
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Amina Tucak-Smajić | + 4533 word(s) | 4533 | 2021-07-09 11:58:34 | | | |
| 2 | Lindsay Dong | Meta information modification | 4533 | 2021-07-28 15:14:25 | | |
Video Upload Options
Microneedles (MNs) represent the concept of attractive, minimally invasive puncture devices of micron-sized dimensions that penetrate the skin painlessly and thus facilitate the transdermal administration of a wide range of active substances. MNs have been manufactured by a variety of production technologies, from a range of materials, but most of these manufacturing methods are time-consuming and expensive for screening new designs and making any modifications. Additive manufacturing (AM) has become one of the most revolutionary tools in the pharmaceutical field, with its unique ability to manufacture personalized dosage forms and patient-specific medical devices such as MNs.
1. Introduction
Microneedles (MNs) represent skin-friendly puncturing devices of microscale dimensions [1] that are designed to efficiently and painlessly bypass the outermost layer of the skin, the stratum corneum (SC), which acts as a barrier for transdermal penetration of APIs, in particular for those with log p values below 1 and greater than 3 [2], by forming microchannels and thus releasing the drug into the skin’s microcirculation [3][4].
Additive manufacturing (AM), commonly known as three-dimensional (3D) printing, rapid prototyping, or solid free form fabrication (SFF), represents a family of techniques launched in the 1980s that revolutionized not only the pharmaceutical industry [5][6] but also the majority of industrial and scientific fields such as automotive [7], aerospace [8], construction [9], and consumer electronics industries [10]. The term “3D printing” was defined by the International Standard Organization (ISO) as “the fabrication of objects through the deposition of a material using a print head, nozzle, or another printer technology” [11]. The reason for the great interest in 3D printing was the possibility for fast, cost-effective, and time-saving prototyping of complex structures with high production rates, reduced material waste, and increased productivity [3][12]. More than 10 different AM technologies have been proposed since Chuck Hull’s first development and commercialization of stereolithography apparatus (SLA) back in 1986. They include material extrusion, vat photopolymerization, material and binder jetting, powder bed fusion (PBF), directed energy deposition, and sheet lamination [13][14][15], which have been used for the precise manufacture of various drug dosage forms for oral [16][17][18], transdermal [13][14][19][20], vaginal [21], and subcutaneous [22] applications, as well as implants and prosthetics, whose high tunability and complexity are unattainable by conventional techniques [3][6]. More recently, the wide range of 3D printing technologies has opened an interesting new field of research to produce MNs.
2. Microneedles: Characteristics, Classification, and Delivery Strategies
The ultimate success of MN-based drug delivery is founded on the critical parameters, which include their dimensions (shape, size, geometry), manufacturing method, and materials, as well as the type of therapeutics that could be delivered into the skin [23].
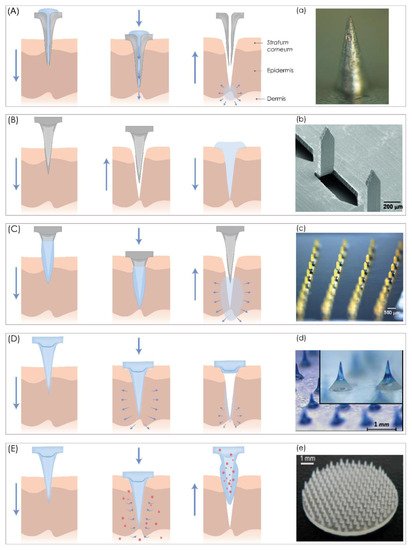
According to their structure and design, MN can be “hollow” or “solid”, but in the context of their application, MNs can be classified into five categories, hollow, solid, coated, dissolving MNs, and swelling MNs, that can be used for both TDD and extraction of interstitial fluids (Figure 1).
3. Fabrication of Microneedles Using 3D Printing Technologies
The process of 3D printing (3DP) of MNs usually contains three main steps. Firstly, a 3D object is designed with computer-aided design (CAD) software, and the geometry is optimized according to printer specifications. Then, the 3D object is exported to a common and printer-recognizable file format such as standard triangulation language (STL), which includes only 3D geometry in the form of each vertex’s position data or an OBJ file in which additional information about polygonal faces or color texture is coded [39]. Finally, the object is printed in a layer-by-layer manner [40].
3.1. Nozzle-Based Deposition Systems
Fused Deposition Modeling (FDM)
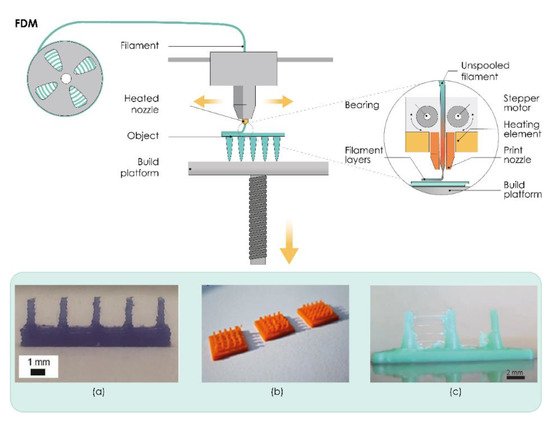
One of the most popular AM techniques is extrusion-based fused filament fabrication (FFF), also referred to as fused deposition modeling (FDM). In this process, the suitable thermoplastic material, in the form of a filament, is melted in a liquefier head at a temperature above its melting point and then selectively deposited layer-by-layer through a nozzle on a build plate, where it is cooled and solidified in less than a second, as shown in Figure 2. The printer’s head moves within the x- and y-axes, whereas the platform can move within the z-axis, thus creating 3D structures [41].
The quality and mechanical properties of the fabricated part can be attributed to the proper selection of process parameters such as nozzle diameter, feed rate, the temperature of both the nozzle and the building plate, printing speed, the height of the layers, and orientation of the built part. All these process parameters need to be studied and optimized in the FDM process to improve surface finish, strength, and other properties of the printed part [42][43].
Initial attempts to use FDM processes in the production of MNs were reported by Luzuriaga et al. (Figure 2a). Tang et al. investigated the effects of FDM process parameters, such as printing temperature, layer thickness, extrusion width, infill width, and nozzle orifice diameter on the final print quality of MNs printed with different types of PLA (Figure 2c).
3.2. Laser-Based Writing Systems
3.2.1. Stereolithography (SLA)
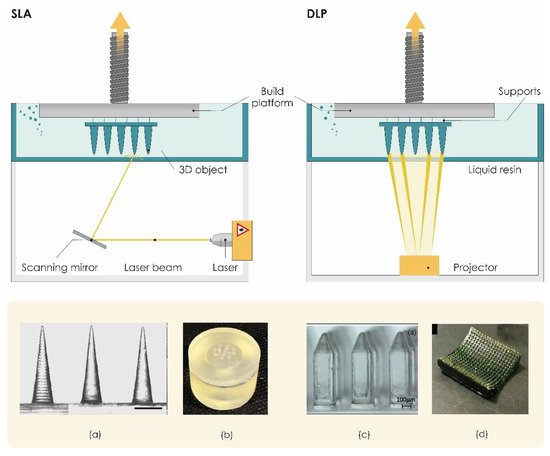
Photopolymerization-based or photocuring 3DP technologies, which are based on the ability to selectively polymerize photosensitive polymers through laser emissions or projections of light, are one of the oldest and the most widely used AM technologies in the current manufacturing industry [45]. Vat photopolymerization technologies such as stereolithography (SLA) are liquid-based processes characterized by high precision and accuracy.
SLA printers usually include a printing platform and a resin tank (Figure 3). A UV laser draws the cross-section onto a photopolymer resin bath that solidifies the cross-section. Once the first layer is completed, the platform is typically lifted about 0.05–0.15 mm according to the layer thickness [46], the laser then solidifies the next cross-section, and the process repeats until the entire part is finished. Resin that is not touched by the laser remains in the vat and can be reused. A post-process treatment may be used to achieve the desired mechanical performance [15]. The SLA processes can be divided into two main categories based on different filling mechanisms: free surface and constrained surface. In the free surface approach, structures are built bottom-up from a support platform that rests just below the resin surface while the constrained surface approach, also called the “bat” configuration, has a building platform which can be suspended above the resin bath [47].
3.2.2. Digital Light Processing (DLP)
3.2.3. Liquid Crystal Display (LCD)
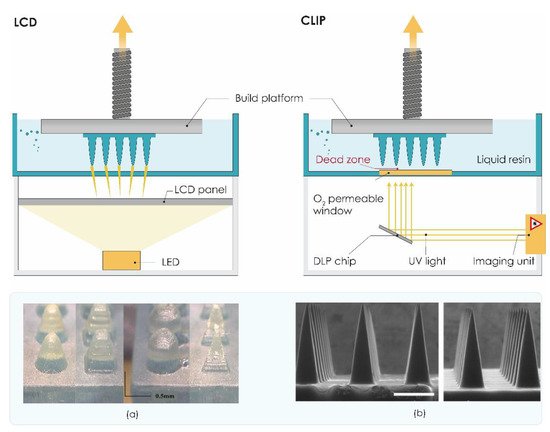
3.2.4. Continuous Liquid Interface Production (CLIP)
3.2.5. Two-Photon Polymerization (TPP/2PP)
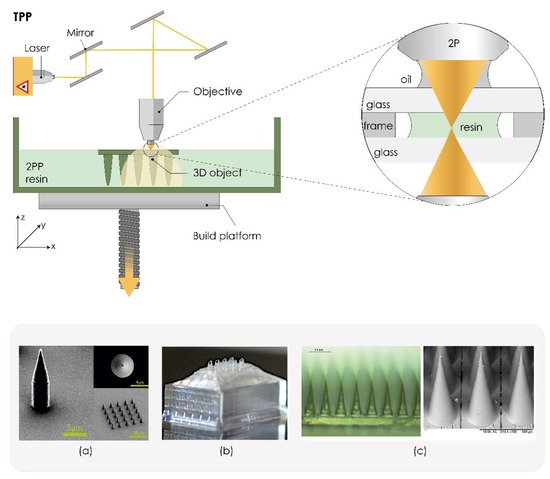
3.2.6. Powder Bed Technologies (SLS, DLMS, SLM)
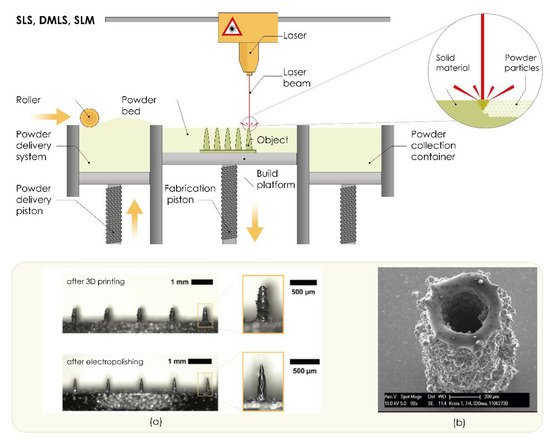
4. Parameters Affecting the Mechanical Properties of 3D Printed Microneedles
4.1. Material Selection
It is well known that materials used for MN manufacturing must have sufficient strength to penetrate biological barriers, must be easily manufactured, and be compatible with drug molecules. Material properties affect critical parameters such as stability, tensile strength, drug loading, and biocompatibility of MNs [70][71]. Biocompatibility and safety are of the utmost importance when it comes to material selection for MN manufacturing. Although the AM technology allows the use of different materials, all materials must fulfill properties such as biocompatibility, biodegradability without toxic products, mechanical strength, and scalability to be used in the development of TDD systems such as MNs [72]. This can seriously limit the expansion of 3D printing in MN manufacturing, as the material selection can be challenging.
4.2. Precision of 3D Printing
4.3. Microneedle Design

5. Evaluation of 3D printed MNs
5.1. Physical Characterization
Geometry, dimensions, surface morphology, and distribution of MNs on the array can be determined and evaluated by visual inspection, stereomicroscopy, and optical or scanning electron microscopy [77]. Properties of the surface of MN patches can be evaluated by drop shape analysis and contact angle determination [53].
Fluorescent labeling or dyeing the molecules incorporated in the MN patch can be used for their identification. Successfully incorporated molecules may be visualized by confocal laser scanning microscopy (CLSM), fluorescent microscopy, or even visual inspection. Visualization is useful for the localization of molecules incorporated within an MN patch whether it is the tip, shaft, or the backing layer [78]. Coated MNs can be evaluated by FTIR spectroscopy [53].
5.2. Mechanical Characterization
Mechanical characterization is necessary to ensure the safe use of MNs. MNs tend to bend, fracture, or buckle due to inelastic or elastic instability during insertion or removal, so it is of great importance to evaluate their mechanical properties. Adequate mechanical strength is required to penetrate the SC and deliver the drug. The term mechanical characterization comprises a range of tests that provide the simulation of MNs’ insertion in vivo [2].
5.2.1. Failure Force Tests
Axial Fracture Force Tests
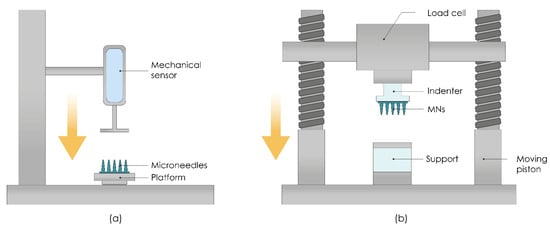
This type of test involves measuring the failure of MNs caused by axial or transverse loading. The test station presses the MN array parallel against a rigid metal surface followed by the measurement of force and displacement while generating the stress against strain curves (Figure 8a). If MNs fail, the force drops suddenly and the maximum force applied immediately before the drop is the force of the MNs’ failure. Subsequently, MN arrays are visually observed by microscope and compared with scans taken before the failure to determine the failure mode [79]. Axial fracture force tests that use only a single MN should be cautiously interpreted because those results cannot always be correlated with ones taken from an MN array [80].
Transverse Fracture Force
Transverse fracture force tests are important for the assessment of the application of MNs, and they can be applied to a single MN, and a row of MNs as well, for which the force should be divided by the number of MNs in a row to calculate the transverse fracture force per individual MN. The main limitation of this test is the required manual alignment of the metal probe with a defined length, which can cause inaccuracies [83].
Baseplate Strength and Flexibility Tests
5.2.2. Insertion Force Tests
Measuring the insertion force required to pierce the skin by MNs is also important to ensure a complete and equivalent assessment of fracture forces. Fracture forces should be significantly higher than the insertion forces required for inserting MNs into the skin. MNs insertion can be done manually or by using different applicators (Figure 8b).
5.2.3. Skin Irritation and Recovery Studies
5.3. Permeation Studies
5.3.1. In Vitro Permeation Studies
The amount of the drug delivered into the skin can be determined by diffusion cells. Usually, the test is performed on ex vivo human or animal skin or an artificial polymeric membrane placed between receptor and donor compartments. The observed permeant is inserted into the donor compartment, which diffuses into the receptor compartment via the membrane of choice [85]. Transdermal delivery of investigated permeant from MNs can be analyzed from the receptor compartment by high-performance liquid chromatography (HPLC). Intradermal delivery can be examined by extraction and analysis of the compound of interest from the skin sample after permeation [77].
5.3.2. In Vivo Permeation Studies
Neither in vitro nor ex vivo models can fully replicate the in vivo conditions and all skin characteristics. In vivo studies are necessary to investigate the absorption, disposition, and permeation of drugs delivered by MNs. Thickness and elasticity of the skin are crucial parameters that must be considered while selecting the appropriate in vivo model.
Another important factor to consider when developing in vivo models for MN performance assessments is the application site. Different anatomical sites offer a varying extent of permeability and barrier function of the skin. In addition, the thickness and rigidness of the injection site, which can be affected by lymphatic uptake, play an important role in achieving reproducible results. Measuring plasma levels of tested drugs provides information about their transdermal delivery from MNs, while skin extraction provides information about intradermal delivery. In vivo permeation studies can also provide information about the metabolism of delivered drugs [77].
6. Conclusions
References
- Hong, X.; Wu, Z.; Chen, L.; Wu, F.; Wei, L.; Yuan, W. Hydrogel Microneedle Arrays for Transdermal Drug Delivery. Nano-Micro Lett. 2014, 6, 191–199.
- Larrañeta, E.; Lutton, R.E.M.; Woolfson, A.D.; Donnelly, R.F. Microneedle arrays as transdermal and intradermal drug delivery systems: Materials science, manufacture and commercial development. Mater. Sci. Eng. R Rep. 2016, 104, 1–32.
- Economidou, S.N.; Lamprou, D.A.; Douroumis, D. 3D printing applications for transdermal drug delivery. Int. J. Pharm. 2018, 544, 415–424.
- Tucak, A.; Sirbubalo, M.; Hindija, L.; Rahić, O.; Hadžiabdić, J.; Muhamedagić, K.; Čekić, A.; Vranić, E. Microneedles: Characteristics, Materials, Production Methods and Commercial Development. Micromachines 2020, 11, 961.
- Cordeiro, A.S.; Tekko, I.A.; Jomaa, M.H.; Vora, L.; McAlister, E.; Volpe-zanutto, F.; Nethery, M.; Baine, P.T.; Mitchell, N.; Mcneill, D.W.; et al. Two-Photon Polymerisation 3D Printing of Microneedle Array Templates with Versatile Designs: Application in the Development of Polymeric Drug Delivery Systems. Pharm. Res. 2020, 37, 1–15.
- Wu, M.; Zhang, Y.; Huang, H.; Li, J.; Liu, H.; Guo, Z.; Xue, L.; Liu, S.; Lei, Y. Assisted 3D printing of microneedle patches for minimally invasive glucose control in diabetes. Mater. Sci. Eng. C 2020, 117, 111299.
- Richardson, M. Designer/Maker: The Rise of Additive Manufacturing, Domestic-Scale Production and the Possible Implications for the Automotive Industry. Comput. Aided. Des. Appl. 2012, PACE, 33–48.
- Ahmed, N.A.; Page, J.R. Manufacture of an unmanned aerial vehicle (UAV) for advanced project design using 3D printing technology. In Proceedings of the Applied Mechanics and Materials; Trans Tech Publications Ltd.: Bäch SZ, Switzerland, 2013; Volume 397–400, pp. 970–980.
- Henke, K.; Treml, S. Wood based bulk material in 3D printing processes for applications in construction. Eur. J. Wood Prod. 2013, 71, 139–141.
- MacDonald, E.; Salas, R.; Espalin, D.; Perez, M.; Aguilera, E.; Muse, D.; Wicker, R.B. 3D printing for the rapid prototyping of structural electronics. IEEE Access 2014, 2, 234–242.
- International Organization for Standardization. ISO/ASTM 52900: Additive Manufacturing-General Principles-Terminology; ISO/ASME International: Geneva, Switzerland, 2015; Volume 5, pp. 1–26.
- Goole, J.; Amighi, K. 3D printing in pharmaceutics: A new tool for designing customized drug delivery systems. Int. J. Pharm. 2016, 499, 376–394.
- Luzuriaga, M.A.; Berry, D.R.; Reagan, J.C.; Smaldone, R.A.; Gassensmitha, J.J. Biodegradable 3D printed polymer microneedles for transdermal drug delivery. Lab Chip 2018, 18, 1223–1230.
- Lim, S.H.; Kathuria, H.; Tan, J.J.Y.; Kang, L. 3D printed drug delivery and testing systems—a passing fad or the future? Adv. Drug Deliv. Rev. 2018, 132, 139–168.
- Ngo, T.D.; Kashani, A.; Imbalzano, G.; Nguyen, K.T.Q.; Hui, D. Additive manufacturing (3D printing): A review of materials, methods, applications and challenges. Compos. Part B 2018, 143, 172–196.
- Maroni, A.; Melocchi, A.; Parietti, F.; Foppoli, A.; Zema, L.; Gazzaniga, A. 3D printed multi-compartment capsular devices for two-pulse oral drug delivery. J. Control. Release 2017, 268, 10–18.
- Melocchi, A.; Parietti, F.; Loreti, G.; Maroni, A.; Gazzaniga, A.; Zema, L. 3D printing by fused deposition modeling (FDM) of a swellable/erodible capsular device for oral pulsatile release of drugs. J. Drug Deliv. Sci. Technol. 2015, 30, 360–367.
- Goyanes, A.; Wang, J.; Buanz, A.; Martínez-Pacheco, R.; Telford, R.; Gaisford, S.; Basit, A.W. 3D Printing of Medicines: Engineering Novel Oral Devices with Unique Design and Drug Release Characteristics. Mol. Pharm. 2015, 12, 4077–4084.
- Camović, M.; Biščević, A.; Brčić, I.; Borčak, K.; Bušatlić, S.; Ćenanović, N.; Dedović, A.; Mulalić, A.; Osmanlić, M.; Sirbubalo, M.; et al. Coated 3D Printed PLA Microneedles as Transdermal Drug Delivery Systems. In Proceedings of the CMBEBIH 2019. IFMBE Proceedings; Badnjević, A., Škrbić, R., Gurbeta Pokvić, L., Eds.; Springer Nature: Cham, Switzerland, 2020; Volume 73, pp. 735–742.
- Pere, C.P.P.; Economidou, S.N.; Lall, G.; Ziraud, C.; Boateng, J.S.; Alexander, B.D.; Lamprou, D.A.; Douroumis, D. 3D printed microneedles for insulin skin delivery. Int. J. Pharm. 2018, 544, 425–432.
- Fu, J.; Yu, X.; Jin, Y. 3D printing of vaginal rings with personalized shapes for controlled release of progesterone. Int. J. Pharm. 2018, 539, 75–82.
- Gowers, S.A.N.; Curto, V.F.; Seneci, C.A.; Wang, C.; Anastasova, S.; Vadgama, P.; Yang, G.Z.; Boutelle, M.G. 3D Printed Microfluidic Device with Integrated Biosensors for Online Analysis of Subcutaneous Human Microdialysate. Anal. Chem. 2015, 87, 7763–7770.
- Guillot, A.J.; Cordeiro, A.S.; Donnelly, R.F.; Montesinos, M.C.; Garrigues, T.M.; Melero, A. Microneedle-based delivery: An overview of current applications and trends. Pharmaceutics 2020, 12, 569.
- Norman, J.J.; Choi, S.O.; Tong, N.T.; Aiyar, A.R.; Patel, S.R.; Prausnitz, M.R.; Allen, M.G. Hollow microneedles for intradermal injection fabricated by sacrificial micromolding and selective electrodeposition. Biomed. Microdevices 2013, 15, 203–210.
- Gupta, J.; Gill, H.S.; Andrews, S.N.; Prausnitz, M.R. Kinetics of skin resealing after insertion of microneedles in human subjects. J. Control. Release 2011, 154, 148–155.
- Gill, H.S.; Prausnitz, M.R. Coating formulations for microneedles. Pharm. Res. 2007, 24, 1369–1380.
- Kim, J.D.; Kim, M.; Yang, H.; Lee, K.; Jung, H. Droplet-born air blowing: Novel dissolving microneedle fabrication. J. Control. Release 2013, 170, 430–436.
- Yang, S.; Wu, F.; Liu, J.; Fan, G.; Welsh, W.; Zhu, H.; Jin, T. Phase-Transition Microneedle Patches for Efficient and Accurate Transdermal Delivery of Insulin. Adv. Funct. Mater. 2015, 25, 4633–4641.
- Nagarkar, R.; Singh, M.; Nguyen, H.X.; Jonnalagadda, S. A review of recent advances in microneedle technology for transdermal drug delivery. J. Drug Deliv. Sci. Technol. 2020, 59, 101923.
- Van Der Maaden, K.; Jiskoot, W.; Bouwstra, J. Microneedle technologies for (trans)dermal drug and vaccine delivery. J. Control. Release 2012, 161, 645–655.
- Ito, Y.; Hagiwara, E.; Saeki, A.; Sugioka, N.; Takada, K. Feasibility of microneedles for percutaneous absorption of insulin. Eur. J. Pharm. Sci. 2006, 29, 82–88.
- Kathuria, H.; Kang, K.; Cai, J.; Kang, L. Rapid microneedle fabrication by heating and photolithography. Int. J. Pharm. 2020, 575, 118992.
- Wilke, N.; Mulcahy, A.; Ye, S.R.; Morrissey, A. Process optimization and characterization of silicon microneedles fabricated by wet etch technology. Microelectron. J. 2005, 36, 650–656.
- Liu, Y.; Eng, P.F.; Guy, O.J.; Roberts, K.; Ashraf, H.; Knight, N. Advanced deep reactive-ion etching technology for hollow microneedles for transdermal blood sampling and drug delivery. IET Nanobiotechnology 2013, 7, 59–62.
- Camović, M.; Biščević, A.; Brčić, I.; Borčak, K.; Bušatlić, S.; Ćenanović, N.; Dedović, A.; Mulalić, A.; Sirbubalo, M.; Tucak, A.; et al. Acid-resistant capsules with sugar microneedles for oral delivery of ascorbic acid. In Proceedings of the CMBEBIH 2019. IFMBE Proceedings; Badnjević, A., Škrbić, R., Gurbeta Pokvić, L., Eds.; Springer: Cham, Switzerland, 2019; pp. 749–753.
- Donnelly, R.F.; Raj Singh, T.R.; Woolfson, A.D. Microneedle-based drug delivery systems: Microfabrication, drug delivery, and safety. Drug Deliv. 2010, 17, 187–207.
- Vranić, E.; Tucak, A.; Sirbubalo, M.; Rahić, O.; Elezović, A.; Hadžiabdić, J. Microneedle-based sensor systems for real-time continuous transdermal monitoring of analytes in body fluids. In Proceedings of the CMBEBIH 2019, IFMBE Proceedings; Badnjević, A., Škrbić, R., Gurbeta Pokvić, L., Eds.; Springer: Cham, Switzerland, 2019; pp. 167–172.
- Donnelly, R.F.; McCrudden, M.T.C.; Alkilani, A.Z.; Larrañeta, E.; McAlister, E.; Courtenay, A.J.; Kearney, M.C.; Raj Singh, T.R.; McCarthy, H.O.; Kett, V.L.; et al. Hydrogel-forming microneedles prepared from “super swelling” polymers combined with lyophilised wafers for transdermal drug delivery. PLoS ONE 2014, 9.
- Jamróz, W.; Szafraniec, J.; Kurek, M.; Jachowicz, R. 3D Printing in Pharmaceutical and Medical Applications–Recent Achievements and Challenges. Pharm. Res. 2018, 35, 176.
- Quan, H.; Zhang, T.; Xu, H.; Luo, S.; Nie, J.; Zhu, X. Photo-curing 3D printing technique and its challenges. Bioact. Mater. 2020, 5, 110–115.
- Carneiro, O.S.; Silva, A.F.; Gomes, R. Fused deposition modeling with polypropylene. Mater. Des. 2015, 83, 768–776.
- Mohamed, O.A.; Masood, S.H.; Bhowmik, J.L. Optimization of fused deposition modeling process parameters: A review of current research and future prospects. Adv. Manuf. 2015, 3, 42–53.
- Camposeco-Negrete, C. Optimization of printing parameters in fused deposition modeling for improving part quality and process sustainability. Int. J. Adv. Manuf. Technol. 2020, 108, 2131–2147.
- Tang, T.O.; Holmes, S.; Dean, K.; Simon, G.P. Design and fabrication of transdermal drug delivery patch with milliprojections using material extrusion 3D printing. J. Appl. Polym. Sci. 2020, 137, 1–17.
- ASTM Committee. F42 ISO/ASTM 52921-2013: Standard Terminology for Additive Manufacturing-Coordinate Systems and Test Methodologies. ASTM Int. 2013, 1, 1–13.
- Rahmati, S.; Ghadami, F. Process Parameters Optimization to Improve Dimensional Accuracy of Stereolithography Parts. Int. J. Adv. Des. Manuf. Technol. 2014, 7, 59–65.
- Schmidleithner, C.; Kalaskar, D.M. Stereolithography. In 3D Printing; Cvetković, D., Ed.; InTech: London, UK, 2018; pp. 3–22.
- Krieger, K.J.; Bertollo, N.; Dangol, M.; Sheridan, J.T.; Lowery, M.M.; O’Cearbhaill, E.D. Simple and customizable method for fabrication of high-aspect ratio microneedle molds using low-cost 3D printing. Microsyst. Nanoeng. 2019, 5, 42.
- Farias, C.; Lyman, R.; Hemingway, C.; Chau, H.; Mahacek, A.; Bouzos, E.; Mobed-Miremadi, M. Three-dimensional (3D) printed microneedles for microencapsulated cell extrusion. Bioengineering 2018, 5, 59.
- Lu, Y.; Mantha, S.N.; Crowder, D.C.; Chinchilla, S.; Shah, K.N.; Yun, Y.H.; Wicker, R.B.; Choi, J.W. Microstereolithography and characterization of poly(propylene fumarate)-based drug-loaded microneedle arrays. Biofabrication 2015, 7, 045001.
- Lim, S.H.; Tiew, W.J.; Zhang, J.; Ho, P.C.L.; Kachouie, N.N.; Kang, L. Geometrical optimisation of a personalised microneedle eye patch for transdermal delivery of anti-wrinkle small peptide. Biofabrication 2020, 12.
- Mohamed, M.; Kumar, H.; Wang, Z.; Martin, N.; Mills, B.; Kim, K. Rapid and Inexpensive Fabrication of Multi-Depth Microfluidic Device using High-Resolution LCD Stereolithographic 3D Printing. J. Manuf. Mater. Process. 2019, 3, 26.
- Xenikakis, I.; Tsongas, K.; Tzimtzimis, E.K.; Zacharis, C.K.; Theodoroula, N.; Kalogianni, E.P.; Demiri, E.; Vizirianakis, I.S.; Tzetzis, D.; Fatouros, D.G. Fabrication of hollow microneedles using liquid crystal display (LCD) vat polymerization 3D printing technology for transdermal macromolecular delivery. Int. J. Pharm. 2021, 597, 120303.
- Johnson, A.R.; Caudill, C.L.; Tumbleston, J.R.; Bloomquist, C.J.; Moga, K.A.; Ermoshkin, A.; Shirvanyants, D.; Mecham, S.J.; Luft, J.C.; De Simone, J.M. Single-step fabrication of computationally designed microneedles by continuous liquid interface production. PLoS ONE 2016, 11, 1–17.
- Tumbleston, J.R.; Shirvanyants, D.; Ermoshkin, N.; Janusziewicz, R.; Johnson, A.R.; Kelly, D.; Chen, K.; Pinschmidt, R.; Rolland, J.P.; Ermoshkin, A.; et al. Continuous liquid interface production of 3D objects. Science 2015, 347, 1349–1352.
- Balli, J.; Kumpaty, S.; Anewenter, V. Continuous liquid interface production of 3D objects: An unconventional technology and its challenges and opportunities. In Proceedings of the ASME International Mechanical Engineering Congress and Exposition, Proceedings (IMECE); American Society of Mechanical Engineers (ASME): New York, NY, USA, 2017; Volume 5.
- Xing, J.F.; Zheng, M.L.; Duan, X.M. Two-photon polymerization microfabrication of hydrogels: An advanced 3D printing technology for tissue engineering and drug delivery. Chem. Soc. Rev. 2015, 44, 5031–5039.
- Farsari, M.; Chichkov, B.N. Materials processing: Two-photon fabrication. Nat. Photonics 2009, 3, 450–452.
- Kavaldzhiev, M.; Perez, J.E.; Ivanov, Y.; Bertoncini, A.; Liberale, C.; Kosel, J. Biocompatible 3D printed magnetic micro needles. Biomed. Phys. Eng. Express 2017, 3, 025005.
- Moussi, K.; Bukhamsin, A.; Hidalgo, T.; Kosel, J. Biocompatible 3D Printed Microneedles for Transdermal, Intradermal, and Percutaneous Applications. Adv. Eng. Mater. 2020, 22, 1901358.
- Beg, S.; Almalki, W.H.; Malik, A.; Farhan, M.; Aatif, M.; Rahman, Z.; Alruwaili, N.K.; Alrobaian, M.; Tarique, M.; Rahman, M. 3D printing for drug delivery and biomedical applications. Drug Discov. Today 2020, 25, 1668–1681.
- Deckard, C.R..; Beaman, J.J..; Darrah, J.F. Method and Apparatus for Producing Parts by Selective Sintering. U.S. Patent 4,863,538, 5 September 1989.
- Mazzoli, A. Selective laser sintering in biomedical engineering. Med. Biol. Eng. Comput. 2013, 51, 245–256.
- Singh, R.; Singh, S.; Hashmi, M.S.J. Implant Materials and Their Processing Technologies; Elsevier Ltd.: Amsterdam, The Netherlands, 2016; ISBN 9780128035818.
- Gardan, J. Additive manufacturing technologies: State of the art and trends. Int. J. Prod. Res. 2016, 54, 3118–3132.
- Charoo, N.A.; Barakh Ali, S.F.; Mohamed, E.M.; Kuttolamadom, M.A.; Ozkan, T.; Khan, M.A.; Rahman, Z. Selective laser sintering 3D printing–an overview of the technology and pharmaceutical applications. Drug Dev. Ind. Pharm. 2020, 46, 869–877.
- Rahmati, S. 10.12. Direct Rapid Tooling. In Comprehensive Materials Processing; Hashmi, S., Batalha, G., Van Tyne, C., Yilbas, B., Eds.; Elsevier: Amsterdam, The Netherlands, 2014; Volume 10, pp. 303–344. ISBN 9780080965338.
- Krieger, K.J.; Liegey, J.; Cahill, E.M.; Bertollo, N.; Lowery, M.M.; O’Cearbhaill, E.D. Development and Evaluation of 3D-Printed Dry Microneedle Electrodes for Surface Electromyography. Adv. Mater. Technol. 2020, 5, 1–13.
- Gieseke, M.; Senz, V.; Vehse, M.; Fiedler, S.; Irsig, R.; Hustedt, M.; Sternberg, K.; Nölke, C.; Kaierle, S.; Wesling, V.; et al. Additive manufacturing of drug delivery systems. Biomed. Tech. 2012, 57, 398–401.
- Bhatnagar, S.; Gadeela, P.R.; Thathireddy, P.; Venuganti, V.V.K. Microneedle-based drug delivery: Materials of construction. J. Chem. Sci. 2019, 131.
- Ali, R.; Mehta, P.; Arshad, M.; Kucuk, I.; Chang, M.W.; Ahmad, Z. Transdermal Microneedles—A Materials Perspective. AAPS PharmSciTech 2020, 21.
- Lee, K.J.; Jeong, S.S.; Roh, D.H.; Kim, D.Y.; Choi, H.K.; Lee, E.H. A practical guide to the development of microneedle systems–In clinical trials or on the market. Int. J. Pharm. 2020, 573, 118778.
- Lee, P.; Chung, H.; Won Lee, S.; Yoo, J.; Ko, J. Review: Dimensional accuracy in additive manufacturing processes. In Proceedings of the ASME 2014 International Manufacturing Science and Engineering Conference, Detroit, MI, USA, 9–13 June 2014; pp. 1–8.
- Kjar, A.; Huang, Y. Application of micro-scale 3D printing in pharmaceutics. Pharmaceutics 2019, 11, 390.
- Economidou, S.N.; Pissinato Pere, C.P.; Okereke, M.; Douroumis, D. Optimisation of Design and Manufacturing Parameters of 3D Printed Solid Microneedles for Improved Strength, Sharpness, and Drug Delivery. Micromachines 2021, 12, 117.
- Ovsianikov, A.; Chichkov, B.; Mente, P.; Monteiro-Riviere, N.A.; Doraiswamy, A.; Narayan, R.J. Two photon polymerization of polymer-ceramic hybrid materials for transdermal drug delivery. Int. J. Appl. Ceram. Technol. 2007, 4, 22–29.
- Sabri, A.H.; Kim, Y.; Marlow, M.; Scurr, D.J.; Segal, J.; Banga, A.K.; Kagan, L.; Lee, J.B. Intradermal and transdermal drug delivery using microneedles–Fabrication, performance evaluation and application to lymphatic delivery. Adv. Drug Deliv. Rev. 2020, 153, 195–215.
- Van Der Maaden, K.; Sekerdag, E.; Schipper, P.; Kersten, G.; Jiskoot, W.; Bouwstra, J. Layer-by-Layer Assembly of Inactivated Poliovirus and N-Trimethyl Chitosan on pH-Sensitive Microneedles for Dermal Vaccination. Langmuir 2015, 31, 8654–8660.
- Park, J.-H.; Allen, M.G.; Prausnitz, M.R. Biodegradable polymer microneedles: Fabrication, mechanics and transdermal drug delivery. J. Control. Release 2005, 104, 51–66.
- Davis, S.P.; Landis, B.J.; Adams, Z.H.; Allen, M.G.; Prausnitz, M.R. Insertion of microneedles into skin: Measurement and prediction of insertion force and needle fracture force. J. Biomech. 2004, 37, 1155–1163.
- Machekposhti, S.; Soltani, M.; Najafizadeh, P.; Ebrahimi, S.A.; Chen, P. Biocompatible polymer microneedle for topical/dermal delivery of tranexamic acid. J. Control. Release 2017, 261, 87–92.
- Ryan, E.; Garland, M.J.; Singh, T.R.R.; Bambury, E.; O’Dea, J.; Migalska, K.; Gorman, S.P.; McCarthy, H.O.; Gilmore, B.F.; Donnelly, R.F. Microneedle-mediated transdermal bacteriophage delivery. Eur. J. Pharm. Sci. 2012, 47, 297–304.
- Donnelly, R.F.; Majithiya, R.; Singh, T.R.R.; Morrow, D.I.J.; Garland, M.J.; Demir, Y.K.; Migalska, K.; Ryan, E.; Gillen, D.; Scott, C.J.; et al. Design, optimization and characterisation of polymeric microneedle arrays prepared by a novel laser-based micromoulding technique. Pharm. Res. 2011, 28, 41–57.
- Kusamori, K.; Katsumi, H.; Sakai, R.; Hayashi, R.; Hirai, Y.; Tanaka, Y.; Hitomi, K.; Quan, Y.-S.; Kamiyama, F.; Yamada, K.; et al. Development of a drug-coated microneedle array and its application for transdermal delivery of interferon alpha. Biofabrication 2016, 8, 15006.
- Gupta, J.; Gupta, R. Vanshita Microneedle Technology: An Insight into Recent Advancements and Future Trends in Drug and Vaccine Delivery. Assay Drug Dev. Technol. 2021, 19, 97–114.





