| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Konrad Mendrala | + 1341 word(s) | 1341 | 2021-07-06 05:15:19 | | | |
| 2 | Konrad Mendrala | + 167 word(s) | 1508 | 2021-08-02 10:24:57 | | | | |
| 3 | Dean Liu | Meta information modification | 1508 | 2021-08-03 03:04:30 | | |
Video Upload Options
While standard Deep Hypothermia (DH) is defined as a body core temperature below 28°C, this value is lower in cardiac surgery, below 20°C. DH is used not only to improve the technical aspect of surgery but primarily to prevent ischemic injury of the central nervous system and crucial organs. The protective effect of hypothermia is provided mainly by slowing the cellular metabolism and thus decreasing its oxygen consumption and energy demand.
1. Introduction
Hypothermia, defined as a decrease in body core temperature to below 35 °C, is a supportive technique in cardiac surgery. While standard deep hypothermia is defined as a body core temperature below 28 °C, this value is lower in cardiac surgery, below 20 °C (Table 1). Metabolic protection offered by hypothermia enables safe circulatory arrest during cardiac surgical intervention. Despite DH being an “old” technique of organ protection, it remains indispensable in specific types of cardiac surgical procedures.
| Stages | Body Core Temperature | |
|---|---|---|
| Standard Values | Cardiac Surgery | |
| Mild (°C) | 35–32 | 34–28.1 |
| Moderate (°C) | 31.9–28 | 28–20.1 |
| Deep (°C) | 27.9–20.1 | 20–14.1 |
| Profound (°C) | ≤20 | ≤14 |
2. Indications for the Use of DH in Cardiac Surgery
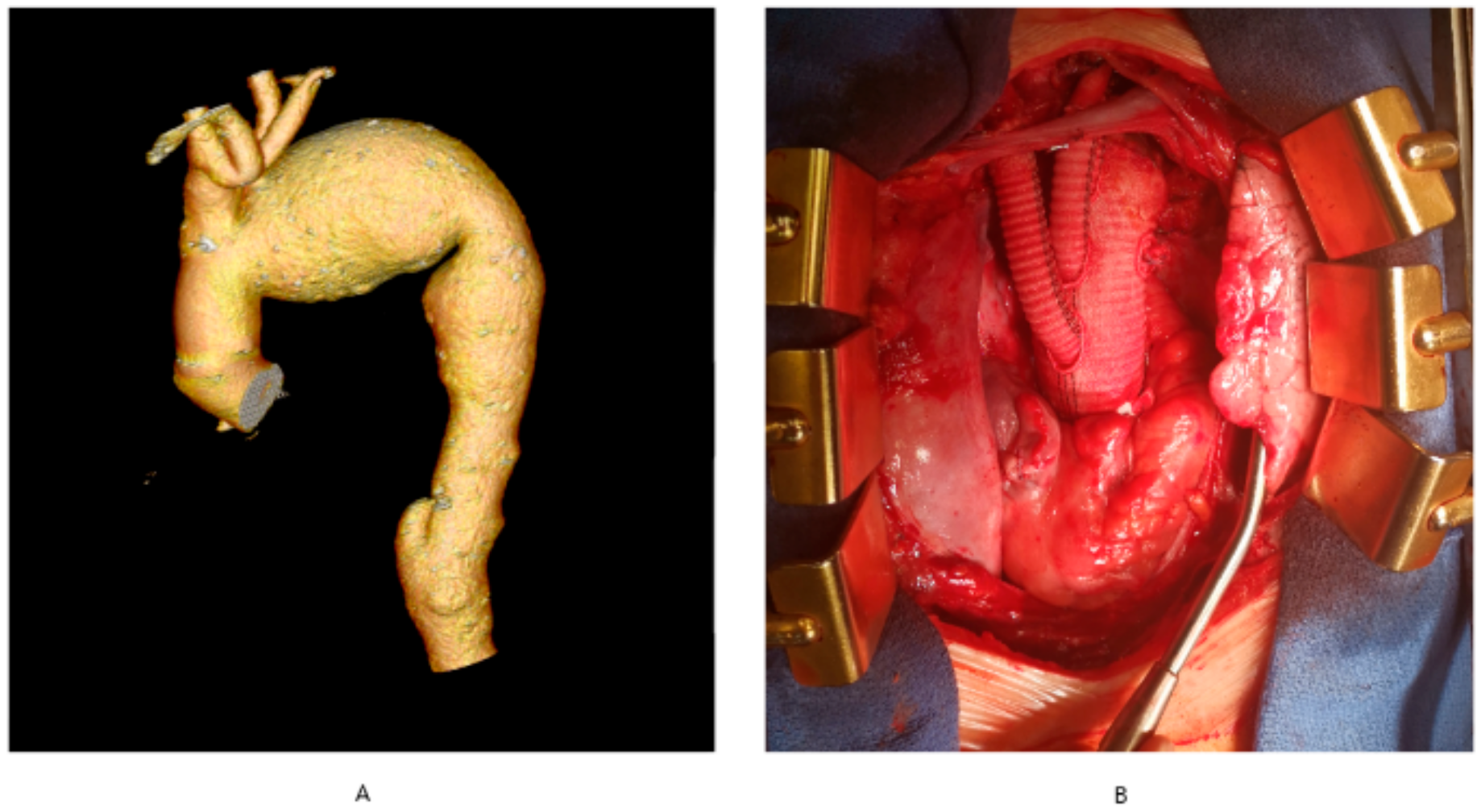
Developments in surgical techniques and advances in myocardial protection and extracorporeal circulation have resulted in a substantial narrowing of indications for DH in cardiac surgery. In adult cardiac surgery, DH is currently used both in elective and emergent interventions. Elective interventions include: complex aortic arch surgery (Figure 1A,B), chronic type-A aortic dissection, pulmonary embolism surgery, complex thoraco-abdominal aneurysm surgery and surgery with co-existing massive calcifications of the ascending aorta precluding cross-clamping (porcelain aorta). Emergent interventions include surgery for acute type-A aortic dissection.
Figure 1. Aortic arch aneurysm. (A) computerized tomography angiography of the thoracic aorta with aortic arch aneurysm, (B) total arch replacement involving aortic arch replacement with concomitant re-implantation of arch vessels.
Infant cardiac surgery is the exception to the rule, as hypothermia represents the mainstay of cerebral protection in most interventions for congenital heart defects. In infant cardiac surgery mild hypothermia is used for corrections of simple defects, where circulatory arrest is not required, and cardiac arrest does not exceed 20 min. Moderate hypothermia without circulatory arrest is used in older infants with a bodyweight above 10 kg and a complex heart defect, in whom the cardiac arrest duration needed for correction exceeds 20 min. Deep hypothermia is used to correct congenital heart defects in infants below 10 kg in bodyweight, and selected older infants. In infant cardiac surgery, DH still represents the gold standard in procedures involving aortic arch reconstruction, left hypoplastic heart syndrome, and interrupted aortic arch, as well as the multi-stage treatment of infants with a single ventricle in the neonatal period.
3. Physiological Effects of Hypothermia
Understanding the role of hypothermia is not a simple matter, as complex systemic and cellular mechanisms mediate its effects. The assumption of benefits from using hypothermia in clinical practice was provided by hypothesizing that the “safe” circulatory arrest period is inversely proportional to one’s body temperature. The term “safe” is reserved for such a period of circulatory arrest, after which no adverse effects can be noted on the structure and function of the internal organs and particularly on the brain, which is most susceptible to ischemia (Table 2).
Table 2. Pathophysiology of hypothermia.
| Symptoms | Mild | Moderate | Deep |
|---|---|---|---|
| Neuro-muscular | ataxia dysarthria shivering |
stiffness of muscles and joints | muscle contraction |
| Neurological | confusion amnesia apathy limited awareness |
limited consciousness | dilated pupils coma loss of self-regulation |
| Circulatory | tachycardia vascular constriction blood pressure increase |
bradycardia widening of QRS complexes elevation/depression of ST segment T-wave inversion AV block QT segment prolongation |
serious bradycardia asystole ventricular fibrillation |
| Respiratory | tachypnae HbO2 curve shifts to the left |
bradypnae bronchial constriction |
lactic acidosis HbO2 curve shifts to the right |
With a gradual decrease in body temperature, chemical reactions, oxygen consumption, and energy requirements are reduced. The human brain is subject to self-regulatory mechanisms, which couple the cerebral blood flow to cerebral oxygen consumption and metabolic activity. This permits safe circulatory arrest in normothermia for only 5 min, after which irreversible changes in brain tissue occur. In his study, Mezrow found that lowering body temperature by 10 degrees Celsius decreases cerebral metabolic activity four-fold and thus increases the tolerance of ischemia [1].
Based on McCullough’s studies [2], safe periods of circulatory arrest in humans for specific temperatures were established: 30 min at 15 °C ; 40 min at 10 °C.
Other study findings indicate that although the period of 40 min in deep hypothermic circulatory arrest is “safe”, exceeding this limit is associated with a substantially elevated risk of neurological adverse events [3][4]. In addition to the effect on reducing metabolic requirements, other mechanisms of hypothermia may be relevant, such as suppression of free radicals, inhibition of destructive enzymatic reactions, and inhibition of the biosynthesis, release, and uptake of excitatory neurotransmitters [5][6][7]. Through these mechanisms, hypothermia provides a favorable balance between oxygen supply and demand, slows the onset of ischemic depolarization, decreases the release of ischemia-induced intracellular calcium influx, and suppresses nitric oxide synthase activity.
4. Neuroprotection during DH
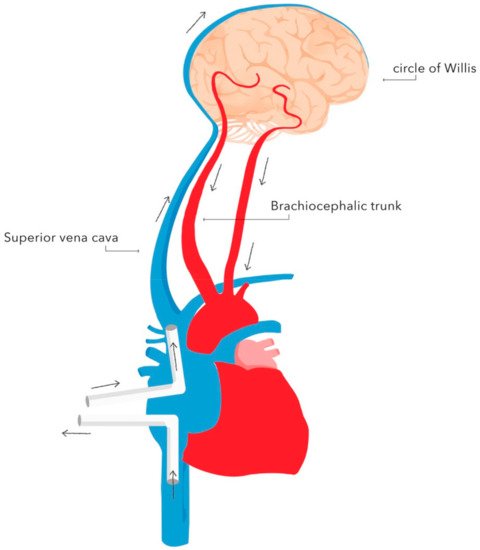
The basic techniques of neuroprotection supporting DH during circulatory arrest include selective antegrade cerebral perfusion (SACP) (Figure 2), retrograde cerebral perfusion (RCP) (Figure 3), cerebrospinal fluid drainage and pharmacotherapy.
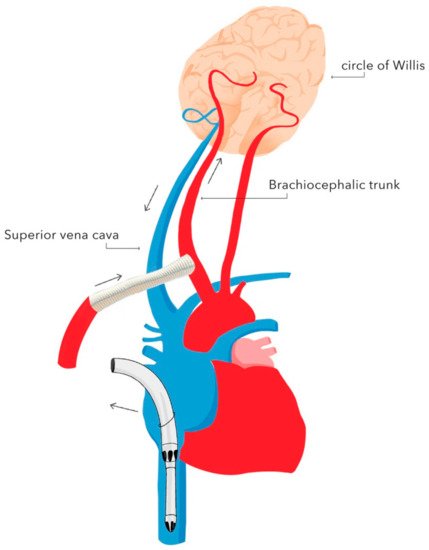
Bilateral SACP provides blood supply to the brain through both the left and right carotid arteries by cannulating the brachiocephalic trunk and the left carotid artery. Unilateral SACP provides blood supply to the brain through one of the carotid arteries. Alternatively, the right subclavian artery can be used as vascular access for the blood supply. SACP is preferred as it provides continuous blood supply to the brain throughout surgery, which enables one to accept higher body temperatures in the range of 25–26 °C—the duration of cooling and rewarming are shortened, and consequently the CPB duration and CPB-associated adverse events are reduced [8].
Retrograde cerebral perfusion (RCP) consists of brain perfusion by reversing blood flow in the cerebral vessels. The brain is supplied retrogradely - oxygenated blood is delivered through the superior vena cava, and deoxygenated blood returns through the cerebral arteries. This method of perfusion is possible as there are no valves in the cerebral venous system. The implementation of RCP enables safe prolongation of circulatory arrest during DH to 60 min [9].
Another neuroprotective intervention during DHCA in complex interventions involving the descending aorta is cerebrospinal fluid drainage. The most beneficial effect of cerebrospinal fluid drainage is reducing pressure in the spinal canal, thus decreasing the forces pressurizing the spinal cord [10]. A drainage catheter is placed prior to surgery (before administering anticoagulants and antiplatelet agents) in the 2nd or 3rd lumbar space and remains in place for 2–3 days after surgery. The recommended target values for cerebrospinal fluid pressure are 8–10 mmHg during surgery and 10–12 mmHg in the postoperative period [10].
Neuroprotective pharmacotherapy includes several groups of medications that are used during DH. As studies regarding the benefits of general anesthetics remain inconsistent, the choice of anesthetic agent depends mainly on the anesthesiologist’s personal preferences. It has been suggested that propofol- and opioid- based anesthetics decrease cerebral metabolism without affecting cerebral flow metabolism coupling, which may occur during the use of volatile agents [11]. In contrast, data from animal studies suggest that the use of desflurane may improve the neurological outcome after DHCA [46]. However, it should be noted that the solubility of gases and volatile agents in the blood increases at low temperatures, which may significantly increase the time to achieve adequate partial pressure in the central nervous system. An intravenous anesthetic that significantly affects brain metabolic activity by inducing burst suppression is thiopental. Its beneficial effects include reducing energy requirements for synaptic transmission and preventing focal brain injury [47–49]. However, it should be remembered that thiopental has a negative inotropic effect, which can impact the early postoperative course. Although there is still inconclusive evidence of a neuroprotective effect of corticosteroids, their use during DHCA should be considered. Steroids reduce the systemic inflammatory response syndrome (SIRS), which can be triggered by CPB [12]. Moreover, they reduce the concentration of inflammatory cytokines [13]. Some studies indicate that the use of steroids after DH may have a beneficial effect on cerebral metabolism, enhance brain tissue blood perfusion, and reduce capillary permeability, thus preventing tissue edema [13][14]. The use of other potentially neuroprotective drugs, such as calcium channel blockers, aprotinin, nafamostat, mannitol, deferoxamine, magnesium, lidocaine, fosphenytoin, and thromboxane A2 receptor blockers, is still a matter of debate and clinical research.
5. Advantages of deep hypothermia
- enables complex surgical interventions involving the aortic arch during circulatory arrest
- indispensable for pulmonary embolism surgery
- in infant congenital defect surgery, it improves the technical aspect, as the CPB cannulas limiting the operating field can be temporarily removed during circulatory arrest
- protects the tissues (particularly nervous tissue) from ischemic damage by reducing the cellular metabolism.
6. Drawbacks of deep hypothermia
- as DH does not stop the cellular metabolism completely, the time of its application is limited
- DH requires advanced monitoring of physical and biochemical parameters
- Risk of excessive bleeding due to coagulopathy
- to limit the rate of neurological adverse events, the use of neuroprotective measures such as pharmacotherapy and selective organ perfusion techniques is necessary.
References
- Mezrow, C.K.; Midulla, P.S.; Sadeghi, A.M.; Gandsas, A.; Wang, W.; Dapunt, O.E.; Zappulla, R.; Griepp, R.B. Evaluation of Cerebral Metabolism and Quantitative Electroencephalography after Hypothermic Circulatory Arrest and Low-Flow Cardiopulmonary Bypass at Different Temperatures. J. Thorac. Cardiovasc. Surg. 1994, 107, 1006–1019.
- McCullough, J.N.; Zhang, N.; Reich, D.L.; Juvonen, T.S.; Klein, J.J.; Spielvogel, D.; Ergin, M.A.; Griepp, R.B. Cerebral Metabolic Suppression during Hypothermic Circulatory Arrest in Humans. Ann. Thorac. Surg. 1999, 67, 1895–1899; discussion 1919–1921.
- Englum, B.R.; Andersen, N.D.; Husain, A.M.; Mathew, J.P.; Hughes, G.C. Degree of Hypothermia in Aortic Arch Surgery—Optimal Temperature for Cerebral and Spinal Protection: Deep Hypothermia Remains the Gold Standard in the Absence of Randomized Data. Ann. Cardiothorac. Surg. 2013, 2, 184–193.
- Kayatta, M.O.; Chen, E.P. Optimal Temperature Management in Aortic Arch Operations. Gen. Thorac. Cardiovasc. Surg. 2016, 64, 639–650.
- Gambert, S.; Bès-Houtmann, S.; Vandroux, D.; Tissier, C.; Vergely-Vandriesse, C.; Rochette, L.; Athias, P. Deep Hypothermia during Ischemia Improves Functional Recovery and Reduces Free-Radical Generation in Isolated Reperfused Rat Heart. J. Heart Lung Transplant. 2004, 23, 487–491.
- Alva, N.; Palomeque, J.; Carbonell, T. Oxidative Stress and Antioxidant Activity in Hypothermia and Rewarming: Can RONS Modulate the Beneficial Effects of Therapeutic Hypothermia? Oxid. Med. Cell. Longev. 2013, 2013, 957054.
- González-Ibarra, F.P.; Varon, J.; López-Meza, E.G. Therapeutic Hypothermia: Critical Review of the Molecular Mechanisms of Action. Front. Neurol. 2011, 2, 4.
- Halkos, M.E.; Kerendi, F.; Myung, R.; Kilgo, P.; Puskas, J.D.; Chen, E.P. Selective Antegrade Cerebral Perfusion via Right Axillary Artery Cannulation Reduces Morbidity and Mortality after Proximal Aortic Surgery. J. Thorac. Cardiovasc. Surg. 2009, 138, 1081–1089.
- Ueda, Y. A Reappraisal of Retrograde Cerebral Perfusion. Ann. Cardiothorac. Surg. 2013, 2, 316–325.
- Wortmann, M.; Böckler, D.; Geisbüsch, P. Perioperative Cerebrospinal Fluid Drainage for the Prevention of Spinal Ischemia after Endovascular Aortic Repair. Gefässchirurgie 2017, 22, 35–40.
- Slupe, A.M.; Kirsch, J.R. Effects of Anesthesia on Cerebral Blood Flow, Metabolism, and Neuroprotection. J. Cereb. Blood Flow Metab. 2018, 38, 2192–2208.
- Finn, A.; Naik, S.; Klein, N.; Levinsky, R.J.; Strobel, S.; Elliott, M. Interleukin-8 Release and Neutrophil Degranulation after Pediatric Cardiopulmonary Bypass. J. Thorac. Cardiovasc. Surg. 1993, 105, 234–241.
- Bronicki, R.A.; Backer, C.L.; Baden, H.P.; Mavroudis, C.; Crawford, S.E.; Green, T.P. Dexamethasone Reduces the Inflammatory Response to Cardiopulmonary Bypass in Children. Ann. Thorac. Surg. 2000, 69, 1490–1495.
- Shum-Tim, D.; Tchervenkov, C.I.; Jamal, A.M.; Nimeh, T.; Luo, C.Y.; Chedrawy, E.; Laliberte, E.; Philip, A.; Rose, C.P.; Lavoie, J. Systemic Steroid Pretreatment Improves Cerebral Protection after Circulatory Arrest. Ann. Thorac. Surg. 2001, 72, 1465–1471; discussion pp. 1471–1472.