Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Da-Jeng Yao | + 2075 word(s) | 2075 | 2021-07-29 03:27:41 | | | |
| 2 | Bruce Ren | -21 word(s) | 2054 | 2021-08-09 03:33:05 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Yao, D. Microfluidics on Assisted Reproductive Technology. Encyclopedia. Available online: https://encyclopedia.pub/entry/12901 (accessed on 09 May 2026).
Yao D. Microfluidics on Assisted Reproductive Technology. Encyclopedia. Available at: https://encyclopedia.pub/entry/12901. Accessed May 09, 2026.
Yao, Da-Jeng. "Microfluidics on Assisted Reproductive Technology" Encyclopedia, https://encyclopedia.pub/entry/12901 (accessed May 09, 2026).
Yao, D. (2021, August 07). Microfluidics on Assisted Reproductive Technology. In Encyclopedia. https://encyclopedia.pub/entry/12901
Yao, Da-Jeng. "Microfluidics on Assisted Reproductive Technology." Encyclopedia. Web. 07 August, 2021.
Copy Citation
Infertility is a state of the male or female reproductive system that is defined as the failure to achieve pregnancy even after 12 or more months of regular unprotected sexual intercourse. Assisted reproductive technology (ART) plays a crucial role in addressing infertility. Various ART are now available for infertile couples. Fertilization in vitro (IVF), intracytoplasmic sperm injection (ICSI) and intrauterine insemination (IUI) are the most common techniques in this regard. Various microfluidic technologies can incorporate various ART procedures such as embryo and gamete (sperm and oocyte) analysis, sorting, manipulation, culture and monitoring.
droplet microfluidics
assisted reproductive technology
fertilization in vitro
sperm and oocyte
embryo culture
1. Introduction
About 7% of couples worldwide suffer from infertility conditions [1]. Assisted reproductive technology (ART) includes all treatments and medical procedures that include the handling of eggs and embryos to address infertility. Through ART, part of infertility in women can be resolved by manual processing. In recent reproductive medicine technology, various methods are available for infertile couples. ART can increase the genetic quality of livestock, produce transgenic animals, assisting in cloning and artificial insemination, reduce disease transmission, preserve endangered germplasm, produce chimeric animals for disease research and treat infertility. It can improve fertility through artificial treatments, including fertilization in vitro (IVF) [2], gamete intra-fallopian transfer (GIFT), pronuclear-stage tubal transfer (PROST), tubal embryo transfer (TET), zygote intra-fallopian transfer (ZIFT), intrauterine insemination (IUI) and intracytoplasmic sperm injection (ICSI) [3]. ART makes a continuing endeavor to improve outcomes and to optimize efficiency. The most typical ART methods are fertilization in vitro (IVF) and intracytoplasmic injection of sperm (ICSI) [4].
In the case of the male reproductive system, infertility is most commonly caused by problems of semen ejection, absence or low levels of sperm, or abnormal morphology or motility of sperm. In the female reproductive system, infertility might be caused by abnormalities of the ovaries, uterus, fallopian tubes and endocrine system. ICSI is a technique in which a single sperm is injected directly into an oocyte for fertilization, which might cause unexpected damage due to manipulation of the sperm and oocytes [5].
Fertilization in vitro (IVF) became an exciting scientific achievement of the twentieth century that made a significant impact on human lives [6]. IVF involves procedures, in a complicated series, that are used to treat infertility and genetic problems that assist in childbirth. It is an effective form of ART. The rates of success of fertilization in vitro and embryo transfer in humans have risen dramatically, since Louise Brown was born in 1978, through improvements in ovulation induction medications and regimens and progress in culture technology, including culture media [7]. In the face of widening ART applications, there are still compelling issues associated with it, such as small rates of implantation and large rates of multiple pregnancy [8].
IVF treatment is an emotional and physical burden for women and their partners. Research results show that couples that set foot in an IVF-treatment program are, in general, psychologically well-adjusted [9]. Chiware et al. [10] investigated the current availability of IVF in low- or middle-income countries (LMIC).
2. Conventional IVF & ICSI
During IVF, mature oocytes are retrieved from a patient’s ovaries and fertilized with a sperm in the laboratory. The fertilized oocyte is subsequently implanted into the patient’s uterus. Current IVF procedures include selecting motile sperm, retrieval and processing of oocytes, insemination, embryo culture in vitro and cryopreservation. Two main approaches of insemination in a laboratory are conventional IVF and intracytoplasmic sperm injection (ICSI). During conventional IVF, every oocyte is placed in an oil-covered micro drop in a culture dish with an optimal concentration of sperm, and fertilization occurs when one sperm penetrates the oocyte in a natural way. In contrast, in ICSI, a single sperm is injected directly into the oocyte’s cytoplasm through a thin needle. ICSI is the presiding treatment for almost all forms of male infertility [11]. There is no need to undertake ICSI in most cases unless there is a male factor or history of previous failure in fertilization, but the truth is that many groups use ICSI in women who have poor prognosis, women who are undergoing PGS, or for fertilization of vitrified oocytes. The results show that ICSI is used regularly in a laboratory for >70% of cases. Two essential steps in the early stages of IVF could be standardized using robotics: removing cumulus cells surrounding the oocyte at retrieval and sperm injection into the cytoplasm [12].
An early insightful opinion on miniaturization and mini-machines for biological applications was presented by Professor Richard P. Feynman (Nobel Laureate in Physics, 1965) [13].
3. Droplet Microfluidics
Droplet-based microfluidics manipulates and controls individual sections of fluid, rather than the bulk fluid flow. In continuous microfluidic flow, the fluid mixing is dominated by molecular diffusion, and hence cross contamination may occur. The main attractions of droplet based microfluidics include a reduction in sample/reagents, faster reaction times, and increased control over dispersion, mixing, and separation [14].
Droplet-based microfluidic systems are compatible with many chemical and biological reagents and are capable of performing a variety of digital fluidic techniques as programmable and reconfigurable operations. This technology has the potential to provide novel solutions to today’s biomedical engineering challenges for advanced diagnostics. Droplet-based microfluidic systems are sometimes referred to as digital microfluidics. This term aims to emphasize the utilization of discrete and distinct volumes of fluids and to contrast these with the continuous nature of other systems. Digital or droplet-based microfluidics involves the generation and manipulation of discrete droplets inside micro devices. This method produces highly monodisperse droplets in the nanometer to micrometer diameter range, at rates of up to twenty thousand per second, used as micro reactors in the nano-liter to femto-liter range [15]. Based on the fabrication and handling technique, the field itself can be further divided into two groups: (i) emulsion- based droplet microfluidics and (ii) digital microfluidics.
3.1. Emulsion-Based Droplet Microfluidics
In emulsion-based droplet microfluidics, micro droplets are formed from the interaction of two immiscible fluids (such as oil and water-in continuous and dispersed phases, respectively). For ART application, this branch of microfluidics is very similar to what embryologists perform in clinical IVF, i.e., encapsulating the gamete in microdroplets covered with an oil overlay. Moreover, the technology can overcome many handling difficulties involving microdroplets in such clinical settings [14].
3.2. Digital Microfluidics
Digital microfluidics (DMF) is a popular technique for diverse applications. Digital microfluidics is an emerging and very promising field in droplet-based microfluidics. Here, individual droplets (either in open or confined channels) can be precisely manipulated by non-contact forces such as electrical, magnetic or thermal ones. The non-contact forces are of great benefit, especially for ART and IVF applications. DMF has a range of discrete fluidic operations for the manipulation of nano-litre to microliter droplets containing samples and reagents. Here, electric potential has been applied in series to array of patterned electrodes which are coated with a hydrophobic insulator. DMF can address each reagent individually with no need for complicated networks of tubing or micro-valves; it has the ability to control liquids relative to solids with no risk of clogging, and it is compatible with a large range of volumes, making it useful for preparative-scale sample handling [16]. The DMF device has been widely applied for biomedical applications. The advantages of DMF include minimized consumption of reagents, rapid reactions and modest cost (e.g., DNA-based applications, protein-based applications, cell-based applications and clinical applications [17][18][19][20][21][22][23][24]). A review of analogue and digital microfluidics is shown in Figure 1 [25]. Electro-wetting-on-dielectric (EWOD) is an example of a DMF platform.
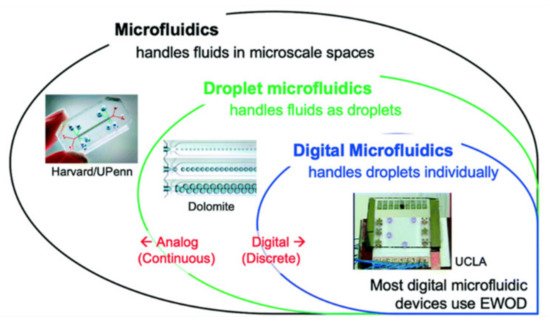
Figure 1. A road to analogue microfluidics to digital microfluidics. Image is adapted from ref. [25] with permission from the Royal Society of Chemistry.
Electro-Wetting-On-Dielectric
EWOD is a method to manipulate discrete droplets on a solid (usually hydrophobic) surface, in a programmable manner, using electrodes made of a conductor such as indium tin oxide (ITO). At least one of the surfaces that is in contact with the liquid is insulated with a layer of dielectric, e.g., Parylene or SU8, and a layer of a hydrophobic material, e.g., Teflon or Cytop. The electrode pattern defines the desired droplet path. Therefore, by arranging the electrodes in the desired way, a variety of parallel or sequential analyses can be accomplished [14].
4. Microfluidics towards ART
Microfluidic techniques have become a powerful tool that can closely recreate the physiological conditions of organ systems in vivo [12]. Microfluidics can be considered as both a science and a technology. Scientifically, it is the study of fluid behavior at a sub-microliter level and the investigation of its application to the fields of cell biology, chemistry, genetics, molecular biology and medicine [26]. Technologically, it relates to applications within analytics and diagnostics, cell biology and single-cell genomics [27]. Microfluidics broadly represents a multi- and trans-disciplinary field of study comprising engineering, physics, chemistry, biology and biotechnology. Systems with small fluid volumes used to multiplex, to automate, to integrate and to facilitate cell manipulation and high-throughput analysis or screening can be designed for practical applications [28]. There are at least two microfluidics—of mechanical and biochemical characteristics—that can influence mammalian gamete and preimplantation embryo biology. Microfluidic mechanical and biochemical characteristics might have practical and technical applications towards ART [28]. Microfluidic devices for biological assays perform analysis with efficient throughput; their advantages include the use of samples or reagents in small amounts and the analysis of single cells [29][30][31].
Fluids at the microscale are likely to suffer from forces that are typically insignificant at scales present in our everyday lives. Fluid at the scale of our typical environment is turbulent (undergoing irregular fluctuations), which leads to a particle within a stream of fluid moving in an unpredictable pattern. Turbulent flow depends on specific fluid characteristics (viscosity, density and velocity) and the geometry of the channel, leading to the calculation of a value known as Reynold’s number. As the scale of the channel reaches micro-meter levels, the Reynold’s number decreases. A decrease in Reynold’s number below a threshold value brings the fluid flow in a laminar fashion so that the flow within a channel becomes streamlined and predictable [32]. Some of the advantages of utilizing a microfluidics platform include scalability, increased automation, and reduced turnaround times for male infertility diagnosis and treatment [33].
Basic Functions of a Microfluidic Bio Chip
The fabrication process of a PDMS-based microfluidic device is explained in [34]. Microfluidic systems serve to handle gametes, mature oocytes, culture embryos, and to perform other basic procedures in a microenvironment that closely imitates conditions in vivo [35]. To perform the above stages, a microfluidic biomedical chip must integrate four essential functions—generation of a droplet of an emulsion, sorting, expansion and restoration. A microfluidic biomedical chip, with a PDMS substrate that has all these essential functions, employed in a mouse embryo system to assess reproductive medicine, is explained in [36]. Here, a droplet carries an embryo, proceeds towards an enlargement area, and another droplet without an embryo is abandoned and directed towards the waste area outlet using a manual sorting method.
A PDMS flow-focusing microfluidic device which was used to generate microliter medium-in-oil droplets is explained in [37]. The device contains two inlets for the continuous injection of oil and medium with oocytes. The volume of micro-droplets was controlled by adjusting the flow rates of both oil and medium inlets. A single mouse oocyte was encapsulated in each dispersed micro-droplet for further in vitro fertilization (IVF) research.
5. Microfluidic IVF
The current research to regulate various stages of assisted reproductive programs with microfluidic techniques, with their benefits and shortcomings, is explained in [11]. IVF is a technique in which sperm and oocytes are conventionally inseminated and cultured in specialized laboratory conditions. In a traditional IVF operation, unnecessary damage to oocytes occurs on account of the di-electrophoretic force manipulation of the sperm and oocytes [38]. Microfluidic chips might have multiple layers of varied materials for a specific purpose [39][40], i.e., an elastomer bound to glass for a product with increased rigidity. Polydimethylsiloxane (PDMS) is a popular elastomer material of one kind for the fabrication of microfluidic chips. As PDMS is cheap and possesses gas permeability and biocompatibility, most microfluidic devices related to biological research are made of PDMS [41]. Some studies, concerned with microfluidic application in the IVF field, were performed to evaluate the real biocompatibility of the materials required to build a device [42][43][44][45]; these initial studies were performed using mouse embryos [45] and pig oocytes [46]. The results showed that many materials such as silicon nitrate, silicon oxide, borosilicate glass, chromium, gold, titanium and polydimethylsiloxane (PDMS) have no negative impact on embryo development [42][45][46][47].
References
- Pourakbari, R.; Ahmadi, H.; Yousefi, M.; Aghebati-Maleki, L. Cell therapy in female infertility-related diseases: Emphasis on recurrent miscarriage and repeated implantation failure. Life Sci. 2020.
- Nikshad, A.; Aghlmandi, A.; Safaralizadeh, R.; Aghebati-Maleki, L.; Warkiani, M.E.; Khiavi, F.M.; Yousefi, M. Advances of microfluidic technology in reproductive biology. Life Sci. 2020.
- Huang, H.-Y.; Lu, C.-Y.; Wang, I.-W.; Yao, D.-J. Motility-driven Sperm-sorting Microfluidic Chip with Little Cell Damage for Oligozoospermia Patients. Sens. Mater. 2020, 32, 2585–2596.
- Dickey, R.P. The relative contribution of assisted reproductive technologies and ovulation induction to multiple births in the United States 5 years after the Society for Assisted Reproductive Technology/American Society for Reproductive Medicine recommendation to limit the number of embryos transferred. Fertil. Steril. 2007, 88, 1554–1561.
- Ma, R.; Xie, L.; Han, C.; Su, K.; Qiu, T.; Wang, L.; Huang, G.; Xing, W.; Qiao, J.; Wang, J. In vitro fertilization on a single-oocyte positioning system integrated with motile sperm selection and early embryo development. Anal. Chem. 2011, 83, 2964–2970.
- Steptoe, P.C.; Edwards, R.G. Birth after the reimplantation of a human embryo. Lancet 1978, 312, 366.
- Pool, T.B. Recent advances in the production of viable human embryos in vitro. Reprod. Biomed. Online 2002, 4, 294–302.
- Botros, L.; Sakkas, D.; Seli, E. Metabolomics and its application for non-invasive embryo assessment in IVF. Mol. Hum. Reprod. 2008, 14, 679–690.
- Eugster, A.; Vingerhoets, A. Psychological aspects of in vitro fertilization: A review. Soc. Sci. Med. 1999, 48, 575–589.
- Chiware, T.M.; Vermeulen, N.; Blondeel, K.; Farquharson, R.; Kiarie, J.; Lundin, K.; Matsaseng, T.C.; Ombelet, W.; Toskin, I. IVF and other ART in low-and middle-income countries: A systematic landscape analysis. Hum. Reprod. Update 2020.
- Weng, L. IVF-on-a-Chip: Recent advances in microfluidics technology for in vitro fertilization. SLAS Technol. Transl. Life Sci. Innov. 2019, 24, 373–385.
- Meseguer, M.; Kruhne, U.; Laursen, S. Full in vitro fertilization laboratory mechanization: Toward robotic assisted reproduction? Fertil. Steril. 2012, 97, 1277–1286.
- Feynman, R.P. There’s plenty of room at the bottom. Calif. Inst. Technol. Eng. Sci. Mag. 1960. Available online: https://www.ias.ac.in/article/fulltext/reso/016/09/0890-0905 (accessed on 6 November 2020).
- Kashaninejad, N.; Shiddiky, M.J.A.; Nguyen, N.T. Advances in microfluidics-based assisted reproductive technology: From sperm sorter to reproductive system-on-a-chip. Adv. Biosyst. 2018, 2, 1700197.
- Teh, S.-Y.; Lin, R.; Hung, L.-H.; Lee, A.P. Droplet microfluidics. Lab Chip 2008, 8, 198–220.
- Jebrail, M.J.; Bartsch, M.S.; Patel, K.D. Digital microfluidics: A versatile tool for applications in chemistry, biology and medicine. Lab Chip 2012, 12, 2452–2463.
- Shen, H.-H.; Fan, S.-K.; Kim, C.-J.; Yao, D.-J. EWOD microfluidic systems for biomedical applications. Microfluid. Nanofluidics 2014, 16, 965–987.
- Vergauwe, N.; Witters, D.; Ceyssens, F.; Vermeir, S.; Verbruggen, B.; Puers, R.; Lammertyn, J. A versatile electrowetting-based digital microfluidic platform for quantitative homogeneous and heterogeneous bio-assays. J. Micromech. Microeng. 2011, 21, 054026.
- Rui, X.; Song, S.; Wang, W.; Zhou, J. Applications of electrowetting-on-dielectric (EWOD) technology for droplet digital PCR. Biomicrofluidics 2020, 14, 061503.
- Liu, Y.-J.; Yao, D.-J.; Chang, H.-Y.; Liu, C.-M.; Chen, C. Magnetic bead-based DNA detection with multi-layers quantum dots labeling for rapid detection of Escherichia coli O157: H7. Biosens. Bioelectron. 2008, 24, 558–565.
- Lapierre, F.; Piret, G.; Drobecq, H.; Melnyk, O.; Coffinier, Y.; Thomy, V.; Boukherroub, R. High sensitive matrix-free mass spectrometry analysis of peptides using silicon nanowires-based digital microfluidic device. Lab Chip 2011, 11, 1620–1628.
- Witters, D.; Vergauwe, N.; Vermeir, S.; Ceyssens, F.; Liekens, S.; Puers, R.; Lammertyn, J. Biofunctionalization of electrowetting-on-dielectric digital microfluidic chips for miniaturized cell-based applications. Lab Chip 2011, 11, 2790–2794.
- Sista, R.S.; Wang, T.; Wu, N.; Graham, C.; Eckhardt, A.; Winger, T.; Srinivasan, V.; Bali, D.; Millington, D.S.; Pamula, V.K. Multiplex newborn screening for Pompe, Fabry, Hunter, Gaucher, and Hurler diseases using a digital microfluidic platform. Clin. Chim. Acta 2013, 424, 12–18.
- Mousa, N.A.; Jebrail, M.J.; Yang, H.; Abdelgawad, M.; Metalnikov, P.; Chen, J.; Wheeler, A.R.; Casper, R.F. Droplet-scale estrogen assays in breast tissue, blood, and serum. Sci. Transl. Med. 2009, 1, 1ra2.
- Li, J. Current commercialization status of electrowetting-on-dielectric (EWOD) digital microfluidics. Lab Chip 2020, 20, 1705–1712.
- Smith, G.D.; Takayama, S. Application of microfluidic technologies to human assisted reproduction. MHR: Basic Sci. Reprod. Med. 2017, 23, 257–268.
- Whitesides, G.M. The origins and the future of microfluidics. Nature 2006, 442, 368–373.
- Volpatti, L.R.; Yetisen, A.K. Commercialization of microfluidic devices. Trends Biotechnol. 2014, 32, 347–350.
- Verpoorte, E. Microfluidic chips for clinical and forensic analysis. Electrophoresis 2002, 23, 677–712.
- Huang, W.-H.; Cheng, W.; Zhang, Z.; Pang, D.-W.; Wang, Z.-L.; Cheng, J.-K.; Cui, D.-F. Transport, location, and quantal release monitoring of single cells on a microfluidic device. Anal. Chem. 2004, 76, 483–488.
- Lin, T.-H.; Yao, D.-J. Applications of EWOD systems for DNA reaction and analysis. J. Adhes. Sci. Technol. 2012, 26, 1789–1804.
- Suh, R.S.; Phadke, N.; Ohl, D.A.; Takayama, S.; Smith, G.D. Rethinking gamete/embryo isolation and culture with microfluidics. Hum. Reprod. Update 2003, 9, 451–461.
- Vaughan, D.A.; Sakkas, D. Sperm selection methods in the 21st century. Biol. Reprod. 2019, 101, 1076–1082.
- Thapa, S.; Heo, Y.S. Microfluidic technology for in vitro fertilization (IVF). JMST Adv. 2019, 1–11.
- Wheeler, M.B.; Rubessa, M. Integration of microfluidics in animal in vitro embryo production. MHR: Basic Sci. Reprod. Med. 2017, 23, 248–256.
- Chiu, Y.-L.; Yadav, R.A.K.; Huang, H.-Y.; Wang, Y.-W.; Yao, D.-J. Unveiling the Potential of Droplet Generation, Sorting, Expansion, and Restoration in Microfluidic Biochips. Micromachines 2019, 10, 756.
- Shen, H.-H.; Tsai, H.-Y.; Yao, D.-J. Single mouse oocyte encapsulated in medium-in-oil microdroplets by using a polydimethylsiloxane microfluidic device. Sens. Mater. 2014, 26, 85–94.
- Huang, H.-Y.; Lai, Y.-L.; Yao, D.-J. Dielectrophoretic microfluidic device for in vitro fertilization. Micromachines 2018, 9, 135.
- Villar, G.; Graham, A.D.; Bayley, H. A tissue-like printed material. Science 2013, 340, 48–52.
- Chen, C.-C.; Chen, Y.-A.; Liu, Y.-J.; Yao, D.-J. A multilayer concentric filter device to diminish clogging for separation of particles and microalgae based on size. Lab Chip 2014, 14, 1459–1468.
- Lo, S.-J.; Yao, D.-J. Get to understand more from single-cells: Current studies of microfluidic-based techniques for single-cell analysis. Int. J. Mol. Sci. 2015, 16, 16763–16777.
- Choi, S.; Glasgow, I.; Zeringue, H.; Beebe, D.; Wheeler, M. Development of microelectromechanical systems to analyze individual mammalian embryos: Embryo biocompatability systems to analyze individual mammalian embryos: Embryo biocompatability. Biol. Reprod. 1998, 58, 96.
- Chan, N.; Lyman, J.; Choi, S.; Zeringue, H.; Glasgow, I.; Beebe, D.; Wheeler, M. Development of an embryo transport and analysis system: Material biocompatablity. Theriogenology 1999, 51, 234.
- Chan, N.; Raty, S.; Zeringue, H.; Beebe, D.; Wheeler, M. Abstracts for Poster Presentation-Embryo Culture-Development of Microfabricated Devices for Embryo Production: Embryo Biocompatibility. Theriogenology 2001, 55, 332.
- Glasgow, I.K.; Zeringue, H.C.; Beebe, D.J.; Choi, S.-J.; Lyman, J.T.; Chan, N.G.; Wheeler, M.B. Handling individual mammalian embryos using microfluidics. IEEE Trans. Biomed. Eng. 2001, 48, 570–578.
- Hester, P.; Roseman, H.; Clark, S.; Walters, E.; Beebe, D.; Wheeler, M. Enhanced cleavage rates following in vitro maturation of pig oocytes within polydimethylsiloxane-borosilicate microchannels. Theriogenology 2002, 57, 723.
- Walters, E.M.; Clark, S.G.; Beebe, D.J.; Wheeler, M.B. Mammalian embryo culture in a microfluidic device. In Germ Cell Protocols; Springer: Berlin/Heidelberg, Germany, 2004; pp. 375–381.
More
Information
Subjects:
Biochemistry & Molecular Biology
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
985
Revisions:
2 times
(View History)
Update Date:
09 Aug 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





