
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Bo Li | + 2715 word(s) | 2715 | 2021-08-04 14:36:30 | | | |
| 2 | Conner Chen | Meta information modification | 2715 | 2021-08-16 05:47:23 | | | | |
| 3 | Conner Chen | Meta information modification | 2715 | 2021-09-22 04:17:51 | | |
Video Upload Options
Suture mesenchymal stem cells (SuSCs), a heterogeneous stem cell population, belong to mesenchymal stem cells (MSCs) or skeletal stem cells (SSCs), with the ability to self-renew and undergo multi-lineage differentiation. Unlike the well-established perivascular niche of SSCs in the long bone, stem cells of the cranial bone are generally located and confined within the cranial suture mesenchyme, subsequently defined as SuSCs. In the long bone, SSCs play an essential role in plenty of physiological processes, such as growth and development, life-long homeostasis, and fracture healing. Similarly, as the major stem cell population of cranial bones, the physiological significance of SuSCs is undoubted and self-evident.
1. The Anatomy of Cranial Sutures
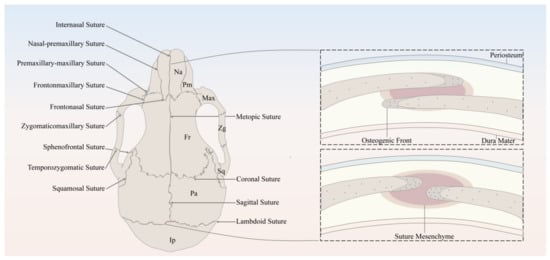
2. Temporal and Spatial Distribution Features of SuSCs Subpopulations
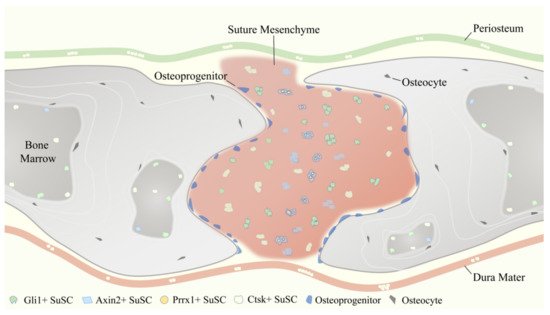
2.1. Temporal and Spatial Distribution Features of Gli1+ SuSCs
2.2. Temporal and Spatial Distribution Features of Axin2+ SuSCs
2.3. Temporal and Spatial Distribution Features of Prrx1+ SuSCs
2.4. Temporal and Spatial Distribution Features of Ctsk+ SuSCs
3. Characteristics of SuSCs and Their Role in Injury Repair
3.1. Biological Characteristics of Different SuSCs Subpopulations
3.2. Contribution of SuSCs in Calvarial Bone Injury
References
- Lenton, K.A.; Nacamuli, R.P.; Wan, D.C.; Helms, J.A.; Longaker, M.T. Cranial Suture Biology. Curr. Top. Dev. Biol. 2005, 66, 287–328.
- Behr, B.; Longaker, M.T.; Quarto, N. Differential activation of canonical Wnt signaling determines cranial sutures fate: A novel mechanism for sagittal suture craniosynostosis. Dev. Biol. 2010, 344, 922–940.
- White, H.E.; Goswami, A.; Tucker, A.S. The Intertwined Evolution and Development of Sutures and Cranial Morphology. Front. Cell Dev. Biol. 2021, 9, 690.
- Opperman, L.A. Cranial Sutures as Intramembranous Bone Growth Sites. Dev. Dyn. 2000, 219, 472–485.
- Morriss-Kay, G.M.; Wilkie, A.O.M. Growth of the normal skull vault and its alteration in craniosynostosis: Insights from human genetics and experimental studies. J. Anat. 2005, 207, 637–653.
- Zhao, H.; Feng, J.; Ho, T.V.; Grimes, W.; Urata, M.; Chai, Y. The suture provides a niche for mesenchymal stem cells of craniofacial bones. Nat. Cell Biol. 2015, 17, 386–396.
- Maruyama, T.; Jeong, J.; Sheu, T.J.; Hsu, W. Stem cells of the suture mesenchyme in craniofacial bone development, repair and regeneration. Nat. Commun. 2016, 7, 10526.
- Wilk, K.; Yeh, S.C.A.; Mortensen, L.J.; Ghaffarigarakani, S.; Lombardo, C.M.; Bassir, S.H.; Aldawood, Z.A.; Lin, C.P.; Intini, G. Postnatal Calvarial Skeletal Stem Cells Expressing PRX1 Reside Exclusively in the Calvarial Sutures and Are Required for Bone Regeneration. Stem Cell Rep. 2017, 8, 933–946.
- Debnath, S.; Yallowitz, A.R.; McCormick, J.; Lalani, S.; Zhang, T.; Xu, R.; Li, N.; Liu, Y.; Yang, Y.S.; Eiseman, M.; et al. Discovery of a periosteal stem cell mediating intramembranous bone formation. Nature 2018, 562, 133–139.
- Jing, D.; Li, C.; Yao, K.; Xie, X.; Wang, P.; Zhao, H.; Feng, J.Q.; Zhao, Z.; Wu, Y.; Wang, J. The vital role of Gli1+ mesenchymal stem cells in tissue development and homeostasis. J. Cell. Physiol. 2021, 236, 6077–6089.
- Shi, Y.; He, G.; Lee, W.C.; McKenzie, J.A.; Silva, M.J.; Long, F. Gli1 identifies osteogenic progenitors for bone formation and fracture repair. Nat. Commun. 2017, 8, 2043.
- Schneider, R.K.; Mullally, A.; Dugourd, A.; Ebert, B.L.; Humphreys, B.D.; Schneider, R.K.; Mullally, A.; Dugourd, A.; Peisker, F.; Hoogenboezem, R. Gli1+ Mesenchymal Stromal Cells Are a Key Driver of Bone Marrow Fibrosis and an Important Cellular Therapeutic Target. Cell Stem Cell 2017, 20, 785–800.
- Zhao, H.; Feng, J.; Seidel, K.; Shi, S.; Klein, O.; Sharpe, P.; Chai, Y.; Sharpe, P.; Chai, Y. Secretion of Shh by a neurovascular bundle niche supports mesenchymal stem cell homeostasis in the adult mouse incisor. Cell Stem Cell 2014, 14, 160–173.
- Chen, S.; Jing, J.; Yuan, Y.; Feng, J.; Han, X.; Wen, Q.; Ho, T.V.; Lee, C.; Chai, Y. Runx2+ Niche Cells Maintain Incisor Mesenchymal Tissue Homeostasis through IGF Signaling. Cell Rep. 2020, 32, 108007.
- Men, Y.; Wang, Y.; Yi, Y.; Dian, J.; Luo, W.; Shen, B.; Stenberg, W.; Chai, Y.; Ge, W.-P.; Feng, J.Q.; et al. Gli1 + periodontium stem cells are regulated by osteocytes and occlusal force. Dev. Cell 2020, 54, 639–654.
- Degirmenci, B.; Valenta, T.; Dimitrieva, S.; Hausmann, G.; Basler, K. GLI1-expressing mesenchymal cells form the essential Wnt-secreting niche for colon stem cells. Nature 2018, 558, 449–453.
- Cassandras, M.; Wang, C.; Kathiriya, J.; Tsukui, T.; Matatia, P.; Matthay, M.; Wolters, P.; Molofsky, A.; Sheppard, D.; Chapman, H.; et al. Gli1 + mesenchymal stromal cells form a pathological niche to promote airway progenitor metaplasia in the fibrotic lung. Nat. Cell Biol. 2020, 22, 1295–1306.
- Dian, J.; Zhao, H.; Yuan, Q.; Feng, J.Q.; Luo, W.; Gao, X.; Men, Y.; Zhao, Z.; Gao, L.; Ge, W.-P.; et al. Tissue clearing of both hard and soft tissue organs with the PEGASOS method. Cell Res. 2018, 28, 803–818.
- Luo, W.; Yi, Y.; Jing, D.; Zhang, S.; Men, Y.; Ge, W.P.; Zhao, H. Investigation of Postnatal Craniofacial Bone Development with Tissue Clearing-Based Three-Dimensional Imaging. Stem Cells Dev. 2019, 28, 1310–1321.
- Maruyama, T.; Mirando, A.J.; Deng, C.X.; Hsu, W. The balance of WNT and FGF signaling influences mesenchymal stem cell fate during skeletal development. Sci. Signal. 2010, 3, ra40.
- Martin, J.F.; Olson, E.N. Identification of a prx1 limb enhancer. Genesis 2000, 26, 225–229.
- Ten Berge, D.; Brouwer, A.; Korving, J.; Martin, J.F.; Meijlink, F. Prx1 and Prx2 in skeletogenesis: Roles in the craniofacial region, inner ear and limbs. Development 1998, 125, 3831–3842.
- Boyle, W.J.; Simonet, W.S.; Lacey, D.L. Osteoclast differentiation and activation. Nature 2003, 423, 337–342.
- Yang, W.; Wang, J.; Moore, D.C.; Liang, H.; Dooner, M.; Wu, Q.; Terek, R.; Chen, Q.; Ehrlich, M.G.; Quesenberry, P.J.; et al. Ptpn11 deletion in a novel progenitor causes metachondromatosis by inducing hedgehog signalling. Nature 2013, 499, 491–495.
- Horwitz, E.M.; Le Blanc, K.; Dominici, M.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.C.; Deans, R.J.; Krause, D.S.; Keating, A. Clarification of the nomenclature for MSC: The International Society for Cellular Therapy position statement. Cytotherapy 2005, 7, 393–395.
- Dominici, M.; Le Blanc, K.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.C.; Krause, D.S.; Deans, R.J.; Keating, A.; Prockop, D.J.; Horwitz, E.M. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 2006, 8, 315–317.
- Penninger, J.M.; Irie-Sasaki, J.; Sasaki, T.; Oliveira-dos-Santos, A.J. CD45: New jobs for an old acquaintance. Nat. Immunol. 2001, 2, 389–396.
- Kina, T.; Ikuta, K.; Takayama, E.; Wada, K.; Sen Majumdar, A.; Weissman, I.L.; Katsura, Y. The monoclonal antibody TER-119 recognizes a molecule associated with glycophorin A and specifically marks the late stages of murine erythroid lineage. Br. J. Haematol. 2000, 109, 280–287.
- Chan, C.K.F.; Lindau, P.; Jiang, W.; Chen, J.Y.; Zhang, L.F.; Chen, C.C.; Seita, J.; Sahoo, D.; Kim, J.B.; Lee, A.; et al. Clonal precursor of bone, cartilage, and hematopoietic niche stromal cells. Proc. Natl. Acad. Sci. USA 2013, 110, 12643–12648.
- Doro, D.H.; Grigoriadis, A.E.; Liu, K.J. Calvarial suture-derived stem cells and their contribution to cranial bone repair. Front. Physiol. 2017, 8, 956.





