
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Lok Shrestha | + 2573 word(s) | 2573 | 2021-08-03 05:44:27 | | | |
| 2 | Amina Yu | Meta information modification | 2573 | 2021-08-12 10:12:32 | | |
Video Upload Options
As generally known dimensionality of materials is a crucial factor to determine functions and properties of the materials. In addition to zero-dimensional, one-dimensional, three-dimensional, and further integrated functional materials, various two-dimensional materials have been paid special attention. Two-dimensional materials have their unique electronic propertiesand play important roles in interfacial sciences.
1. Introduction
As a game-changer of science and technology in the late 20th to 21st century, nanotechnology has great contributions in explorations on nanoscale materials and their phenomena. Developments in nanotechnology-based observation/evaluation techniques [1][2][3] and manipulation methods even in atom/molecular scales [4][5][6] open ways to control nanoscale structures and create nano-specific functions such as highly anisotropic electronic and optical properties. These research trends make us realize the importance of nanoscale structures in material functions. In parallel, basic areas and emerging fields for material production have continuously progressed. Functional materials in huge varieties have been prepared and fabricated through efforts in organic synthesis [7][8][9][10], polymer chemistry [11][12][13], coordination chemistry [14][15][16][17], supramolecular chemistry [18][19][20][21], and the other materials science especially for nanostructured materials [22][23][24][25]. Various fabrication techniques including the self-assembled monolayer (SAM) method [26][27][28], Langmuir–Blodgett (LB) technique [29][30], and layer-by-layer (LbL) assembly [ 31,32] have indispensable roles in the developments of functional materials as well as contributions with emerging materials such as nanocarbon materials [31][32] and nanoporous materials [33][34].
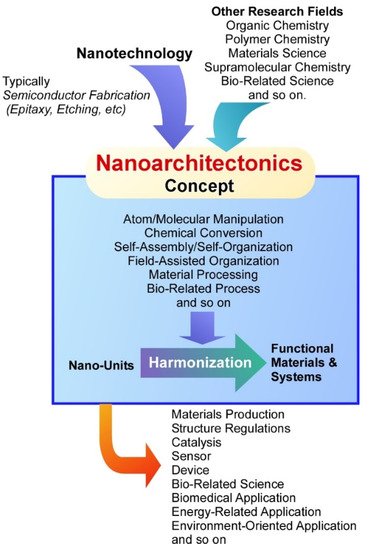
On the basis of these developments of science and technology, various functional materials and systems have been continuously produced upon important social demands in energy [35][36][37][38], environmental [39][40][41][42], and biomedical fields [43][44][45][46]. It becomes realized that regulation of nanostructures within materials systems is crucially important for getting better performances with higher efficiencies. Interactions in well-coordinated components leads to efficient processes in many events including conductivity, reactivity, adhesion, and optical response. Both materials’ intrinsic qualities and their precise internal structures are indispensable factors to produce more desirable materials for target functions. Therefore, knowledge and techniques in nanotechnology and the other research fields have to be combined into a unified new paradigm. This task is assigned to an emerging concept, nanoarchitectonics [47].
As the nanotechnology concept is said to be initiated by a lecture by Richard Feynman [48][49], nanoarchitectonics concept in this meaning was first proposed by Masakazu Aono [50][51] at the conference held in 2000. Nanoarchitectonics unifies nanotechnology with other research fields such as organic chemistry, polymer chemistry, materials science, supramolecular chemistry, and bio-related science [52] ( Figure 1 ). Based on the nanoarchitectonics strategies, functional material systems can be produced from nanoscale unit components through the combinations and selections of various effects and the processes including atom/molecular manipulation, chemical conversion, self-assembly/self-organization, field-assisted organization, material processing, and bio-related process [53]. Because molecular and material interactions at the nanoscale cannot ignore the influences of various ambiguities such as thermal fluctuations, statistical distributions, and quantum effects, nanoarchitectonics fabrications are often based on the harmonization of unit processes rather than their simple summation [54]. In many cases of functional materials production based on nanoarchitectonics procedures, several processes are used together and sequential assembly and fabrications are applied. Therefore, the nanoarchitectonics processes are advantageous to produce hierarchical and asymmetrical structures rather than equilibrated self-assembly often used in supramolecular chemistry [55]. These features can be commonly applied to a wide range of materials. Therefore, the nanoarchitectonics concept has been applied in various research fields including materials production [56][57], structure regulations [58][59], catalysis [60][61], sensor [62][63], device [64][65], bio-related science [66][67][68], biomedical applications [69][70], energy-related applications [71][72], and environment-oriented applications [73][74].
As generally known dimensionality of materials is a crucial factor to determine functions and properties of the materials. In addition to zero-dimensional [75][76][77], one-dimensional [78][79][80], three-dimensional [81][82][83], and further integrated functional materials [84][85], various two-dimensional materials have been paid special attention [86][87][88]. Two-dimensional materials have their unique electronic properties [89][90] and play important roles in interfacial sciences [91][92]. As typically seen in graphene as a representative two-dimensional material, many attractive properties such as optical transparency, mechanical strength, thermal conductivity, and carrier mobility are highly expected. These features can be potentially used for device applications. Unlike zero-dimensional and one-dimensional materials, two-dimensional materials can efficiently form interfacial environments that are useful as interface for devices such as sensors. The method of synthesis has strong influence on these properties. Therefore, two-dimensional materials are selected as subjects of nanoarchitectonics research in this review article.
2. Two-Dimensional Fullerene Nanoarchitecture
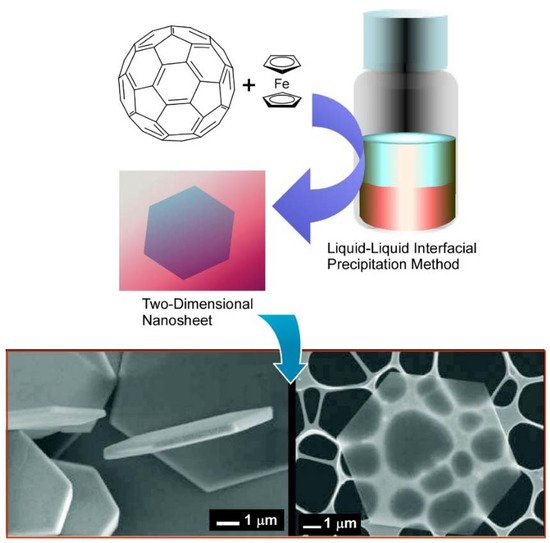
For the preparation of self-assembled fullerene nanocrystals, a simple liquid–liquid interface precipitation method is often used [93][94]. In the liquid–liquid interface precipitation method, the crystal formation mechanism is supposed to be driven by supersaturation of fullerene molecules at interfaces between good solvent (with higher fullerene solubility) and bad solvent (with lower fullerene solubility). Although the preparation procedure of this technique is very simple, fullerene assemblies in various dimensions—including one-dimensional rods /tubes [95][96], three-dimensional cubes [97][98], and their hierarchical nanoarchitectures [99][100]—can be obtained. For example, Sathish and Miyazawa synthesized size-tunable C 60 hexagonal thin crystalline nanosheets through the liquid–liquid interface precipitation method at the carbon tetrachloride (CCl 4)/alcohol interface for the first time [101]. The size of the hexagonal nanosheets could be tailored merely by selecting proper alcohol for the interfacial precipitation. Hexagonal nanosheet with ~7.5 μm diameter was observed for the CCl 4/isopropyl alcohol interface, whereas the hexagonal nanosheets with ~2.5 μm diameter and 500 nm diameter at the CCl 4/ethanol and CCl 4/methanol interface, respectively. This work opens a new door for synthesizing fullerene nanosheets by simple self-assembly.
It was demonstrated in further studies that variations in the shape and size of two-dimensional fullerene assemblies can be fabricated with the appropriate choice of solvent combinations for the liquid–liquid interfacial precipitation method ( Figure 2 A) [102]. Formation of hexagonal C 60 nanosheets was achieved at the interfaces between toluene and isopropyl alcohol, while two-dimensional nanosheets with uniform rhombi and hexagonal shapes were obtained through the self-assembly of C 60 molecules at the interfaces of toluene/ tert -butyl alcohol and CCl 4/isopropyl alcohol, respectively. The lattice formation favoring these two-dimensional crystalline assemblies may be the most influential factor in determining the shape of the two-dimensional nanosheets. Under certain stimuli, such as contact with water, two-dimensional rhombi-shaped nanosheets were transformed into one-dimensional nanorods. The latter transformation would be occurred accompanied by the release of high surface energy.
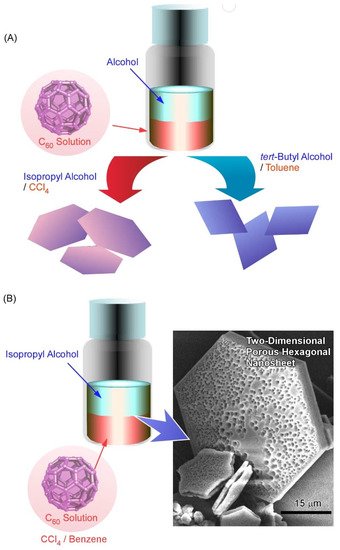
Pore formation within a two-dimensional C 60 nanosheet was achieved by faint adaptations of solvent systems to the liquid–liquid interfacial precipitation system [103]. Two-dimensional nanosheets possessing dimorphic macroporous/interporous structures were fabricated via C 60 assembly at interfaces between isopropyl alcohol and CCl 4/benzene mixtures ( Figure 2 B). At high CCl 4 contents, macropores were formed in the two-dimensional hexagonal nanosheets. In addition, smaller mesopores were also formed within the macropores under suitable mixing conditions between CCl 4 and benzene. The average mesopore sizes came to be 15 to 25 nm and 20 to 40 nm at CCl 4/benzene mixing ratios of 80:20 and 90:10, respectively. The latter two-dimensional structures with bimodal features were accompanied by mixed fcc and hcp crystal structures. It is expected that the evaporation of solvents trapped in them would be the cause of mesopore formation. The formed bimodal nanoarchitectures have high surface areas with enhanced electrochemical activities, which would be useful for energy-related applications.
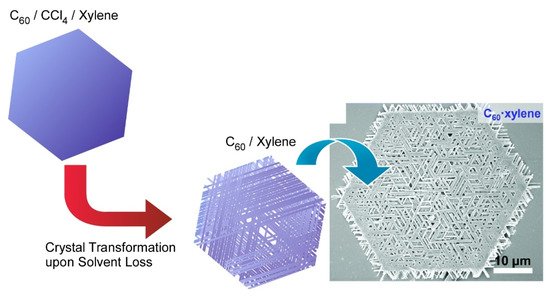
Chen and co-workers developed a simple but effective co-solvent induced crystal transformation strategy to transform two-dimensional C 60 microplates to nano-meshes made of ordered nanorods ( Figure 3 ) [104]. The transformation was triggered by removing the solvent molecule and then re-incorporating another solvent molecule. Specifically, C 60 plates were obtained first by adding isopropyl alcohol to the CCl 4/ m -xylene mixture with gentle swirling. The C 60 plates were concentrated by centrifugation and then diluted with isopropyl alcohol to transform to two-dimensional mesh networks. After diluting with isopropyl alcohol, the incorporated CCl 4/ m -xylene would then leave the C 60 plate, leading to the formation of pores in the C 60 microplates. Under appropriate conditions, m -xylene can be re-incorporated during the C 60 recrystallization and eventually gives rise to the mesh network. This work opens a new field for designing hierarchical two-dimensional superstructures. The fabricated complicated two-dimensional mesh-like fullerene superstructures would be useful separation and filtration such as oil/water separation and biomolecular segregations.
3. Two-Dimensional Nanoarchitecture from Fullerene with Other Component
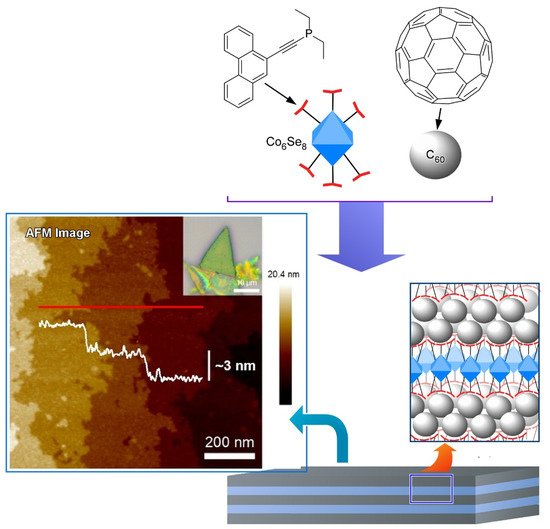
Steigerwald, Nuckolls, Roy, and coworkers proposed a method that utilizes dimensionally confined fullerene as a strippable layer to obtain two-dimensional fullerene ( Figure 4 ) [105]. They firstly synthesized a layered van der Waals solid from a structure-directing building block and C 60 fullerene by self-assembly method. Transition metal chalcogenide cluster ( Co 6Se 8(PEt 2phen) 6) was employed as the structure-directing building block that associated with C 60 and thereby directed the spontaneous assembly of C 60 into crystalline solid of [ Co 6Se 8(PEt 2phen) 6][C 60] 5. The structure of [Co 6Se 8(PEt 2phen) 6][C 60] 5 is composed of two layers stacked along the hexagonal c -axis in which a slightly corrugated trigonal array of C 60 was separated by a layer comprised of a molecular cluster with stoichiometry [ Co 6Se 8(PEt 2phen) 6][C 60] 2. The intralayer bonding of [ Co 6Se 8(PEt 2phen) 6][C 60] 5 is strong enough while interlayer bonding is weak enough that the layered solid may be mechanically exfoliated. Therefore, they exfoliated the layered crystals to produce two-dimensional sheets as thin as ~130 nm with smooth surfaces and transferred them onto Si wafers. In this example, flexible molecular recognition between fullerene and ligand units enables us to nanoarchitect van der Waals materials from multiple components in desired layered structures.
Wakahara, Tateyama, and coworkers prepared C 60/ferrocene nanosheets by a simple liquid–liquid interfacial precipitation method through the formation of an interface between isopropyl alcohol and toluene solution containing C 60 and ferrocene ( Figure 6 ) [106]. A strong charge-transfer band of diffuse reflectance spectra between ferrocene and C 60 was observed at 782 nm, indicating the presence of donor−acceptor interaction in the nanosheets. The average size of the C 60/ ferrocene hexagonal nanosheets was 9.1 ± 6.2 μm and the thickness is about 250−550 nm. Size would be controlled by conditions at the liquid–liquid interfacial precipitation method. In the case of C 60/ferrocene nanosheets, each C 60 is surrounded by two ferrocene molecules, showing a triclinic C 60(ferrocene) 2 structure. Upon heating the nanosheets to 150 °C to sublimate ferrocene, the C 60/ferrocene hexagonal nanosheets can be converted to fcc C 60 hexagonal nanosheets with the same shape and size as the C 60/ferrocene nanosheets. The successful preparation of C 60/ferrocene nanosheets based on the co-crystallization in the liquid–liquid interface is expected to be an important steppingstone to the fabrication of novel fullerene nanosheets and to lead to a new field relative to fullerene-derived co-crystals engineering.
In addition to synthesizing C 60/ferrocene hexagonal nanosheets co-crystals nanosheets, Wakahara, Bradley, Anthopoulos, and coworkers also reported the preparation of novel co-crystals nanosheets comprising C 60 and 5,10,15,20-tetrakis(4-methoxyphenyl) porphyrinato cobalt(II) (CoTMPP) using liquid–liquid interfacial precipitation method [107]. The hybrid nanosheets were formed at an interface between isopropyl alcohol and toluene solution containing C 60/CoTMPP. The thickness of the nanosheets is ~50–200 nm. The C 60/CoTMPP co-crystals nanosheets exhibited ambipolar transport characteristics with nearly balanced hole/electron mobilities. The successful preparation of fullerene-derived co-crystals nanosheets by using the liquid–liquid interfacial precipitation method to co-crystallization would enrich the fabrication of two-dimensional fullerene.
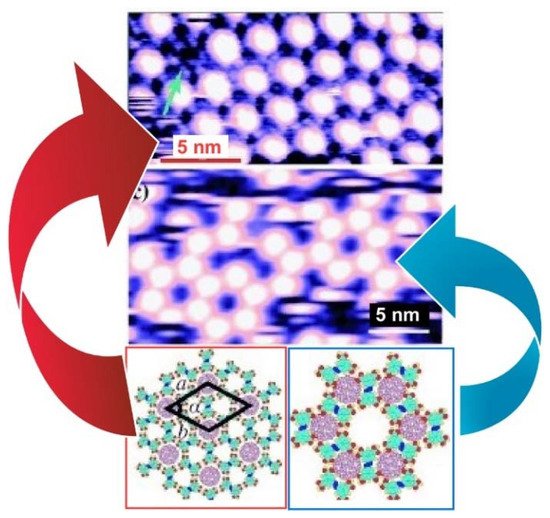
Zeng, Wang, and coworkers reported a two-dimensional molecular template with two types of cavities that have different sizes and symmetry at the liquid–solid interface formed by an azobenzene derivative (NN4A) [108] ( Figure 7 ). Both the A- (diameter: 12.0 Å) and B-type (diameter: 8.6 Å) cavities were large enough in diameter to accommodate guest molecules such as fullerene molecules. Therefore, highly ordered fullerene arrays were constructed at the liquid–solid interface of graphite by coadsorption of fullerenes and NN4A. The experimental and theoretical results indicate that C 60 molecules (diameter:6.80 Å) occupy both types of the cavity, while the larger C 80 (diameter: 8.22 Å) and Sc 3N@C 80 molecules were trapped exclusively in the A-type cavities, which implies appreciable site selectivity. These template-directed fabrications of fullerene arrays provide a new method for the formation of two-dimensional fullerene.
4. Two-Dimensional Hierarchical Fullerene Nanoarchitecture from Fullerene
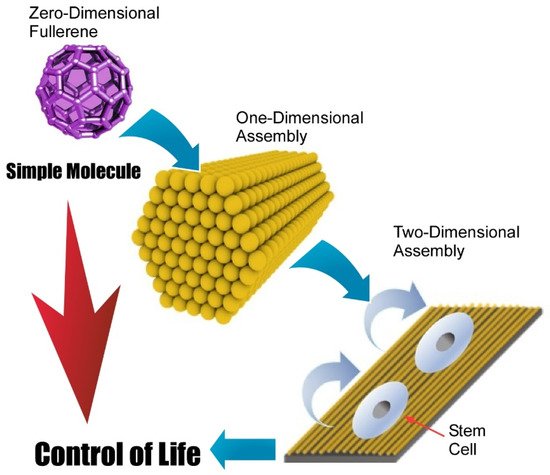
Fabrication methods of two-dimensional structures from zero-dimensional fullerene molecules are not limited to direct zero-to-two dimensional nanoarchitectonics. A multi-phase strategy such as zero-to-one to two nanoarchitectonics is also possible. In these cases, hierarchical two-dimensional structures are often obtainable. Minami et al. proposed the fabrication of two-dimensional films of one-dimensional fullerene (C 60) nanowhiskers that were used as a scaffold for living cells [109]. In the initial step, zero-dimensional C 60 molecules were converted into one-dimensional fullerene nanowhiskers through the liquid–liquid interfacial precipitation method using toluene as a good solvent and isopropyl alcohol as a poor solvent. The obtained one-dimensional fullerene nanowhiskers were then assembled and aligned at the air–water interface and transferred as a two-dimensional film onto a solid substrate through the conventional LB method. This nanoarchitectonics method provides hierarchical structures of two-dimensional structures in a centimeter area with highly aligned one-dimensional fullerene nanowhiskers. The resulting structures were subjected to cell cultures to control cellular orientation and differentiation to muscle cells. Because of the simplicity and low-cost nature of the proposed approach, this hierarchical nanoarchitectonics strategy would be a promising cell scaffold for tissue engineering.
Krishnan et al. demonstrated a modified method to modulate aligning curvature of one-dimensional fullerene ( C 60) nanowhiskers within the two-dimensional film. Instead of using a conventional LB technique, a newly developed vortex LB method was employed for this purpose [110]. The C 60 nanowhiskers floated on a water surface were spontaneously assembled according to rotational flows of water subphase. Their alignment curvatures can be controlled at the position from a center of rotating motions. The aligned fullerene nanowhiskers with controlled alignment curvatures were then transferred onto glass substrates used for the culture of bone-forming human osteoblast MG63 cells. It was observed that the growth of the MG63 cells was highly oriented with an axis of aligned one-dimensional C 60 nanowhiskers. Cell proliferation experiments confirmed the low toxicity nature of the used hierarchical two-dimensional fullerene scaffold, suggesting the potential availability of the proposed method in various biomedical applications.
Very recently, Song et al. investigated the regulation of adhesion and function of human mesenchymal stem cells on two-dimensional fullerene (C 60) nanowhisker nanopatterns with controlled alignments that were prepared with conventional LB technique ( Figure 8 ) [111]. It was found that regenerative capacity and long-term multipotency were enhanced on two-dimensional nanopatterns with highly aligned fullerene nanowhisker arrays. Appropriately architected nanotopographic surface with certain hydrophobicity gave limited contact area between material surface and cells. This situation induced decreased cell spreading and focal adhesion areas of human mesenchymal stem cells. Enhancement of stemness retention of human mesenchymal stem cells would be related to the diminished cytoskeletal tensions. Regulated contact with surfaces resulted in the appropriate cell contractility with localization of Yes-associated proteins in the nucleus. The proposed strategy would be useful in biomedical applications, such as tissue engineering as a platform for in vitro stem cell expansion. This example demonstrates important roles of two-dimensional assembly to bridge single molecules and life control.
References
- Shimizu, T.; Lungerich, D.; Stuckner, J.; Murayama, M.; Harano, K.; Nakamura, E. Real-time video imaging of mechanical motions of a single molecular shuttle with sub-millisecond sub-angstrom precision. Bull. Chem. Soc. Jpn. 2020, 93, 1079–1085.
- Kazuma, E. Real-space studies of plasmon-induced dissociation reactions with an STM. Bull. Chem. Soc. Jpn. 2020, 93, 1552–1557.
- Kamei, K.; Shimizu, T.; Harano, K.; Nakamura, E. Aryl radical addition to curvatures of carbon nanohorns for single-molecule-level molecular imaging. Bull. Chem. Soc. Jpn. 2020, 93, 1603–1608.
- Kaur, G.; Lewis, J.S.; van Oijen, A.M. Shining a spotlight on DNA: Single-molecule methods to visualise DNA. Molecules 2019, 24, 491.
- Hishikawa, A.; Matsuda, A.; Fushitani, M. Ultrafast reaction imaging and control by ultrashort intense laser pulses. Bull. Chem. Soc. Jpn. 2020, 93, 1293–1304.
- Muraoka, T. Biofunctional molecules inspired by protein mimicry and manipulation. Bull. Chem. Soc. Jpn. 2020, 93, 138–153.
- Sun, Z.; Ikemoto, K.; Fukunaga, T.M.; Koretsune, T.; Arita, R.; Sato, S.; Isobe, H. Finite phenine nanotubes with periodic vacancy defects. Science 2019, 363, 151–155.
- Niwa, T.; Hosoya, T. Molecular renovation strategy for expeditious synthesis of molecular probes. Bull. Chem. Soc. Jpn. 2020, 93, 230–248.
- Muramatsu, W.; Hattori, T.; Yamamoto, H. Game change from reagent- to substrate-controlled peptide synthesis. Bull. Chem. Soc. Jpn. 2020, 93, 759–767.
- Yamada, H.; Kuzuhara, D.; Suzuki, M.; Hayashi, H.; Aratani, N. Synthesis and morphological control of organic semiconducting materials using the precursor approach. Bull. Chem. Soc. Jpn. 2020, 93, 1234–1267.
- Akagi, K. Interdisciplinary chemistry based on integration of liquid crystals and conjugated polymers: Development and progress. Bull. Chem. Soc. Jpn. 2019, 92, 1509–1655.
- Tanaka, M.; Kobayashi, S.; Murakami, D.; Aratsu, F.; Kashiwazaki, A.; Hoshiba, T.; Fukushima, K. Design of polymeric biomaterials: The “intermediate water concept”. Bull. Chem. Soc. Jpn. 2019, 92, 2043–2057.
- Yamago, S. Photoactivation of organotellurium compounds in precision polymer synthesis: Controlled radical polymerization and radical coupling reactions. Bull. Chem. Soc. Jpn. 2020, 93, 287–298.
- Mashima, K. Redox-active α-diimine complexes of early transition metals: From bonding to catalysis. Bull. Chem. Soc. Jpn. 2020, 93, 799–820.
- Hosono, N. Design of porous coordination materials with dynamic properties. Bull. Chem. Soc. Jpn. 2021, 94, 60–69.
- Domoto, Y.; Abe, M.; Yamamoto, K.; Kikuchi, T.; Fujita, M. “Eggs in egg cartons”: Co-crystallization to embed molecular cages into crystalline lattices. Chem. Sci. 2020, 11, 10457–10460.
- Yamashita, M. Next generation multifunctional nano-science of advanced metal complexes with quantum effect and nonlinearity. Bull. Chem. Soc. Jpn. 2021, 94, 209–264.
- Fukunaga, K.; Tsutsumi, H.; Mihara, H. Self-assembling peptides as building blocks of functional materials for biomedical applications. Bull. Chem. Soc. Jpn. 2019, 92, 391–399.
- Roy, B.; Govindaraju, T. Amino acids and peptides as functional components in arylenediimide-based molecular architectonics. Bull. Chem. Soc. Jpn. 2019, 92, 1883–1901.
- Datta, S.; Kato, Y.; Higashiharaguchi, S.; Aratsu, K.; Isobe, A.; Saito, T.; Prabhu, D.D.; Kitamoto, Y.; Hollamby, M.J.; Smith, A.J.; et al. Self-assembled poly-catenanes from supramolecular toroidal building blocks. Nature 2020, 583, 400–405.
- Percec, V.; Xiao, Q. Helical self-organizations and emerging functions in architectures, biological and synthetic macromolecules. Bull. Chem. Soc. Jpn. 2021, 94, 900–928.
- Wang, X.-B.; Jiang, X.-F.; Bando, Y. Blowing route towards advanced inorganic foams. Bull. Chem. Soc. Jpn. 2019, 92, 245–263.
- Pileni, M.P. Au supracrystal growth processes: Unexpected morphologies. Bull. Chem. Soc. Jpn. 2019, 92, 312–329.
- Kanao, E.; Kubo, T.; Otsuka, K. Carbon-based nanomaterials for separation media. Bull. Chem. Soc. Jpn. 2020, 93, 482–489.
- Li, M.-T.; Liu, M.; Yu, Y.-H.; Li, A.-W.; Sun, H.-B. Laser-structured graphene/reduced graphene oxide films towards bio-inspired superhydrophobic surfaces. Bull. Chem. Soc. Jpn. 2019, 92, 283–289.
- Kise, R.; Fukumi, A.; Shioya, N.; Shimoaka, T.; Sonoyama, M.; Amii, H.; Takagi, T.; Kanamori, T.; Eda, K.; Hasegawa, T. Fluorous property of a short perfluoroalkyl-containing compound realized by self-assembled monolayer technique on a silicon substrate. Bull. Chem. Soc. Jpn. 2019, 92, 785–789.
- Akiyama, T. Development of fullerene thin-film assemblies and fullerene-diamine adducts towards practical nanocarbon-based electronic materials. Bull. Chem. Soc. Jpn. 2019, 92, 1181–1199.
- Kuodis, Z.; Matulaitienė, I.; Špandyreva, M.; Labanauskas, L.; Stončius, S.; Eicher-Lorka, O.; Sadzevičienė, R.; Niaura, G. Reflection absorption iInfrared spectroscopy characterization of SAM formation from 8-mercapto-N-(phenethyl)octanamide thiols with Phe ring and amide groups. Molecules 2020, 25, 5633.
- Ariga, K.; Mori, T.; Li, J. Langmuir nanoarchitectonics from basic to frontier. Langmuir 2019, 35, 3585–3599.
- Ariga, K. Don’t forget Langmuir–Blodgett films 2020: Interfacial nanoarchitectonics with molecules, materials, and living objects. Langmuir 2020, 36, 7158–7180.
- Nakanishi, W.; Minami, K.; Shrestha, L.K.; Ji, Q.; Hill, J.P.; Ariga, K. Bioactive nanocarbon assemblies: Nanoarchitectonics and applications. Nano Today 2014, 9, 378–394.
- Sugimoto, Y.; Irisawa, T.; Hatori, H.; Inagaki, M. Yarns of carbon nanotubes and reduced graphene oxides. Carbon 2020, 165, 358–377.
- Mao, Y.; Zhang, Y.; Hu, W.; Ye, W. Carbon dots-modified nanoporous membrane and Fe3O4@Au magnet nanocomposites-based FRET assay for ultrasensitive histamine detection. Molecules 2019, 24, 3039.
- Kim, G.; Shiraki, T.; Fujigaya, T. Thermal conversion of triazine-based covalent organic frameworks to nitrogen-doped nanoporous carbons and their capacitor performance. Bull. Chem. Soc. Jpn. 2020, 93, 414–420.
- Guo, D.; Shibuya, R.; Akiba, C.; Saji, S.; Kondo, T.; Nakamura, J. Active sites of nitrogen-doped carbon materials for oxygen reduction reaction clarified using model catalysts. Science 2016, 351, 361–365.
- Roy, N.; Suzuki, N.; Terashima, C.; Fujishima, A. Recent improvements in the production of solar fuels: From CO2 reduction to water splitting and artificial photosynthesis. Bull. Chem. Soc. Jpn. 2019, 92, 178–192.
- Yamada, Y. Concentrated battery electrolytes: Developing new functions by manipulating the coordination states. Bull. Chem. Soc. Jpn. 2020, 93, 109–118.
- Li, Y.; Henzie, J.; Park, T.; Wang, J.; Young, C.; Xie, H.; Yi, J.W.; Li, J.; Kim, M.; Kim, J.; et al. Fabrication of flexible microsupercapacitors with binder-free ZIF-8 derived carbon films via electrophoretic deposition. Bull. Chem. Soc. Jpn. 2020, 93, 176–181.
- Sai-Anand, G.; Sivanesan, A.; Benzigar, M.R.; Singh, G.; Gopalan, A.-I.; Baskar, A.V.; Ilbeygi, H.; Ramadass, K.; Kambala, V.; Vinu, A. Recent progress on the sensing of pathogenic bacteria using advanced nanostructures. Bull. Chem. Soc. Jpn. 2019, 92, 216–244.
- Pang, P.; Lai, Y.; Zhang, Y.; Wang, H.; Conlan, X.A.; Barrow, C.J.; Yang, W. Recent advancement of biosensor technology for the detection of microcystin-LR. Bull. Chem. Soc. Jpn. 2020, 93, 637–646.
- Zhang, N.; Ye, C.; Yan, H.; Li, L.; He, H.; Wang, D.; Li, Y. Single-atom site catalysts for environmental catalysis. Nano Res. 2020, 13, 3165–3182.
- Singh, B.; Na, J.; Konarova, M.; Wakihara, T.; Yamauchi, Y.; Salomon, C.; Gawande, M.B. Functional mesoporous silica nanomaterials for catalysis and environmental applications. Bull. Chem. Soc. Jpn. 2020, 93, 1459–1496.
- Kobayashi, J.; Okano, T. Design of temperature-responsive polymer-grafted surfaces for cell sheet preparation and manipulation. Bull. Chem. Soc. Jpn. 2019, 92, 817–824.
- Paris, J.I.; Vallet-Regí, M. Ultrasound-activated nanomaterials for therapeutics. Bull. Chem. Soc. Jpn. 2020, 93, 220–229.
- Kankala, R.K.; Han, Y.; Na, J.; Lee, C.; Sun, Z.; Wang, S.; Kimura, T.; Ok, Y.S.; Yamauchi, Y.; Chen, A.; et al. Nanoarchitectured structure and surface biofunctionality of mesoporous silica nanoparticles. Adv. Mater. 2020, 32, 1907035.
- Arora, H.; Ramesh, M.; Rajasekhar, K.; Govindaraju, T. Molecular tools to detect alloforms of Aβ and Tau: Implications for multiplexing and multimodal diagnosis of Alzheimer’s disease. Bull. Chem. Soc. Jpn. 2020, 93, 507–546.
- Ariga, K.; Ji, Q.; Nakanishi, W.; Hill, J.P.; Aono, M. Nanoarchitectonics: A new materials horizon for nanotechnology. Mater. Horiz. 2015, 2, 406–413.
- Feynman, R.P. There’s plenty of room at the bottom. Eng. Sci. 1960, 23, 22–36.
- Roukes, M. Plenty of room, indeed. Sci. Am. 2001, 285, 48–51.
- Ariga, K.; Ji, Q.; Hill, J.P.; Bando, Y.; Aono, M. Forming nanomaterials as layered functional structures toward materials nanoarchitectonics. NPG Asia Mater. 2012, 4, e17.
- Ariga, K. Nanoarchitectonics revolution and evolution: From small science to big technology. Small Sci. 2021, 1, 2000032.
- Ariga, K.; Li, J.; Fei, J.; Ji, Q.; Hill, J.P. Nanoarchitectonics for dynamic functional materials from atomic-/molecular-level manipulation to macroscopic action. Adv. Mater. 2016, 28, 1251–1286.
- Ariga, K. Nanoarchitectonics: What’s coming next after nanotechnology? Nanoscale Horiz. 2021, 6, 364–378.
- Aono, M.; Ariga, K. The way to nanoarchitectonics and the way of nanoarchitectonics. Adv. Mater. 2015, 28, 989–992.
- Ariga, K.; Jia, X.; Song, J.; Hill, J.P.; Leong, D.T.; Jia, Y.; Li, J. Nanoarchitectonics beyond self-assembly: Challenges to create bio-like hierarchic organization. Angew. Chem. Int. Ed. 2020, 59, 15424–15446.
- Azhar, A.; Zakaria, M.B.; Kim, J.; Na, J.; Kaneti, Y.V.; Fatehmulla, A.; Aldhafiri, A.M.; Farooq, W.A.; Bando, Y.; Yamauchi, Y.; et al. Single crystal growth of two-dimensional cyano-bridged coordination polymer of Co(H2O)2Ni(CN)4·4H2O using trisodium citrate dihydrate. Bull. Chem. Soc. Jpn. 2019, 92, 1263–1267.
- Ariga, K.; Shionoya, M. Nanoarchitectonics for coordination asymmetry and related chemistry. Bull. Chem. Soc. Jpn. 2021, 94, 839–859.
- Dutta, S.; Kim, J.; Hsieh, P.-H.; Hsu, Y.-S.; Kaneti, Y.V.; Shieh, F.-K.; Yamauchi, Y.; Wu, K.C.-W. Nanoarchitectonics of biofunctionalized metal-organic frameworks with biological macromolecules and living cells. Small Methods 2019, 3, 1900213.
- Ariga, K.; Mori, T.; Kitao, T.; Uemura, T. Supramolecular chiral nanoarchitectonics. Adv. Mater. 2020, 32, 1905657.
- Deka, R.C.; Deka, A.; Deka, P.; Saikia, S.; Baruah, J.; Sarma, P.J. Recent advances in nanoarchitectonics of SnO₂ clusters and their applications in catalysis. J. Nanosci. Nanotechnol. 2020, 20, 5153–5161.
- Datta, K.K.R.; Madakannu, I.; Prasanthi, I. Hetero atom doped graphene nanoarchitectonics as electrocatalysts towards the oxygen reduction and evolution reactions in acidic medium. J. Inorg. Organomet. Polym. 2021, 31, 1859–1876.
- Ishihara, S.; Labuta, J.; Van Rossom, W.; Ishikawa, D.; Minami, K.; Hill, J.P.; Ariga, K. Porphyrin-based sensor nanoarchitectonics in diverse physical detection modes. Phys. Chem. Chem. Phys. 2014, 16, 9713–9746.
- Komiyama, M.; Mori, T.; Ariga, K. Molecular imprinting: Materials nanoarchitectonics with molecular information. Bull. Chem. Soc. Jpn. 2018, 91, 1075–1111.
- Giussi, J.M.; Cortez, M.L.; Marmisollé, W.A.; Azzaroni, O. Practical use of polymer brushes in sustainable energy applications: Interfacial nanoarchitectonics for high-efficiency devices. Chem. Soc. Rev. 2019, 48, 814–849.
- Ariga, K.; Ito, M.; Mori, T.; Watanabe, S.; Takeya, J. Atom/molecular nanoarchitectonics for devices and related applications. Nano Today 2019, 28.
- Ariga, K.; Ji, Q.; Mori, T.; Naito, M.; Yamauchi, Y.; Abe, H.; Hill, J.P. Enzyme nanoarchitectonics: Organization and device application. Chem. Soc. Rev. 2013, 42, 6322–6345.
- Stulz, E. Nanoarchitectonics with porphyrin functionalized DNA. Acc. Chem. Res. 2017, 50, 823–831.
- Liang, X.; Li, L.; Tang, J.; Komiyama, M.; Ariga, K. Dynamism of supramolecular DNA/RNA nanoarchitectonics: From interlocked structures to molecular machines. Bull. Chem. Soc. Jpn. 2020, 93, 581–603.
- Zhao, L.; Zou, Q.; Yan, X. Self-assembling peptide-based nanoarchitectonics. Bull. Chem. Soc. Jpn. 2019, 92, 70–79.
- Banerjee, S.; Pillai, J. Solid lipid matrix mediated nanoarchitectonics for improved oral bioavailability of drug. Expert Opin. Drug Metab. Toxicol. 2019, 15, 499–515.
- Kim, J.; Kim, J.H.; Ariga, K. Redox-active polymers for energy storage nanoarchitectonics. Joule 2017, 1, 739–768.
- Li, Y.; Dang, Q.; Chen, W.; Tang, L.; Hu, M. Recent advances in rechargeable batteries with prussian blue analogs nanoarchitectonics. J. Inorg. Organomet. Polym. 2021, 31, 1877–1893.
- Pandeeswar, M.; Senanayak, S.P.; Govindaraju, T. Nanoarchitectonics of small molecule and DNA for ultrasensitive detection of mercury. ACS Appl. Mater. Interfaces 2016, 8, 30362–30371.
- Boukhalfa, N.; Darder, M.; Boutahala, M.; Aranda, P.; Ruiz-Hitzky, E. Composite nanoarchitectonics: Alginate beads encapsulating sepiolite/magnetite/prussian blue for removal of cesium ions from water. Bull. Chem. Soc. Jpn. 2021, 94, 122–132.
- Imaoka, T.; Yamamoto, K. Wet-chemical strategy for atom-precise metal cluster catalysts. Bull. Chem. Soc. Jpn. 2019, 92, 941–948.
- Nagaura, T.; Park, T.; Lim, H.; Lin, J.; Iqbal, M.; AlShehri, S.M.; Ahamad, T.; Kaneti, Y.V.; Yi, J.W.; Kim, Y.; et al. Controlled synthesis of mesoporous Pt, Pt-Pd and Pt-Pd-Rh nanoparticles in aqueous nonionic surfactant solution. Bull. Chem. Soc. Jpn. 2020, 93, 455–460.
- Yoshida, H.; Kawakami, Y.; Tokuzumi, W.; Shimokawa, Y.; Hirakawa, T.; Ohyama, J.; Machida, M. Low-temperature NO reduction over Fe-Ni alloy nanoparticles using synergistic effects of Fe and Ni in a catalytic NO-CO-C3H6-O2 reaction. Bull. Chem. Soc. Jpn. 2020, 93, 1050–1055.
- Glotov, A.; Stavitskaya, A.; Chudakov, Y.; Ivanov, E.; Huang, W.; Vinokurov, V.; Zolotukhina, A.; Maximov, A.; Karakhanov, E.; Lvov, Y. Mesoporous metal catalysts templated on clay nanotubes. Bull. Chem. Soc. Jpn. 2019, 92, 61–69.
- Fujigaya, T. Development of thermoelectric conversion materials using carbon nanotube sheets. Bull. Chem. Soc. Jpn. 2019, 92, 400–408.
- Toshimitsu, F.; Ishimaru, W.; Nakashima, N. Individual solubilization behavior of single-walled carbon nanotubes by riboflavin (vitamin B2) in water and its analyses using regression approach and computational simulations. Bull. Chem. Soc. Jpn. 2019, 92, 1679–1683.
- Mizuno, A.; Shuku, Y.; Awaga, K. Recent developments in molecular spin gyroid research. Bull. Chem. Soc. Jpn. 2019, 92, 1068–1093.
- Hippler, M.; Blasco, E.; Qu, J.; Tanaka, M.; Barner-Kowollik, C.; Wegener, M.; Bastmeyer, M. Controlling the shape of 3D microstructures by temperature and light. Nat. Commun. 2019, 10, 232.
- Kageyama, H.; Yajima, T.; Tsujimoto, Y.; Yamamoto, T.; Tassel, C.; Kobayashi, Y. Exploring structures and properties through anion chemistry. Bull. Chem. Soc. Jpn. 2019, 92, 1349–1357.
- Zhao, R.; Liang, Z.; Gao, S.; Yang, C.; Zhu, B.; Zhao, J.; Qu, C.; Zou, R.; Xu, Q. Puffing up energetic metal-organic frameworks to large carbon networks with hierarchical porosity and atomically dispersed metal sites. Angew. Chem. Int. Ed. 2019, 58, 1975–1979.
- Watanabe, Y.; Aiba, Y.; Ariyasu, S.; Abe, S. Molecular design and regulation of metalloenzyme activities through two novel approaches: Ferritin and P450s. Bull. Chem. Soc. Jpn. 2020, 93, 379–392.
- Tan, S.M.; Pumera, M. Two-dimensional materials on the rocks: Positive and negative role of dopants and impurities in electrochemistry. ACS Nano 2019, 13, 2681–2728.
- Maeda, K.; Mallouk, T.E. Two-dimensional metal oxide nanosheets as building blocks for artificial photosynthetic assemblies. Bull. Chem. Soc. Jpn. 2019, 92, 38–54.
- Eom, S.; Choi, G.; Nakamura, H.; Choy, J.-H. 2-Dimensional nanomaterials with imaging and diagnostic functions for nanomedicine; a review. Bull. Chem. Soc. Jpn. 2020, 93, 1–12.
- Rao, C.N.R.; Pramoda, K. Borocarbonitrides, BxCyNz, 2D nanocomposites with novel properties. Bull. Chem. Soc. Jpn. 2019, 92, 441–468.
- Ruan, D.; Fujitsuka, M.; Majima, T. Exfoliated Mo2C nanosheets hybridized on CdS with fast electron transfer for efficient photocatalytic H2 production under visible light irradiation. Appl. Catal. B Environ. 2020, 264, 118541.
- Kahn, E.; Liu, M.; Zhang, T.; Liu, H.; Fujisawa, K.; Bepete, G.; Ajayan, P.M.; Terrones, M. Functional hetero-interfaces in atomically thin materials. Mater. Today 2020, 37, 74–92.
- Hu, Y.; Li, Y.; Cheng, J.; Chen, M.-S.; Fu, W.; Liu, B.; Zhang, M.; Shen, Z. Intercalation of carbon nanosheet into layered TiO2 grain for highly interfacial lithium storage. ACS Appl. Mater. Interfaces 2020, 12, 21709–21719.
- Shrestha, L.K.; Ji, Q.; Mori, T.; Miyazawa, K.; Yamauchi, Y.; Hill, J.P.; Ariga, K. Fullerene nanoarchitectonics: From zero to higher dimensions. Chem. Asian J. 2013, 8, 1662–1679.
- Ariga, K.; Shrestha, L.K. Zero-to-one (or more) nanoarchitectonics: How to produce functional materials from zero-dimensional single-element unit, fullerene. Mater. Adv. 2021, 2, 582–597.
- Minato, J.; Miyazawa, K. Solvated structure of C60 nanowhiskers. Carbon 2005, 43, 2837–2841.
- Shrestha, L.K.; Shrestha, R.G.; Yamauchi, Y.; Hill, J.P.; Nishimura, T.; Miyazawa, K.; Kawai, T.; Okada, S.; Wakabayashi, K.; Ariga, K. Nanoporous carbon tubes from fullerene crystals as the π-electron carbon source. Angew. Chem. Int. Ed. 2015, 54, 951–955.
- Shrestha, L.K.; Sathish, M.; Hill, J.P.; Miyazawa, K.; Tsuruoka, T.; Sanchez-Ballester, N.M.; Honma, I.; Ji, Q.; Ariga, K. Alcohol-induced decomposition of Olmstead’s crystalline Ag(I)-fullerene heteronanostructure yields ‘bucky cubes’. J. Mater. Chem. C 2013, 1, 1174–1181.
- Peng, Z.; Hu, Y.; Wang, J.; Liu, S.; Li, C.; Jiang, Q.; Lu, J.; Zeng, X.; Peng, P.; Li, F.-F. Fullerene-based in situ doping of N and Fe into a 3D cross-like hierarchical carbon composite for high-performance supercapacitors. Adv. Energy Mater. 2019, 9, 1802928.
- Bairi, P.; Minami, K.; Nakanishi, W.; Hill, J.P.; Ariga, K.; Shrestha, L.K. Hierarchically structured fullerene C70 cube for sensing volatile aromatic solvent vapors. ACS Nano 2016, 10, 6631–6637.
- Bairi, P.; Minami, K.; Hill, J.P.; Ariga, K.; Shrestha, L.K. Intentional closing/opening of “hole-in-cube” fullerene crystals with microscopic recognition properties. ACS Nano 2017, 11, 7790–7796.
- Sathish, M.; Miyazawa, K. Size-tunable hexagonal fullerene (C60) nanosheets at the liquid-liquid interface. J. Am. Chem. Soc. 2007, 129, 13816–13817.
- Sathish, M.; Miyazawa, K.; Hill, J.P.; Ariga, K. Solvent engineering for shape-shifter pure fullerene (C60). J. Am. Chem. Soc. 2009, 131, 6372–6373.
- Shrestha, L.K.; Yamauchi, Y.; Hill, J.P.; Miyazawa, K.; Ariga, K. Fullerene crystals with bimodal pore architectures consisting of macropores and mesopores. J. Am. Chem. Soc. 2013, 135, 586–589.
- Lei, Y.; Wang, S.; Lai, Z.; Yao, X.; Zhao, Y.; Zhang, H.; Chen, H. Two-dimensional C60 nano-meshes via crystal transformation. Nanoscale 2019, 11, 8692–8698.
- Choi, B.; Yu, J.; Paley, D.W.; Trinh, M.T.; Paley, M.V.; Karch, J.M.; Crowther, A.C.; Lee, C.-H.; Lalancette, R.A.; Zhu, X.; et al. van der Waals solids from self-assembled nanoscale building blocks. Nano Lett. 2016, 16, 1445–1449.
- Wakahara, T.; Sathish, M.; Miyazawa, K.; Hu, C.; Tateyama, Y.; Nemoto, Y.; Sasaki, T.; Ito, O. Preparation and optical properties of fullerene/ferrocene hybrid hexagonal nanosheets and large-scale production of fullerene hexagonal nanosheets. J. Am. Chem. Soc. 2009, 131, 9940–9944.
- Wakahara, T.; D’Angelo, P.; Miyazawa, K.; Nemoto, Y.; Ito, O.; Tanigaki, N.; Bradley, D.D.C.; Anthopoulos, T.D. Fullerene/cobalt porphyrin hybrid nanosheets with ambipolar charge transporting characteristics. J. Am. Chem. Soc. 2012, 134, 7204–7206.
- Li, M.; Deng, K.; Lei, S.-B.; Yang, Y.-L.; Wang, T.-S.; Shen, Y.-T.; Wang, C.-R.; Zeng, Q.-D.; Wang, C. Site-selective fabrication of two-dimensional fullerene arrays by using a supramolecular template at the liquid-solid interface. Angew. Chem. Int. Ed. 2008, 47, 6717–6721.
- Minami, K.; Kasuya, Y.; Yamazaki, T.; Ji, Q.; Nakanishi, W.; Hill, J.P.; Sakai, H.; Ariga, K. Highly ordered 1D fullerene crystals for concurrent control of macroscopic cellular orientation and differentiation toward large-scale tissue engineering. Adv. Mater. 2015, 27, 4020–4026.
- Krishnan, V.; Kasuya, Y.; Ji, Q.; Sathish, M.; Shrestha, L.K.; Ishihara, S.; Minami, K.; Morita, H.; Yamazaki, T.; Hanagata, N.; et al. Vortex-aligned fullerene nanowhiskers as a scaffold for orienting cell growth. ACS Appl. Mater. Interfaces 2015, 7, 15667–15673.
- Song, J.; Jia, X.; Minami, K.; Hill, J.P.; Nakanishi, J.; Shrestha, L.K.; Ariga, K. Large-area aligned fullerene nanocrystal scaffolds as culture substrates for enhancing mesenchymal stem cell self-renewal and multipotency. ACS Appl. Nano Mater. 2020, 3, 6497–6506.





