
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | He Huang | + 1412 word(s) | 1412 | 2021-07-26 12:21:21 | | | |
| 2 | Vicky Zhou | Meta information modification | 1412 | 2021-07-28 07:03:09 | | |
Video Upload Options
Cellulose is a macromolecular polysaccharide linked by glucose via a β-1,4-glycosidic bond, is insoluble in water and organic solvents, and forms the plant cell wall together with hemicellulose, pectin, and lignin. Cellulases are a group of enzymes that can hydrolyze the glycosidic bonds of cellulose to produce glucose, and the members of this group include exoglucanase, endoglucanase, and β-glucosidase. The synergistic action of these enzymes can break down cellulose into glucose.
1. Introduction
Cellulases are already widely used in various biological industries, including food, wine, animal feed, laundry, pulp, and agriculture. In the food industry, cellulase can be used to extract fruit and vegetable juice, produce nectar and fruit puree, etc. The use of cellulase in the wine industry can decompose starch and cellulose into sugar and increase the wine yield. Cellulase is also a feed additive, which can reduce the nutrient loss of feed and promote digestion and absorption. Cellulase can also be added to washing powder to enhance the performance of detergents, remove small and fuzzy fibrils on the surface of the fabric, and improve appearance and color brightness. Cellulase can improve the drainage, beating, and running properties of paper mills. The mixture and separated components of cellulase, hemicellulose, and pectinase have potential applications in agriculture, and can be used to control plant diseases and promote plant growth and development [1].
Due to the continuous consumption of fossil fuels, the shortage of energy has become a global problem, accompanied by serious environmental pollution and global warming. There is therefore an urgent need to develop alternative energy sources to reduce dependence on fossil fuels and ease environmental stress. The biochemical conversion of biomass mainly includes three steps: first remove lignin and hemicellulose, then decompose cellulose into glucose, and finally ferment with glucose to produce ethanol. Cellulases and other enzymes can be used to transform natural renewable biomass (such as agricultural and forestry waste) into biofuel [2]. Cellulases can also be used to extract bioactive ingredients for natural medicines. Traditional methods for extracting and separating bioactive ingredients, such as decoction, dipping, percolation, and reflux, all have their shortcomings. Low extraction rate, high impurity content, energy consumption, and long production cycles directly restrict the development of the pharmaceutical industry. Alongside the rapid development of modern industrial engineering technology, novel technologies have been continuously applied to natural medicine production to improve extraction efficiency [3]. Enzymes have been used since the mid-1990s to extract and separate traditional natural medicines. Although enzyme use in the traditional natural medicine pharmaceutical industry started late, it has since been shown to have unique advantages and broad application prospects [4].
Cellulases are widely used in the extraction of natural medicine because they can destroy plant cell walls and facilitate bioactive ingredient extraction. The catalytic activity of any cellulase depends on its spatial structure and is often affected by any physical and chemical factors that can lead to denaturation and inactivation. Although research on cellulase-assisted extraction technology has made rapid progress, its focus is mainly on the exploration of process conditions using existing enzymes. The lack of cellulases with new functional properties has become the bottleneck in cellulase-assisted extraction of natural active substances. Current research focuses on finding new and efficient cellulases suitable for economical industrial production. We discuss the strategies to obtain novel cellulases based on sequence alignment and gene function screening, methods to improve cellulases performance through protein engineering, and the application of cellulase in bioactive substance extraction ( Figure 1 ).
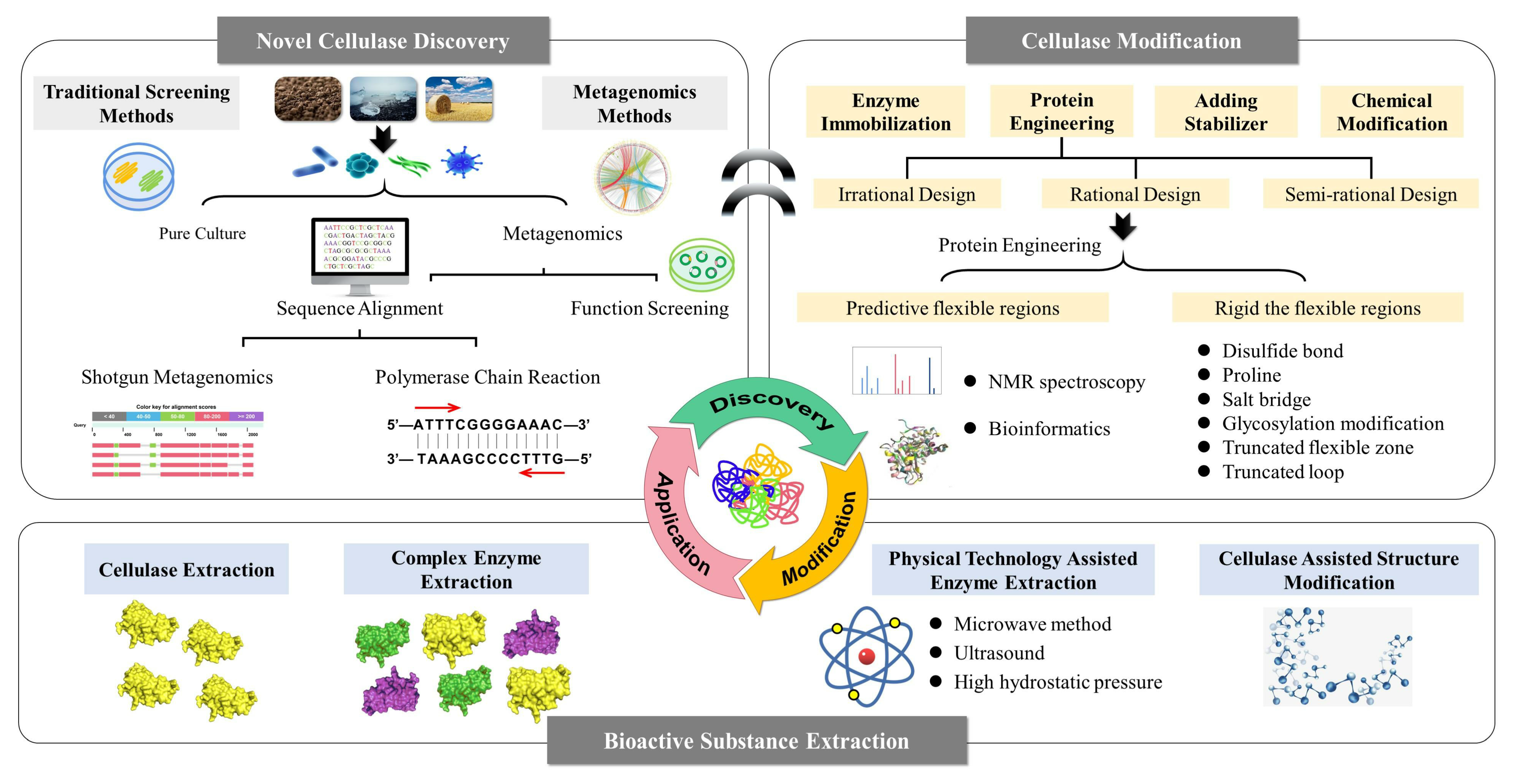 Figure 1. Strategies for obtaining high-performance cellulase and its application in natural medicine extraction.
Figure 1. Strategies for obtaining high-performance cellulase and its application in natural medicine extraction.
2. Cellulase Use in Extraction of Natural Active Substances
3. Conclusions and Outlook
Cellulase-assisted extraction technology provides new ideas and methods for producing and researching bioactive medicinal ingredients and can significantly increase the extraction rate of effective ingredients and overcome the complex procedures and time consumption of traditional methods. Cellulase was initially usually used alone or in combination with other enzymes for the pretreatment of plant materials. Using some appropriate enzymes (including cellulase, hemicellulase, and pectinase) on plant cells can degrade cellulose, hemicellulose, pectin, and other substances in the cell wall and the intercellular space. It can destroy the dense structure of the cell wall to reduce the mass transfer resistance of the cell wall and other mass transfer barriers. In general, treating medicinal materials with complex enzymes is better than cellulase alone. With the advancement of technology and research, using physical methods, such as ultrasound, microwave, and HHP, in combination with enzymatic hydrolyses to extract bioactive substances has become a viable approach. The reaction conditions of enzymatic hydrolysis are mild, which can maintain the conformation of the natural product without destroying its three-dimensional structure and biological activity and will also reduce pollutant emissions. Although enzymatic hydrolysis also has limitations and requires high industrial application conditions, the broad application prospects and economic benefits should encourage researchers and engineers to develop further and optimize related manufacturing techniques.
With the continuing research on cellulase-assisted extraction technology, the limitations of cellulase have gradually emerged. Its high cost restricts the development of this technology, and finding new cellulases is a way to reduce production costs. Molecular biology methods and DNA recombination technology can be used to screen special microbial strains for enzyme-producing genes (including new enzymes in extreme environmental conditions) and chemical methods or genetic engineering can be used to modify existing enzymes to construct specifically engineered bacteria. As enzymes are extremely sensitive to reaction conditions, it is necessary to determine the optimum temperature, pH, and reaction time to maximize cellulase activity throughout experiments. The influence of enzyme concentration, substrate concentration, agonists, and inhibitors should also be considered. At the moment, most of the cellulases used are processed by heating and inactivation and cannot be recovered. This not only increases cost but also has an impact on extract safety and effectiveness due to cellulase residue. There are some profound problems that need to be solved: whether the enzyme residue will degrade, precipitate, or form a complex with the bioactive ingredients in the preparation; whether it will affect the quality and quantity of the bioactive ingredients; whether it will produce adverse reactions; and whether it will affect the quality of the preparation or interfere with detection and affect safety or effectiveness. Research into nonaqueous mediator enzyme reactions and immobilized enzymes is an effective way to improve enzyme stability and potential recycling. In conclusion, cellulase-assisted extraction technology cannot solve all the problems in the extraction of bioactive ingredients. As a new technology, it must be applied in conjunction with other technologies to allow exploitation of its advantages, and we consider that these issues will become the focus of future research.
References
- Bhat, M.K. Cellulases and related enzymes in biotechnology. Biotechnol. Adv. 2000, 18, 355–383.
- Zhang, X.; Zhang, Y.P. Cellulases: Characteristics, Sources, Production, and Applications; John Wiley Sons Inc.: Hoboken, NJ, USA, 2013; pp. 131–146.
- Sticher, O. Natural product isolation. Nat. Prod. Rep. 2008, 25, 517–554.
- Lu, Y.; Jiang, J.G. Application of enzymatic method in the extraction and transformation of natural botanical active ingredients. Appl. Biochem. Biotechnol. 2013, 169, 923–940.
- Chemat, F.; Vian, M.A.; Ravi, H.K.; Khadhraoui, B.; Hilali, S.; Perino, S.; Tixier, A.S.F. Review of alternative solvents for green extraction of food and natural products: Panorama, principles, applications and prospects. Molecules 2019, 24, 3007.
- Ren, Q.; Xing, H.; Bao, Z.; Su, B.; Yang, Q.; Yang, Y.; Zhang, Z. Recent advances in separation of bioactive natural products. Chin. J. Chem. Eng. 2013, 21, 937–952.





