
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Ajay K Dalai | + 1599 word(s) | 1599 | 2021-07-29 08:44:09 | | | |
| 2 | Beatrix Zheng | + 197 word(s) | 1796 | 2021-08-07 16:15:45 | | |
Video Upload Options
Light olefins as one the most important building blocks in chemical industry can be produced via Fischer–Tropsch synthesis (FTS) from syngas. FT synthesis conducted at high temperature would lead to light paraffins, carbon dioxide, methane, and C5+ longer chain hydrocarbons. The present work focuses on providing a critical review on the light olefin production using Fischer–Tropsch synthesis.
1. Introduction
Olefins including ethylene, propylene, and butylene are considered the most widely used petrochemical feedstocks used as chemical intermediates for production of solvents, polymers, plastics, fibers, and detergents. The demand for ethylene as one of the important derivatives of olefins is over 155 million tons annually. The common method for olefin production is steam cracking (SC) of hydrocarbons. The trend of ethylene production using thermal cracking had a growth rate of 4% between 2007 and 2012 [1]. The ethylene production in Canada is based on ethane as a feed through steam cracking at high temperatures. From 2000 to 2010, it was reported that the propylene production declined while the ethylene production increased [2].
Heavy petroleum oil with low API gravity containing different impurities such as sulfur, nitrogen, and metals brings about many challenges during its processing to the light olefins. Petroleum oil is estimated as the main energy source up to 2040 based on the OPEC’s World Oil Outlook in 2016, with the energy demand reaching to 382 million barrels of oil equivalent per day (mboe/d). As olefin production depends on oil fractions and steam cracking, the increasing demand for light olefins can cause strain on crude oil resources [3]. The non-oil routes for olefin production can be categorized in four groups, namely methanol to olefin (MTO), ethanol to olefin (ETO), dimethyl ether to olefin (DMTO), and the Fischer–Tropsch synthesis (FTS), which was developed in 1922 [4].
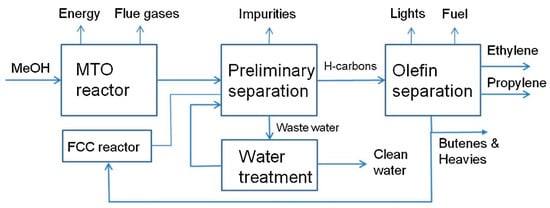
The technologies including MTO and FTS use methane as a feedstock for higher hydrocarbon production in an indirect way, while oxidative coupling of methane (OCM) is a direct route for ethylene and higher hydrocarbons production using methane. The H 2/CO ratio in CO-rich syngas needs to be adjusted by water–gas shift (WGS) reactions to eliminate the CO level, while resulting in CO 2 emissions [2]. However, the adjustment of H 2/CO ratio is not necessary for the H 2-rich syngas. Synthesis of an intermediate like methanol or dimethyl ether is the basis of MTO and dimethyl ether-to-olefin (DMTO) processes to produce light olefins indirectly, while Fischer–Tropsch to olefins process is based on a one-step reaction consuming synthesis gas without adjustment of H 2/CO ratio [2]. An MTO plant is shown in Figure 1 where methanol is consumed in an MTO reactor and light olefins are separated within two stages of separation.
Coal, natural gas, and biomass are the main source of synthesis gas for olefins production. Coal and biomass are converted to syngas via gasification, while the conversion of natural gas to syngas is carried out through steam reforming. The established FTS plants presented in Table 1 show a tendency toward natural gas compared to coal over the years [6]. FTS provides two advantages compared to MTO. The lower cost of olefin production in FTS involves a one-step process. Additionally, a wide range of raw materials can be used as feedstocks in FTS, with less wastewater production throughout the process [7]. Oil resource depletion and high cost of exploration has led scientists to develop alternative feedstocks for the olefins production. Biomass and waste streams including solid plastic waste or municipal waste are good candidates for future use in olefins production, indicating that FTS can be regarded as an environmentally friendly and economical process for light olefins production [8].
| Company | Carbon Source | Capacity (bpd) | Commissioning Date |
|---|---|---|---|
| Sasol | Coal | 2500 | 1955 |
| Sasol | Coal | 85,000 | 1980 |
| Sasol | Coal | 85,000 | 1982 |
| MossGas | Natural gas | 30,000 | 1992 |
| Shell | Natural gas | 12,500 | 1993 |
| Sasol/Qatar Petroleum | Natural gas | 34,000 | 2006 |
| Sasol Chevron | Natural gas | 34,000 | 2007 |
| Shell | Natural gas | 140,000 | 2009 |
| Sasol/USA | Natural gas | 96,000 | 2018 |
| Sasol/Canada | Natural gas | 96,000 | 2020 |
2. Catalysts for Fischer–Tropsch to Olefins (FTO)
The decrease in metal particle diameter greatly affects the chemisorption behavior of both hydrogen and carbon monoxide, indicating the dispersion of catalyst active metal. Taking it into account, CO-chemisorption and H 2− temperature programmed desorption (H 2− TPD) in conjunction with O 2-titration are proved effective methods to determine active metal dispersion in catalysts. Table 2 illustrates active metal dispersion of different FTS catalysts for light olefin production.
| Catalyst | Technique | Dispersion (%) | C2-C4 Selectivity (%) |
Note | Reference |
|---|---|---|---|---|---|
| Co/TiO2 Co/TiO2@mSiO2 a CoRu/TiO2@mSiO2 |
Pulse Chemisorption | 4.5–1.9 3.6–3.7 5.0–6.7 |
10.6–20.9 5.2–21.7 12.1–23.3 |
TChemisorption = 350–450 °C FTS (T = 220–250 °C, P = 10 bar, H2/CO = 2, GHSV = 800 mLg−1h−1) |
[10] |
| Co/CNT | H2−TPD | 8.2–10.8 | 29.5–18.9 | FTS (T = 220 °C, P = 20 bar, H2/CO = 2, GHSV = 40 mLg−1h−1) | [11] |
| CoPt/TiO2-C b CoPt/TiO2-P1 c CoPt/TiO2-P3 CoPt/TiO2-P4 |
H2−TPD O2−titration |
20.4 26.9 27.8 73.7 |
5.4 6.2 6.5 7.0 |
FTS (T = 210 °C, P = 10 bar, H2/CO = 2, GHSV = 4 SLg−1h−1) | [12] |
| 0CTAB-Co@C d 2CTAB-Co@C 4CTAB-Co@C 8CTAB-Co@C |
H2−TPD | 32.05 20.07 37.07 38.51 |
10.87 11.87 11.21 11.27 |
FTS (T = 230 °C, P = 20 bar, H2/CO = 2, GHSV = 6.75 SLg−1h−1) | [13] |
Cheng et al. [14] studied the effects of support pore size on silica supported iron catalysts in high-temperature Fischer–Tropsch synthesis. It was reported that larger pore size of silica supported iron catalyst offers higher olefin and C 5+ selectivity due to easier iron carbidization [14].
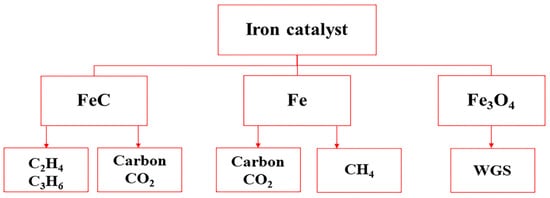
Transformation of the active iron carbide phase into a less or non-active iron carbide has a direct role in deactivation of Fe-based catalysts. Carbon may have a beneficial protecting effect on iron catalyst used for olefin production in FTS. The catalytic performances of FeO x supported on Al 2O 3, ZrO 2, and anodized plates for production of C 1-C 4 hydrocarbons using synthesis gas were studied by Lodeng et al. [15]. A model was suggested to describe the state of iron catalyst phases before deactivation ( Figure 2 ). It was reported that iron carbide would lead to light olefin production. The iron carbide is more likely to provide more contact between Fe and promoters due to high mobility of promoters [16]. However, the Ɵ-Fe 3C can cause catalyst deactivation through undesired carbide formation. The reduced form of iron suggests methane formation, while the iron oxide is selective to the WGS reaction.
Ordomsky et al. [17] evaluated the catalytic performance of supported cobalt and iron catalysts in FTS using ammonia in syngas. It was reported that the deactivation of the Co catalyst occurs at ammonia concentrations higher than 400 ppm. On cobalt catalysts, acetonitrile and NH 3 led to an irreversible catalyst deactivation. In the case of iron catalysts, the CO conversion is influenced by nitrogen addition. During the exposure of cobalt catalyst to acetonitrile, the formation of cobalt nitride is the main reason of catalyst deactivation. However, ammonia addition under syngas flow leads to conversion of metallic iron and iron oxides into iron nitride and iron carbide, respectively [17]. It was reported that addition of Na to a series of supported iron catalysts at high concentration can cause a drop in FT reaction rate due to the strong interaction between iron and sodium and site blocking [18].
3. Summary and Conclusions
Production of light olefins through Fischer–Tropsch synthesis using syngas as feedstock is an issue of great importance. Development of catalytic systems in terms of activity, selectivity, and stability is required to consider it feasible that light olefins can be produced on an industrial scale via FTO. Iron can be suggested as the promising metal for light olefin synthesis as it is more tolerant of sulfur contaminants present in hydrogen-deficient syngas obtained from biomass, inexpensive, and highly selective toward light olefins. Activity of Fe for water–gas-shift (WGS) reaction can compensate H 2 deficiency in CO-rich syngas. Fe exists in different forms (χ-Fe 5C 2, ε-Fe 2C, ε’-Fe 2.2 C, and Fe 7C 3) as iron carbide which is known to be the active phase in typical FTO process. Among these carbide phases, the highest activity and the lowest methane selectivity belong to Fe 7C 3 and ε-Fe 2C for medium range of temperature in FTO. Compared to Fe, Co-based catalysts exhibit high catalytic activity, low WGS activity, and superior stability. Due to low activity for WGS reaction, Co catalysts require higher ratios of H 2/CO in comparison with Fe-based catalyst. Formation of light olefins over Co-based catalysts is mainly attributed to the reactions taking place at the Co/Co 3C interface. In terms of crystal phase, hexagonal close-packed (HCP) structure of Co is believed to have higher FTO activity than face-centered cubic (FCC) structure.
Selection of support, promoter, catalyst synthesis method, and process conditions can significantly affect the output. The support should be capable of providing desirable interaction with active metal and promoters. Promoters should be used cautiously to avoid catalyst poisoning. Basicity of catalysts can be improved by adding alkali metals providing the strength for syngas hydrogenation. Strong basicity sites would offer an enhanced dissociative adsorption of CO and higher olefin selectivity compared to medium basicity sites. Phase transition in catalyst reducibility is also affected by adding alkali metals. That is to say, the low-temperature reduction would be inhibited, while that of high-temperature will be enhanced by increasing alkali metal promoters. It might be suggested that Fe-based catalysts possessing terrace or hierarchical sites with pores being molecular sieve are feasible to increase light olefin production during FTS. Surface modification of Fe catalysts supported on carbon materials with nitrogen-containing functionalities is also observed to increase light olefins selectivity in FTO.
Catalyst deactivation can be intensified by inadequate selection of the support, promoter, and synthesis method resulting in the catalyst poor structure. Fe particles as active metal requires protection against re-oxidation, carbidization, carbon deposition, and sintering through designing a robust catalytic system specially by incorporating Fe particles within support porous network. Confinement of Fe nanoparticles inside carbon materials like carbon nanotubes would offer high dispersion of active metal within carbon nanotubes protecting metals from deactivation. Promoted core–shell Co-based catalysts with enhanced dispersion of active metals would also be another good candidate for Fischer–Tropsch to light olefins due to sintering resistance.
The economic, energy, and environmental aspects of the FTS process as well as carbon and energy balance of the process can be calculated applying a techno-economic and lifecycle analysis (TEA/LCA), thus suggesting a decrease in both the waste released and raw material consumption. Although the FTS-based plants are mostly environmentally friendly with pollutant emissions and nonrenewable source energy demand being reduced, the minimum biofuel selling price cannot still compete with the market prices of petroleum fuels. Therefore, it is necessary for governments to provide renewable FTS plants with governmental subsidy and tax concession or exemption, helping them to survive and patronize in the global market.
References
- Sadrameli, S. Thermal/catalytic cracking of hydrocarbons for the production of olefins: A state-of-the-art review I: Thermal cracking review. Fuel 2015, 140, 102–115.
- Amghizar, I.; Vandewalle, L.A.; Van Geem, K.M.; Marin, G.B. New trends in olefin production. Engineering 2017, 3, 171–178.
- Alotaibi, F.M.; Gonzalez-Cortes, S.; Alotibi, M.F.; Xiao, T.; Al-Megren, H.; Yang, G.; Edwards, P.P. Enhancing the production of light olefins from heavy crude oils: Turning challenges into opportunities. Catal. Today 2018, 317, 86–98.
- van der Laan, G.P.; Beenackers, A.A. Hydrocarbon selectivity model for the gas–solid Fischer–Tropsch synthesis on precipitated iron catalysts. Ind. Eng. Chem. Res. 1999, 38, 1277–1290.
- Dimian, A.C.; Bildea, C.S. Energy efficient methanol-to-olefins process. Chem. Eng. Res. Des. 2018, 131, 41–54.
- Ail, S.S.; Dasappa, S. Biomass to liquid transportation fuel via Fischer Tropsch synthesis–Technology review and current scenario. Renew. Sustain. Energy Rev. 2016, 58, 267–286.
- Zafari, R.; Abdouss, M.; Zamani, Y. Effect of Mn and reduced graphene oxide for the Fischer–Tropsch reaction: An efficient catalyst for the production of light olefins from syngas. React. Kinet. Mech. Catal. 2020, 129, 707–724.
- Di, Z.; Zhao, T.; Feng, X.; Luo, M. A Newly Designed Core-Shell-Like Zeolite Capsule Catalyst for Synthesis of Light Olefins from Syngas via Fischer–Tropsch Synthesis Reaction. Catal. Lett. 2019, 149, 441–448.
- Lewis, P.E. Gas to Liquids: Beyond Fischer Tropsch. In Proceedings of the SPE Asia Pacific Oil and Gas Conference and Exhibition, Jakarta, Indonesia, 12–14 October 2013.
- Phaahlamohlaka, T.N.; Dlamini, M.W.; Mogodi, M.W.; Kumi, D.O.; Jewell, L.L.; Billing, D.G.; Coville, N.J. A sinter resistant Co Fischer-Tropsch catalyst promoted with Ru and supported on titania encapsulated by mesoporous silica. Appl. Catal. A Gen. 2018, 552, 129–137.
- Pour, A.N.; Karimi, J.; Taghipoor, S.; Gholizadeh, M.; Hashemian, M. Fischer–Tropsch synthesis over CNT-supported cobalt catalyst: Effect of magnetic field. J. Iran. Chem. Soc. 2017, 14, 1477–1488.
- Hong, J.; Du, J.; Wang, B.; Zhang, Y.; Liu, C.; Xiong, H.; Sun, F.; Chen, S.; Li, J. Plasma-assisted preparation of highly dispersed cobalt catalysts for enhanced Fischer–Tropsch synthesis performance. ACS Catal. 2018, 8, 6177–6185.
- Chen, Y.; Li, X.; Nisa, M.U.; Lv, J.; Li, Z. ZIF-67 as precursor to prepare high loading and dispersion catalysts for Fischer-Tropsch synthesis: Particle size effect. Fuel 2019, 241, 802–812.
- Cheng, K.; Virginie, M.; Ordomsky, V.V.; Cordier, C.; Chernavskii, P.A.; Ivantsov, M.I.; Paul, S.; Wang, Y.; Khodakov, A.Y. Pore size effects in high-temperature Fischer–Tropsch synthesis over supported iron catalysts. J. Catal. 2015, 328, 139–150.
- Lødeng, R.; Lunder, O.; Lein, J.-E.; Dahl, P.I.; Svenum, I.-H. Synthesis of light olefins and alkanes on supported iron oxide catalysts. Catal. Today 2018, 299, 47–59.
- Gu, B.; Ordomsky, V.V.; Bahri, M.; Ersen, O.; Chernavskii, P.A.; Filimonov, D.; Khodakov, A.Y. Effects of the promotion with bismuth and lead on direct synthesis of light olefins from syngas over carbon nanotube supported iron catalysts. Appl. Catal. B Environ. 2018, 234, 153–166.
- Ordomsky, V.V.; Carvalho, A.; Legras, B.; Paul, S.; Virginie, M.; Sushkevich, V.L.; Khodakov, A.Y. Effects of co-feeding with nitrogen-containing compounds on the performance of supported cobalt and iron catalysts in Fischer–Tropsch synthesis. Catal. Today 2016, 275, 84–93.
- Cheng, K.; Ordomsky, V.V.; Legras, B.; Virginie, M.; Paul, S.; Wang, Y.; Khodakov, A.Y. Sodium-promoted iron catalysts prepared on different supports for high temperature Fischer–Tropsch synthesis. Appl. Catal. A Gen. 2015, 502, 204–214.





