
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Mahdieh Shokrollahi Barough | + 3805 word(s) | 3805 | 2021-07-07 12:10:35 | | | |
| 2 | Lindsay Dong | Meta information modification | 3805 | 2021-07-29 11:59:07 | | |
Video Upload Options
Viral-associated respiratory infectious diseases are one of the most prominent subsets of respiratory failures, known as viral respiratory infections (VRI). VRIs are proceeded by an infection caused by viruses infecting the respiratory system. Due to their specific physical and biological properties, nanoparticles hold promising opportunities for both anti-viral treatments and vaccines against viral infections.
1. Introduction
Viral infectious diseases and respiratory viral infections are among the most severe global health threats. According to the World Health Organization (WHO), millions of people are globally affected by viral diseases annually [1]. VRIs caused by different viral sources can infect the human upper and lower respiratory tracts, making the respiratory mucosa the primary gate of entry. Many viruses that could potentially lead to VARID have been reported in the current literature. Table 1 summarizes these viruses and their associated respiratory infectious disease. In this regard, SARS-CoV-2 and the influenza virus H1N1 are among the most recent to have caused global pandemics.
| Virus | VARID | Ref |
|---|---|---|
| Adenoviruses | Common Cold, Pneumonia | [2] |
| Coronaviruses | Common Cold, SARS, MERS, COVID-19 | [3] |
| Enteroviruses | Common Cold | [4] |
| Influenza Virus (Types A and B) | Influenza, Pneumonia | [5] |
| Metapneumovirus | Common Cold, Pneumonia, Bronchiolitis | [6] |
| Parainfluenza Virus (Type 3) | Common Cold, Croup, Pneumonia, Bronchiolitis | [7] |
| Parainfluenza Viruses (Types 1, 2) | Croup | [7] |
| Respiratory Syncytial viruses | Pneumonia, Bronchiolitis | [8] |
| Rhinoviruses | Common Cold | [9] |
2. Anti-Viral Responses of the Immune System in VARID and Evidence of Nanomedicine
2.1. Physical-Mucosal Barriers from Saliva to Bronchus-Associated Lymphoid Tissue
The physical-mucosal barriers from the oral and nasal cavities to the deepest regions of the lungs are considered the first line of defense in the innate immune system [10]. Alongside these physicochemical barriers, scattered lymphatic regions in the basal side of the respiratory tract (e.g., Nasal Associated Lymphoid Tissue (NALT) in the nasal cavities and mucosal associated lymphoid tissue in the mucosal layer of the respiratory tract) have a critical role in tropic anti-viral immunity responses [11].
The oral cavity is another physical barrier with enzyme-containing saliva and other NALT layers [12]. The contents of the saliva and nose mucosa in the respiratory tract determine the type of immune response. Given that the mouth and nose are the primary gateways for virus entry into the pulmonary tract, passing the virus through the saliva barrier, nasal mucosa, and ciliated layers of the upper respiratory tract can lead to acute viral infection in the lungs [13]. Some periodontal therapies using silver and gold nanoparticles can improve the immunity of the oral cavity and prevent the penetration of pathogens into the epithelium [14].
Saliva content, as an element of oral immunity, includes peptides, enzymes, and immune/epithelial cells-derived cytokines. Host defense peptides (HDP or antimicrobial peptides (AMP)) of the innate immune system, such as Cathelicidin (LL-37), α, β defensins, lactoferrin, lysozyme, and heterotypic salivary agglutinin (gp340, DMBT1) are secreted from the epithelial cells of the mouth, can consequently block the virus entry into epithelial cells or inhibit virus pathogenesis [15].
One of the most important AMPs in saliva and mucus is Cathelicidin (LL37); known for its antimicrobial role via lipopolysaccharide (LPS)-binding function, the anti-viral impacts were recently established in rhinoviruses and influenza [16][17][18]. Although saliva-derived HDPs and other antimicrobial elements might be used in the formulation of some nanoparticles, the saliva content can impact the fate of oral nanoparticles [19]. Nowadays, some nano-antibiotics have been developed, containing membrane-active human LL37 and synthetic compounds that mimic antimicrobial peptides, such as ceragenins [20].
Alveolar macrophages (AMQ) are the most prevalent immune cells located in all parts of the respiratory tract. AMQs stabilize the hemostasis of the alveolar regions through rapid recognition of infections and activation of immune responses, such as DCs, T cells, and B cells. AMQ has an important immunomodulatory role in mucosal immunity and can cause the cytokine storm initiation via TNF-α, IFN-g, and IL-6 production, or cascade repair beginning through TGF-β and culminating in IDO release due to viral infection [21]. The pathogenesis of IL-6 and AMQs in alveolar thickness and fibrosis induction is important for immunopathogenesis in VARIDS, which is described in SARS-CoV-2 infection and illustrated in Figure 1.
2.2. Surfactant Role in Viral Infection
Pulmonary surfactant is a layer of phospho-lipoprotein compound secreted by Type 2 alveolar epithelial cells that cover the surface of the alveoli. This surfactant layer has amphipathic properties and traps water in the mucosal barrier to regulate the alveoli's size and tension. There are four important functions of surfactants in anti-viral immunity and homeostasis, including anti-viral immunity enhancement, inhibition of viral infectivity, inflammation regulation, and virus entry facilitating. The pulmonary surfactants are categorized into four subtypes (SP-A, SP-B, SP-C, and SP-D) depending on their hydrophobic features and protein contents. Generally, SP-B and SP-C are more hydrophobic and smaller, while the anti-viral immunity of surfactants is more related to SP-A and SP-D. These compounds can bind to the viral glycoproteins and facilitate phagocytosis; the loss of SP-A would lead to delayed virus clearance. The amphipathic properties of these surfactants make them suitable candidates for anti-viral nanoparticle decoration [23].
2.3. Antiviral IFN Route
It seems that either an appropriate underlying genetic background showing a specific anti-viral response or the utilization of anti-sera or PEGylated IFNα to stimulate the immune response is significant at the incubation stage in people infected with SARS-CoV-2. The response to Type I interferon in the patients with a poor prognosis has been significantly lower than that in the recovered patients during adaptive immune responses. When using a Type I IFN for treatment in a mouse model of SARS-CoV or MERS-CoV infection, the timing of administration is essential to obtain a protective response.
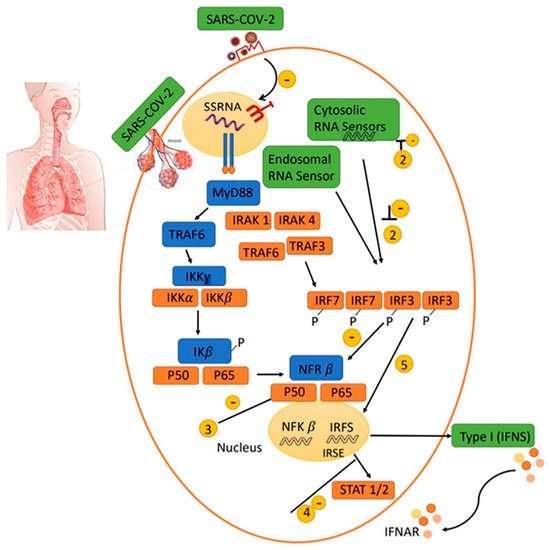
2.4. Natural and Secondary IgA
Dimeric IgA production can modulate the inflammation in the alveolar area and protect the respiratory epithelium from high inflammatory response-related damage. It occurs due to viral particles blocking and modulating the respiratory dysfunction, especially in chlamydia-dependent infections in neonates [25]. The uptake of IgA-loaded nanoparticles especially in chitin/chitosan nanoparticles within the nasal membranes following intranasal administration shows passive immunity in some respiratory diseases. Chitosan (CS) -dextran sulfate (DS) nanoparticles potentially increase the IgA-loaded combinations into nasal membranes and are widely used in intranasal formulations [26]. Some DNA vaccines have coupling capabilities with poly-lactide-co-glycolide (PLGA) and boost IgA production against the respiratory syncytial virus (RSV) in acute respiratory disease caused by RSV in children [27].
2.5. Cell-Mediated Immunity (CMI)
Cell-mediated immunity (CMI) is a specific immune response for the destruction of cells infected with viruses and subsequently protects the body against cancers, fungi, protozoa, and bacteria. Generally, virus-infected cells activate CMI, causing CD4 or T helper cells to affect the appearance of phagocytes, antigen-specific cytotoxic T lymphocytes (CTLs), and the secretion of various cytokines against the antigen. CTL activation is dependent on DC interactions and antigen presentation. In this regard, some polymeric nanoparticles such as PEI-coated PLGA NPs can stimulate the specific DC generation to stimulate specific anti-viral CTL [28].
3. Anti-Viral Systemic or Local Nano-Vaccination and Immunotherapy
Vaccination is a general strategy for the control of infectious diseases and is considered a significant choice for fighting viral diseases [29]. Due to several limitations (e.g., failure to trigger the immune system, potential of high toxicity, invasive administration, low in vivo stability, storage, and transport temperatures requirement), the clinical outcomes of some vaccines against different viral infections are not significant enough [30]. However, with emerging new formulations of vaccines (i.e., nano-vaccines), many of the shortcomings of conventional vaccination protocols are successfully addressed. Nano-vaccines can induce and enhance both humoral and cell-mediated immune responses in a more effective way than their former generations [31].
VARID nano-vaccines lead to a specific immune response using inactivated pathogens, attenuated virus, or subunit protein antigens. Examples of inactivated virus vaccine formulations for seasonal respiratory diseases, such as influenza include Influvac® [32], Vaxigrip® [33], and Fluzone® [34] against influenza Type A and Type B viruses. Examples of attenuated virus vaccine formulations include Nasovac® and Flumist® [35][36]. During the ongoing SARS-CoV-2 pandemic, a significant number of vaccines have been designed using nanotechnologies such as Pfizer®, Moderna, NovaVax, Sinopharm, Sanofi–GSK, and others.
The most common strategies in viral respiratory vaccine solutions are to encapsulate antigens/epitopes within the nanoparticles to protect the structure of antigens from proteolytic degradation, as well as to deliver the antigen/epitopes to APCs and NALT. Another efficient reported strategy is the conjugation of antigens or epitopes on the surface of the polymer nanoparticles through which the viral behavior is mimicked [37][38][39].
The SARS-CoV-2 receptor-binding domain (RBD) on S protein binds strongly to human and bat angiotensin-converting enzyme 2 (ACE2) receptors, resulting in specific humoral responses through RBD-specific antibodies secretion neutralization. This method has exciting potential for use in developing RBD-based vaccines against SARS-CoV-2 infections [40].
In general, there are two types of vaccination methods in VARID: Systemic vaccination and intranasal vaccination. Alternatively, in intranasal vaccination, stable vaccines are formulated, such as mRNA loaded LNPs and are introduced to the nasal cavity through swaps or sprays.
Compared to conventional approaches, recent advances in vaccine nanomedicine offer superior therapeutic potential for viral respiratory diseases [41]. The unique features of the nanoparticles, including small particle size (100–200 nm), adjustable surface charge, and specific surfaces, resulting in a powerful platform for pharmaceutical applications and medicine.
3.1. Nanocarriers for Targeted Anti-Viral Drug Delivery and Nano-Vaccine Design
3.1.1. Liposomes
Liposomes are spherical shape lipid nanoparticles (20–200 nm) composed of a synthetic or natural phospholipid bilayer. Liposomes mimic cell membrane’s structure and can carry both hydrophilic and lipophilic pharmaceutics compounds [42]. The vesicle size of the liposomes for drug and vaccine delivery can have a significant role in triggering the immune activation process as soon as introducing to the body through the development of different pathways, such as Th1- or Th2 responses [43]. Additionally, they can stimulate the APCs uptake rate based on their size and surface charge. The surface charge of the liposomes can also have a giant effect on the rate of Ag loading efficacy through either entrapment or electrostatic adsorption methods [44].
3.1.2. Polymeric Nanoparticles
Polymeric nanoparticles (PNs) are another example of nanocarriers for anti-viral therapeutic delivery systems. PNs are generally composed of monomeric units in the form of a colloidal phase, categorized into synthetic polymers, natural polymers, and copolymers. Besides the application of polymeric nanostructures in the pharmaceutical industry, PNs have multiple advantages for vaccine delivery applications, including controlled release of antigens, intracellular persistence in APCs, and adjustable properties such as size, composition, and surface properties [45].
Commonly used polymeric nanoparticles for such applications are PLGA, poly-ε caprolactone (PCL), poly-(γ-glutamic acid) (γ-PGA), polymethylmethacrylate (PMMA), poly-alkyl-cyanoacrylates, polyvinyl pyridine, polygluteraldehyde, polyacrylamides, polyethyleneimine (PEI), gelatin chitosan, and human serum albumin (HSA) [46]. Similar to liposomes, PNs are quickly taken up by the RES and Kupffer cells, with similar effects [47]. In one study, a poly (ethylene oxide)-modified poly (ε-caprolactone) (PEO-PCL) nanoparticulate system was developed for the encapsulation of saquinavir (SQV), an antiretroviral agent, using a solvent displacement process. THP-1 cells of the monocyte/macrophage origin demonstrated rapid cellular uptake of the encapsulated PEO-PCL nanoparticles. Intracellular SQV concentrations of the PEO-PCL-SQV nanoparticles were significantly higher than that of aqueous SQV solutions, indicating their benefits in viral therapy [48]. As previously mentioned, modified PEG is used in the final compound of both Pfizer® and Moderna® mRNA-based vaccines. NVX-CoV2373 (Novavax) is a protein-based vaccine containing saponin Matrix-M™ adjuvant [49]. With the use of polymeric nanoparticles, drug molecules are protected and both therapy and imaging can be combined [50][51]. Other promising characteristics of the polymeric nanoparticles such as biocompatibility [52], long-time spatiotemporal stability [53][54], and pathogen-like characteristics [55] make them a suitable candidate for intranasal vaccine administration [56].
3.1.3. Dendrimers
Dendrimers are a group of star-shaped three-dimensional macromolecular networks with some particular properties that make them a lucrative nanocarrier for anti-viral therapy. KK-46 dendrimer is a peptide-based compound used for intracellular delivery of anti-SARS-CoV-2 siRNA for inhibition of virus replication [57]. Finally, astrodimer sodium is a four-lysine dendrimer with a polyanionic charge that has been shown to inhibit viral infections in VeroE6 cells and reduce the replication of the virus [58].
3.1.4. Quantum Dots and Inorganic Nanoparticle
Quantum dots (QDs) are semiconductor inorganic nanocrystals with size-dependent optical and electronic properties and have been widely used for virus detection and imaging, given their inherent fluorescent emission [59]. QDs can also be used in viral replication inhibition approaches due to their inherent additional anti-viral capabilities.
3.1.5. SAPNs and VLPs
Self-assembling protein and peptide nanoparticles (SAPNs) are complexes made from monomeric protein oligomerization using recombinant technologies and are considered suitable candidates for pharmaceutical nanocarriers [63]. They can be formed in nano-diameter ranges and used as nano-vaccine candidates against viruses, making them suitable for intranasal delivery [63][64]. They can be designed to mimic viruses or bacteria in size and surface antigenicity and have been reported to elicit CD8+ T cell responses.
VLPs are another type of nano-vaccines that mimic the structure and the antigenic epitopes of their virus without including genetic material. They also promote efficient phagocytosis by APCs and immune response activation [65][66][67]. Today, ‘smart’ VLPs are often created using immunoinformatic strategies, the identification of epitopes, and artificially and genetically modifications. Construct design and viral vector engineering usually plays a very important role in this regard. Combining the VLPs with other nanoparticles is the basis of an effective vaccine [68].
4. Local Airway Delivery of Nanoparticles in VARID
4.1. Intranasal Airway Delivery of Therapeutic Nano-Carriers in VARID
Nanotechnology can potentially facilitate the efficacy of advanced therapeutics or vaccines by encapsulation inside the micro/nano-carriers to be administered using intranasal inhalation, as opposed to systematic delivery. In this way, Broichsitter et al. claimed that the anti-inflammatory corticosteroid Salbutamol could be effectively loaded in a polymeric nanocarrier composed of poly (vinyl sulfonate-co-vinyl alcohol)- graft-poly (D, L-lactide-co-glycolide, PLGA) for sustained pulmonary drug release [69]. To further enhance the selectivity of vaccine/drug delivery, the nanocarrier can be designed to have a targeted and smart release approach through stimuli-responsive delivery systems. As an example, the anti-inflammatory therapeutic hydroxy benzyl alcohol was incorporated into polyoxalate, which response to hydrogen peroxide. The drug incorporated polymer was then encapsulated inside PLGA nanoparticles. The results showed that the cleavage of peroxalate ester links between the drug and the polyoxalate polymer in the presence of hydrogen peroxide releases the drug to improve selectivity and environmental responsivity in drug delivery [70][71].
Other specific drugs, such as antibiotics can also be encapsulated inside nano-carriers to enhance the efficacy of therapy against bacterial lung infections through intranasal administration [72][73]. In the same way, the co-encapsulation of multiple antimicrobial agents can potentially improve the efficacy of the VARID treatment process [73].
Airway delivery of therapeutic nucleic acids (DNA and RNA) is also a practical approach for the treatment of side effect diseases caused by a viral infection.
Airway delivery of drug-loaded nano-carriers to the lungs and other parts of the respiratory tract could be performed by various methods such as nasal or oral spray, nebulization, dry powder inhaler devices or pressurized metered-dose inhalers [74].
In the case of SARS-CoV-2 as a type of VARID, various FDA-approved prescribed drugs have been evaluated for the treatment of infected patients [75][76][77][78]. However, despite considerable nanotechnology research and patent publications on various aspects of the coronavirus treatments [79][80], there is only one report on the utilization of nanotechnology-based design to address SARS-CoV-2 VARID using a localized airway delivery route. This nano-formulation of pharmaceutics design suggests the application of a previously developed nano-carrier based on chitosan (Novochizol™) for delivery of potential anti-COVID-19 drugs to the lungs [81]. Therefore, there is a remarkable capacity for the development of new drug formulations to prevent and/or treat the newly emerged SARS-CoV-2 virus, using nanotechnology enhanced airway delivery drugs through modulation of molecular targets or treatments of VARID. Aerosol liposomal therapy has also been used for several years with acceptable and safe clinical results [82][83], in terms of potential SARS-CoV-2 infection prevention and treatment, some reports claimed the efficiency of inhalation and oral use of a liposomal formulation of lactoferrin [84].
4.2. Intranasal Airway Delivery of Nano-Vaccines in VARID
Examples of intranasal vaccines against VARID are summarized in Table 3.
| Type of Nanoparticle | Main Material | Size (nm) | Target Respiratory Virus | Antigen/Epitope | Ref. |
|---|---|---|---|---|---|
| Polymeric | PLGA | 225 | Bovine parainfluenza 3 virus (BPI3V) |
BPI3V proteins | [85] |
| PLGA | 200–300 | Swine influenza virus (H1N2) | Inactivated virus H1N2 antigen | [82] | |
| γ-PGA | 100–200 | Influenza (H1N1) | Hemagglutinin | [86] | |
| Chitosan | 140 | Influenza (H1N1) | H1N1 antigen | [83] | |
| Chitosan | 300–350 | Influenza (H1N1) | HA-Split | [87] | |
| Chitosan | 572 | Swine influenza virus (H1N2) | Killed swine influenza antigen | [84] | |
| Chitosan | 200–250 | Influenza (H1N1) | M2e peptide | [88] | |
| HPMA/NIPAM | 12–25 | RSV | F protein | [89] | |
| PEG | 40–500 | RSV | F protein | [76] | |
| SA-CPH copolymer | 348–397 | RSV | Eα peptide | [90] | |
| CPH-CPTEG copolymer | - | RSV | F and G glycoproteins | [91] | |
| Self-assembled proteins and peptides (SANP) | Nucleocapsid (N) protein of RSV | 15 | RSV | RSV phosphoprotein | [92] |
| Nucleocapsid (N) protein of RSV | 15 | RSV | FsII epitope | [92] | |
| Nucleocapsid (N) protein of RSV | 15 | Influenza (H1N1) | M2e peptide | [93] | |
| Ferritin | 12.5 | Influenza (H1N1) | M2e peptide | [94] | |
| Influenza acid polymerase and the Q11 self-assembly domain | - | Influenza (H1N1) | Acid polymerase | [95] | |
| Inorganic | gold | 12 | Influenza (H1N1, H3N2, H5N1) | M2e peptide | [37] |
| VLP | - | - | Influenza (H1N1) | Hemagglutinin | [96] |
| - | 80–120 | Influenza (H1N1, H3N2, H5N1) | M2e5x peptide | [67] | |
| - | 60–80 | RSV | F protein et G glycoprotein of RSV and M1 protein of Influenza |
[97] | |
| Liposome | DLPC | 30–100 | Influenza (H1N1) | M2, HA, NP | [98] |
| Liposome, Polymer | 10:1:1:1 of DPPC, DPPG, Cholesterol (Chol), and DPPE-PEG2000 | 89 | SARS-COV 2 | S+ STING agonist | [99] |
| LNP | ChAdenovirus (S) | - | SARS-COV 2 | ChAd-S | [81] |
5. Conclusions and Future Perspectives
References
- Sohrabi, C.; Alsafi, Z.; O’Neill, N.; Khan, M.; Kerwan, A.; Al-Jabir, A.; Iosifidis, C.; Agha, R. World Health Organization declares global emergency: A review of the 2019 novel coronavirus (COVID-19). Int. J. Surg. 2020, 76, 71–76.
- Lynch, J.P., 3rd; Kajon, A.E. Adenovirus: Epidemiology, Global Spread of Novel Serotypes, and Advances in Treatment and Prevention. Semin. Respir. Crit. Care Med. 2016, 37, 586–602.
- Singhal, T. A Review of Coronavirus Disease-2019 (COVID-19). Indian J. Pediatrics 2020, 87, 281–286.
- Garcia, J.; Espejo, V.; Nelson, M.; Sovero, M.; Villaran, M.V.; Gomez, J.; Barrantes, M.; Sanchez, F.; Comach, G.; Arango, A.E.; et al. Human rhinoviruses and enteroviruses in influenza-like illness in Latin America. Virol. J. 2013, 10, 305.
- Macias, A.E.; McElhaney, J.E.; Chaves, S.S.; Nealon, J.; Nunes, M.C.; Samson, S.I.; Seet, B.T.; Weinke, T.; Yu, H. The disease burden of influenza beyond respiratory illness. Vaccine 2021, 39 (Suppl. 1), A6–A14.
- Williams, J.V. Human metapneumovirus: An important cause of respiratory disease in children and adults. Curr. Infect. Dis. Rep. 2005, 7, 204–210.
- Branche, A.R.; Falsey, A.R. Parainfluenza Virus Infection. Semin. Respir. Crit. Care Med. 2016, 37, 538–554.
- Meng, J.; Stobart, C.C.; Hotard, A.L.; Moore, M.L. An overview of respiratory syncytial virus. PLoS Pathog. 2014, 10, e1004016.
- Gwaltney, J.M., Jr.; Jordan, W.S., Jr. Rhinoviruses and Respiratory Disease. Bacteriol. Rev. 1964, 28, 409–422.
- Tacchi, L.; Musharrafieh, R.; Larragoite, E.T.; Crossey, K.; Erhardt, E.B.; Martin, S.A.M.; LaPatra, S.E.; Salinas, I. Nasal immunity is an ancient arm of the mucosal immune system of vertebrates. Nat. Commun. 2014, 5, 5205.
- Porzia, A.; Cavaliere, C.; Begvarfaj, E.; Masieri, S.; Mainiero, F. Human nasal immune system: A special site for immune response establishment. J. Biol. Regul. Homeost. Agents 2018, 32 (Suppl. 1), 3–8.
- Moutsopoulos, N.M.; Konkel, J.E. Tissue-Specific Immunity at the Oral Mucosal Barrier. Trends Immunol. 2018, 39, 276–287.
- Feller, L.; Altini, M.; Khammissa, R.A.G.; Chandran, R.; Bouckaert, M.; Lemmer, J. Oral mucosal immunity. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2013, 116, 576–583.
- Osorio, R.; Alfonso-Rodríguez, C.A.; Medina-Castillo, A.L.; Alaminos, M.; Toledano, M. Bioactive Polymeric Nanoparticles for Periodontal Therapy. PLoS ONE 2016, 11, e0166217.
- Corstjens, P.L.A.M.; Abrams, W.R.; Malamud, D. Saliva and viral infections. Periodontology 2000 2016, 70, 93–110.
- Sousa, F.H.; Casanova, V.; Findlay, F.; Stevens, C.; Svoboda, P.; Pohl, J.; Proudfoot, L.; Barlow, P.G. Cathelicidins display conserved direct antiviral activity towards rhinovirus. Peptides 2017, 95, 76–83.
- Tripathi, S.; Tecle, T.; Verma, A.; Crouch, E.; White, M.; Hartshorn, K.L. The human cathelicidin LL-37 inhibits influenza A viruses through a mechanism distinct from that of surfactant protein D or defensins. J. Gen. Virol. 2013, 94 Pt 1, 40–49.
- Larrick, J.W.; Morgan, J.G.; Palings, I.; Hirata, M.; Yen, M.H. Complementary DNA sequence of rabbit CAP18—A unique lipopolysaccharide binding protein. Biochem. Biophys. Res. Commun. 1991, 179, 170–175.
- Teubl, B.J.; Stojkovic, B.; Docter, D.; Pritz, E.; Leitinger, G.; Poberaj, I.; Prassl, R.; Stauber, R.H.; Fröhlich, E.; Khinast, J.G.; et al. The effect of saliva on the fate of nanoparticles. Clin. Oral Investig. 2018, 22, 929–940.
- Wnorowska, U.; Fiedoruk, K.; Piktel, E.; Prasad, S.V.; Sulik, M.; Janion, M.; Daniluk, T.; Savage, P.B.; Bucki, R. Nanoantibiotics containing membrane-active human cathelicidin LL-37 or synthetic ceragenins attached to the surface of magnetic nanoparticles as novel and innovative therapeutic tools: Current status and potential future applications. J. NanobioTechnol. 2020, 18, 3.
- Bowden, D.H. The alveolar macrophage. Environ. Health Perspect. 1984, 55, 327–341.
- Wiley, J.A.; Richert, L.E.; Swain, S.D.; Harmsen, A.; Barnard, D.L.; Randall, T.D.; Jutila, M.; Douglas, T.; Broomell, C.; Young, M.; et al. Inducible Bronchus-Associated Lymphoid Tissue Elicited by a Protein Cage Nanoparticle Enhances Protection in Mice against Diverse Respiratory Viruses. PLoS ONE 2009, 4, e7142.
- Glasser, J.R.; Mallampalli, R.K. Surfactant and its role in the pathobiology of pulmonary infection. Microbes Infect. 2012, 14, 17–25.
- Prompetchara, E.; Ketloy, C.; Palaga, T. Immune responses in COVID-19 and potential vaccines: Lessons learned from SARS and MERS epidemic. Asian Pac. J. Allergy Immunol. 2020, 38, 1–9.
- Lanka, G.K.K.; Yu, J.-J.; Gong, S.; Gupta, R.; Mustafa, S.B.; Murthy, A.K.; Zhong, G.; Chambers, J.P.; Guentzel, M.N.; Arulanandam, B.P. IgA modulates respiratory dysfunction as a sequela to pulmonary chlamydial infection as neonates. Pathog. Dis. 2016, 74, ftv121.
- Sharma, S.; Benson, H.A.; Mukkur, T.K.; Rigby, P.; Chen, Y. Preliminary studies on the development of IgA-loaded chitosan-dextran sulphate nanoparticles as a potential nasal delivery system for protein antigens. J. Microencapsul. 2013, 30, 283–294.
- Jorquera, P.A.; Tripp, R.A. Synthetic Biodegradable Microparticle and Nanoparticle Vaccines against the Respiratory Syncytial Virus. Vaccines 2016, 4, 45.
- Al-Halifa, S.; Gauthier, L.; Arpin, D.; Bourgault, S.; Archambault, D. Nanoparticle-based vaccines against respiratory viruses. Front. Immunol. 2019, 10, 1–11.
- Singh Sekhon, B.; Saluja, V. Nanovaccines-an overview. Int. J. Pharm. Front. Res. 2011, 1, 101–109.
- Marasini, N.; Skwarczynski, M.; Toth, I. Intranasal delivery of nanoparticle-based vaccines. Ther. Deliv. 2017, 8, 151–167.
- Laval, J.M.; Mazeran, P.E.; Thomas, D. NanobioTechnology and its role in the development of new analytical devices. Analyst 2000, 125, 29–33.
- Grohskopf, L.A.; Sokolow, L.Z.; Olsen, S.J.; Bresee, J.S.; Broder, K.R.; Karron, R.A. Prevention and control of influenza with vaccines: Recommendations of the Advisory Committee on Immunization Practices, United States, 2015–2016 influenza season. MMWR. Morb. Mortal. Wkly. Rep. 2015, 64, 818.
- Carter, N.J.; Curran, M.P. Live attenuated influenza vaccine (FluMist®; Fluenz™). Drugs 2011, 71, 1591–1622.
- Dhere, R.; Yeolekar, L.; Kulkarni, P.; Menon, R.; Vaidya, V.; Ganguly, M.; Tyagi, P.; Barde, P.; Jadhav, S. A pandemic influenza vaccine in India: From strain to sale within 12 months. Vaccine 2011, 29, A16–A21.
- Chattopadhyay, S.; Chen, J.; Chen, H.; Hu, C. Nanoparticle vaccines adopting virus-like features for enhanced immune potentiation. Nanotheranostics 2017, 1, 244–260.
- Fromen, C.A.; Robbins, G.R.; Shen, T.W.; Kai, M.P.; Ting, J.P.; DeSimone, J.M. Controlled analysis of nanoparticle charge on mucosal and systemic antibody responses following pulmonary immunization. Proc. Natl. Acad. Sci. USA 2015, 112, 488–493.
- Tao, W.; Hurst, B.L.; Shakya, A.K.; Uddin, M.J.; Ingrole, R.S.; Hernandez-Sanabria, M.; Arya, R.P.; Bimler, L.; Paust, S.; Tarbet, E.B. Consensus M2e peptide conjugated to gold nanoparticles confers protection against H1N1, H3N2 and H5N1 influenza A viruses. Antivir. Res. 2017, 141, 62–72.
- Tai, W.; He, L.; Zhang, X.; Pu, J.; Voronin, D.; Jiang, S.; Zhou, Y.; Du, L. Characterization of the receptor-binding domain (RBD) of 2019 novel coronavirus: Implication for development of RBD protein as a viral attachment inhibitor and vaccine. Cell Mol. Immunol. 2020, in press.
- Bhattacharya, M.; Sharma, A.R.; Patra, P.; Ghosh, P.; Sharma, G.; Patra, B.C.; Lee, S.-S.; Chakraborty, C. Development of epitope-based peptide vaccine against novel coronavirus 2019 (SARS-COV-2): Immunoinformatics approach. J. Med. Virol. 2020, 92, 618–631.
- Svindland, S.C.; Jul-Larsen, Å.; Pathirana, R.; Andersen, S.; Madhun, A.; Montomoli, E.; Jabbal-Gill, I.; Cox, R.J. The mucosal and systemic immune responses elicited by a chitosan-adjuvanted intranasal influenza H5N1 vaccine. Influenza Other Respir. Viruses 2012, 6, 90–100.
- Mallick, S.; Choi, J.S. Liposomes: Versatile and biocompatible nanovesicles for efficient biomolecules delivery. J. Nanosci. Nanotechnol. 2014, 14, 755–765.
- Perrie, Y.; Mohammed, A.R.; Kirby, D.J.; McNeil, S.E.; Bramwell, V.W. Vaccine adjuvant systems: Enhancing the efficacy of sub-unit protein antigens. Int. J. Pharm. 2008, 364, 272–280.
- Wang, N.; Chen, M.; Wang, T. Liposomes used as a vaccine adjuvant-delivery system: From basics to clinical immunization. J. Control. Release 2019, 303, 130–150.
- Henriksen-Lacey, M.; Devitt, A.; Perrie, Y. The vesicle size of DDA: TDB liposomal adjuvants plays a role in the cell-mediated immune response but has no significant effect on antibody production. J. Control. Release 2011, 154, 131–137.
- Meijer, D.; Jansen, R.; Molema, G. Drug targeting systems for antiviral agents: Options and limitations. Antivir. Res. 1992, 18, 215–258.
- Shah, L.K.; Amiji, M.M. Intracellular delivery of saquinavir in biodegradable polymeric nanoparticles for HIV/AIDS. Pharm. Res. 2006, 23, 2638–2645.
- Tian, J.-H.; Patel, N.; Haupt, R.; Zhou, H.; Weston, S.; Hammond, H.; Logue, J.; Portnoff, A.D.; Norton, J.; Guebre-Xabier, M.; et al. SARS-CoV-2 spike glycoprotein vaccine candidate NVX-CoV2373 immunogenicity in baboons and protection in mice. Nat. Commun. 2021, 12, 372.
- Krasia-Christoforou, T.; Georgiou, T. Polymeric theranostics: Using polymer-based systems for simultaneous imaging and therapy. J. Mater. Chem. B 2013, 1, 3002–3025.
- Tang, Z.; He, C.; Tian, H.; Ding, J.; Hsiao, B.S.; Chu, B.; Chen, X. Polymeric nanostructured materials for biomedical applications. Prog. Polym. Sci. 2016, 60, 86–128.
- Vela-Ramirez, J.E.; Goodman, J.T.; Boggiatto, P.M.; Roychoudhury, R.; Pohl, N.L.B.; Hostetter, J.M.; Wannemuehler, M.J.; Narasimhan, B. Safety and biocompatibility of carbohydrate-functionalized polyanhydride nanoparticles. AAPS J. 2015, 17, 256–267.
- Carrillo-Conde, B.; Schiltz, E.; Yu, J.; Chris Minion, F.; Phillips, G.J.; Wannemuehler, M.J.; Narasimhan, B. Encapsulation into amphiphilic polyanhydride microparticles stabilizes Yersinia pestis antigens. Acta Biomater. 2010, 6, 3110–3119.
- Petersen, L.K.; Phanse, Y.; Ramer-Tait, A.E.; Wannemuehler, M.J.; Narasimhan, B. Amphiphilic polyanhydride nanoparticles stabilize Bacillus anthracis protective antigen. Mol. Pharm. 2012, 9, 874–882.
- Ulery, B.D.; Petersen, L.K.; Phanse, Y.; Kong, C.S.; Broderick, S.R.; Kumar, D.; Ramer-Tait, A.E.; Carrillo-Conde, B.; Rajan, K.; Wannemuehler, M.J.; et al. Rational design of pathogen-mimicking amphiphilic materials as nanoadjuvants. Sci. Rep. 2011, 1, 1–9.
- Ross, K.A.; Haughney, S.L.; Petersen, L.K.; Boggiatto, P.; Wannemuehler, M.J.; Narasimhan, B. Lung deposition and cellular uptake behavior of pathogen-mimicking nanovaccines in the first 48 hours. Adv. Healthc. Mater. 2014, 3, 1071–1077.
- Acharya, S.; Sahoo, S.K. PLGA nanoparticles containing various anticancer agents and tumour delivery by EPR effect. Adv. Drug Deliv. Rev. 2011, 63, 170–183.
- Silva, A.L.; Soema, P.C.; Slütter, B.; Ossendorp, F.; Jiskoot, W. PLGA particulate delivery systems for subunit vaccines: Linking particle properties to immunogenicity. Hum. Vaccin. Immunother. 2016, 12, 1056–1069.
- Zhang, L.-J.; Wang, S.; Xia, L.; Lv, C.; Tang, H.-W.; Liang, Z.; Xiao, G.; Pang, D.-W. Lipid-specific labeling of enveloped viruses with quantum dots for single-virus tracking. mBio 2020, 11, e00135-20.
- Huang, S.; Gu, J.; Ye, J.; Fang, B.; Wan, S.; Wang, C.; Ashraf, U.; Li, Q.; Wang, X.; Shao, L. Benzoxazine monomer derived carbon dots as a broad-spectrum agent to block viral infectivity. J. Colloid Interface Sci. 2019, 542, 198–206.
- Loczechin, A.; Séron, K.; Barras, A.; Giovanelli, E.; Belouzard, S.; Chen, Y.-T.; Metzler-Nolte, N.; Boukherroub, R.; Dubuisson, J.; Szunerits, S. Functional Carbon Quantum Dots as Medical Countermeasures to Human Coronavirus (HCoV). ACS Appl. Mater. Interfaces 2019, 11, 42964–42974.
- Haimov, E.; Weitman, H.; Polani, S.; Schori, H.; Zitoun, D.; Shefi, O. meso-Tetrahydroxyphenylchlorin-conjugated gold nanoparticles as a tool to improve photodynamic therapy. ACS Appl. Mater. Interfaces 2018, 10, 2319–2327.
- Osminkina, L.; Timoshenko, V.Y.; Shilovsky, I.; Kornilaeva, G.; Shevchenko, S.; Gongalsky, M.; Tamarov, K.; Abramchuk, S.; Nikiforov, V.; Khaitov, M. Porous silicon nanoparticles as scavengers of hazardous viruses. J. Nanopart. Res. 2014, 16, 2430.
- Huo, C.; Xiao, J.; Xiao, K.; Zou, S.; Wang, M.; Qi, P.; Liu, T.; Hu, Y. Pre-treatment with zirconia nanoparticles reduces inflammation induced by the pathogenic H5N1 influenza virus. Int. J. Nanomed. 2020, 15, 661.
- Deng, L.; Mohan, T.; Chang, T.Z.; Gonzalez, G.X.; Wang, Y.; Kwon, Y.-M.; Kang, S.-M.; Compans, R.W.; Champion, J.A.; Wang, B.-Z. Double-layered protein nanoparticles induce broad protection against divergent influenza A viruses. Nat. Commun. 2018, 9, 359.
- He, L.; Lin, X.; Wang, Y.; Abraham, C.; Sou, C.; Ngo, T.; Zhang, Y.; Wilson, I.A.; Zhu, J. Single-component, self-assembling, protein nanoparticles presenting the receptor binding domain and stabilized spike as SARS-CoV-2 vaccine candidates. Sci. Adv. 2021, 7, eabf1591.
- Pimentel, T.A.P.F.; Yan, Z.; Jeffers, S.A.; Holmes, K.V.; Hodges, R.S.; Burkhard, P. Peptide nanoparticles as novel immunogens: Design and analysis of a prototypic severe acute respiratory syndrome vaccine. Chem. Biol. Drug Des. 2009, 73, 53–61.
- Xu, R.; Shi, M.; Li, J.; Song, P.; Li, N. Construction of SARS-CoV-2 Virus-Like Particles by Mammalian Expression System. Front. Bioeng. Biotechnol. 2020, 8, 862.
- Lee, Y.; Ko, E.; Lee, Y.; Kim, K.; Kim, M.; Lee, Y.; Kang, S. Intranasal vaccination with M2e5x virus-like particles induces humoral and cellular immune responses conferring cross-protection against heterosubtypic influenza viruses. PLoS ONE 2018, 13, e0190868.
- Swann, H.; Sharma, A.; Preece, B.; Peterson, A.; Eldridge, C.; Belnap, D.M.; Vershinin, M.; Saffarian, S. Minimal system for assembly of SARS-CoV-2 virus like particles. Sci. Rep. 2020, 10, 21877.
- Alhajlan, M.; Alhariri, M.; Omri, A. Efficacy and safety of liposomal clarithromycin and its effect on Pseudomonas aeruginosa virulence factors. Antimicrob. Agents Chemother. 2013, 57, 2694–2704.
- Silva, L.F.C.; Kasten, G.; de Campos, C.E.M.; Chinelatto, A.L.; Lemos-Senna, E. Preparation and characterization of quercetin-loaded solid lipid microparticles for pulmonary delivery. Powder Technol. 2013, 239, 183–192.
- Liu, C.; Shi, J.; Dai, Q.; Yin, X.; Zhang, X.; Zheng, A. In-vitro and in-vivo evaluation of ciprofloxacin liposomes for pulmonary administration. Drug Dev. Ind. Pharm. 2015, 41, 272–278.
- Troy, N.M.; Bosco, A. Respiratory viral infections and host responses; insights from genomics. Respir. Res. 2016, 17, 156.
- Mehta, M.; Tewari, D.; Gupta, G.; Awasthi, R.; Singh, H.; Pandey, P.; Chellappan, D.K.; Wadhwa, R.; Collet, T.; Hansbro, P.M.; et al. Oligonucleotide therapy: An emerging focus area for drug delivery in chronic inflammatory respiratory diseases. Chem. Biol. Interact. 2019, 308, 206–215.
- Kandil, R.; Merkel, O.M. Pulmonary delivery of siRNA as a novel treatment for lung diseases. Ther. Deliv. 2019, 10, 203–206.
- Liu, C.; Zhou, Q.; Li, Y.; Garner, L.V.; Watkins, S.P.; Carter, L.J.; Smoot, J.; Gregg, A.C.; Daniels, A.D.; Jervey, S.; et al. Research and development on therapeutic aents and vaccines for COVID-19 and related human coronavirus diseases. ACS Cent. Sci. 2020, 6, 315–331.
- Francica, J.R.; Lynn, G.M.; Laga, R.; Joyce, M.G.; Ruckwardt, T.J.; Morabito, K.M.; Chen, M.; Chaudhuri, R.; Zhang, B.; Sastry, M. Thermoresponsive polymer nanoparticles co-deliver RSV F trimers with a TLR-7/8 adjuvant. Bioconjug. Chem. 2016, 27, 2372–2385.
- Chan, W.C. Nano Research for COVID-19; ACS Publications: Washington, DC, USA, 2020.
- Taylor, K.M.; Fan, S.J. Liposomes for drug delivery to the respiratory tract. Drug Dev. Ind. Pharm. 1993, 19, 123–142.
- Wyde, P.R.; Six, H.R.; Wilson, S.Z.; Gilbert, B.E.; Knight, V. Activity against rhinoviruses, toxicity, and delivery in aerosol of enviroxime in liposomes. Antimicrob. Agents Chemother. 1988, 32, 890–895.
- Serrano, G.; Kochergina, I.; Albors, A.; Diaz, E.; Oroval, M.; Hueso, G.; Serrano, J.M. Liposomal Lactoferrin as Potential Preventative and Cure for COVID-19. Int. J. Res. Health Sci. 2020, 8, 8–15.
- Hassan, A.O.; Kafai, N.M.; Dmitriev, I.P.; Fox, J.M.; Smith, B.K.; Harvey, I.B.; Chen, R.E.; Winkler, E.S.; Wessel, A.W.; Case, J.B.; et al. A Single-Dose Intranasal ChAd Vaccine Protects Upper and Lower Respiratory Tracts against SARS-CoV-2. Cell 2020, 183, 169–184.e13.
- Dhakal, S.; Hiremath, J.; Bondra, K.; Lakshmanappa, Y.S.; Shyu, D.-L.; Ouyang, K.; Kang, K.-I.; Binjawadagi, B.; Goodman, J.; Tabynov, K. Biodegradable nanoparticle delivery of inactivated swine influenza virus vaccine provides heterologous cell-mediated immune response in pigs. J. Control. Release 2017, 247, 194–205.
- Liu, Q.; Zheng, X.; Zhang, C.; Shao, X.; Zhang, X.; Zhang, Q.; Jiang, X. Conjugating influenza a (H1N1) antigen to n-trimethylaminoethylmethacrylate chitosan nanoparticles improves the immunogenicity of the antigen after nasal administration. J. Med. Virol. 2015, 87, 1807–1815.
- Dhakal, S.; Renu, S.; Ghimire, S.; Shaan Lakshmanappa, Y.; Hogshead, B.T.; Feliciano-Ruiz, N.; Lu, F.; HogenEsch, H.; Krakowka, S.; Lee, C.W. Mucosal immunity and protective efficacy of intranasal inactivated influenza vaccine is improved by chitosan nanoparticle delivery in pigs. Front. Immunol. 2018, 9, 934.
- Mansoor, F.; Earley, B.; Cassidy, J.P.; Markey, B.; Doherty, S.; Welsh, M.D. Comparing the immune response to a novel intranasal nanoparticle PLA vaccine and a commercial BPI3V vaccine in dairy calves. BMC Vet. Res. 2015, 11, 220.
- Okamoto, S.; Matsuura, M.; Akagi, T.; Akashi, M.; Tanimoto, T.; Ishikawa, T.; Takahashi, M.; Yamanishi, K.; Mori, Y. Poly (γ-glutamic acid) nano-particles combined with mucosal influenza virus hemagglutinin vaccine protects against influenza virus infection in mice. Vaccine 2009, 27, 5896–5905.
- Sawaengsak, C.; Mori, Y.; Yamanishi, K.; Mitrevej, A.; Sinchaipanid, N. Chitosan nanoparticle encapsulated hemagglutinin-split influenza virus mucosal vaccine. AAPS PharmSciTech 2014, 15, 317–325.
- Dabaghian, M.; Latifi, A.M.; Tebianian, M.; NajmiNejad, H.; Ebrahimi, S.M. Nasal vaccination with r4M2e.HSP70c antigen encapsulated into N-trimethyl chitosan (TMC) nanoparticulate systems: Preparation and immunogenicity in a mouse model. Vaccine 2018, 36, 2886–2895.
- Lynn, G.M.; Laga, R.; Darrah, P.A.; Ishizuka, A.S.; Balaci, A.J.; Dulcey, A.E.; Pechar, M.; Pola, R.; Gerner, M.Y.; Yamamoto, A.; et al. In vivo characterization of the physicochemical properties of polymer-linked TLR agonists that enhance vaccine immunogenicity. Nat. Biotechnol. 2015, 33, 1201–1210.
- Ulery, B.D.; Phanse, Y.; Sinha, A.; Wannemuehler, M.J.; Narasimhan, B.; Bellaire, B.H. Polymer chemistry influences monocytic uptake of polyanhydride nanospheres. Pharm. Res. 2009, 26, 683–690.
- McGill, J.L.; Kelly, S.M.; Kumar, P.; Speckhart, S.; Haughney, S.L.; Henningson, J.; Narasimhan, B.; Sacco, R.E. Efficacy of mucosal polyanhydride nanovaccine against Respiratory Syncytial Virus infection in the neonatal calf. Sci. Rep. 2018, 8, 3021.
- Hervé, P.-L.; Deloizy, C.; Descamps, D.; Rameix-Welti, M.-A.; Fix, J.; McLellan, J.S.; Eléouët, J.-F.; Riffault, S. RSV N-nanorings fused to palivizumab-targeted neutralizing epitope as a nanoparticle RSV vaccine. Nanomedicine 2017, 13, 411–420.
- Hervé, P.-L.; Raliou, M.; Bourdieu, C.; Dubuquoy, C.; Petit-Camurdan, A.; Bertho, N.; Eléouët, J.-F.; Chevalier, C.; Riffault, S. A novel subnucleocapsid nanoplatform for mucosal vaccination against influenza virus that targets the ectodomain of matrix protein 2. J. Virol. 2014, 88, 325–338.
- Qi, M.; Zhang, X.-E.; Sun, X.; Zhang, X.; Yao, Y.; Liu, S.; Chen, Z.; Li, W.; Zhang, Z.; Chen, J.; et al. Intranasal nanovaccine confers homo- and hetero-subtypic influenza protection. Small 2018, 14, 1703207.
- Kim, T.W.; Lee, J.H.; Hung, C.F.; Peng, S.; Roden, R.; Wang, M.C.; Viscidi, R.; Tsai, Y.C.; He, L.; Chen, P.J.; et al. Generation and characterization of DNA vaccines targeting the nucleocapsid protein of severe acute respiratory syndrome coronavirus. J. Virol. 2004, 78, 4638–4645.
- Tao, W.; Ziemer, K.S.; Gill, H.S. Gold nanoparticle-M2e conjugate coformulated with CpG induces protective immunity against influenza A virus. Nanomedicine 2014, 9, 237–251.
- Quan, F.-S.; Huang, C.; Compans, R.W.; Kang, S.-M. Virus-like particle vaccine induces protective immunity against homologous and heterologous strains of influenza virus. J. Virol. 2007, 81, 3514–3524.
- Tai, W.; Roberts, L.; Seryshev, A.; Gubatan, J.M.; Bland, C.S.; Zabriskie, R.; Kulkarni, S.; Soong, L.; Mbawuike, I.; Gilbert, B.; et al. Multistrain influenza protection induced by a nanoparticulate mucosal immunotherapeutic. Mucosal Immunol. 2011, 4, 197–207.
- An, X.; Martinez-Paniagua, M.; Rezvan, A.; Fathi, M.; Singh, S.; Biswas, S.; Pourpak, M.; Yee, C.; Liu, X.; Varadarajan, N. Single-dose intranasal vaccination elicits systemic and mucosal immunity against SARS-CoV-2. bioRxiv 2020.





