Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Harekrushna Sutar | + 8876 word(s) | 8876 | 2021-07-12 04:23:02 | | | |
| 2 | Bruce Ren | -21 word(s) | 8855 | 2021-07-20 03:18:02 | | | | |
| 3 | Lindsay Dong | Meta information modification | 8855 | 2021-10-09 11:39:25 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Sutar, H. Graphene, Graphene-Derivatives and Composites. Encyclopedia. Available online: https://encyclopedia.pub/entry/12185 (accessed on 08 June 2026).
Sutar H. Graphene, Graphene-Derivatives and Composites. Encyclopedia. Available at: https://encyclopedia.pub/entry/12185. Accessed June 08, 2026.
Sutar, Harekrushna. "Graphene, Graphene-Derivatives and Composites" Encyclopedia, https://encyclopedia.pub/entry/12185 (accessed June 08, 2026).
Sutar, H. (2021, July 19). Graphene, Graphene-Derivatives and Composites. In Encyclopedia. https://encyclopedia.pub/entry/12185
Sutar, Harekrushna. "Graphene, Graphene-Derivatives and Composites." Encyclopedia. Web. 19 July, 2021.
Copy Citation
Graphene has accomplished huge notoriety and interest from the universe of science considering its exceptional mechanical, physical and thermal properties. Graphene is an allotrope of carbon having one atom thick size and planar sheets thickly stuffed in a lattice structure resembling a honeycomb structure. Numerous methods to prepare graphene have been created throughout a limited span of time. Due to its fascinating properties, it has found some extensive applications to a wide variety of fields. So, we believe there is a necessity to produce a document of the outstanding methods and some of the novel applications of graphene.
graphene
graphene-derivatives
composites
synthesis
applications
1. Introduction
Graphene is an allotrope of carbon which has a two-dimensional structure. It is in the form of a hexagonal lattice (see Figure 1) that resembles honeycomb structure [1]. It has some unique properties that have interested the researchers since its discovery by Geim and Noveselov [2]. Due to its high versatility and relatively huge specific surface region [3], graphene is sought as a good option in sensing applications [4]. Main advantages of graphene are: (a) it is the finest and toughest material known; (b) it has carbon monolayered atoms that are both flexible and transparent in colour; (c) it is an excellent thermal and electrical conductor; (d) its main usage is in the manufacture of high-speed electronic gadgets; (e) explosives detection through chemical sensors; (f) membranes for more proficient separation of gases and is produced using sheets from which nanoscale pores have been made; (g) for manufacturing of transistors operating at high frequencies; (h) it has boosted the manufacture of low-cost display screens of cell phones replacing the indium-based electrodes in organic light emitting devices (OLED); (i) to produce lithium-ion batteries that use graphene on the anode surface and these batteries recharge faster; (j) stockpiling hydrogen for cars powered by fuel cells; (k) cheaper water desalination techniques by using graphene films with nanoscale holes to separate water from ions in brine; and (l) Graphene condoms are produced to increase the sensation and is thinner than conventional latex condoms.
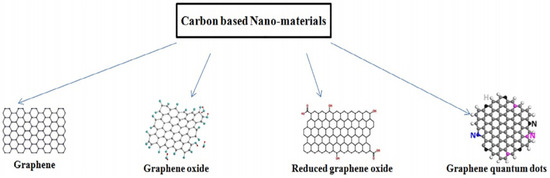
Figure 1. Various forms of Graphene. “Reproduced with permission from [5], [FlatChem]; published by Elsevier, 2020”.
The very first examinations on exfoliated graphene done by Schedin et al. [6] have showcased graphene’s capability on identifying single gas particles based on estimations under Hall effect. The basic guideline behind the usage of graphene in gas sensors is the transfer of charge between the molecule adsorbed to its surface and the material [7]. The carrier concentration of graphene is changed by the absorbed particles which cause the electrical properties to change and the concentration of particles is determined. The response of the system largely depends on the nature of the particle donor or the acceptor [6][8]. Graphene electrical conductivity increases as a result of adsorption of the acceptor compounds (e.g., H2O or NO2). On the other hand, conductivity decreases when the donor compounds get absorbed to the graphene surface (e.g., CO, NH3). Due to the 2D structure, every carbon atom in graphene lattice turns into a surface atom. It makes graphene extremely delicate to the outside environment, yet in addition also restricts its gas selectivity simultaneously [9][10]. Its properties like high intrinsic mobility, enormous surface area, excellent conductor of electricity and heat, and its ability to resist a current density of 108A/cm2 is the main driving basis of utilizing graphene and its subsidiaries [5]. Properties shown by graphene, make them ideal for biomedical application as well.
Graphene was isolated by passing power through crystals that were cut off from mass graphite and were moved onto fine SiO2 or silicon wafer [5]. Further, with the current emergence of green chemistry and sustainable, non-polluting approaches, graphene was manufactured by using biomolecules as a substrate. Recently, biomolecules and biosimilars are being capitalized in a wide reach due to the non-poisonous and biocompatibility nature forthe production of graphene. According to the underlying property of graphene-based nanomaterials (GBNs), graphene is a water resisting material. So, it needs a modification in its functional groups making it bio-medically suitable. The modifications are covalent and non-covalent functionalization. The non-covalent functionalization assists detection, biocompatibility, dispersibility, reactivity and mixing efficiency [11]. Between the polar points present on the surface of graphene oxide and water molecule, hydrogen bonds are available which makes it suitable for biomedical packings [12][13]. In the course of the most recent twenty years there has been a tremendous and sudden development in the utilization of nanotechnology in analysis and therapeutics areas. In addition to this, GBNs have an affable physicochemical property which makes them a great therapeutic molecule. The partially soluble drugs are aided in loading with high productivity and intensity by pi-pi stacking, electrostatic collaboration, and huge explicit area of GBNs [14]. Hence GBNs hold a substantial position in the applications in biomedical field. Since GBNs manage the rate at which drugs are released, it has an incredible practicality in drug delivery frameworks [15]. GBNs-based nanocomposites utilizing polymers, biomolecules, are less toxic in nature, and enhances proficiency against microorganisms. Materials required for bio imaging must have properties likehigh specificity, non-poisonous, and sensitivity which are displayed by graphene quantum dots (GQDs). Besides, they demonstrate extraordinary photophysical and spectrofluorometric properties needed for producing images of biological segments [16]. Graphene also found applications in photodynamic therapy (PDT), which is a non-intrusive technique for cancer therapy. PDT technique uses hydrophobic photosensitizer (PS) and near infrared (NIR) light to form an active form of oxygen inside the tumour cells to destroy the malignant cells [17][18]. It is additionally helpful in the detection and treatment of infections and in transporting explicit medications to the target organs. Being a lightweight material that can be synthesised, it helps inconveyance of medications and genes easily without any critical convolutions. The extraordinary mechanical and other useful properties of graphene shows that it has a high potential to create predominant gadgets and has grabbed the title of “wonder material” [19]. Graphene materials are widely used in the field of electromechanical devices [20], field-effect semiconductors (FET) [21], strain sensing devices [22], electronics devices [23], super-capacitors [24][25], H2 storage [26] and solar power-driven cells [27][28]. It is typically converted into quantum dots [29] and nanoribbons [30] for its utilization in hydrogels [31], froths [32][33] and semiconductor gadgets for various energy applications. The most promising effect in the latest innovations have been created from the creative transformations of various graphene supported polymers and metal-based grid nanocomposite. Henceforth, over the most recent few decades, various innovative work identified with graphene-based nanocomposite have been expanded at a quick rate. These researches showed that various graphene materials can be added to the polymer framework to enhance electrical, mechanical, thermal and numerous other functional properties of thecomposite substances which in turn boosts the total efficacy of the substances manufactured using these composites [34]. The polymer-graphene nanocomposite working greatly improves as a result from the creation of large particle interfacial areas. This is done so by the fusion of nanofiller in the matrix, thus changing their significant properties and rearranging the nanoparticles [19]. Graphene materials operate as either agile medium or as well distributed and aligned electrode material in light to other energy conversion fields such as solar to thermal power, solar to electricity conversion, catalysis in presence of light and so forth, contributing widely in renewable energy production. The technological improvements have developed a 3D graphene material having cross linking bonds which absorbs radiations from the sun and converts it to thermal energy [35]. It produces productivity as high as around 87% in one sun intensity and over 80% in the surrounding daylight. However, this impressive proficiency can be further evolved by changing the underlying model of the material.
2. Synthesis of Graphene
2.1. Graphene: Conception and Manufacture
All graphitic allotropes of carbon are derived from graphene. The basic structure of the graphene is same as those of its carbon allotropic counterparts. The structure of graphene is illustrated in Figure 2. The structure of graphene is organised in the form of planar sheets of one atom thick carbon layer. The carbon atoms are uniformly aligned in the form of hexagonal grids and closely bonded by sp2 hybridised bonds [36].
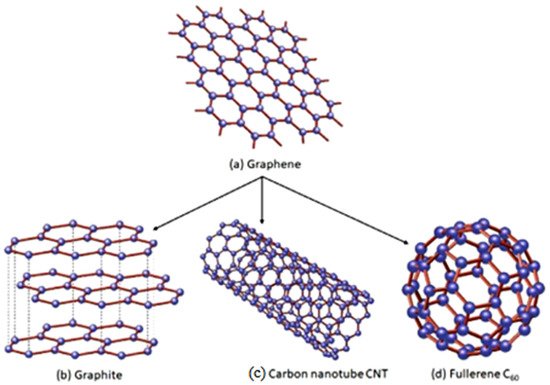
Figure 2. The Fundamental structure of Graphene. “Reproduced with permission from [19], [Materialia]; published by Elsevier, 2020”.
Andre Geim and Kostya Novoselov [2] isolated the slimmest and world’s first 2D material in 2004. Graphene, earlier known by the tag name of ‘academic material’, came to be widely associated with industrial use presently due to its excellent characteristics [37]. Because of the sheet like special structure of the graphene, it possesses interesting properties such as great mechanical strength (Young’s modulus: 2.4 ± 0.4 TPa—single layer and 2 ± 0.5 TPa—bilayered). Graphene has an ultimate tensile strength of 130 GPa [38], extremely high stiffness, extraordinary heat conducting property (5000 W·m−1·K−1) [39], high specific surface area (2630 m2·g−1) [40][41], quantum hall effect [23] required in proximity sensing, speed detection etc., exceptional optical transmission (97.7%) and outstanding electronic transportation [42][43]. It also excellently inhibits the outbreak of fire due to its zero band gaps [2][44][45][46][47][48]. Usually, pure graphene is not used because of difficulty in its production at large quantities at a decent quality. However, graphene derivatives such as graphene oxide (GO), reduced graphene oxide (rGO) and functionalized reduced graphene oxide (frGO) are being widely used due to their similarity in properties with that of graphene. Figure 3 illustrates the arrangement of carbon in graphene and its derivatives.
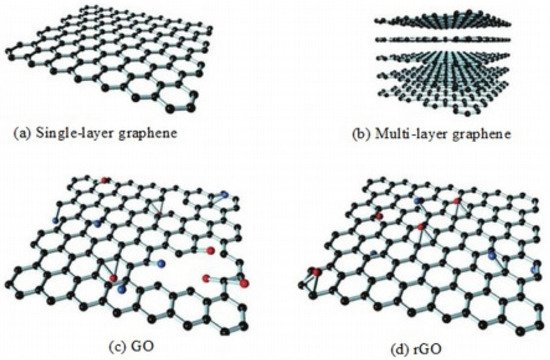
Figure 3. Structure of graphene and its derivatives. “Reproduced with permission from [19], [Materialia]; published by Elsevier, 2020”.
Graphene materials and its derivatives’ unique properties and requirements make its production to be continued for many more decades and accelerate its applications especially in certain focus areas. It has become quite popular for industrial purposes as well as for the academic researches as they exhibit extraordinary properties, increasing their market demand. Graphene has undoubtedly increased the competition standards for similar kinds of materials due to which fabrication advancements have been made to successfully synthesize graphene and its derivatives.
2.2. Synthesis of Graphene and Graphene Derivatives
The two main methods: top-down (destruction) and bottom-up (construction) techniques [49][50] as illustrated in Figure 4 are the backbone of graphene production. These two methods can further be narrowed down into multiple synthesising techniques as shown in Figure 4. The process of exfoliation of graphite and graphite derivatives producing nano-sized graphene sheets is known as the top-down synthesising approach which can also be referred as the destruction approach. Mechanical exfoliation [37], liquid phase exfoliation [51][52], arc discharge [53], oxidative exfoliation reduction [54][55] and unzipping of carbon nanotube (CNT) [54][55] are a few top-down methods. These methods separate and divides the graphite layers into one layer (mono-), two layer (bi-) and multiple layer graphene. Even if the graphene produced by destruction method are very expandable and of top-notch quality, they possess some limitations. Since top-down approach produces graphene by exfoliating graphite, the quality of graphene mainly depends upon the quality of the parent graphite and hence some limitations like inconsistent properties and low yield can be seen. There is another method of graphene synthesis which uses atomic sized carbon precursor rather than graphite to develop graphene and its derivatives. This method is known as the bottom-up technique which is also regarded as the construction technique. Chemical vapour deposition (CVD) [56][57], substrate-free gas-phase synthesis (SFGP) [58] epitaxial growth [59][60], template route [61] and total organic synthesis [62] are some of the exemplars of the construction method. The bottom-up technique has some advantages over the top-down approach as the former is capable of producing near to perfect graphene having a huge surface area. However, these techniques need well developed operational facilities and techniques and are comparatively expensive than the top-down approach. Generally, the graphene synthesis technique decides the amount and quality of the graphene produced. Different synthesis methods for graphene and its derivatives are mentioned below by different researchers.
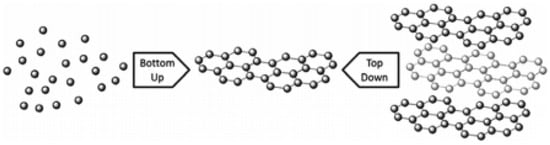
Figure 4. Top-down model and bottom-up synthesis of graphene. “Reproduced with permission from [19], [Materialia]; published by Elsevier, 2020”.
-
According to He-nan Mao and Xiao-gong Wang [63] there are various methods for graphene production. The methods incorporated in synthesis of graphene can be categorised as: “top-down” and “bottom-up” [64], as shown in Figure 5. For the top-down approach, single-layer graphene is obtained by exfoliating graphite to decrease the van der waals interaction between the graphite layers. Some of the widely used top-down approaches are: the mechanical peeling method [37], redox method [65][66][67], solvent peeling method [68][69][70], and electrochemical peeling method [71][72][73][74], whereas, the chemical vapor deposition (CVD) method [47], chemical synthesis method [75][76] and SiC substrate epitaxial growth method are some of the examples of bottom-up approach [77][78]. However, the redox method and CVD methods are most widely used.
-
Graphene oxide (GO)—the precursor of graphene, is full of O2-containing groups on the exterior and sides of the graphene-based sheets. It is made by the oxidation reaction of the graphite. Graphene oxide is made by a powerful oxidation of graphite using a strong oxidising agent in a concentrated acid. Preparation of Graphene Oxide (GO) can be classified into three methods on the basis of the oxidant chosen, acidic medium and the concentration of the acid. They are: the Bordie method [79], Staudenmaier method [80] and Hummer’s method [65]. Strong oxidizing agent like potassium chlorate in nitric acid is used to oxidize graphite to produce Graphene Oxide in the Brodie method. This method helps in graphene oxidation as it uniformly distributes the functional groups over the surface of graphene oxide flakes and also results in a higher relative amount of hydroxyl groups.
-
N.I. Zaaba, K.L. Foo et al. [81] conducted some research in which GO was obtained from graphite flakes by using a modified Hummer’s method, a method different from the conventional Hummer’s method. Contrasting to the traditional Hummer’s method, this method was carried out without the use of Sodium Nitrate (NaNO3) and instead of an ice bath, this method was performed at normal room temperature. The GO obtained undergoes the following steps: (i) The GO obtained is dissolved into variety of solvents, mainly acetone and ethanol. (ii) After dissolving, silicon wafer and IDE is uniformly spreaded on the GO surface by the process of spin-coating to produce acetone-GO (A-GO) and ethanol-GO (E-GO).
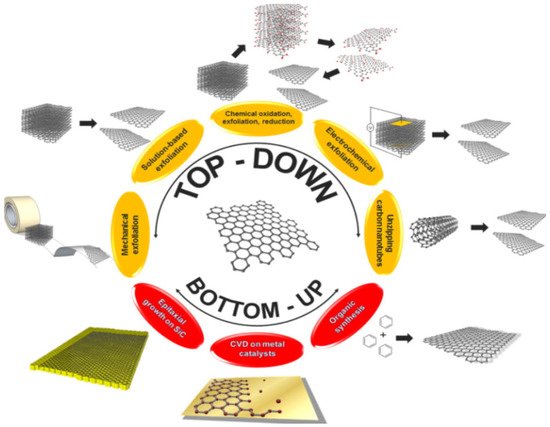
Figure 5. Illustration of different approaches of graphene production. “Reproduced with permission from [63], [New Carbon Materials]; published by Elsevier, 2020”.
Even without using NaNO3 and ice bath, the modified Hummer’s process still produces the same characteristics of GO. This modified process has advantages over cost reduction and does not emit toxic gases. Dissolving GO in ethanol has advantage over acetone because of higher electrical conductivity and solubility of GO. Marcano et al. [82] completed the study of difference between the traditional Hummer’s method and modified Hummer’s method. Their study improved graphene oxidation process without the use of NaNO3 as using NaNO3 for oxidation would emit toxic gases. Thus, the conventional uses of NaNO3 were replaced with H2SO4, H3PO4 and double the amount of KMnO4 in the improvised Hummer’s method by Marcano et al. [82]. Improvising Hummer’s method was advantageous in many ways, such as: more hydrophilic carbon material, equivalent conductivity and reduction in toxic gas emission and also exponentially increases the GO production [82].
- 4.
-
Roberto Muñoz and Cristina Gómez-Aleixandre [83] has written about the chemical vapour deposition (CVD) process of synthesizing graphene in their article and illustrated in Figure 6. In the review paper the CVD “bottom-up” synthesis method is considered as a wide scale production method of graphene with large surface area and superior quality [84][85][86]. Till now the best quality graphene in terms of robustness has been produced by CVD method, but the layers cannot be controlled with ease. But if we compare the properties exhibited by graphene synthesized by the CVD method, to that of flaked graphene, the exfoliation method produces better quality until now.
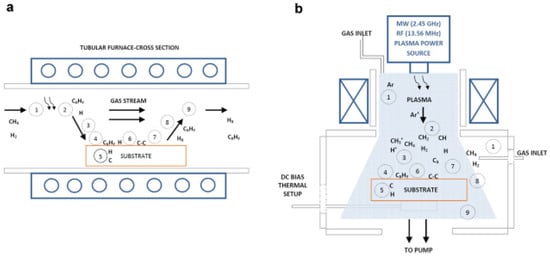 Figure 6. (a) Thermal CVD (b) plasma assisted CVD(PACVD). (1) Forced convection of reactants. (2) (a) thermal (b) plasma activation. (3) Gaseous diffusion of reactants. (4) Adsorption. (5) Dissolution and bulk diffusion. (6) Thermally assisted surface processes. (7) Desorption. (8) Diffusion of by-products. (9) Forced convection of product. “Reproduced with permission from [83], [Chemical Vapor Deposition]; published by Wiley, 2013”.
Figure 6. (a) Thermal CVD (b) plasma assisted CVD(PACVD). (1) Forced convection of reactants. (2) (a) thermal (b) plasma activation. (3) Gaseous diffusion of reactants. (4) Adsorption. (5) Dissolution and bulk diffusion. (6) Thermally assisted surface processes. (7) Desorption. (8) Diffusion of by-products. (9) Forced convection of product. “Reproduced with permission from [83], [Chemical Vapor Deposition]; published by Wiley, 2013”.
In CVD process gaseous reactants undergo chemical reaction forming a firm deposition of solids over a substrate [83]. The energy requirements of this process are aided by various sources paving the way for plasma assisted CVD(PACVD), thermal and laser assisted CVD. The deposition cycle can incorporate two types of reactions: homogeneous gas-stage reactions, that takes place in the gaseous stage, and heterogeneous chemical reactions which takes place over/close to a hot surface. In both cases, powders/films are formed.
The detailed stepwise fundamental procedure is as follows:
-
Pyrolysis stage: The catalyst-substrate (in a controlled surrounding) and reaction gases (in hot-wall reactors) is heated up to a certain fixed temperature.
-
Toughening stage: The temperature and the gaseous environment is maintained for the catalyst surface to undergo reduction. This step is performed to clean the catalyst surfaces and adjust the surface morphology like roughness, and particle dimensions of the metal catalyst. Dissipation of metals by vaporization must be omitted as far as possible.
-
Growth stage: In this stage, new precursors are introduced into the reaction system and graphene development takes place over the catalyst. Growth of graphene completely depends on the nature of the catalyst (solubility, catalytic action etc.). The pressure of the blending gases, residence time etc. can be altered as per the reaction requirements.
-
Cooling stage: After the growing step, the reactor is then allowed to cool down to prevent oxidation of the uncovered catalyst. The reactor temperature is kept below 200 °C. The reactor atmosphere is kept similar to the previous steps. The elements involved in the cooling step are to be minutely monitored as they are critical in controlling the development of graphene.
-
Final step: Backfilling with non-reactive gases (Argon, Nitrogen) up to 1 atm pressure and opening the reactor chamber.
- 5.
-
Pan et al. [87] synthesized graphene quantum dots (GQD) through hydrothermal method. Concentrated H2SO4 and HNO3 was poured over graphene sheets (GSs) and kept like that for 15–20 h under gentle ultrasonication, causing oxidation of GS. Xie et al. [88] used the solvothermal method for the synthesis of GQDs from carbon nanotubes (CNTs). In this process H2O2 was taken as the oxidant and ethanol as solvent. Shin et al. [89] reported the synthesis of GQDs from carbon fibres (CFs) by a solvothermal method where described synthesis of GQDs by solvothermal process from carbon fibres (CFs) by utilizing Oxone as the oxidant. Huang et al. [90] introduced a bottom-up procedure to fabricate the GQDs from polythiophene derivatives. Umrao et al. [91] described a bottom-up pathway to synthesise GQDs from acetylacetone as the initial organic solvent via carbonization using microwave radiations and converting them into aromatic compounds. But those methods have certain disadvantages: (1) harmful for environmental, as the strong oxidants or organic solvents need to be used; (2) expensive precursors like graphene, GSs, GFs and CNTs; (3) requires long durations due to the post-processes, in which the impurities like metallic ions, acid residues need to be discarded by constant washing and sometimes even dialysis. So, an environment friendly artificial process was initiated by Weifeng Chen et al. [92] as illustrated in Figure 7 that led to the discovery of a replenishable asset as the raw material, which improved the scope of GQDs manifolds in medical areas.
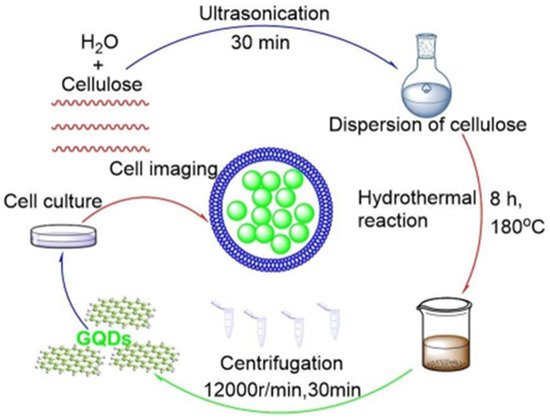 Figure 7. The renewable pathway for eco-friendly synthesis of GQD. “Reproduced with permission from [92], [ChemistrySelect]; published by Wiley, 2019”.
Figure 7. The renewable pathway for eco-friendly synthesis of GQD. “Reproduced with permission from [92], [ChemistrySelect]; published by Wiley, 2019”.
The synthetic mechanism consists of certain steps wherein the development of GQDs comprises of two steps. First step involves cellulose hydrolyzation to generate glucose, followed by cyclic condensation of glucose to form GQDs. The entire process can be depicted as follows: Firstly, a lot of H+ and OH− are produced at an elevated temperature and pressure by the deionized water under hydrothermal condition. Cellulose hydrolysis took place under the activity of H+, without the presence of cellulase and acids. Later glucose, levoglucosan and cellobiose, and were obtained from cellulose. As the hydrolysis reaction progressed, the production of glucose increased manifolds, subsequently turning the glucose into the main product. Later on, reaction occurred between the formyl groups and the hydroxyl groups; and the OH− groups of glucose with the H2 atoms. This resulted to glucose dehydration under the hydrothermal condition. Simultaneously, aromatic rings (the rudimentary unit of the graphene structure) were formed by covalent linkage of the carbon atoms of the glucose with one another. Thus, GQDs are formed by the cyclic condensation of the glucose. Simultaneously, the carbide amorphous insolubles were also formed from the carbonization remnants of the carbohydrates. In the end, the centrifugation was performed segregating the insoluble out from the reaction products and thus GQDs solution was obtained.
- 6.
-
Yan Zhao, Minyue Wen et al. [93] devised a new method for graphene synthesis wherein the fermentation of straw/acid hydrolysis of straw was done to produce lignin residue. This residue served as the carbon source to produce graphene. Tetrahydrofuran was added as a modifier to lignin and ferric nitrate as a catalyst for pre-treatment. The resultant product was thermally treated at 900 °C for 3 h. The characterization of the prepared graphene was done by XRD, Raman, HRTEM, and its electrochemical properties were tested. The prepared lignin-based graphene was found to be a good electrode material for supercapacitors. The results of this study open up a new method for efficient graphene preparation based on renewable resources lignin.
- 7.
-
In the experimental procedure approached by Albert Dato and Michael Frenklach [94], the effects of gas flow rate, applied microwave power, and precursor composition on the substrate-free gas-phase synthesis of graphene were investigated. For the first time free-standing graphene has been created without the use of substrates or graphite through gas-phase microwave synthesis [95]. Liquid ethanol droplets and aerosols consisting of argon gas were sent directly into argon plasmas. These argon plasmas were generated using an atmospheric-pressure microwave plasma reactor. Over a time, of order of 10−1 s the ethanol droplets evaporated and dissociated in the plasmas and formed graphene that showed a highly ordered, oxygen-free structure [96]. In contrast to the other alternative approaches, this technique is highly capable of rapidly and continuously synthesizing ultrahigh-quality graphene in a single step at an ambient pressure.
- 8.
-
A facile and fast approach to the synthesis of high-quality graphene nanosheets was conducted by Hui-Lin Guo et al. [97]. They performed an electrochemical reduction of the exfoliated GO at a graphite electrode (see Figure 8). The reaction rate was increased by increasing the temperature. According to the modified method of Hummer the starting material for the synthesis of exfoliated graphene is graphite flakes that are oxidised [98][99]. The GO dispersion in water is increased by the O2 groups bonded on the GO surface by increasing its charged capacity. Then electrochemical reduction of the exfoliated GO takes place on a graphite electrode in a GO dispersion with magnetic stirring but a different voltage.
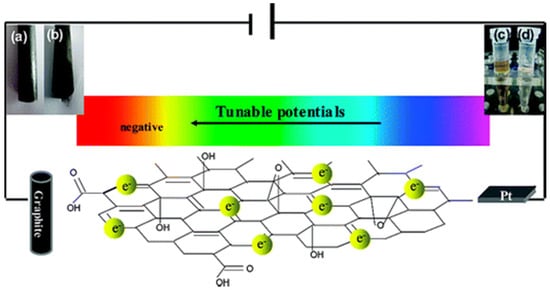 Figure 8. (a,c)Optical illustration of graphite electrode and GO suspension before and (b,d) after reduction. “Reproduced with permission from [97], [ACS Nano]; published by [American Chemical Society], 2009”.
Figure 8. (a,c)Optical illustration of graphite electrode and GO suspension before and (b,d) after reduction. “Reproduced with permission from [97], [ACS Nano]; published by [American Chemical Society], 2009”.
Kovtyukhova and colleagues [98][99] prepared a graphite oxide from spectral graphite by using modified Hummer’s method. The prepared GO formed a brown colored dispersion upon suspension in water. The dispersion underwent dialysis for a week for complete removal of the residual salts and acids. Sonifier was used to perform ultrasound of 0.5 weight % of GO dispersion to produce exfoliated GO. The brown dispersion was centrifuged for 5 min at 3000 rpm to remove the un-exfoliated GO. CRGO (chemical reduced graphite oxide) was prepared using ammonia and hydrazine hydrate as reducing agents. On a working graphite electrode, the GO suspension was electrochemically reduced that was performed on a 3-electrode system having platinum as counter electrode, saturated calomel as reference with power supply stirring of 2 h. Alumina powders were used to polish glassy carbon electrode (GCE). GCE is washed properly and dried by blowing N2. A thin film of exfoliated GO suspension formed on a graphite disk or bare GCE is dried in a vacuum desiccator and then electrochemically reduced. All experiments were conducted at room temperature. This method is eco-friendly and does not cause graphene contamination. But the graphene produced by electrochemical reduction contains some defects due to the increased reduction rate.
- 9.
-
Top-down approach: This methodology involves the techniques to synthesize a solitary graphene sheet by dismantling the stacked layers of graphite [100][101]. Figure 9 reveals the detailed flowchart of various top-down techniques and also explained in brief.
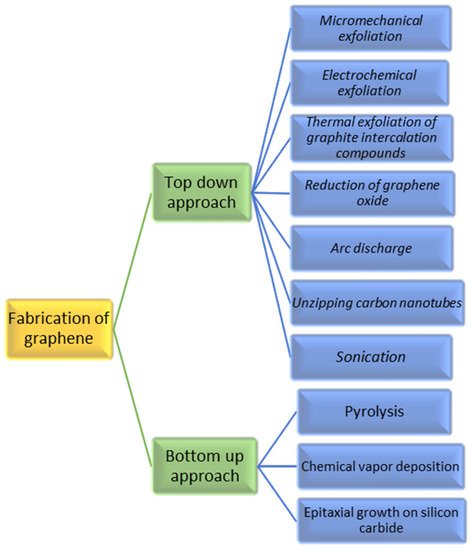 Figure 9. Detailed flowchart of various graphene synthesis techniques in top down and bottom-up 363 approaches. “Reproduced with permission from [101], [TrAC Trends in Analytical Chemistry]; 364 published by [Elsevier], [2019]”.
Figure 9. Detailed flowchart of various graphene synthesis techniques in top down and bottom-up 363 approaches. “Reproduced with permission from [101], [TrAC Trends in Analytical Chemistry]; 364 published by [Elsevier], [2019]”.
-
Mechanical Exfoliation: Developed by Geim and Novoselov [2], this method utilizes HOPG (high oriented pyrolytic graphite) as an antecedent. Mesas-like structures were formed when HOPG was exposed to an O2 plasma etching. These mesas were 5 μm profound which were then squeezed into a layer of photoresist [102]. The photoresist was baked. From these baked resists the HOPG was cleaved. To peel off the graphite flakes from the mesas scotch tape was utilized. The thin flakes formed from the mesas were then dipped in acetone. After that it was caught on the Si/SiO2 wafer surface [37]. The few layer graphene (FLG) flakes possess high mobilities of 15 × 103 cm2·V−1·s−1 on Si/SiO2 wafers [46]. The illustration below shows (Figure 10) that the scotch tape technique can generate flakes with sides of up to 10−3 m long [103][104], of great quality. But the process is restricted to small sizes, not for large scale industrial production.
-
Micromechanical exfoliation: This methodology utilizes adhesive tapes (e.g., scotch tape) forstripping off the graphite layers to synthesize mono-, bi- and FLGs. The resultant graphene is of good quality however the technique is long standing and tedious.
-
Electrochemical exfoliation: Earlier the graphite was utilized as a sacrificial electrode within the electrochemical set-up, and the exfoliated material used to be gathered from the electrolyte solution. Presently with latest technology, surfactant (to prevent re-agglomeration of the graphene) and H2SO4-KOH solutions (H2SO4 is a strong oxidant and causes oxidisation of graphite and KOH reduces the acidity of the electrolyte solution) are blended within electrolytes [102]. Graphite flakes with variable thickness are often made by this process and FLGs are segregated by centrifugation.
-
Thermal exfoliation of graphite intercalation compounds: By this method graphite is broken down to graphene through intercalation by applying heat. Graphite intercalation compounds (GIC) can be manufactured by employing various chemical species in between the graphite interlayer spacing. At first GICs are produced from graphite treated in strong acids, then exposed to microwave radiation for thermal decomposition of the intercalates into gaseous species. Different intercalates produce different GICs having unique properties to improve electrical and thermal performance [104][105].
-
Arc discharge: In this technique few layers graphene is synthesized by passing direct current in between highly pure graphite electrodes in a highly pressurized hydrogen gas atmosphere. Shen et al. [106] found that out of all different gases considered the combination of He and H2 gas delivers the noteworthy crystallinity of the material. The growth mechanism involves a cumulative process of graphite evaporation and reactive-gas-restrictive crystallization of the evaporated carbon molecules. Kim et al. [107] revealed production of bi- and tri layer graphene by an aqueous arc discharge process. It was acquired from the graphite electrodes by means of thermal expansion and water cavitation employing quick heating. A medium numbers of graphene layers were obtained having less O2-related imperfections by modifying the power of the arc discharge.
-
Unzipping carbon nanotubes: Oxidized graphene nanoribbons were formed by longitudinally cutting multi-walled carbon nanotubes by means of laser irradiation, plasma etching techniques or by wet chemistry methods. This unfastening produces graphene nanoribbons. The nanoribbon widths are governed by the tube diameter. Before cutting the nanotubes, they were first suspended in H2SO4 and then treated in KMnO4. To unzip the nanotubes, fission via CeC is often started at the deformity sites, leading to irregular sides [108]. The resultant graphene nanoribbons are good conductors but were of electronically inferior quality as compared to industrial scale graphene sheets because of presence of O2 deformity sites [109]. Later, the developments of nanoribbons having smooth edges were performed [110] by unzipping of flattened CNTs, where attack occurs along the bent edges.
-
Sonication: This technique as shown in Figure 11 employs ultrasonic energy to obtain the unmodified, natural flake graphite to isolate graphene pieces scattered in solvents [111]. Experiments revealed that NMP (N-methyl-pyrrolidone) is the best solvent when dispersion percentage of monolayer graphene is considered, but cyclopentanone offers the most elevated absolute concentration (mono layer and FLG). By increasing the sonication time, graphene concentration can be enhanced. Aqueous surfactant solutions can be utilized for performing sonication. The surfactant solution discourages the use of costly solvents and prevents re-aggregation of the graphene.
-
Graphene Oxide Reduction: Another technique dealing with the production of graphene is sonication and reduction of graphene oxide (GO). The presence of polar O and hydroxyl groups make the graphite oxide hydrophilic during the oxidation process. Chemical peeling of this GO is done in various solvents including H2O [112]. Several GO nanoplatelets are obtained from the sonicated graphite oxide solution. The oxygen groups are eliminated in the reduction process using those reducing agents. Stankovich et al. [113] utilised this method by using a hydrazine reducing agent, but the reduction process was seen to be inadequate, leaving some remnant O2. Graphene synthesis involves Graphene oxide (GO) production as a precursor. In contrast to graphite, hydrophilic nature of GO renders it useful. GO is suspended in water by sonication [114][115]. Then it is deposited onto surfaces by spin coating or filtration to make single- or double layer GO. Then this GO is thermally or chemically reduced to produce graphene films [116]. Some of the techniques to synthesize GO are listed out below.
-
Wet Chemical Synthesis: According to Hummers procedure [65] graphite is treated in an anhydrous mixture of H2SO4, NaNO3for several hours, then water is added to it. The resulting product is graphite oxide hydrate containing around 23% water. The heat and electricity conducting properties of along the axis of the surface of graphene oxide is strongly affected by the degree of oxidation. Reduction of the epoxide groups is doneby applying heat energy or by reacting with potassium iodide (KI). And this results in a similar structure, containing hydroxyl groups that lead to improved electrical conductivity and unaffected hydrophilicity. In both cases, stringent temperature control and a long reaction time is strictly required.
-
Plasma Functionalization: Plasma treatment is another such technique that incorporates O2 species into the lattices of graphitic materials (e.g., CNTs, graphene, carbon nanofibers, and graphite) [117]. The structure of graphite oxides synthesized by this method possess very similar structure to Hummers GO. To decrease the epoxides, present in it, thermal treatment is required. This method has the advantage of occurring very quickly, conducted at room temperature and no requirement of strong acids. Plasma provides room for adjusting the electrical conductivity of graphene. This provides a scope for band gap engineering as well as photoluminescence (PL).
-
RF Plasma: Radio frequency (RF) plasma incorporates the usage of capacitive plasma ignited in an isolated volume. Compared to DC discharges ion bombardments are quite low in this method that makes it a good option for surface treatment of graphene. The AC frequency requirement for this technique is 13.57 MHz, power range between 10 W to 50 W and electrodes must not be in contact with the plasma gas. RF treatment has been seen to specifically influence the outermost surface of graphene [118].
-
Photoluminescence: GO does have a photoluminescent response due to the negligible band gap of native graphene. So, RF plasma oxidation has been preferred for GO synthesis. Typically, graphene is prepared by micro cleavage process using the scotch tape or other methods. Then electrical contact or other additions are put in place then RF plasma treatment in Ar-O2 mixes is applied in one-to-six second intervals. The plasma power of 10 W, pressure of 0.04 mbar, and the gas composition ratio of 2:1 Ar to O2 was reported. A significant finding was reported in which plasma treatment time was increased [119]. As a result, 2D and 2D peaks intensity drastically reduced. This suggests sp2 hybridization. This suggests the introduction of oxygen groups and GO formation by disruption of the graphene lattice structure. Photoluminescence occurs in succession of the plasma treatment due to the introduction of imperfections in the graphene structure. Such defects disturb the electrical properties of fresh graphene and introduce a band gap. Band gaps are desirable for optoelectronic applications and for more PL [120][121]. Comparative discoveries were accounted for in other works utilizing pulsed RF plasma method. Bulk graphite is oxidized and isolated in H2O, then thermally broken down to produce single-layer graphene. Figure 12 illustrates various method to produce GO.
-

Figure 10. Single layered graphene synthesised by mechanical exfoliation on Si/SiO2 [102].
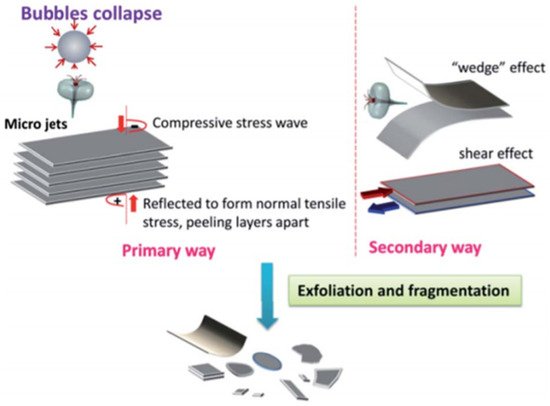
Figure 11. Schematic figure of mechanical exfoliation of graphene by sonication method. “Reproduced with permission from [111], [Journal of Materials Chemistry A]; published by [Royal Society of Chemistry], 2015”.
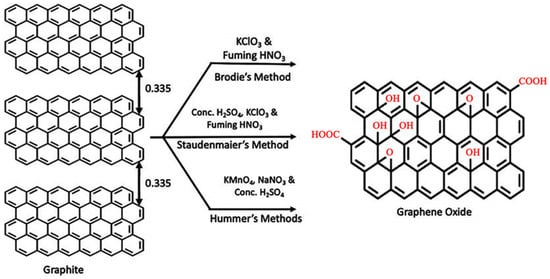
Figure 12. Fabrication techniques of Graphene oxide [121].
- 10.
-
Bottom-up methods: These methods include preparation of large sized graphene utilizing alternative carbon containing sources of smaller sizes. The detailed explanation is given below.
-
Pyrolysis: Sodium-Ethanol Pyrolysis: Graphene synthesis was confirmed by heating Na and C2H5OH at a 1:1 molar ratio in a sealed vessel. This solvothermal product of sodium and ethanol was heated in absence of oxygen to form a substance consisting of fused graphene layers. This substance is separated by sonication in ethanol. Graphene sheets measuring 10 μm in sizes are formed. The crystalline and graphitic nature, individual layer, of the samples was confirmed by selected area electron diffraction (SAED), TEM, and Raman spectroscopy [122]. This method had an advantage of low-cost production of high purity graphene at reduced temperatures without using costly graphitic precursors. But this method was further improvised by D.K Singh et al. [123] by using a medium pressure autoclave and mild catalyst sodium borohydride instead of sodium metal. Addition of a surfactant such as sodium dodecyl sulphate (SDS) showed improved quality of graphene. The product yield increased when surfactant was added.This made this process safer and commercially viable for bulk production of mono layered graphene and FLGs.
-
Chemical vapor deposition: This method employs the breakdown of hydrocarbon gases on a catalyst surface or the use of carbon dissolved in some metal masses obtained by surface separation. The key determining factor in the dominant growth process is the metal’s carbon solubility limit. In the growth process, the solubility limit determines whether the metal or carbon will be dominant. By decomposing hydrocarbon gases on catalytic surfaces or surface segregating the carbon dissolved in the entire mass of the specific metals, chemical vapour deposition (CVD) is used to fabricate graphene and few-layer graphene (FLG). The carbon solubility limit in the metal determines whether the former or latter will be the dominant growth mechanism. Various substrates, such as Co and Fe, are used. The metals Ni and Cu continue to be the most common. At 1000 °C, C is less than 0.001% atom percent soluble in Cu. As a result, the graphene growth mechanism in Cu is surface catalysed, resulting in monolayer graphene under UHV conditions. Since C is 1.3 percent soluble in Ni at 1000 °C, the growth of graphene occurs through a segregation mechanism using a variety of metal substrates such as Ni, Fe, Ru, Co, and R Cu, Au, semiconductors like Ge, and even insulators [124] have all been used to develop graphene, but Cu and Ni remain the most common. Cu and Ni polycrystalline films can be used to grow graphene indefinitely. At 1000 °C, Cu has a solubility of less than 0.001 atom percent, whereas Ni has a solubility of 1.3 atom percent. The different solubility limits resulted in different growth rates. For Cu and Ni, the difference in solubility limits resulted in different graphene growth mechanisms. Under UHV conditions, a surface catalysed mechanism is responsible for graphene growth on Cu, resulting in monolayer graphene growth. The growth of graphene on Ni substrate was performed through segregation growth mechanism inabsence of UHV conditions, but the process is problematic to monitor. In the case of graphene, substrate-free growth has also been recorded [125], and this process produces large amounts of graphene.
-
Epitaxial growth on silicon carbide: The uppermost layers of SiC crystal (3CeSiC, 4HeSiC, or 6HeSiC) are thermally decomposed during high temperature annealing under vacuum for graphitisation, resulting in desorption of Si atoms. The remaining C atoms on the surface rearrange and re-bond, forming an epitaxial graphene layer. The presence of argon or small amounts of disilane slows the rate of silicon sublimation, allowing for higher temperatures and better graphene efficiency. Graphene formation begins on the top surface of SiC, where approximately three SieC bilayers decompose (0.75 nm) to form one graphene layer (0.344 nm) and then progresses inwards.
3. Use of Graphene Substances and Its Composites
Since graphene supported nanocomposite has exceptional multifunctional properties, its use has exploded in a variety of fields. Typically, the addition of nanofiller in the matrix improves the efficiency of graphene-based polymer nanocomposites bringing about significant changes in their properties and the positioning of nanoparticles thus creating a huge molecular interfacial area [19]. The following are some of the promising areas in which graphene nanocomposites are being utilized and Figure 13 highlights these promising areas in pictorial form.
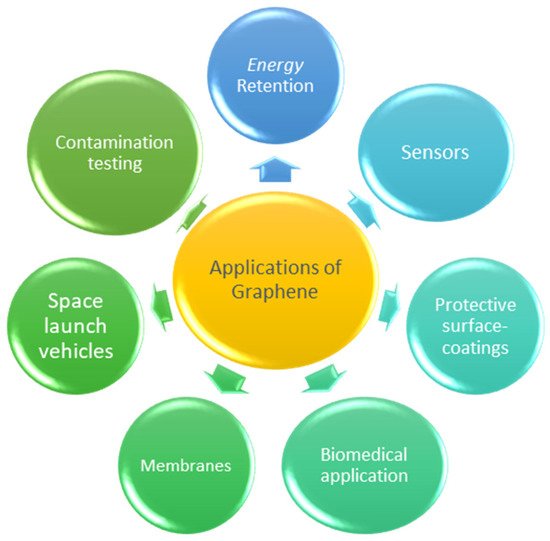
Figure 13. Schematic diagram of different applications of graphene.
-
Energy Retention: Graphene is now extensively used in the production of replenishable power, as in light to energy applications such as solar-heat energy form conversion, solar-electrical energy conversion, and catalysis in presence of light. A three-dimensional cross-connected graphene material that acts as an ideal solar-thermal converter has been created [126]. It has a performance of about 87 percent at 1 KW/m2 solar intensity and more than 80 percent in surrounding sunlight. Depending on the material’s structural design, the performance of energy conversion increases even further. An innovative structure was created by means of the usage of one-step plasma-enhanced chemical vapour deposition (PECVD) process to create a hierarchical graphene foam [127]. The GNPs form an arrangement on the three-dimensional framework allowing the graphene foam to possess a large heat exchange region. The conversion efficacy of solar energy to thermal energy increases to 93 percent as a result of this. Dye-sensitized solar cells (DSSC) are commonly adopted to transform solar energy to electrical energy and are made up of a combination of a redox couple and an auxiliary electrode [128]. Graphene containing electrodes are nowadays being added to DSSC. The modification in DSSC has increased the conductivity, chemical stability, porosity, surface area, catalytic power thereby improving the energy conversion efficiency while lowering costs significantly [129][130][131][132]. Stroller et al. [133] produced a super-capacitor using customized graphene of specific capacitances of 135 F/g in aqueous electrolytes, 99 F/g in organic electrolytes, and 75 F/g in ionic electrolytes. These super capacitors have a longlife-cycle and high power. Zhang et al. [134] used mechanically exfoliated graphene and chemical treatment to create a stretchable electrode. These electrodes are very versatile and can be used in a number of electrolytes. Conductive polymer composites with graphene filler or graphene-based transition metal oxide composites may also be used to make supercapacitors to boost power density [134]. Graphene-based batteries are used to improve energy density. In addition, graphene-conducting polymers and graphene-CNTs are used to increase conductivity in such applications [135]. Lithium-ion batteries have grown in popularity as a result of technological advancements. However, due to the constant augmentation and constriction of volume during the lithiation/de-lithiation reaction, the life cycle of the materials used as cathode and anode in lithium-ion batteries is short. Due to their low electrical conducting property or low electrolyte ion transfer rate, these materials show poor performance. So, materials using graphene as base were introduced as a 3D skeleton to build anodes to solve these problems. Now, the newly developed graphene anodes have high electroconductivity and a good permeability. They can provide outstanding lithium-ion battery efficiency. Fe3O4 [136][137], titanium oxide [138][139], manganese oxide [140], tin (IV) oxide [141] and other metal oxide materials are promising materials for lithium-ion batteries used as an anode. Upon reinforcement with graphene, improvement in diffusion and electroconductivity, resulting in improved battery efficiency is observed.
-
Sensors: Graphene’s exceptional properties render it extremely sensitive to the atmosphere, and as a result, graphene and its related composites are widely used in the development of various types of sensors. Graphene sensors can detect small gas molecules and volatile organic chemical compounds. GO and reduced GO-based sensors are being effectively used to identify gaseous NH3, NO2, CH4, H2, H2O, C2H5OH, CH3OH, isopropyl alcohol, and HCN [142][143][144][145][146]. The detection mechanism is mainly based on the process of molecular particles getting adsorbed into the surficial openings, which alters the graphene sheet’s conducting ability. The detection mechanism, on the other hand, can vary from sensor to sensor [143]. In the field of biosensors, graphene and its substrates are gaining a lot of interest. GO nanosheets are extremely hydrophilic due to the presence of oxygenated functional groups, making chemical functionalization simpler [144]. As a result, graphene-based sensors can detect a broad variety of biomolecules, including glucose, proteins, and DNA. Graphene’s outstanding mechanical, optical, and electrical conductivity makes it ideal for biosensor applications. The high charge carrier mobility of graphene allows for an effective movement of electrons from the bioatom to the sensor. Graphene is used in biosensing applications as a current opposing material, an electron transfer material, and a photon transfer material [142]. Temperature sensing is another significant application of graphene. Most thermal sensors are made of a semiconductor material, and since graphene possess semiconducting properties, its use in this form of sensing is a natural progression. The commonly used temperature sensor is reduced graphene oxide (rGO), which exhibits a visible thermal resistivity decrease as the ambient temperature rises. In its non-functionalized form, graphene has a huge surface area and great heat conducting properties, which are some of the few essential properties required for thermal sensors [145]. Graphene is capable of measuring gaseous water, making it useful to calculate the relative dampness of the surrounding. Graphene obtained through chemical vapour deposition method and mechanical exfoliation is utilized in making moisture detecting sensors which are capable in detecting a broad spectrum of relative humidity levels (1–95%) [147][148]. Graphene adsorbs H2O, and as a result of the interaction between the H2O and the graphene sheet, the electrical structure of the graphene sheet changes, resulting in a detectable change in resistivity. This form of sensor often uses single-layered graphene, which are hypersensitive and precisive than multi-layered graphene. This form of sensor requires high-quality graphene [149].
-
Protective surface-coatings: Graphene’s chemical and thermal properties, as well as its special physical structure (2D), make it an excellent medium for coating applications. Many approaches to structural and chemical modification of Gr-based materials have been investigated. These methods are divided into many groups in the literature, including covalent and non-covalent functionalization, nanoparticle immobilization, and substitutional doping. On the surface of Cu [150][151] and Ni foil [151], the CVD process is utilized to synthesize a pure graphene anti-corrosive coating. The CVD approach is advancing, and the growth substrate has been expanded to include more metals, including Rhodium [152], Gold [153], Titanium [154], Platinum [155], Palladium [156], and Cobalt [157].The following are the steps involved in the method: (1) deposition of a polymethyl acrylate (PMMA) resist layer on the graphene surface to form a PMMA/graphene/Cu interlayer; (2) etchant dissolution of the Cu foil; (3) deposition of PMMA/graphene on the target metal surface followed by drying; and (4) dissolution of PMMA with acetone, resulting in a graphene cover on the target metal surface. A CVD-prepared pure graphene anti-corrosive coating demonstrates high durability. A support layer, such as PMMA [158], heat release tape, and polydimethylsiloxane [159], is needed for the transfer process to avoid wrinkles and cracks. Various methods, such as annealing [160][161], using a modern polymer transfer method [162][163], and even a polymer-free transfer method, have been used to eliminate contaminants on the surface and increase the anti-corrosion performance of the pure graphene anti-corrosion coating after mechanical transfer. SEM images of copper and copper+graphene coatings before and after corrosion are presented in Figure 14.

Figure 14. SEM images of pure Cu before and after corrosion, Cu+Gr before and after corrosion. “Reproduced with permission from [163], [Chemical Engineering Journal]; published by [Elsevier], 2019”.
Figure 15 illustrates some of the shielding applications of grapheme [164] in pictorial form. The various areas where graphene-based coatings have found its usage is mentioned below.
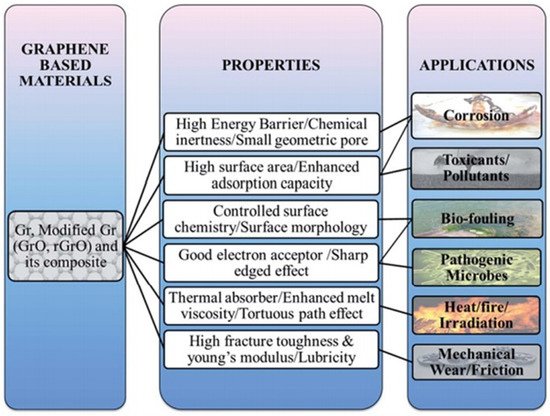
Figure 15. Preventive/shielding applications of graphene. “Reproduced with permission from [164], [Journal of Materials Chemistry A]; published by [Royal Society of Chemistry], 2015”.
-
Graphene as a flame-retardant material
-
Being the thinnest, lightest, and strongest known nano material, it perfectly suited for scratch resistant coatings.
-
Graphene-enhanced antifouling coatings
-
Graphene coatings for pollutant adsorption
-
Graphene-based anti microbial coatings
- d.
-
Biomedical application: Biomedical applications (see Figure 16) are another interesting field in which graphene can be used. Graphene was found to be ideal for bioelectronics, tissue engineering, drug delivery, cancer treatment, biosensing, gene therapy, DNA sequencing etc. [165]. For the first time, graphene was used in biomedical applications to improve drug delivery. The suitableness of GO to be used as a carrier having nano size dimensions used for drug delivery was first recorded in 2008 by Sun et al. [166]. Others were motivated by this discovery to study the biomedical applications of graphene materials further. GO has a wide surface area, and its enhanced O2-containing functional groups have excellent bio compatibleness and are extremely soluble, making it ideal for drug transport [167]. The use of graphene oxide in the oncological therapies and anti-inflammatory drugs is currently being studied [168]. Furthermore, the GO delivery system has made more advances in cancer care using chemo-photothermal therapy [169]. The huge surface area of graphene allows for biomolecule adsorption. Wang et al. [170] reported DNA tethering method by using a chunk of creased film of chemically altered graphene. This DNA-graphene bio interface interaction can be used to determine the presence of DNA with the help of FETs. Graphene is used for bio imaging due to its properties like high thermal, electrical conductivity and large atomic thickness. Graphene materials can also be used in regenerative medicine due to their high mechanical properties [171]. The biocompatibility of graphene with mammalian cells has been demonstrated, which is required for its use as a framework in tissue engineering [171]. Graphene has shown great potential in stabilizing the growth and differentiation of stem cells [165]. Graphene hybrid scaffolds possess phenomenal strength to promote the growth of osteoblasts, finding its ground in bone tissue engineering [171].
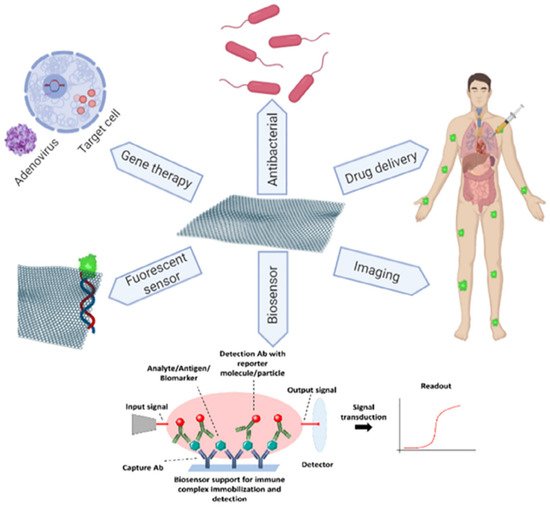 Figure 16. Various applications of graphene in biomedical field. “Reproduced with permission from [5], [FlatChem]; published by [Elsevier], 2020”.
Figure 16. Various applications of graphene in biomedical field. “Reproduced with permission from [5], [FlatChem]; published by [Elsevier], 2020”. - e.
-
Membranes: Membrane separation has recently turned outto be an effective technology for resolving environmental issues. The nanopores and functionalization ability of graphene membrane make it suitable for advanced infiltration flux. The ultimate atomic thickness of graphene results in efficient membranes because there is no scattering of molecules from pore walls during transport. Sint et al. [172] used molecular dynamics simulations to investigate ion diffusion (Lithium ions, Sodium, potassium, chlorine and bromine) through single layered graphene with functionalized nanopores. Their research demonstrated that nano porous graphene monolayers can be used as ion separation membranes for desalination and energy conversion. In addition, Cohen-Tanugi et al. [173] probed H2O desalination by monolayer graphene nanopores. They tested water permeability across nano porous graphene with different pore sizes and functional groups and found that it was 67 L/cm2 which was twice or thrice the times of traditional RO membranes.
- f.
-
Graphene blended materials at cryogenic environments: The current era, especially the automobile and aerospace industry, is focusing more on weightless materials to reduce component weight. As a result, fibre reinforced polymer (FRP) composites have piqued the industrial sector’s interest. Because of their lightweight and high-strength properties, FRP is now being used to replace high pressurized metallic tanks in reusable and non-essential space launch vehicles. Another critical lightweight material, carbon fibre-reinforced polymer (CFRP), is utilized to produce components in space technology. Properties like chemical inertness, thermal stability, medium mechanical property of thermosetting resins make it suitable in manufacturing CFRP. As thermal fatigue loading occurs, they become extremely susceptible to cracking due to its cross-linked structures [174]. Researchers have utilized various reinforcements such as CNTs [175][176], nanoclay [177], and graphene [178] to improve the composite properties in the past to solve this issue. While carbon nanotubes, nano clay have shown promising outcomes in improving characteristics under low-temperature conditions, these materials are prohibitively expensive and difficult to fabricate in the real world. Graphene along with its lineages is currently of considerable importance due to theirnear-perfect mechanical, electrical, and thermal properties. Furthermore, the wide surface area of graphene plays a crucial part in playing the role of excellent nano supporting material for many polymer composites [179][180]. The presence of epoxide, carboxyl, carbonyl, and hydroxyl functional groups in GO, makes it suitable to work as nano-filler in polymeric composites having good chemical reactiveness and easy handling [181][182]. The interfacial bonding is enhanced between the polymeric matrix and the composite by providing a GO coating onto the carbon fabric [183]. Hung et al. [184] performed GO coating using electrophoretic deposition method, increasing inter laminar shear strength, Youngs modulus and ultimate tensile strength in cryogenic conditions. According to Wu et al. [185], GNPs function as a bridging filler, linking crack surfaces together to prevent crack propagation in Al2O3. Just 1.0 vol percent GNPs is used to test the maximum fracture toughness of GNP/Al2O3.
- g.
-
Growth of 2D-materials: Intercalation is one of the methods to tune graphene’s properties. Intercalation includes epitaxial graphene (EG) growth on Silicon Carbide (SiC). The main advantage of this technique is to obtain layers of graphene on semiconducting or semi-insulating substrates. EG on SiC is considered as a novel material for high end electronics. Intercalation through EG on SiC began few years before. This technique modulates graphenes properties to create new 2D forms of 3D materials. The literature reveals the intercalation of different metallic atoms through EG layers [186][187]. The developed 2D material is a function of intercalation temperature, pressure and the intercalant. Thermal evaporation based intercalation methods are the alternatives of complex synthesis techniques. Metal atoms (for example Co, Pt, Fe, Au, Mg and Ag) are deposited on EG surface preferably through thermal or E-beam evaporation. Gas-phase precursors likeH2, O2, air and NH3 can also be used to achieve intercalations. As like metal atoms deposited on EG, gaseous species adsorb on surface of graphene and diffuse into the graphene layers. Here we have reviewed in short, about the creation of 2D Silver as reported by N. Briggs et al. [188]. They disclosed that, the material like Silver finds applications in Plasmon-based technologies in order to produce devices like biosensors and waveguides. Providing Ag films on plasmonic devices is essential, but the protection of the Ag film atoms needs to be realized. So intercalation of Ag atoms using EG layers is one of the promising technique to provide an overlying protection. The Ag atoms are intercalated through EG/SiC. The encapsulated Ag atoms through EG/SiC find places for plasmonic applications. The synthesis of 2D-Ag atoms are shown in Figure 17 in pictorial form. The detailed synthesis method and other aspects of this process can be viewed by following N. Briggs et al. [188].
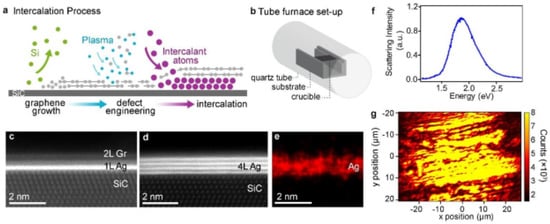 Figure 17. (a) Intercalation schematic illustrating graphene growth, plasma treatment, and metal intercalation (b) Diagram of experimental setup depicting a quartz tube containing an alumina crucible and an EG/SiC substrate placed facing downward on the crucible over the metal source. (c) Cross-sectional STEM showing 1 and (d) 4 layers of Ag between EG and SiC. (e) Corresponding Ag EDS. (f) Dark field scattering spectrum showing a peak at 1.87 eV, reflecting the plasmonic character of the Ag film. (g) Map of SHG peak across the EG/Ag/SiC surface showing the presence of Ag layers across 10 s of microns. “Reproduced with permission from [188], [Nanoscale]; published by [Royal Society of Chemistry], 2019”.
Figure 17. (a) Intercalation schematic illustrating graphene growth, plasma treatment, and metal intercalation (b) Diagram of experimental setup depicting a quartz tube containing an alumina crucible and an EG/SiC substrate placed facing downward on the crucible over the metal source. (c) Cross-sectional STEM showing 1 and (d) 4 layers of Ag between EG and SiC. (e) Corresponding Ag EDS. (f) Dark field scattering spectrum showing a peak at 1.87 eV, reflecting the plasmonic character of the Ag film. (g) Map of SHG peak across the EG/Ag/SiC surface showing the presence of Ag layers across 10 s of microns. “Reproduced with permission from [188], [Nanoscale]; published by [Royal Society of Chemistry], 2019”. - h.
-
Miscellaneous applications: Contamination testing, water treatment, and energy production are only a few of the new usances of graphene that have surfaced recently [189]. The high electron mobility of graphene, which ranges from 10,000 to 50,000 cm2·V−1·s−1, makes it useful for reducing emissions. For environmental refining, graphene-based sorbents and photocatalytic materials are used. GNPs are often used as adsorbents in the marine environment to dissolve organic compounds, mineral components, and gases like CO2. They also act as photocatalytic products, removing contaminants from water. Photo-catalysis and environmental decontamination are also observed in graphene-based silver nitrate nanocomposites. Graphene membranes with nano pores are the ultimate separators of mixed gases. These find applications in biogas production, hydrogen recovery from ammonia plants and oil refineries, carbon capture. Graphene-based nanocomposites are extremely powerful liquid-gas barriers. Water separation was achieved using nano porous graphene membranes and stacked GO membranes. Furthermore, studies have discovered that graphene reacts with the microbial lipid layer and have a major impact on antimicrobial activity [189]. Cho et al. [190] developed graphene-reinforced polyamide nanocomposites for LED lamp production. G. Jiang et al. [191] prepared a flexible LED using laser induced reduced graphene oxide (LIRGO) which has luminescence lifetime of over 60 h and WPE (wall plug efficiency) of 1.4%. In addition, graphene materials can be used to create paints which conducts electricity, shielding of electromagnetic radiations (EMI shielding). Wearable devices, which are commonly used for monitoring health, detecting motion, and environmental monitoring, are some of the recent advancements where graphene has found its applicability [192][193]. Graphene has made quite a stir in the wearable devices market, allowing for precise detection and/or tracking of a variety of critical signals from both humans and the environment. Researches on the thin graphene nanoparticles sheets having large surface area but lesser thickness has revealed that they impart better tensile, flexural strength, morphological and other thermal properties and boosts the performance efficiency of the final composites [194][195][196][197][198][199][200]. Three phase, lead-free, ZnO-graphene-epoxy electroactive nanocomposite thin films has been synthesized to serve as a replacement to toxic perovskite oxides traditionally used in sensors, actuators and biomedical wearable devices [201][202][203]. Graphene has also been used as templates to grow metal oxides on it as described in literature [204][205][206].
References
- Gaj, J.; Clapa, M.; Nowak, D.; Juszczak, J.; Galazka, M.; Pelka, M.; Niedzielski, P. Metallurgical graphene under different gas atmospheres and UV radiation for gas-sensing applications. Sens. Actuators A Phys. 2020, 312, 112152.
- Geim, A.K.; Novoselov, K. The rise of graphene. Nat. Mater. 2007, 6, 183–191.
- Ferrari, A.C.; Bonaccorso, F.; Fal’Ko, V.; Novoselov, K.S.; Roche, S.; Bøggild, P.; Borini, S.; Koppens, F.H.L.; Palermo, V.; Pugno, N.; et al. Science and technology roadmap for graphene, related two-dimensional crystals, and hybrid systems. Nanoscale 2015, 7, 4598–4810.
- Varghese, S.S.; Lonkar, S.; Singh, K.K.; Swaminathan, S.; Abdala, A. Recent advances in graphene based gas sensors. Sens. Actuators B Chem. 2015, 218, 160–183.
- Mathew, T.; Sree, R.A.; Aishwarya, S.; Kounaina, K.; Patil, A.G.; Satapathy, P.; Hudeda, S.; More, S.S.; Muthucheliyan, K.; Kumar, T.N.; et al. Graphene-based functional nanomaterials for biomedical and bioanalysis applications. FlatChem 2020, 23, 100184.
- Schedin, F.; Geim, A.K.; Morozov, S.V.; Hill, E.W.; Blake, P.; Katsnelson, M.I.; Novoselov, K.S. Detection of individual gas molecules adsorbed on graphene. Nat. Mater. 2007, 6, 652–655.
- Cao, G.; Liu, X.; Liu, W.; Li, Q.; Li, X.; Wang, X. Chemical environment dominated Fermi level pinning of a graphene gas sensor. Carbon 2017, 124, 57–63.
- Sakr, M.A.; Elgammal, K.; Delin, A.; Serry, M. Performance-Enhanced Non-Enzymatic Glucose Sensor Based on Graphene-Heterostructure. Sensors 2020, 20, 145.
- Fan, X.; Elgammal, K.; Smith, A.D.; Östling, M.; Delin, A.; Lemme, M.C.; Niklaus, F. Humidity and CO2 gas sensing properties of double-layer graphene. Carbon 2018, 127, 576–587.
- Rodner, M.; Bahonjic, J.; Mathisen, M.; Gunnarsson, R.; Ekeroth, S.; Helmersson, U.; Ivanov, I.G.; Yakimova, R.; Eriksson, J. Performance tuning of gas sensors based on epitaxial graphene on silicon carbide. Mater. Des. 2018, 153, 153–158.
- Zhang, Y.; Zhang, J.; Huang, X.; Zhou, X.; Wu, H.; Guo, S. Assembly of Graphene Oxide-Enzyme Conjugates through Hydrophobic Interaction. Small 2012, 8, 154–159.
- Boukhvalov, D.W.; Katsnelson, M.I. Modeling of Graphite Oxide. J. Am. Chem. Soc. 2008, 130, 10697–10701.
- Zhu, S.-E.; Ghatkesar, M.K.; Zhang, C.; Janssen, G.C.A.M. Graphene based piezoresistive pressure sensor. Appl. Phys. Lett. 2013, 102, 161904.
- Nafiujjaman, M.; Nurunnabi, M.; Kang, S.-H.; Reeck, G.R.; Khan, H.A.; Lee, Y.-K. Ternary graphene quantum dot-polydopamine-Mn3O4 nanoparticles for optical imaging guided photodynamic therapy and T1-weighted magnetic resonance imaging. J. Mater. Chem. B 2015, 3, 5815–5823.
- Moore, T.; Podilakrishna, R.; Rao, A.; Alexis, F. Systemic Administration of Polymer-Coated Nano-Graphene to Deliver Drugs to Glioblastoma. Part. Part. Syst. Charact. 2014, 31, 886–894.
- Savoie, B. On the zero-field orbital magnetic susceptibility of Bloch electrons in graphene-like solids: Some rigorous results. J. Math. Phys. 2012, 53, 073302.
- Schwiertz, J.; Wiehe, A.; Gräfe, S.; Gitter, B.; Epple, M.; Schwiertz, J.; Wiehe, A.; Gräfe, S.; Gitter, B.; Epple, M. Calcium phosphate nanoparticles as efficient carriers for photodynamic therapy against cells and bacteria. Biomaterials 2009, 30, 3324–3331.
- Zhou, J.; Liu, J.; Xia, S.; Wang, X.; Zhang, B. Effect of chelation to lanthanum ions on the photodynamic properties of hypocrellin A. J. Phys. Chem. B 2005, 109, 19529–19535.
- Iqbal, A.A.; Sakib, N.; Nuruzzaman, D.M. Graphene-based nanocomposites and their fabrication, mechanical properties and applications. Materialia 2020, 12, 100815.
- Bunch, J.S.; van der Zande, A.; Verbridge, S.S.; Frank, I.W.; Tanenbaum, D.M.; Parpia, J.M.; Craighead, H.G.; McEuen, P.L. Electromechanical Resonators from Graphene Sheets. Science 2007, 315, 490–493.
- Schwierz, F. Graphene transistors. Nat. Nanotechnol. 2010, 5, 487–496.
- Raju, A.P.A.; Lewis, A.; Derby, B.; Young, R.; Kinloch, I.A.; Zan, R.; Novoselov, K. Wide-Area Strain Sensors based upon Graphene-Polymer Composite Coatings Probed by Raman Spectroscopy. Adv. Funct. Mater. 2014, 24, 2865–2874.
- Eda, G.; Chhowalla, M. Chemically Derived Graphene Oxide: Towards Large-Area Thin-Film Electronics and Optoelectronics. Adv. Mater. 2010, 22, 2392–2415.
- Yoo, J.; Balakrishnan, K.; Huang, J.; Meunier, V.; Sumpter, B.; Srivastava, A.; Conway, M.; Reddy, A.L.M.; Yu, J.; Vajtai, R.; et al. Ultrathin Planar Graphene Supercapacitors. Nano Lett. 2011, 11, 1423–1427.
- Brownson, D.A.C.; Banks, C.E. Fabricating graphene supercapacitors: Highlighting the impact of surfactants and moieties. Chem. Commun. 2011, 48, 1425–1427.
- Patchkovskii, S.; Tse, J.S.; Yurchenko, S.; Zhechkov, L.; Heine, T.; Seifert, G. From The Cover: Graphene nanostructures as tunable storage media for molecular hydrogen. Proc. Natl. Acad. Sci. USA 2005, 102, 10439–10444.
- Wang, X.; Zhi, L.; Müllen, K. Transparent, Conductive Graphene Electrodes for Dye-Sensitized Solar Cells. Nano Lett. 2008, 8, 323–327.
- Miao, X.; Tongay, S.; Petterson, M.K.; Berke, K.; Rinzler, A.G.; Appleton, B.R.; Hebard, A.F. High Efficiency Graphene Solar Cells by Chemical Doping. Nano Lett. 2012, 12, 2745–2750.
- Shen, J.; Zhu, Y.; Yang, X.; Li, C. Graphene quantum dots: Emergent nanolights for bioimaging, sensors, catalysis and photovoltaic devices. Chem. Commun. 2012, 48, 3686–3699.
- Jia, X.; Campos-Delgado, J.; Terrones, M.; Meunier, V.; Dresselhaus, M.S. Graphene edges: A review of their fabrication and characterization. Nanoscale 2010, 3, 86–95.
- Xu, Y.; Sheng, K.; Li, C.; Shi, G. Self-Assembled Graphene Hydrogel via a One-Step Hydrothermal Process. ACS Nano 2010, 4, 4324–4330.
- Xue, Y.; Liu, J.; Chen, H.; Wang, R.; Li, D.; Qu, J.; Dai, L. Nitrogen-Doped Graphene Foams as Metal-Free Counter Electrodes in High-Performance Dye-Sensitized Solar Cells. Angew. Chem. Int. Ed. 2012, 51, 12124–12127.
- Wu, Z.-S.; Sun, Y.; Tan, Y.-Z.; Yang, S.; Feng, X.; Müllen, K. Three-Dimensional Graphene-Based Macro- and Mesoporous Frameworks for High-Performance Electrochemical Capacitive Energy Storage. J. Am. Chem. Soc. 2012, 134, 19532–19535.
- Moghadam, A.D.; Omrani, E.; Menezes, P.L.; Rohatgi, P.K. Mechanical and tribological properties of self-lubricating metal matrix nanocomposites reinforced by carbon nanotubes (CNTs) and graphene—A review. Compos. Part B Eng. 2015, 77, 402–420.
- Tong, X.; Wei, Q.; Zhan, X.; Zhang, G.; Sun, S. The New Graphene Family Materials: Synthesis and Applications in Oxygen Reduction Reaction. Catalysts 2017, 7, 1.
- Duplock, E.J.; Scheffler, M.; Lindan, P.J.D. Hallmark of Perfect Graphene. Phys. Rev. Lett. 2004, 92, 225502.
- Novoselov, K.S.; Geim, A.K.; Morozov, S.V.; Jiang, D.; Zhang, Y.; Dubonos, S.V.; Grigorieva, I.V.; Firsov, A.A. Electric field effect in atomically thin carbon films. Science 2004, 306, 666–669.
- Lee, C.; Wei, X.; Kysar, J.W.; Hone, J. Measurement of the elastic properties and intrinsic strength of monolayer graphene. Science 2008, 321, 385–388.
- Balandin, A.A.; Ghosh, S.; Bao, W.; Calizo, I.; Teweldebrhan, D.; Miao, F.; Lau, C.N. Superior Thermal Conductivity of Single-Layer Graphene. Nano Lett. 2008, 8, 902–907.
- Zhang, L.; Zhang, F.; Yang, X.; Long, G.; Wu, Y.; Zhang, T. Porous 3D graphene based bulk materials with exceptional high surface area and excellent conductivity for supercapacitors. Sci. Rep. 2013, 3, 1408.
- Stankovich, S.; Piner, R.D.; Nguyen, S.; Ruoff, R.S. Synthesis and exfoliation of isocyanate-treated graphene oxide nanoplatelets. Carbon 2006, 44, 3342–3347.
- Becerril, H.A.; Mao, J.; Liu, Z.; Stoltenberg, R.M.; Bao, Z.; Chen, Y. Evaluation of Solution-Processed Reduced Graphene Oxide Films as Transparent Conductors. ACS Nano 2008, 2, 463–470.
- Xu, Y.; Liu, Z.; Zhang, X.; Wang, Y.; Tian, J.; Huang, Y.; Ma, Y.; Zhang, X.; Chen, Y. A Graphene Hybrid Material Covalently Functionalized with Porphyrin: Synthesis and Optical Limiting Property. Adv. Mater. 2009, 21, 1275–1279.
- Guo, Q.; Wang, G.; Pandey, R.; Karna, S. Robust band gaps in the graphene/oxide heterostructure: SnO/graphene/SnO. Phys. Chem. Chem. Phys. 2018, 20, 17983–17989.
- Gadipelli, S.; Guo, Z.X. Graphene-based materials: Synthesis and gas sorption, storage and separation. Prog. Mater. Sci. 2015, 69, 1–60.
- Novoselov, K.S.; Geim, A.K.; Morozov, S.V.; Jiang, D.; Katsnelson, M.I.; Grigorieva, I.V.; Dubonos, S.V.; Firsov, A.A. Two-dimensional gas of massless Dirac fermions in graphene. Nature 2005, 438, 197–200.
- Novoselov, K.S.; Fal′ko, V.I.; Colombo, L.; Gellert, P.R.; Schwab, M.G.; Kim, K. A roadmap for graphene. Nat. Cell Biol. 2012, 490, 192–200.
- Dikin, D.A.; Stankovich, S.; Zimney, E.J.; Piner, R.D.; Dommett, G.H.B.; Evmenenko, G.; Nguyen, S.; Ruoff, R.S. Preparation and characterization of graphene oxide paper. Nat. Cell Biol. 2007, 448, 457–460.
- Edwards, R.S.; Coleman, K.S. Graphene synthesis: Relationship to applications. Nanoscale 2013, 5, 38–51.
- Kudr, J.; Richtera, L.; Nejdl, L.; Xhaxhiu, K.; Vitek, P.; Rutkay-Nedecky, B.; Hynek, D.; Kopel, P.; Adam, V.; Kizek, R. Improved Electrochemical Detection of Zinc Ions Using Electrode Modified with Electrochemically Reduced Graphene Oxide. Materials 2016, 9, 31.
- Güler, Ö.; Güler, S.H.; Selen, V.; Albayrak, M.G.; Evin, E. Production of graphene layer by liquid-phase exfoliation with low sonication power and sonication time from synthesized expanded graphite. Full. Nanotub. Carbon Nanostruct. 2015, 24, 123–127.
- Haar, S.; Bruna, M.; Lian, J.X.; Tomarchio, F.; Olivier, Y.; Mazzaro, R.; Morandi, V.; Moran, J.; Ferrari, A.C.; Beljonne, D.; et al. Liquid-Phase Exfoliation of Graphite into Single- and Few-Layer Graphene with α-Functionalized Alkanes. J. Phys. Chem. Lett. 2016, 7, 2714–2721.
- Wu, Y.; Fang, Y.; Ren, X.; Lu, H. Back propagation neural networks based hysteresis modeling and compensation for a piezoelectric scanner. In Proceedings of the 2016 IEEE International Conference on Manipulation, Manufacturing and Measurement on the Nanoscale (3M-NANO), Chongqing, China, 18–22 July 2016; Institute of Electrical and Electronics Engineers (IEEE): Piscataway, NJ, USA, 2016; pp. 119–124.
- Chua, C.K.; Pumera, M. Chemical reduction of graphene oxide: A synthetic chemistry viewpoint. Chem. Soc. Rev. 2014, 43, 291–312.
- Dimiev, A.M.; Khannanov, A.; Vakhitov, I.; Kiiamov, A.; Shukhina, K.; Tour, J.M. Revisiting the Mechanism of Oxidative Unzipping of Multiwall Carbon Nanotubes to Graphene Nanoribbons. ACS Nano 2018, 12, 3985–3993.
- Jacobberger, R.M.; Machhi, R.; Wroblewski, J.; Taylor, B.; Gillian-Daniel, A.L.; Arnold, M.S. Simple Graphene Synthesis via Chemical Vapor Deposition. J. Chem. Educ. 2015, 92, 1903–1907.
- Son, M.; Ham, M.-H. Low-temperature synthesis of graphene by chemical vapor deposition and its applications. FlatChem 2017, 5, 40–49.
- Koo, J.-H.; Paek, S.-M. Microwave-Assisted Synthesis of Ge/GeO2-Reduced Graphene Oxide Nanocomposite with Enhanced Discharge Capacity for Lithium-Ion Batteries. Nanomaterials 2021, 11, 319.
- Huang, H.; Chen, S.; Wee, A.T.S.; Chen, W. 1-Epitaxial growth of graphene on silicon carbide (SiC). Graphene Prop. Prep. Characterisation Devices 2014, 3–26.
- Yazdi, G.R.; Iakimov, T.; Yakimova, R. Epitaxial Graphene on SiC: A Review of Growth and Characterization. Crystals 2016, 6, 53.
- Yang, Y.; Liu, R.; Wu, J.; Jiang, X.; Cao, P.; Hu, X.; Pan, T.; Qiu, C.; Yang, J.; Song, Y.; et al. Bottom-up Fabrication of Graphene on Silicon/Silica Substrate via a Facile Soft-hard Template Approach. Sci. Rep. 2015, 5, 13480.
- Singh, V.; Joung, D.; Zhai, L.; Das, S.; Khondaker, S.I.; Seal, S. Graphene based materials: Past, present and future. Prog. Mater. Sci. 2011, 56, 1178–1271.
- Mao, H.N.; Wang, X.G. Use of in-situ polymerization in the preparation of graphene/polymer nanocomposites. New Carbon Mater. 2020, 35, 336–343.
- Ambrosi, A.; Chua, C.K.; Bonanni, A.; Pumera, M. Electrochemistry of Graphene and Related Materials. Chem. Rev. 2014, 114, 7150–7188.
- Sieradzka, M.; Ślusarczyk, C.; Biniaś, W.; Fryczkowski, R. The Role of the Oxidation and Reduction Parameters on the Properties of the Reduced Graphene Oxide. Coatings 2021, 11, 166.
- Costa, M.C.F.; Marangoni, V.S.; Ng, P.R.; Nguyen, H.T.L.; Carvalho, A.; Castro Neto, A.H. Accelerated Synthesis of Graphene Oxide from Graphene. Nanomaterials 2021, 11, 551.
- Boehm, H.P.; Clauss, A.; Fischer, G.O.; Hofmann, U. DünnsteKohlenstoff-Folien. Z. Nat. B 1962, 17, 150–153.
- Hernandez, Y.; Nicolosi, V.; Lotya, M.; Blighe, F.M.; Sun, Z.; De, S.; McGovern, I.T.; Holland, B.; Byrne, M.; Gun’Ko, Y.; et al. High-yield production of graphene by liquid-phase exfoliation of graphite. Nat. Nanotechnol. 2008, 3, 563–568.
- Hernandez, Y.; Lotya, M.; Rickard, D.; Bergin, S.D.; Coleman, J. Measurement of Multicomponent Solubility Parameters for Graphene Facilitates Solvent Discovery. Langmuir 2010, 26, 3208–3213.
- Blake, P.; Brimicombe, P.D.; Nair, R.R.; Booth, T.J.; Jiang, D.; Schedin, F.; Ponomarenko, L.A.; Morozov, S.V.; Gleeson, H.F.; Hill, E.W.; et al. Graphene-based liquid crystal device. Nano Lett. 2008, 8, 1704–1708.
- Jnioui, A.; Metrot, A.; Storck, A. Electrochemical production of graphite salts using a three-dimensional electrode of graphite particles. Electrochim. Acta 1982, 27, 1247–1252.
- Takada, Y.; Fujii, R. The Electrochemical Formation of Graphite Intercalation Compound in γ-Butyrolactone. TANSO 1985, 1985, 110–113.
- Noel, M.; Santhanam, R.; Flora, M.F. Effect of polypyrrole film on the stability and electrochemical activity of fluoride based graphite intercalation compounds in HF media. J. Appl. Electrochem. 1994, 24, 455–459.
- Low, C.T.J.; Walsh, F.C.; Chakrabarti, M.H.; Hashim, M.A.; Hussain, M.A. Electrochemical approaches to the production of graphene flakes and their potential applications. Carbon 2013, 54, 1–21.
- Yang, X.; Dou, X.; Rouhanipour, A.; Zhi, L.; Räder, A.H.J.; Müllen, K. Two-Dimensional Graphene Nanoribbons. J. Am. Chem. Soc. 2008, 130, 4216–4217.
- Cai, J.; Ruffieux, P.; Jaafar, R.; Bieri, M.; Braun, T.; Blankenburg, S.; Muoth, M.; Seitsonen, A.P.; Saleh, M.; Feng, X.; et al. Atomically precise bottom-up fabrication of graphene nanoribbons. Nat. Cell Biol. 2010, 466, 470–473.
- Berger, C.; Song, Z.; Li, T.; Li, X.; Ogbazghi, A.Y.; Feng, R.; Dai, Z.; Marchenkov, A.N.; Conrad, E.H.; First, P.N.; et al. Ultrathin Epitaxial Graphite: 2D Electron Gas Properties and a Route toward Graphene-based Nanoelectronics. J. Phys. Chem. B 2004, 108, 19912–19916.
- Berger, C.; Song, Z.; Li, X.; Wu, X.; Brown, N.; Naud, C.; Mayou, D.; Hass, J.; Marchenkov, A.N.; Conrad, E.H.; et al. Electronic Confinement and Coherence in Patterned Epitaxial Graphene. Science 2006, 312, 1191–1196.
- Brodie, B. Note sur un nouveau procédé pour la purification et la désagrégation du graphite. Ann. Chim. Phys. 1855, 45, 351–353.
- Staudenmaier, L. VerfahrenzurDarstellung der Graphitsäur. Ber. Dtsch. Chem. Ges. 1898, 31, 1481–1487.
- Zaaba, N.; Foo, K.; Hashim, U.; Tan, S.; Liu, W.W.; Voon, C. Synthesis of Graphene Oxide using Modified Hummers Method: Solvent Influence. Procedia Eng. 2017, 184, 469–477.
- Kosowska, K.; Domalik-Pyzik, P.; Krok-Borkowicz, M.; Chłopek, J. Synthesis and Characterization of Chitosan/Reduced Graphene Oxide Hybrid Composites. Materials 2019, 12, 2077.
- Muñoz, R.; Gómez-Aleixandre, C. Review of CVD Synthesis of Graphene. Chem Vap. Deposition. 2013, 19, 297–322.
- Kobayashi, T.; Bando, M.; Kimura, N.; Shimizu, K.; Kadono, K.; Umezu, N.; Miyahara, K.; Hayazaki, S.; Nagai, S.; Mizuguchi, Y.; et al. Growth of continuous graphene by open roll-to-roll chemical vapor deposition. Appl. Phys. Lett. 2013, 102, 023112.
- Somanathan, T.; Prasad, K.; Ostrikov, K.; Saravanan, A.; Krishna, V.M. Graphene Oxide Synthesis from Agro Waste. Nanomaterials 2015, 5, 826–834.
- Bae, S.; Kim, H.K.; Lee, Y.; Xu, X.; Park, J.-S.; Zheng, Y.; Balakrishnan, J.; Lei, T.; Kim, H.R.; Song, Y.I.; et al. Roll-to-roll production of 30-inch graphene films for transparent electrodes. Nat. Nanotechnol. 2010, 5, 574–578.
- Pan, D.; Zhang, J.; Li, Z.; Wu, M. Hydrothermal Route for Cutting Graphene Sheets into Blue-Luminescent Graphene Quantum Dots. Adv. Mater. 2010, 22, 734–738.
- Xie, J.-D.; Lai, G.-W.; Huq, M.M. Hydrothermal route to graphene quantum dots: Effects of precursor and temperature. Diam. Relat. Mater. 2017, 79, 112–118.
- Shin, Y.; Park, J.; Hyun, D.; Yang, J.; Lee, J.-H.; Kim, J.-H.; Lee, H. Acid-free and oxone oxidant-assisted solvothermal synthesis of graphene quantum dots using various natural carbon materials as resources. Nanoscale 2015, 7, 5633–5637.
- Huang, D.; Zhou, H.; Wu, Y.; Wang, T.; Sun, L.; Gao, P.; Sun, Y.; Huang, H.; Zhou, G.; Hu, J. Bottom-up synthesis and structural design strategy for graphene quantum dots with tunable emission to the near infrared region. Carbon 2019, 142, 673–684.
- Umrao, S.; Jang, M.-H.; Oh, J.-H.; Kim, G.; Sahoo, S.; Cho, Y.-H.; Srivastva, A.; Oh, I. Microwave bottom-up route for size-tunable and switchable photoluminescent graphene quantum dots using acetylacetone: New platform for enzyme-free detection of hydrogen peroxide. Carbon 2015, 81, 514–524.
- Chen, W.; Shen, J.; Lv, G.; Li, D.; Hu, Y.; Zhou, C.; Liu, X.; Dai, Z. Green Synthesis of Graphene Quantum Dots from Cotton Cellulose. Chem. Sel. 2019, 4, 2898–2902.
- Zhao, Y.; Wen, M.; He, C.; Liu, C.; Li, Z.; Liu, Y. Preparation of graphene by catalytic pyrolysis of lignin and its electrochemical properties. Mater. Lett. 2020, 274, 128047.
- Dato, A.; Frenklach, M. Substrate-free microwave synthesis of graphene: Experimental conditions and hydrocarbon precursors. New J. Phys. 2010, 12, 125013.
- Thomas, D.-G.; Kavak, E.; Hashemi, N.; Montazami, R.; Hashemi, N.N. Synthesis of Graphene Nanosheets through Spontaneous Sodiation Process. C 2018, 4, 42.
- Dato, A.; Lee, Z.; Jeon, K.-J.; Erni, R.; Radmilovic, V.; Richardson, T.J.; Frenklach, M. Clean and highly ordered graphene synthesized in the gas phase. Chem. Commun. 2009, 40, 6095–6097.
- Guo, H.-L.; Wang, X.-F.; Qian, Q.-Y.; Wang, F.-B.; Xia, X.-H. A Green Approach to the Synthesis of Graphene Nanosheets. ACS Nano 2009, 3, 2653–2659.
- Kovtyukhova, N.I.; Ollivier, P.J.; Martin, B.R.; Mallouk, T.E.; Chizhik, S.A.; Buzaneva, E.V.; Gorchinskiy, A.D. Layer-byLayer Assembly of Ultrathin Composite Films from MicronSized Graphite Oxide Sheets and Polycations. Chem. Mater. 1999, 11, 771–778.
- Hummers, W.S.; Offeman, R.E. Preparation of Graphitic Oxide. J. Am. Chem. Soc. 1958, 80, 1339.
- Vesel, A.; Zaplotnik, R.; Primc, G.; Mozetič, M. Synthesis of Vertically Oriented Graphene Sheets or Carbon Nanowalls—Review and Challenges. Materials 2019, 12, 2968.
- Sengupta, J.; Hussain, C.M. Graphene and its derivatives for Analytical Lab on Chip platforms. TrAC Trends Anal. Chem. 2019, 114, 326–337.
- Cooper, D.R.; D’Anjou, B.; Ghattamaneni, N.; Harack, B.; Hilke, M.; Horth, A.; Majlis, N.; Massicotte, M.; Vandsburger, L.; Whiteway, E.; et al. Experimental Review of Graphene. ISRN Condens. Matter Phys. 2012, 2012, 501686.
- Geim, K. Graphene: Status and prospects. Science 2009, 324, 1530–1534.
- Jagiełło, J.; Chlanda, A.; Baran, M.; Gwiazda, M.; Lipińska, L. Synthesis and Characterization of Graphene Oxide and Reduced Graphene Oxide Composites with Inorganic Nanoparticles for Biomedical Applications. Nanomaterials 2020, 10, 1846.
- Brodie, B.C. XXIII.—Researches on the atomic weight of graphite. Q. J. Chem. Soc. 1860, 12, 261–268.
- Shen, B.; Ding, J.; Yan, X.; Feng, W.; Li, J.; Xue, Q. Influence of different buffer gases on synthesis of few-layered graphene by arc discharge method. Appl. Surf. Sci. 2012, 258, 4523–4531.
- Kim, S.; Song, Y.; Wright, J.; Heller, M.J. Graphene bi- and trilayers produced by a novel aqueous arc discharge process. Carbon 2016, 102, 339–345.
- Cho, S.; Kikuchi, K.; Kawasaki, A. Radial followed by longitudinal unzipping of multiwalled carbon nanotubes. Carbon 2011, 49, 3865–3872.
- Kosynkin, D.V.; Higginbotham, A.L.; Sinitskii, A.; Lomeda, J.R.; Dimiev, A.; Price, B.K.; Tour, J. Longitudinal unzipping of carbon nanotubes to form graphene nanoribbons. Nat. Cell Biol. 2009, 458, 872–876.
- Saeed, M.; Alshammari, Y.; Majeed, S.A.; Al-Nasrallah, E. Chemical Vapour Deposition of Graphene—Synthesis, Characterisation, and Applications: A Review. Molecules 2020, 25, 3856.
- Yi, M.; Shen, Z. A review on mechanical exfoliation for the scalable production of graphene. J. Mater. Chem. A 2015, 3, 11700–11715.
- Zhu, Y.; Murali, S.; Cai, W.; Li, X.; Suk, J.W.; Potts, J.R.; Ruoff, R.S. Graphene-based Materials: Graphene and Graphene Oxide: Synthesis, Properties, and Applications. Adv. Mater. 2010, 22, 3906–3924.
- Stankovich, S.; Dikin, D.A.; Piner, R.D.; Kohlhaas, K.A.; Kleinhammes, A.; Jia, Y.; Wu, Y.; Nguyen, S.; Ruoff, R.S. Synthesis of graphene-based nanosheets via chemical reduction of exfoliated graphite oxide. Carbon 2007, 45, 1558–1565.
- Parades, J.I.; Villar-Rodil, S.; Martínez-Alonso, A.; Tascon, J.M.D. Graphene oxide dispersions in organic solvents. Langmuir 2008, 24, 10560–10564.
- McAllister, M.J.; Li, J.-L.; Adamson, D.H.; Schniepp, H.; Abdala, A.; Liu, J.; Herrera-Alonso, M.; Milius, D.L.; Car, R.; Prud’Homme, R.K.; et al. Single Sheet Functionalized Graphene by Oxidation and Thermal Expansion of Graphite. Chem. Mater. 2007, 19, 4396–4404.
- Marcano, D.C.; Kosynkin, D.V.; Berlin, J.M.; Sinitskii, A.; Sun, Z.; Slesarev, A.; Alemany, L.B.; Lu, W.; Tour, J.M. Improved Synthesis of Graphene Oxide. ACS Nano 2010, 4, 4806–4814.
- Vandsburger, L.; Swanson, E.J.; Tavares, J.; Meunier, J.-L.; Coulombe, S. Stabilized aqueous dispersion of multi-walled carbon nanotubes obtained by RF glow-discharge treatment. J. Nanopart. Res. 2009, 11, 1817–1822.
- Hazra, K.S.; Rafiee, J.; Rafiee, M.A.; Mathur, A.; Roy, S.S.; Mclauhglin, J.; Koratkar, N.; Misra, D.S.; Hazra, K.S.; Rafiee, J.; et al. Thinning of multilayer graphene to monolayer graphene in a plasma environment. Nanotechnology 2010, 22, 025704.
- Childres, I.; Jauregui, L.A.; Tian, J.; Chen, Y. Effect of oxygen plasma etching on graphene studied using Raman spectroscopy and electronic transport measurements. New J. Phys. 2011, 13, 025008.
- Nourbakhsh, A.; Cantoro, M.; Vosch, T.; Pourtois, G.; Clemente, F.; van der Veen, M.H.; Hofkens, J.; Heyns, M.M.; De Gendt, S.; Sels, B.F. Bandgap opening in oxygen plasma-treated graphene. Nanotechnology 2010, 21, 435203.
- Adetayo, A.; Runsewe, D. Synthesis and Fabrication of Graphene and Graphene Oxide: A Review. Open J. Compos. Mater. 2019, 9, 207–229.
- Wu, S.; Wang, X.; Li, Z.; Zhang, S.; Xing, F. Recent Advances in the Fabrication and Application of Graphene Microfluidic Sensors. Micromachines 2020, 11, 1059.
- Singh, D.K.; Iyer, P.K.; Giri, P.K. Improved chemical synthesis of graphene using a safer solvothermal route. Int. J. Nanosci. 2011, 10, 39–42.
- Sun, J.; Zhang, Y.; Liu, Z. Direct chemical vapor deposition growth of graphene on insulating substrates. ChemNanoMat 2016, 2.
- Dato, A.; Radmilovic, V.; Lee, Z.; Phillips, J.; Frenklach, M. Substrate-Free Gas-Phase Synthesis of Graphene Sheets. Nano Lett. 2008, 8, 2012–2016.
- Yang, Y.; Zhao, R.; Zhang, T.; Zhao, K.; Xiao, P.; Ma, Y.; Ajayan, P.M.; Shi, G.; Chen, Y.; Yang, Y.; et al. Graphene-Based Standalone Solar Energy Converter for Water Desalination and Purification. ACS Nano 2018, 12, 829–835.
- Ren, H.; Tang, M.; Guan, B.; Wang, K.; Yang, J.; Wang, F.; Wang, M.; Shan, J.; Chen, Z.; Wei, D.; et al. Hierarchical Graphene Foam for Efficient Omnidirectional Solar—Thermal Energy Conversion. Adv. Mater. 2017, 29.
- Mao, S.; Lu, G.; Chen, J. 3 dimensional graphene based composites for energy applications. Nanoscale 2015, 7, 6924–6943.
- Pan, D.; Feng, C.; Wang, L.; Liu, Y.; Chen, Z.; Shi, W.; Li, Z. Binder-Free Graphene Organo-gels as Cost-Efficient Counter Electrodes for Dye-sensitized Solar Cells. Electrochim. Acta 2016, 191, 946–953.
- Chang, Q.; Huang, L.; Wang, J.; Ma, Z.; Li, P.; Yan, Y.; Zhu, J.; Xu, S.; Shen, L.; Chen, Q.; et al. Nanoarchitecture of variable sized graphene nanosheets incorporated into three-dimensional graphene network for dye sensitized solar cells. Carbon 2015, 85, 185–193.
- Wang, G.; Hou, S.; Yan, C.; Zhang, W. A 3D architecture composite of porous vanadium nitride nanoribbons and reduced graphene oxide as a high-efficiency counter electrode for dye-sensitized solar cells. RSC Adv. 2018, 8, 1083–1088.
- Ma, J.; Shen, W.; Yu, F. Graphene-enhanced three-dimensional structures of MoS2 nanosheets as a counter electrode for Pt-free efficient dye-sensitized solar cells. J. Power Sources 2017, 351, 58–66.
- Stoller, M.D.; Park, S.; Zhu, Y.; An, J.; Ruoff, R.S. Graphene based ultra-capacitors. Nano Lett. 2008, 8, 3498–3502.
- Zhang, L.L.; Zhou, R.; Zhao, X. Graphene based materials as super capacitor electrodes. J. Mater. Chem. 2010, 20, 5983–5992.
- Huang, X.; Qi, X.; Boey, F.; Zhang, H. Graphene based composites. Chem. Soc. Rev. 2012, 41, 666–686.
- Luo, J.; Liu, J.; Zeng, Z.; Ng, C.F.; Ma, L.; Zhang, H. Three dimensional graphene foam supported Fe3O4 lithium battery anodes with long cycle life and high rate capability. Nano Lett. 2013, 13, 6136–6143.
- Luo, H.; Ji, D.; Yang, Z.; Huang, Y.; Xiong, G.; Zhu, Y.; Guo, R.; Wan, Y. An ultralight and highly compressible anode for Li-ion batteries constructed from nitrogen-doped carbon enwrapped Fe3O4 nanoparticles confined in a porous 3D nitrogen-doped graphene networ. Chem. Eng. J. 2017, 326, 151–161.
- Yu, S.X.; Yang, L.W.; Tian, Y.; Yang, P.; Jiang, F.; Hu, S.W.; Wei, X.L.; Zhong, J.X. Mesoporous anatase TiO2submicrospheres embedded in self-assembled three-dimensional reduced graphene oxide networks for enhanced lithium storage. J. Mater. Chem. A 2013, 1, 12750–12758.
- Jiang, X.; Yang, X.; Zhu, Y.; Jiang, H.; Yao, Y.; Zhao, P.; Li, C. 3D nitrogen-doped graphene foams embedded with ultrafine TiO2 nanoparticles for high-performance lithium-ion batteries. J. Mater. Chem. A 2014, 2, 11124–11133.
- Sui, Z.-Y.; Wang, C.; Shu, K.; Yang, Q.-S.; Ge, Y.; Wallace, G.G.; Han, B.-H. Manganese dioxide-anchored three-dimensional nitrogen-doped graphene hybrid aerogels as excellent anode materials for lithium ion batteries. J. Mater. Chem. A 2015, 3, 10403–10412.
- Liang, J.; Liu, Y.; Guo, L.; Li, L. Facile one-step synthesis of a 3D macroscopic SnO2–graphene aerogel and its application as a superior anode material for Li-ion batteries. RSC Adv. 2013, 3, 11489–11492.
- Wang, F.; Liu, L.; Li, W.J. Graphene based glucose sensors: A brief review. IEEE Trans. Nano Biosci. 2015, 14, 818–834.
- Hill, E.; Vijayaraghavan, A.; Novoselov, K. Graphene Sensors. IEEE Sens. J. 2011, 11, 3161–3170.
- Krishnan, S.K.; Singh, E.; Singh, P.; Meyyappan, M.; Nalwa, H.S. A review on graphene-based nanocomposites for electrochemical and fluorescent biosensors. RSC Adv. 2019, 9, 8778–8881.
- Bogue, R. Graphene sensors: A review of recent developments. Sens. Rev. 2014, 34, 233–238.
- Singh, E.; Nalwa, H.S. (Eds.) Nanomaterial-Based Flexible and Multifunction Sensors; American Scientific Publisher: Los Angeles, CA, USA, 2019.
- Yong, Z.; Zhou, X.; Li, X.; Zhang, Y. Review on the graphene based optical fiber chemical and biological sensors. Sens. Actuators B 2016, 231, 324–340.
- Shao, Y.; Wang, J.; Wu, H.; Liu, J.; Aksay, I.A.; Lin, Y. Graphene Based Electrochemical Sensors and Biosensors: A Review. Electroanalysis 2010, 22, 1027–1036.
- Zhao, J.; Zhang, G.; Shi, D. Review of graphene-based strain sensors. Chin. Phy. B 2013, 22, 057701.
- Li, X.; Cai, W.; An, J.; Kim, S.; Nah, J.; Yang, D.; Piner, R.; Velamakanni, A.; Jung, I.; Tutuc, E.; et al. Large-Area Synthesis of High-Quality and Uniform Graphene Films on Copper Foils. Science 2009, 324, 1312–1314.
- Wei, D.; Liu, Y.; Wang, Y.; Zhang, H.; Huang, L.; Yu, G. Synthesis of N-Doped Graphene by Chemical Vapor Deposition and Its Electrical Properties. Nano Lett. 2009, 9, 1752–1758.
- Kordatos, A.; Kelaidis, N.; Giamini, S.A.; Marquez-Velasco, J.; Xenogiannopoulou, E.; Tsipas, P.; Kordas, G.; Dimoulas, A. AB stacked few layer graphene growth by chemical vapor deposition on single crystal Rh(1 1 1) and electronic structure characterization. Appl. Surf. Sci. 2016, 369, 251–256.
- Kojima, T.; Bao, Y.; Zhang, C.; Liu, S.; Xu, H.; Nakae, T.; Loh, K.P.; Sakaguchi, H. Orientation and Electronic Structures of Multilayered Graphene Nanoribbons Produced by Two-Zone Chemical Vapor Deposition. Langmuir 2017, 33, 10439–10445.
- Park, B.-J.; Choi, J.-S.; Eom, J.-H.; Ha, H.; Kim, H.Y.; Lee, S.-H.; Shin, H.; Yoon, S.-G. Defect-Free Graphene Synthesized Directly at 150 °C via Chemical Vapor Deposition with No Transfer. ACS Nano 2018, 12, 2008–2016.
- Gao, L.; Ren, W.; Xu, H.; Jin, L.; Wang, Z.; Ma, T.; Ma, L.-P.; Zhang, Z.; Fu, Q.; Peng, L.-M.; et al. Repeated growth and bubbling transfer of graphene with millimetre-size single-crystal grains using platinum. Nat. Commun. 2012, 3, 699.
- Kwon, S.Y.; Ciobanu, C.V.; Petrova, V.; Shenoy, V.B.; Bareño, J.; Gambin, V.; Petrov, I.; Kodambaka, S. Growth of semiconducting graphene on palladium. Nano Lett. 2009, 9, 3985–3990.
- Amato, G. High Temperature Growth of Graphene from Cobalt Volume: Effect on Structural Properties. Materials 2018, 11, 257.
- Kim, H.H.; Kang, B.; Suk, J.W.; Li, N.; Kim, K.S.; Ruoff, R.S.; Lee, W.H.; Cho, K. Clean transfer of wafer-Scale graphene via liquid phase removal of polycyclic aromatic hydrocarbons. ACS Nano 2015, 9, 4726–4733.
- Kim, K.S.; Zhao, Y.; Jang, H.; Lee, S.Y.; Kim, J.M.; Kim, K.S.; Ahn, J.-H.; Kim, P.; Choi, J.-Y.; Hong, B.H. Large-scale pattern growth of graphene films for stretchable transparent electrodes. Nature 2009, 457, 706–710.
- Li, Z.; Wang, Y.; Kozbial, A.; Shenoy, G.J.; Zhou, F.; McGinley, R.; Ireland, P.; Morganstein, B.; Kunkel, A.; Surwade, S.P.; et al. Effect of airborne contaminants on the wettability of supported graphene and graphite. Nat. Mater. 2013, 12, 925–931.
- Lin, Y.-C.; Lu, C.-C.; Yeh, C.-H.; Jin, C.; Suenaga, K.; Chiu, P.-W. Graphene Annealing: How Clean Can It Be? Nano Lett. 2012, 12, 414–419.
- Zhang, Z.; Du, J.; Zhang, D.; Sun, H.; Yin, L.; Ma, L.; Chen, J.; Ma, D.; Cheng, H.-M.; Ren, W. Rosin-enabled ultraclean and damage-free transfer of graphene for large-area flexible organic light-emitting diodes. Nat. Commun. 2017, 8, 14560.
- Cui, G.; Bi, Z.; Zhang, R.; Liu, J.; Yu, X.; Li, Z. A comprehensive review on graphene-based anti-corrosive coatings. Chem. Eng. J. 2019, 373, 104–121.
- Nine, J.; Cole, M.A.; Tran, D.N.H.; Losic, D. Graphene: A multipurpose material for protective coatings. J. Mater. Chem. A 2015, 3, 12580–12602.
- Shen, H.; Zhang, L.; Liu, M.; Zhang, Z. Biomedical Applications of Graphene. Theranostics 2012, 2, 283–294.
- Sun, X.; Liu, Z.; Welsher, K.; Robinson, J.T.; Goodwin, A.; Zaric, S.; Dai, H. Nano-graphene oxide for cellular imaging and drug delivery. Nano Res. 2008, 1, 203–212.
- Su, B.; Tang, J.; Huang, J.; Yang, H.; Qiu, B.; Chen, G.; Tang, D. Graphene and Nanogold-Functionalized Immunosensing Interface with Enhanced Sensitivity for One-Step Electrochemical Immunoassay of Alpha-Fetoprotein in Human Serum. Electroanalysis 2010, 22, 2720–2728.
- Nanda, S.S.; Yi, D.K.; Kim, K. Study of antibacterial mechanism of graphene oxide using Raman spectroscopy. Sci. Rep. 2016, 6, 28443.
- Liu, Z.; Robinson, J.T.; Sun, X.M.; Dai, H. PEGylated nanographene oxide for delivery of water-insoluble cancer drugs. J. Am. Chem. Soc. 2008, 130, 10876–10877.
- Wang, Y.; Li, Z.; Wang, J.; Li, J.; Lin, Y. Graphene and graphene oxide: Bio-functionalization and applications in biotechnology. Trends Biotechnol. 2011, 29, 205–212.
- Shin, S.R.; Li, Y.-C.; Jang, H.L.; Khoshakhlagh, P.; Akbari, M.; Nasajpour, A.; Zhang, Y.S.; Tamayol, A.; Khademhosseini, A. Graphene-based materials for tissue engineering. Adv. Drug Deliv. Rev. 2016, 105, 255–274.
- Sint, K.; Wang, B.; Kral, P. Selective ion passage through functionalized graphene nanopores. J. Am. Chem. Soc. 2008, 130, 16448–16449.
- Cohen-Tanugi, D.; Groassman, J.C. Mechanical strength of nanoporous graphene as a desalination membrane. Nano Lett. 2014, 14, 6171–6178.
- Wu, Y.; Chen, M.; Chen, M.; Ran, Z.; Zhu, C.; Liao, H. The reinforcing effect of polydopaminem functionalized graphene nanoplatelets on the mechanical properties of epoxy resins at cryogenic temperature. Polym. Test. 2017, 58, 262–269.
- Chen, Z.-K.; Yang, J.-P.; Ni, Q.-Q.; Fu, S.-Y.; Huang, Y.-G. Reinforcement of epoxy resins with multi-walled carbon nanotubes for enhancing cryogenic mechanical properties. Polymer 2009, 50, 4753–4759.
- He, Y.; Chen, G.; Zhang, L.; Sang, Y.; Lu, C.; Yao, D. Role of functionalized multi-walled carbon nanotubes on mechanical properties of epoxy-based composites at cryogenic temperature, High Perform. Polymer 2014, 26, 922–934.
- Yang, J.-P.; Yang, G.; Xu, G.; Fu, S.-Y. Cryogenic mechanical behaviors of MMT/epoxy nanocomposites. Compos. Sci. Technol. 2007, 67, 2934–2940.
- Lau, K.-T.; Wong, T.T.; Leng, J.; Hui, D.; Rhee, K. Property enhancement of polymer-based composites at cryogenic environment by using tailored carbon nanotubes. Compos. Part B Eng. 2013, 54, 41–43.
- Sun, X.; Liu, X.; Shen, X.; Wu, Y.; Wang, Z.; Kim, J.-K. Graphene foam/carbon nanotube/poly(dimethyl siloxane) composites for exceptional microwave shielding. Compos. Part A Appl. Sci. Manuf. 2016, 85, 199–206.
- Huang, C.; Fu, S.; Zhang, Y.; Lauke, B.; Li, L.; Ye, L. Cryogenic properties of SiO2/epoxy nanocomposites. Cryogenics 2005, 45, 450–454.
- Li, Z.; Wang, R.; Young, R.; Deng, L.; Yang, F.; Hao, L.; Jiao, W.; Liu, W. Control of the functionality of graphene oxide for its application in epoxy nanocomposites. Polymer 2013, 54, 6437–6446.
- George, J.J.; Bandyopadhyay, A.; Bhowmick, A.K. New generation layered nanocomposites derived from ethylene-co-vinyl acetate and naturally occurring graphite. J. Appl. Polym. Sci. 2008, 108, 1603–1616.
- Wang, C.; Li, J.; Sun, S.; Li, X.; Zhao, F.; Jiang, B.; Huang, Y. Electrophoretic deposition of graphene oxide on continuous carbon fibers for reinforcement of both tensile and interfacial strength. Compos. Sci. Technol. 2016, 135, 46–53.
- Hung, P.-Y.; Lau, K.-T.; Fox, B.; Hameed, N.; Lee, J.H.; Hui, D. Surface modification of carbon fibre using graphene—Related materials for multifunctional composites. Compos. Part B Eng. 2018, 133, 240–257.
- Wu, Y.; Huang, L.; Huang, X.; Guo, X.; Liu, D.; Zheng, D. A room temperature liquid metal based self healing anode foe lithium ion batteries with an ultra long cycle life. Energy Environ. Sci. 2017, 10, 1854–1861.
- Grubišić-Čabo, A.; Kotsakidis, J.C.; Yin, Y.; Tadich, A.; Haldon, M.; Solari, S.; Di Bernardo, I.; Daniels, K.M.; Riley, J.; Huwald, E.; et al. Magnesium-intercalated graphene on SiC: Highly n-doped air-stable bilayer graphene at extreme displacement fields. Appl. Surf. Sci. 2021, 541, 148612.
- Forti, S.; Link, S.; Stöhr, A.; Niu, Y.; Zakharov, A.A.; Coletti, C.; Starke, U. Semiconductor to metal transition in two-dimensional gold and its van der Waals heterostack with graphene. Nat. Commun. 2020, 11, 2236.
- Briggs, N.; Gebeyehu, Z.M.; Vera, A.; Zhao, T.; Wang, K.; Duran, A.D.L.F.; Bersch, B.; Bowen, T.; Knappenberger, K.L.; Robinson, J.A. Epitaxial graphene/silicon carbide intercalation: A minireview on graphene modulation and unique 2D materials. Nanoscale 2019, 11, 15440–15447.
- Perreault, F.; Faria, A.F.; Elimelech, M. Environmental applications of graphene-based nanomaterials. Chem. Soc. Rev. 2015, 44, 5861–5896.
- Cho, E.-C.; Huang, J.-H.; Li, C.-P.; Chang-Jian, C.-W.; Lee, K.-C.; Hsiao, Y.-S.; Huang, J.-H. Graphene-based thermoplastic composites and their application for LED thermal management. Carbon 2016, 102, 66–73.
- Jiang, G.; Tian, H.; Wang, X.; Hirtz, T.; Wu, F.; Qiao, Y.; Gou, G.; Wei, Y.; Yang, J.; Yang, S.; et al. An efficient flexible graphene based light emmitingdevic. Nanoscale Adv. 2019, 1, 4745–4754.
- Singh, E.; Meyyappan, M.; Nalwa, H.S. Flexible Graphene-Based Wearable Gas and Chemical Sensors. ACS Appl. Mater. Interfaces 2017, 9, 34544–34586.
- Liou, Y.-R.; Lin, H.Y.; Shen, T.L.; Cai, S.Y.; Wu, Y.H.; Liao, Y.M.; Lin, H.I.; Chen, T.P.; Lin, T.Y.; Chen, Y.F. Integration of Nanoscale and Macroscale Graphene Heterostructures for Flexible and Multilevel Nonvolatile Photoelectronic Memory. ACS Appl. Nano Mater. 2019, 3, 608–616.
- Sutar, H.; Mishra, B.; Senapati, P.; Murmu, R.; Sahu, D. Mechanical, Thermal, and Morphological Properties of Graphene Nanoplatelet-Reinforced Polypropylene Nanocomposites: Effects of Nanofiller Thickness. J. Compos. Sci. 2021, 5, 24.
- Mahmood, H.; Unterberger, S.H.; Pegoretti, A. Tuning Electrical and Thermal Properties in Epoxy/Glass Composites by Graphene-Based Interphase. J. Compos. Sci. 2017, 1, 12.
- Azizi, S.; Azizi, M.; Sabetzadeh, M. The Role of Multiwalled Carbon Nanotubes in the Mechanical, Thermal, Rheological, and Electrical Properties of PP/PLA/MWCNTs Nanocomposites. J. Compos. Sci. 2019, 3, 64.
- Sultana, T.; Sultana, S.; Nur, H.P.; Khan, W. Studies on Mechanical, Thermal and Morphological Properties of Betel Nut Husk Nano Cellulose Reinforced Biodegradable Polymer Composites. J. Compos. Sci. 2020, 4, 83.
- Martin, I.; Del Castillo, D.S.; Fernandez, A.; Güemes, A. Advanced Thermoplastic Composite Manufacturing by In-Situ Consolidation: A Review. J. Compos. Sci. 2020, 4, 149.
- Zhang, H.; Zhang, X.; Fang, Z.; Huang, Y.; Xu, H.; Liu, Y.; Wu, D.; Zhuang, J.; Sun, J. Recent Advances in Preparation, Mechanisms, and Applications of Thermally Conductive Polymer Composites: A Review. J. Compos. Sci. 2020, 4, 180.
- Rout, D.R.; Senapati, P.; Sutar, H.; Sau, D.C.; Murmu, R. Graphene Oxide (GO) Supported Palladium (Pd) Nanocomposites for Enhanced Hydrogenation. Graphene 2019, 8, 33–51.
- Singh, M.; Kumar, S.; Zoghi, S.; Cervantes, Y.; Sarkar, D.; Ahmed, S.; Chowdhury, S.; Banerjee, S. Fabrication and Characterization of Flexible Three-Phase ZnO-Graphene-Epoxy Electro-Active Thin-Film Nanocomposites: Towards Applications in Wearable Biomedical Devices. J. Compos. Sci. 2020, 4, 88.
- Repetsky, S.; Vyshyvana, I.; Kruchinin, S.; Vlahovic, B.; Bellucci, S. Effect of impurities ordering in the electronic spectrum and conductivity of graphene. Phys. Lett. A 2020, 384, 126401.
- Grushevskaya, H.V.; Krylov, G.G.; Kruchinin, S.P.; Vlahovic, B.; Bellucci, S. Electronic properties and quasi-zero-energy states of graphene quantum dots. Phys. Rev. B 2021, 103, 235102.
- Kim, D.; Coskun, A. Graphene oxide-templated preferential growth of continuous MOF thin films. CrystEngComm 2016, 18, 4013–4017.
- Khan, Z.; Senthilkumar, B.; Park, S.; Park, S.; Yang, J.; Lee, J.H.; Song, H.-K.; Kim, Y.; Kwak, S.K.; Ko, H. Carambola-shaped VO2 nanostructures: A binder-free air electrode for an aqueous Na–air battery. J. Mater. Chem. A 2016, 5, 2037–2044.
- Khan, Z.; Park, S.; Hwang, S.M.; Yang, J.; Lee, Y.; Song, H.-K.; Kim, Y.; Ko, H. Hierarchical urchin-shaped α-MnO2 on graphene-coated carbon microfibers: A binder-free electrode for rechargeable aqueous Na–air battery. NPG Asia Mater. 2016, 8, e294.
More
Information
Subjects:
Materials Science, Composites
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
4.2K
Revisions:
3 times
(View History)
Update Date:
09 Oct 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





