
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Arti Ahluwalia | + 1853 word(s) | 1853 | 2021-08-10 11:54:36 | | | |
| 2 | Ron Wang | + 58 word(s) | 1911 | 2021-08-16 04:21:06 | | |
Video Upload Options
The human placenta brings the maternal and fetal circulatory systems into contact while keeping them independent, due to its intricate structure and adaptation in pregnancy. The placental barrier, composed essentially of trophoblasts, connective tissue and endothelium, separates the fetal and maternal compartments and is a key structure for this organ’s function. Maternal—fetal exchange takes place in the areas where the barrier is an extremely thin membrane (only 3.5 μm thick).
Many of the human placental barrier functions are little understood and more representative models are still needed to better recapitulate the complex architecture and dynamics of the human fetal—placental barrier and provide a more in-depth understanding of the organ’s function.
1. Introduction
The human placenta brings the maternal and fetal circulatory systems into contact while keeping them independent, due to its intricate structure and adaptation in pregnancy. The placental barrier, composed essentially of trophoblasts, connective tissue and endothelium, separates the fetal and maternal compartments and is a key structure for this organ’s function [1][2]. Maternal—fetal exchange takes place in the areas where the barrier is an extremely thin membrane (only 3.5 μm thick) [3]. The rate and amount of human placental transfer are determinants for fetal development, and for that reason, the placenta has been the subject of several studies from different fields. The correct development of the placental structure and function should be maintained for a healthy pregnancy. Importantly, the placenta is permeable to ensure the passage of vital substances (such as nutrients and oxygen); however, it can also be permeable to other exogenous substances and potentially harmful chemicals, such as phthalates, phenols (e.g., bisphenol A (BPA)), polychlorinated biphenyls (PCBs) and heavy metals [4][5].
It is, therefore, crucial to understand placental physiology in humans through the development of reliable models suitable for testing placental function under a multitude of different conditions.
An additional issue with the reliance on animal placental models relates to their ethical and financial implications. Due to stricter animal welfare legislation in the EU, USA and other countries worldwide [6][7], there is a significant drive to replace, reduce and refine (3Rs) the use of animals for scientific purposes. Thus, it is imperative that robust and sensitive alternative placental models are developed, validated and implemented.
In addition to animal models, there are currently other approaches to model the placenta, including ex vivo, in vitro and in silico methods that have provided useful insights into the mechanisms of the fetal—placental barrier.
2. In Vivo Placental Models
With the exception of primates, animal models have been proven to be less than ideal for the study of human placental physiology, as many aspects, such as the high level of invasiveness of trophoblasts, are unique to humans [8][9]. Primate pregnancy has been used as an animal fetal—placental model, as it closely relates to humans in terms of length of gestation, changes in the contractile milieu, mechanisms of steroidogenesis and process of placentation, to name a few [10].
Despite the serious considerations regarding animal models mentioned in the previous section, during the past decade (2010–2020), 8830 manuscripts were published using animal models (ranging from mice and rats to ewes and monkeys) to study placental physiology; 914 were related to preeclampsia, 281 to toxins and 56 to EDCs. Moreover, 211 studies were focused on umbilical cord physiology (source: PubMed.gov ; filters: placenta, 2010–2020, “other animals” with specified search terms). Validation and development of alternative 3D in vitro models or the use of organotypic cultures of human placenta will have a measurable impact as a sound alternative for assessing the effects of any molecule on placental physiology and subsequently contributing towards the 3Rs.
Adoption of alternative models, such as organ-on-a-chip (OOC), human perfused placentas and placental explants, will inevitably drive a quantifiable reduction in and, potentially, replacement of the use of animals as part of the 3Rs. Comparative studies between human placental explants or OOC models and in vivo studies will enable minimal use of animals per experiment, providing there is reproducibility of readouts. It should be noted that when placental function is assessed in mice or rats, apart from the mothers, all offspring are culled, leading to an exponential rise in animal usage, as in Susiarjo et al. ’s study cited above [11]. Besides mouse and rat models, placental studies have also used primates [12]. However, research using nonhuman primates raises serious ethical issues, as reflected by the EU ban (Directive 2010/63) [13] which states: “the use of nonhuman primates should be permitted only in those biomedical areas essential for the benefit of human beings, for which no alternative replacement methods are yet available”. Thus, in Europe as well as in most countries that are adopting legislation to reduce the use of animals for scientific purposes, the nonhuman primate model is no longer an option for studying placental function or structure.
It is expected that OOC models will be used in priority settings during drug development and chemical safety assessment, as they will allow the detection and study of pharmacological and toxicological effects before animal testing is necessary. With the implementation of robust and representative alternative methods, chemicals can be evaluated in more detail than with traditional in vitro systems and reduce the number of ineffective/toxic chemicals that are taken further to in vivo studies. An area where these approaches will have minimum impact will be on the third R: refining the way animal experiments are conducted to ensure minimal suffering.
3. In Vitro Placental Models
Although studies on placental explants from humans benefit from investigating the exact tissues and cells needed to understand human placental physiology, these term placentas represent pathological conditions as mentioned above. Ex vivo perfusion systems represent the whole system and whilst short-term experiments can be conducted, studies over a longer time are required to give a greater understanding of the cell—cell communication. To address these problems, alternative in vitro models using immortalized or primary human trophoblasts are often the model of choice to study placental function. Indeed, they are useful for studying organ or tissue function and at the cellular or subcellular scales, as cells and their organelles can be studied in detail using imaging and molecular techniques.
In order to circumvent small tissue culture time windows, most in vitro studies employ immortalized trophoblastic cell lines, including ones deriving from choriocarcinoma [14]. Importantly, these cell lines have been used extensively to gain a better insight into trophoblast cell biology and placental development, syncytiotrophoblast formation and endocrine activity, as well as immune aspects of the fetal—placental unit [14][15]. For example, cell lines such as BeWo, JEG-3 and JAr (all tumor cells lines derived from trophoblasts) show similar characteristics to primary trophoblasts in terms of hormone secretion (e.g., hCG, estrogens and progesterone) [16]. JEG-3 and BeWo cell lines have distinct fusogenic capacities. The JEG-3 cell line is unable to morphologically differentiate; therefore, it resembles the undifferentiated and hormonally inactive cytotrophoblast cells, making it an appropriate in vitro model to investigate early placental events [17][18]. Additionally, BeWo cells retain the ability to form syncytia when being treated with forskolin (an activator of adenylyl cyclase) or 8-Br-cAMP [9][18][19]. At this stage, the predominant feature of the human placenta is the hormonally active syncytiotrophoblast layer. The capability of BeWo cells to differentiate has established these cells as an in vitro model to study placental physiology including development, immune and endocrine responses and transport mechanisms [16][18][19].
A number of in vitro models that can simulate the human fetal—placental barrier have been developed. Of these, the most commonly used is the Transwell set-up whereby cells are cultured on a microporous membrane, generally polycarbonate, that separates an apical and a basal compartment. The system can be employed to study the transport properties of the trophoblast barrier, and the cell line of choice is BeWo for its ability to form a confluent layer [20]. Two endothelial cell lines are used for coculture with the trophoblast cell lines for more complex Transwell experiments, the most common being a pure vascular endothelial cell line from the human placenta (HPEC) and human umbilical vein endothelial cells (HUVECs). They are derived through enzymatic perfusion of the term placenta and postpartum umbilical vein, respectively. As an example, a coculture model comprising tight layers of BeWo cells and placental endothelial cells has been generated using a 3 µm porous membrane for translocation studies to predict fetal exposure to nanoparticles [21].
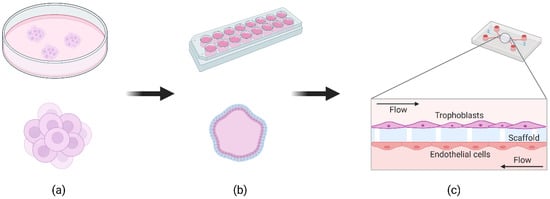
Standard cell culture involves seeding cells in monolayers on tissue culture plates or in transwells. As an alternative, 3D cell models can be used to study human organ physiology in cell culture, especially in the fields of cancer and toxicology. Extracellular parameters are more physiologically relevant in 3D, allowing cells to self-organize and grow into organoid-like structures [22]. The field of 3D placental models has evolved rapidly in the past decade, particularly with the discovery of organoids and advancements in fluidic systems for cell culture. We found 90 reports of 3D placental models in vitro since 2010. About half of them (43) are related to spheroid systems ( Figure 1 a) while 37 (11 were reviews) reports describing a microfluidic placenta-on-a-chip (PoC, Figure 1 c) device of some form have been published (source: PubMed.gov ; filters: 2010–2020; search terms: logical combinations of 3D, spheroid, placenta, model, microfluidic).
4. In Silico Placental Models
In silico models are a useful tool to facilitate the study and prediction of different aspects of placental physiology and pathology since the organ is unreachable for direct analysis during pregnancy.
Some models simulate the hemodynamics of the placenta, scrutinizing the blood circulation in different structures ranging from the spiral arteries of the maternal compartment [23] to the terminal villi on the fetal side [24][25][26][27]. The simulations are based on computational fluid dynamics that recreate the vascular structures of the placenta and model blood flow through them. These allow researchers to understand, for instance, how the development of the correct geometry of the organ can impact transfer processes, since the fetal/maternal exchange of crucial substances is determined by the blood flow of both circulations. Indeed, different flow models address not only the structure and hemodynamics of the placental tissues but also the diffusion of small solutes, providing an approach for linking placental structure and function [28].
The placental structure in some of the most recent models is obtained through imaging of the organ after tissue processing with advanced imaging and 3D reconstruction techniques [29]. Modeling the data gathered through computational simulations allows a noninvasive and faithful representation of the structure and dynamics of the human placenta, which cannot be accomplished with in vitro or in vivo models. In a different perspective, several parameters for the simulations were also acquired from ex vivo studies, which represents a case of a very profitable synergy between ex vivo and in silico , illustrating the potential of the 3Rs for placental models.
In these models, placental transfer has been modeled using different therapeutic substances, specifically antiviral drugs for HIV treatment: tenofovir [30], emtricitabine [30][31], nevirapine [30], darunavir [32] and the benzodiazepine midazolam [33]. One of the most promising features of some of the in silico models is that the same model can be used to predict concentrations of other compounds, by changing drug-related parameters, which confers versatility and “reusability” hardly attainable with in vivo models.
References
- Ramsey, E.M. Biology of Gestation. Vol. 1, The Maternal Organism, 1st ed.; Academic Press: New York, NY, USA, 1968; Volume 162.
- Firth, J.A.; Leach, L. Structure and Permeability in Human Placental Capillaries. Placenta 1997, 18, 205–213.
- Donnelly, L.; Campling, G. Functions of the Placenta. Anaesth. Intensive Care Med. 2014, 15, 136–139.
- Needham, L.L.; Grandjean, P.; Heinzow, B.; Jørgensen, P.J.; Nielsen, F.; Sjödin, A.; Patterson, D.G.; Turner, W.E.; Weihe, P. Partition of Environmental Chemicals between Maternal and Fetal Blood and Tissues. Environ. Sci. Technol. 2011, 45, 1121–1126.
- Mitro, S.D.; Johnson, T.; Zota, A.R. Cumulative Chemical Exposures During Pregnancy and Early Development. Curr. Environ. Health Rep. 2015, 2, 367–378.
- Animal Welfare Act|Animal Welfare Information Center|NAL|USDA. Available online: https://www.nal.usda.gov/awic/animal-welfare-act (accessed on 13 March 2021).
- Legislation for the Protection of Animals Used for Scientific Purposes—Environment—European Commission. Available online: https://ec.europa.eu/environment/chemicals/lab_animals/legislation_en.htm (accessed on 13 March 2021).
- Carter, A.M. Animal Models of Human Placentation—A Review. Placenta 2007, 28, S41–S47.
- Orendi, K.; Kivity, V.; Sammar, M.; Grimpel, Y.; Gonen, R.; Meiri, H.; Lubzens, E.; Huppertz, B. Placental and Trophoblastic in Vitro Models to Study Preventive and Therapeutic Agents for Preeclampsia. Placenta 2011, 32, S49–S54.
- Grigsby, P.L. Animal Models to Study Placental Development and Function throughout Normal and Dysfunctional Human Pregnancy. Semin. Reprod. Med. 2016, 34, 11–16.
- Susiarjo, M.; Sasson, I.; Mesaros, C.; Bartolomei, M.S. Bisphenol A Exposure Disrupts Genomic Imprinting in the Mouse. PLoS Genet. 2013, 9, e1003401.
- Patterson, T.A.; Twaddle, N.C.; Roegge, C.S.; Callicott, R.J.; Fisher, J.W.; Doerge, D.R. Concurrent Determination of Bisphenol A Pharmacokinetics in Maternal and Fetal Rhesus Monkeys. Toxicol. Appl. Pharmacol. 2013, 267, 41–48.
- DeGrazia, D. Nonhuman Primates, Human Need, and Ethical Constraints. Hastings Cent. Rep. 2016, 46, 27–28.
- King, A.; Thomas, L.; Bischof, P. Cell Culture Models of Trophoblast II: Trophoblast Cell Lines—A Workshop Report. Placenta 2000, 21, S113–S119.
- Berkowitz, R.S.; Goldstein, D.P. Chorionic Tumors. N. Engl. J. Med. 1996, 335, 1740–1748.
- Wolfe, M.W. Culture and Transfection of Human Choriocarcinoma Cells. Methods Mol. Med. 2006, 121, 229–239.
- Ntrivalas, E.; Kwak-Kim, J.; Beaman, K.; Mantouvalos, H.; Gilman-Sachs, A. An in Vitro Coculture Model to Study Cytokine Profiles of Natural Killer Cells during Maternal Immune Cell-Trophoblast Interactions. J. Soc. Gynecol. Investig. 2006, 13, 196–202.
- Mparmpakas, D.; Zachariades, E.; Foster, H.; Kara, A.; Harvey, A.; Goumenou, A.; Karteris, E. Expression of MTOR and Downstream Signalling Components in the JEG-3 and BeWo Human Placental Choriocarcinoma Cell Lines. Int. J. Mol. Med. 2010, 25, 65–69.
- de Aguiar Greca, S.-C.; Kyrou, I.; Pink, R.; Randeva, H.; Grammatopoulos, D.; Silva, E.; Karteris, E. Involvement of the Endocrine-Disrupting Chemical Bisphenol A (BPA) in Human Placentation. J. Clin. Med. 2020, 9, 405.
- Bode, C.J.; Jin, H.; Rytting, E.; Silverstein, P.S.; Young, A.M.; Audus, K.L. In Vitro Models for Studying Trophoblast Transcellular Transport. Methods Mol. Med. 2006, 122, 225–239.
- Aengenheister, L.; Keevend, K.; Muoth, C.; Schönenberger, R.; Diener, L.; Wick, P.; Buerki-Thurnherr, T. An Advanced Human in Vitro Co-Culture Model for Translocation Studies across the Placental Barrier. Sci. Rep. 2018, 8, 5388.
- Lelièvre, S.A.; Kwok, T.; Chittiboyina, S. Architecture in 3D Cell Culture: An Essential Feature for in Vitro Toxicology. Toxicol. Vitr. 2017, 45, 287–295.
- Cotter, S.L.; Klika, V.; Kimpton, L.; Collins, S.; Heazell, A.E.P. A Stochastic Model for Early Placental Development. J. R. Soc. Interface 2014, 11, 20140149.
- Gill, J.S.; Salafia, C.M.; Grebenkov, D.; Vvedensky, D.D. Modeling Oxygen Transport in Human Placental Terminal Villi. J. Theor. Biol. 2011, 291, 33–41.
- Plitman Mayo, R.; Olsthoorn, J.; Charnock-Jones, D.S.; Burton, G.J.; Oyen, M.L. Computational Modeling of the Structure-Function Relationship in Human Placental Terminal Villi. J. Biomech. 2016, 49, 3780–3787.
- Plitman Mayo, R.; Charnock-Jones, D.S.; Burton, G.J.; Oyen, M.L. Three-Dimensional Modeling of Human Placental Terminal Villi. Placenta 2016, 43, 54–60.
- Pearce, P.; Brownbill, P.; Janáček, J.; Jirkovská, M.; Kubínová, L.; Chernyavsky, I.L.; Jensen, O.E. Image-Based Modeling of Blood Flow and Oxygen Transfer in Feto-Placental Capillaries. PLoS ONE 2016, 11, e0165369.
- Erlich, A.; Pearce, P.; Mayo, R.P.; Jensen, O.E.; Chernyavsky, I.L. Physical and Geometric Determinants of Transport in Feto-Placental Microvascular Networks. Sci. Adv. 2019, 5, eaav6326.
- Plitman Mayo, R. Advances in Human Placental Biomechanics. Comput. Struct. Biotechnol. J. 2018, 16, 298–306.
- De Sousa Mendes, M.; Lui, G.; Zheng, Y.; Pressiat, C.; Hirt, D.; Valade, E.; Bouazza, N.; Foissac, F.; Blanche, S.; Treluyer, J.M.; et al. A Physiologically-Based Pharmacokinetic Model to Predict Human Fetal Exposure for a Drug Metabolized by Several CYP450 Pathways. Clin. Pharmacokinet. 2017, 56, 537–550.
- Abduljalil, K.; Johnson, T.N.; Jamei, M. Application of Feto-Maternal Physiologically-Based Pharmacokinetic Model To Predict Emtricitabine Concentration during Pregnancy. J. Pharmacokinet. Pharmacodyn. 2018, 45, S76.
- Schalkwijk, S.; Buaben, A.O.; Freriksen, J.J.M.; Colbers, A.P.; Burger, D.M.; Greupink, R.; Russel, F.G.M. Prediction of Fetal Darunavir Exposure by Integrating Human Ex-Vivo Placental Transfer and Physiologically Based Pharmacokinetic Modeling. Clin. Pharmacokinet. 2018, 57, 705–716.
- Zhang, Z.; Imperial, M.Z.; Patilea-Vrana, G.I.; Wedagedera, J.; Gaohua, L.; Unadkat, J.D. Development of a Novel Maternal-Fetal Physiologically Based Pharmacokinetic Model I: Insights into Factors That Determine Fetal Drug Exposure through Simulations and Sensitivity Analyses. Drug Metab. Dispos. 2017, 45, 920–938.





