and Hybrid QPIs
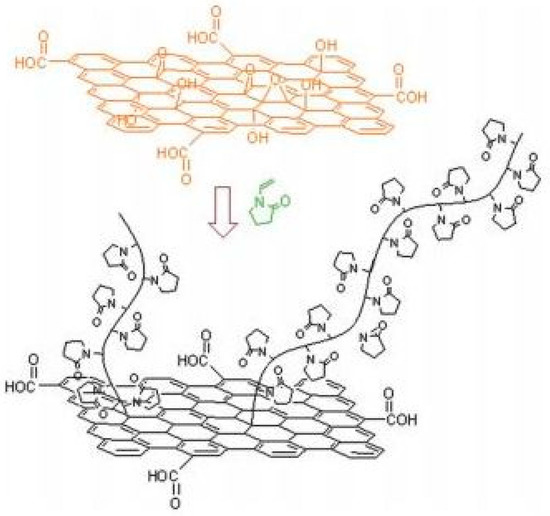
2.5. Polymer–Hybrid QPIs
References
- Waiskopf, N.; Ben-Shahar, Y.; Banin, U. Photocatalytic hybrid semiconductor–metal nanoparticles; from synergistic properties to emerging applications. Adv. Mater. 2018, 30, 1706697.
- Schmitt, M. ZnO Nanoparticle Induced Photo-Kolbe Reaction, Fragment Stabilization and Effect on Photopolymerization Monitored by Raman–UV-Vis Measurements. Macromol. Chem. Phys. 2012, 213, 1953–1962.
- Schmitt, M.; Lalevée, J. ZnO nanoparticles as polymerisation Photo-Initiator: Levulinic acid/NaOH content variation. Colloids Surf. A Physicochem. Eng. Asp. 2017, 532, 189–194.
- Ojah, R.; Dolui, S.K. Solar radiation-induced polymerization of methyl methacrylate in the presence of semiconductor-based photocatalyst. Sol. Energy. Mater. Sol. Cells 2006, 90, 1615–1620.
- Strandwitz, N.C.; Khan, A.; Boettcher, S.W.; Mikhailovsky, A.A.; Hawker, C.J.; Nguyen, T.Q.; Stucky, G.D. One-and two-photon induced polymerization of methylmethacrylate using colloidal CdS semiconductor quantum dots. J. Am. Chem. Soc. 2008, 130, 8280–8288.
- Zhang, D.; Yang, J.; Bao, S.; Wu, Q.; Wang, Q. Semiconductor nanoparticle-based hydrogels prepared via self-initiated polymerization under sunlight, even visible light. Sci. Rep. 2013, 3, 1–7.
- Verbitsky, L.; Waiskopf, N.; Magdassi, S.; Banin, U. A clear solution: Semiconductor nanocrystals as photoinitiators in solvent free polymerization. Nanoscale 2019, 11, 11209–11216.
- Kuriacose, J.C.; Markham, M.C. Mechanism of the Photo-Initiated Polymerization of Methyl Methacrylate at Zinc Oxide Surfaces. J. Phys. Chem. 1961, 65, 2232–2236.
- Waiskopf, N.; Magdassi, S.; Banin, U. Quantum Photoinitiators: Toward Emerging Photocuring Applications. J. Am. Chem. Soc. 2021, 143, 577–587.
- Pinkas, A.; Waiskopf, N.; Gigi, S.; Naor, T.; Layani, A.; Banin, U. Morphology Effect on Zinc Oxide Quantum Photoinitiators for Radical Polymerization. Nanoscale 2021, 3, 7152–7160.
- Zhou, J.; Allonas, X.; Ibrahim, A.; Liu, X. Progress in the development of polymeric and multifunctional photoinitiators. Prog. Polym. Sci. 2019, 99, 101165.
- Feng, J.; Ye, D. Polymerizable ZnO photoinitiators of surface modification with hydroxyl acrylates and photopolymerization with UV-curable waterborne polyurethane acrylates. Eur. Polym. J. 2019, 120, 109252.
- Dadashi-Silab, S.; Atilla Tasdelen, M.; Mohamed Asiri, A.; Bahadar Khan, S.; Yagci, Y. Photoinduced atom transfer radical polymerization using semiconductor nanoparticles. Macromol. Rapid Comm. 2014, 35, 454–459.
- Huang, Y.; Zhu, Y.; Egap, E. Semiconductor quantum dots as photocatalysts for controlled light-mediated radical polymerization. ACS Macro Lett. 2018, 7, 184–189.
- Liang, E.; Liu, M.S.; He, B.; Wang, G.X. ZnO as photocatalyst for photoinduced electron transfer–reversible addition–fragmentation chain transfer of methyl methacrylate. Adv. Polym. Technol. 2018, 37, 2879–2884.
- Liao, J.; Ye, D. Improving ZnO photoinitiation efficiency by surface reaction with 2-hydroxy-2-methylpropiophenone. Eur. Polym. J. 2021, 147, 110293.
- Han, Y.; Wang, F.; Lim, C.Y.; Chi, H.; Chen, D.; Wang, F.; Jiao, X. High-performance nano-photoinitiators with improved safety for 3D printing. ACS Appl. Mater. Interfaces. 2017, 9, 32418–32423.
- Huang, Y.; Zeng, M.; Ren, J.; Wang, J.; Fan, L.; Xu, Q. Preparation and swelling properties of graphene oxide/poly (acrylic acid-co-acrylamide) super-absorbent hydrogel nanocomposites. Colloids Surf. A Physicochem. Eng. Asp. 2012, 401, 97–106.
- Feng, R.; Zhou, W.; Guan, G.; Li, C.; Zhang, D.; Xiao, Y.; Zheng, L.; Zhu, W. Surface decoration of graphene by grafting polymerization using graphene oxide as the initiator. J. Mater. Chem. 2012, 22, 3982–3989.
- Andryushina, N.S.; Stroyuk, O.L.; Dudarenko, G.V.; Kuchmiy, S.Y.; Pokhodenko, V.D. Photopolymerization of acrylamide induced by colloidal graphene oxide. J. Photochem. Photobiol. A 2013, 256, 1–6.
- Feng, Y.; Peng, C.; Li, Y.; Hu, J.; Deng, Q.; Wu, Q.; Xu, Z. Superhydrophobic nanocomposite coatings with photoinitiated three-dimensional networks based on reactive graphene nanosheet-induced self-wrinkling patterned surfaces. J. Colloid. Interface. Sci. 2019, 536, 149–159.
- Sangermano, M.; Marchi, S.; Valentini, L.; Bon, S.B.; Fabbri, P. Transparent and conductive graphene oxide/poly (ethylene glycol) diacrylate coatings obtained by photopolymerization. Macromol. Mater. Eng. 2011, 296, 401–407.
- Wang, X.; Xing, W.; Song, L.; Yu, B.; Hu, Y.; Yeoh, G.H. Preparation of UV-curable functionalized graphene/polyurethane acrylate nanocomposite with enhanced thermal and mechanical behaviors. React Funct. Polym. 2013, 73, 854–858.
- Chen, Z.; Oprych, D.; Xie, C.; Kutahya, C.; Wu, S.; Strehmel, B. Upconversion-nanoparticle-assisted radical polymerization at λ= 974 nm and the generation of acidic cations. ChemPhotoChem 2017, 1, 499–503.
- Li, Z.; Chen, H.; Wang, C.; Chen, L.; Liu, J.; Liu, R. Efficient photopolymerization of thick pigmented systems using upconversion nanoparticles-assisted photochemistry. J. Polym. Sci. A Polym. Chem. 2018, 56, 994–1002.
- Rocheva, V.V.; Koroleva, A.V.; Savelyev, A.G.; Khaydukov, K.V.; Generalova, A.N.; Nechaev, A.V.; Guller, A.E.; Semchishen, V.A.; Chichkov, B.N.; Khaydukov, E.V. High-resolution 3D photopolymerization assisted by upconversion nanoparticles for rapid prototyping applications. Sci. Rep. 2018, 8, 1–10.
- Qin, X.; Carneiro Neto, A.N.; Longo, R.L.; Wu, Y.; Malta, O.L.; Liu, X. Surface Plasmon–Photon Coupling in Lanthanide-Doped Nanoparticles. J. Phys. Chem. Lett. 2021, 12, 1520–1541.
- Wang, K.; Peña, J.; Xing, J. Upconversion Nanoparticle-Assisted Photopolymerization. Photochem. Photobiol. 2020, 96, 741–749.
- Bagheri, A.; Arandiyan, H.; Adnan, N.N.M.; Boyer, C.; Lim, M. Controlled direct growth of polymer shell on upconversion nanoparticle surface via visible light regulated polymerization. Macromolecules 2017, 50, 7137–7147.
- Oprych, D.; Schmitz, C.; Ley, C.; Allonas, X.; Ermilov, E.; Erdmann, R.; Strehmel, B. Photophysics of Up-Conversion Nanoparticles: Radical Photopolymerization of Multifunctional Methacrylates Comprising Blue-and UV-Sensitive Photoinitiators. ChemPhotoChem 2019, 3, 1119–1126.
- Zhang, J.; Huang, Y.; Jin, X.; Nazartchouk, A.; Liu, M.; Tong, X.; Jiang, Y.; Ni, L.; Sun, S.; Sang, Y.; et al. Plasmon enhanced upconverting [email protected] triple-shell nanoparticles as recyclable panchromatic initiators (blue to infrared) for radical polymerization. Nanoscale Horiz. 2019, 4, 907–917.
- Jee, H.; Chen, G.; Prasad, P.N.; Ohulchanskyy, T.Y.; Lee, J. In Situ Ultraviolet Polymerization Using Upconversion Nanoparticles: Nanocomposite Structures Patterned by Near Infrared Light. Nanomaterials 2020, 10, 2054.
- Ding, C.; Wang, J.; Zhang, W.; Pan, X.; Zhang, Z.; Zhang, W.; Zhu, J.; Zhu, X. Platform of near-infrared light-induced reversible deactivation radical polymerization: Upconversion nanoparticles as internal light sources. Polym. Chem. 2016, 7, 7370–7374.
- Hu, L.; Hao, Q.; Wang, L.; Cui, Z.; Fu, P.; Liu, M.; Qiao, X.; Pang, X. The in situ “grafting from” approach for the synthesis of polymer brushes on upconversion nanoparticles via NIR-mediated RAFT polymerization. Polym. Chem. 2021, 12, 545–553.
- Nehlig, E.; Schneider, R.; Vidal, L.; Clavier, G.; Balan, L. Silver nanoparticles coated with thioxanthone derivative as hybrid photoinitiating systems for free radical polymerization. Langmuir 2012, 28, 17795–17802.
- Yang, Y.; Zhao, Y.; Yan, Y.; Wang, Y.; Guo, C.; Zhang, J. Preparation of AgCl nanocubes and their application as efficient photoinitiators in the polymerization of N-isopropylacrylamide. J. Phys. Chem. B 2015, 119, 14807–14813.
- Kiskan, B.; Zhang, J.; Wang, X.; Antonietti, M.; Yagci, Y. Mesoporous graphitic carbon nitride as a heterogeneous visible light photoinitiator for radical polymerization. ACS Macro Lett. 2012, 1, 546–549.
- Dadashi-Silab, S.; Tasdelen, M.A.; Kiskan, B.; Wang, X.; Antonietti, M.; Yagci, Y. Photochemically mediated atom transfer radical polymerization using polymeric semiconductor mesoporous graphitic carbon nitride. Macromol. Chem. Phys. 2014, 215, 675–681.
- Fu, Q.; Ruan, Q.; McKenzie, T.G.; Reyhani, A.; Tang, J.; Qiao, G.G. Development of a robust PET-RAFT polymerization using graphitic carbon nitride (g-C3N4). Macromolecules 2017, 50, 7509–7516.
- Würth, C.; Kaiser, M.; Wilhelm, S.; Grauel, B.; Hirsch, T.; Resch-Genger, U. Excitation power dependent population pathways and absolute quantum yields of upconversion nanoparticles in different solvents. Nanoscale 2017, 9, 4283–4294.
- Du, Y.; Jia, S.; Chen, Y.; Zhang, L.; Tan, J. Type I Photoinitiator-Functionalized Block Copolymer Nanoparticles Prepared by RAFT-Mediated Polymerization-Induced Self-Assembly. ACS Macro Lett. 2021, 10, 297–306.
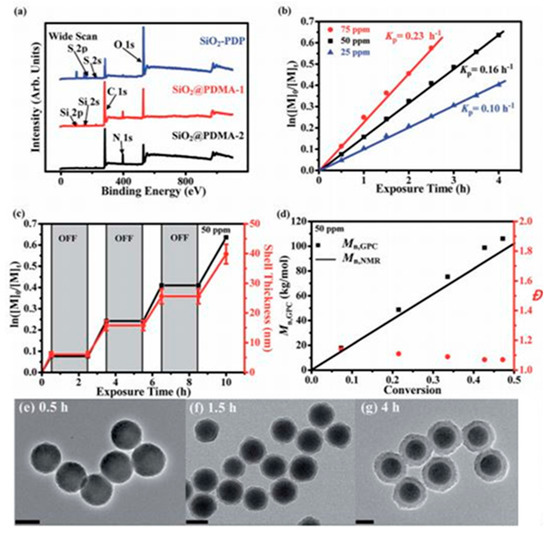
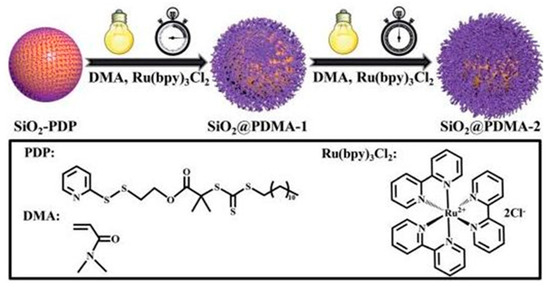
- Li, X.; Ye, S.; Huang, Y.; Le Li, J.; Cai, T. Precise growth of polymer brushes on silica-based nanocomposites via visible-light-regulated controlled radical polymerization. J. Mater. Chem. A 2019, 7, 6173–6179.





