Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Kin Wah Terence Lee | + 1146 word(s) | 1146 | 2021-07-16 09:44:41 |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Lee, K.W.T. Cancer Stem Cell-Targeted Therapies. Encyclopedia. Available online: https://encyclopedia.pub/entry/12145 (accessed on 31 March 2026).
Lee KWT. Cancer Stem Cell-Targeted Therapies. Encyclopedia. Available at: https://encyclopedia.pub/entry/12145. Accessed March 31, 2026.
Lee, Kin Wah Terence. "Cancer Stem Cell-Targeted Therapies" Encyclopedia, https://encyclopedia.pub/entry/12145 (accessed March 31, 2026).
Lee, K.W.T. (2021, July 19). Cancer Stem Cell-Targeted Therapies. In Encyclopedia. https://encyclopedia.pub/entry/12145
Lee, Kin Wah Terence. "Cancer Stem Cell-Targeted Therapies." Encyclopedia. Web. 19 July, 2021.
Copy Citation
Cancer stem cell-targeted therapies means therapies targeted at cancer stem cells.
cancer
cancer stem cells
immune cells
stromal cells
tumor microenvironment
1. Introduction
Targeting stromal cells has become one of the strategies to destem CSCs. Many preclinical and clinical trials are being conducted to evaluate the therapeutic efficacies of various small molecule inhibitors/neutralizing antibodies in disrupting the interactions between stromal cells and cancer cells. The current direction in this field is to dissect how stromal cells recruit and regulate various immune suppressive cells to create an immunosuppressive TME. Single-cell RNA sequencing analysis provides a dynamic stromal niche that supports cancer stemness and immune evasion in various cancer types. Strikingly, this technique will provide mechanistic insight and a novel strategy for current immune checkpoint therapy. Traditionally, CSCs were regarded as subpopulations within the tumor that promote tumor recurrence and therapeutic resistance. Accumulating evidence has demonstrated the distinct role of CSCs in immune evasion. Immense effort has currently been made to dissect the crosstalk between CSCs and various immune suppressive cells, including MDSCs and Tregs. In addition, increasing interest is directed towards understanding the direct interaction between CSCs and macrophages and CD8+ T cells. Based on these interesting findings, CSC-targeted immunotherapy may be a promising approach for cancer therapy, and its therapeutic efficacy needs further investigation.
2. Interplay of Various Cellular Factors within the TME in the Regulation of Cancer Stemness and Immune Evasion
In the previous sections, we discussed how individual stromal cells and immune cells interact with CSCs within the TME. However, stromal cells do not regulate the plasticity of CSCs in isolation and have a highly context-dependent mechanism of action. As one of the major stromal cells in the TME, CAFs interact with various stromal cells and immune cells in the TME to regulate CSC plasticity and immune evasion. First, a number of reports have demonstrated the crosstalk between CAFs and macrophages in the promotion of cancer stemness. CAFs play a crucial role in the promotion of cancer stemness in HCC by reciprocally inducing the activity of TAMs [1]. Overexpression of these markers, including α-SMA and CD68+, is associated with HCC recurrence and shorter overall survival [1]. Recently, Yang et al. showed that CAFs express endosialin, which regulates macrophage recruitment and polarization to support HCC progression [2]. In prostate cancer (PCa), Comito et al. identified crosstalk among different cellular components, including CAFs, TAMs and PCa cells, leading to the promotion of cancer stemness [3].
Apart from TAMs, CAFs also interact with endothelial cells in the TME. CAFs regulate the endothelial lipoma-preferred partner (LPP) gene in endothelial cells, rendering a chemoresistant phenotype in ovarian cancer [4]. Moreover, Song et al. reported novel cytokine-mediated crosstalk among CAFs, HCC cells and TANs, augmenting cancer stemness and TAN recruitment in HCC [5]. Specifically, CAF-derived CLCF1 recruited and promoted N2 polarization of TANs via induced secretion of CXCL6 and TGF-β in HCC cells [5]. Accumulating evidence has also demonstrated the interaction between CAFs and immune cells in promoting an immunosuppressive environment. CAFs promoted recruitment of CCR2+ monocytes and conversion to the MDSC phenotype via preferential secretion of CCL2, which created an immunosuppressive environment [6]. Targeting the CAF-MDSC axis by CCR2 inhibition may open a promising therapeutic avenue for converting from a non-T-cell-inflamed TME to a T-cell-inflamed counterpart in lung carcinoma [6]. In addition to MDSCs, the CAF-neutrophil axis was reported to be a promising approach for the development of stromal treatments in pancreatic cancer. CAF-derived CXCL12 recruited neutrophil infiltration, which led to resistance to T-cell mediated killing [7]. In addition, CAFs recruit and enrich the Treg population via IL6 secretion in esophageal cancer [8]. Last, CD73+ CAFs suppressed T cell activity in a colon cancer model via A2A-mediated immune suppression, and thus targeting the CD73-adenosine pathway is a promising approach for complementing PD1 therapy [9].
3. Conclusions
The dynamic interactions between stromal cells and CSCs are not simply unidirectional but reciprocal, promoting the expansion of stem cell markers, migration, invasion, drug resistance and self-renewal properties of CSCs (Figure 1).
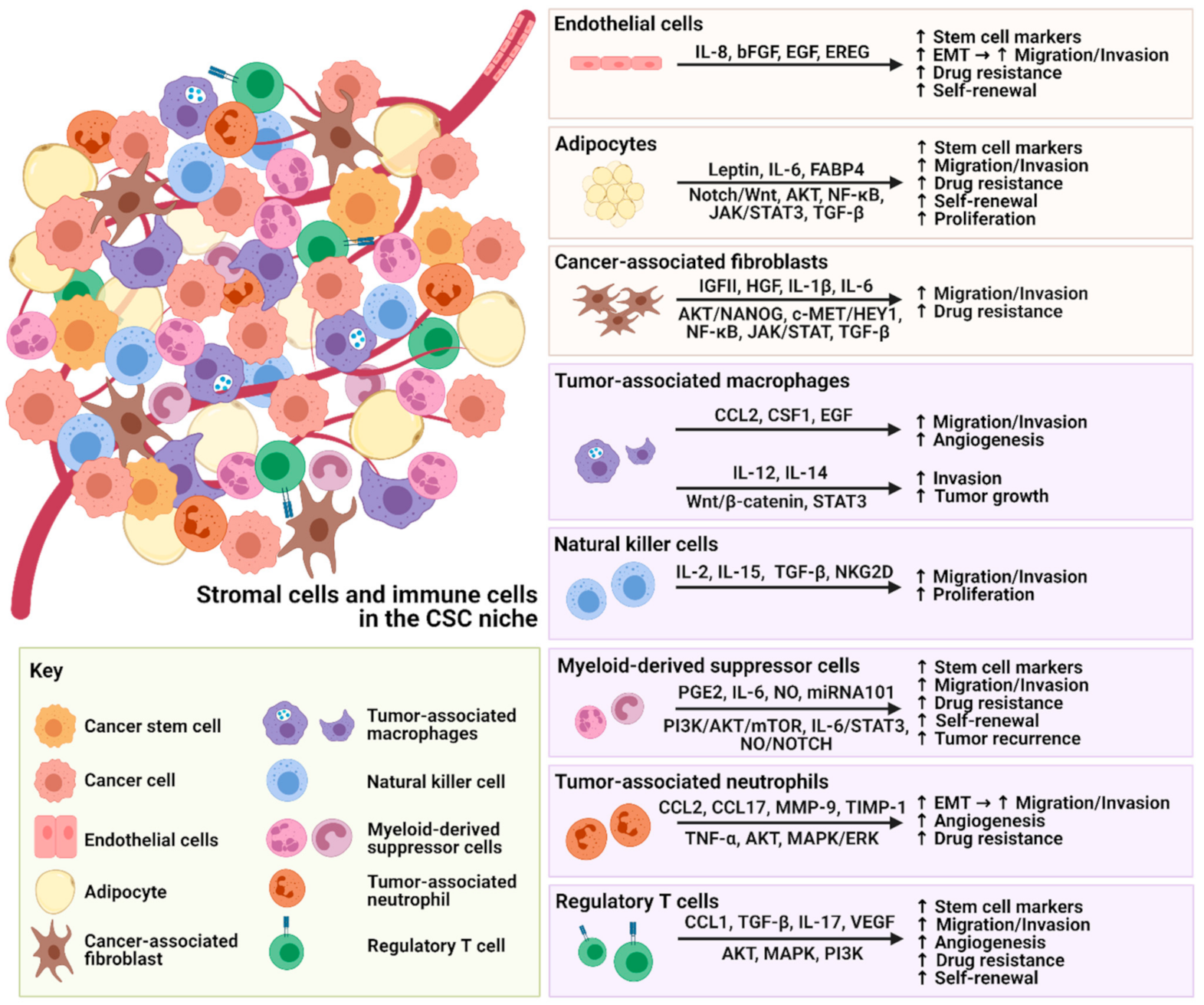 Figure 1. Illustration of the crosstalk between stromal cells, immune cells and CSCs.
Figure 1. Illustration of the crosstalk between stromal cells, immune cells and CSCs.Similar tumor-promoting crosstalk is also observed with immune cells, leading to immune evasion of CSCs and tumor recurrence. An increasing number of studies have focused on targeting the major secretomes or molecules related to stromal cells and immune cells in the TME, which are summarized in Table 1. These therapeutic inhibitors or neutralizing antibodies could potentially be used as single treatments or in combination with current therapies for better treatment outcomes. However, it is not yet clear whether these promising preclinical strategies that enhance our understanding of cancer development will translate into effective treatment for cancers. More knowledge needs to be gained, and convincing preclinical results need to be evaluated in clinical trials—there is still a long way to go. Although these targets have been reported, their effects on cancer phenotypes may be tumor-type specific. Some solid tumors and liquid tumors may have different responses to stromal cells and immunotherapy. Therefore, hampering stromal cells as a therapeutic strategy to destem CSCs needs further investigation.
Table 1. Therapeutic strategies targeting the interactions between CSCs and stromal cells for cancer treatments.
| Type of Targeted Cell | Cancer Type | Novel Therapeutic Strategies | Mode of Action | Effects on Cancer Stemness | References |
|---|---|---|---|---|---|
| Targeted Stromal Cells | - | ||||
| Endothelial cells | Glioblastoma | IL-8 neutralizing antibody | Blockade of endothelial cell-secreted IL-8 | Reduced spheroid size and tumor growth in vivo | [10] |
| Adipocytes | Breast/Mammary cancer | BMS309403 | Inhibiting FABP4 functions | Suppressed tumor growth and tumor volume with decreased IL-6 level and tumor ALDH1 activity | [11] |
| Ovarian cancer | Anti-OB-R blocking peptide | Blockade of leptin receptor | Decreased leptin-induced cell migration and invasion abilities | [12] | |
| CAFs | Breast Cancer | GW4064 | Agonist of FXR | Reduced progression and motility of tumors | [13][14] |
| Pirfenidone (PFD) and doxorubicin | Inhibiting collagen production and tumor growth | Reduced components of ECM, inhibited tumor growth and lung metastasis | [15] | ||
| Targeted Immune Cells | - | ||||
| TAMs | Pancreatic carcinoma | CD40 Agonists | Activating and inducing macrophages | Degraded ECM and improved tumor infiltration of immune cells | [16] |
| Non-small cell lung cancer (NSCLC) | IL-33 neutralizing antibody | Blockade of IL-33 | Inhibited M2-like macrophages polarization via inhibition of IL-10 and VEGF as well as reduced accumulation of Treg cells | [17] | |
| NK cells | Colon cancer | Chondrocytes | Expressing a high level of IL-12 | Increased the infiltrations of both T cells and NK cells, reduced cancer cells and tumor angiogenesis | [18] |
| MDSCs | Breast cancer | Combination of anti-IL-6 antibody and iNOS inhibitor | Targeting MDSC-derived IL-6 and nitric oxide | Reduced spheroid formation stimulated by MDSCs | [19] |
| Endometrial cancer | Doxorubicin and Gr-1 neutralizing antibody or celecoxib | Blockade of MDSCs or inhibiting MDSC functions | Reduced ALHD+ expression and sensitized tumor cells to chemotherapy | [20] | |
| TANs | Breast cancer | Zileuton | Inhibiting neutrophil Alox5 | Reduced lung metastasis | [21] |
| Treg cells | Glioma | Tocilizumab | Blockade of IL-6 receptor | Inhibited tumor growth and CD133 expression induced by Treg cells | [22] |
| Acute myeloid leukemia (AML) | IL-10R neutralizing antibody | Neutralizing IL-10 receptor functions | Reduced side population, sphere formation ability, and expression of OCT4 and NANOG | [23] | |
| Ovarian cancer | CD25 neutralizing antibody | Inhibiting CD25+ Treg cells | Reduced angiogenesis and inhibited tumor growth | [24] | |
References
- Ji, J.; Eggert, T.; Budhu, A.; Forgues, M.; Ji, J.; Eggert, T.; Budhu, A.; Forgues, M.; Takai, A.; Dang, H.; et al. Hepatic stellate cell and monocyte interaction contributes to poor prognosis in hepatocellular carcinoma. Hepatology 2015, 62, 481–495.
- Yang, F.; Wei, Y.; Han, D.; Li, Y.; Shi, S.; Jiao, D.; Wu, J.; Zhang, Q.; Shi, C.; Yang, L.; et al. Interaction with CD68 and regulation of GAS6 expression by endosialin in fibroblasts drives recruitment and polarization of marophages in hepatocellular carcinoma. Cancer Res. 2020, 80, 3892–3905.
- Comito, G.; Giannoni, E.; Segura, C.P.; Barcellos-de-Souza, P.; Raspollini, M.R.; Baroni, G.; Lanciotti, M.; Serni, S.; Chiarugi, P. Cancer-associated fibroblasts and M2-polarized macrophages synergize during prostate carcinoma progression. Oncogene 2014, 33, 2423–2431.
- Leung, C.S.; Yeung, T.L.; Yip, K.P.; Wong, K.K.; Ho, S.Y.; Mangala, L.S.; Sood, A.K.; Lopez-Berestein, G.; Sheng, J.; Wong, S.T.; et al. Cancer-associated fibroblasts regulate endothelial adhesion protein LPP to promote ovarian cancer chemoresistance. J. Clin. Investig. 2018, 128, 589–606.
- Song, M.; He, J.; Pan, Q.Z.; Yang, J.; Zhao, J.; Zhang, Y.J.; Huang, Y.; Tang, Y.; Wang, Q.; He, J.; et al. Cancer-associated fibroblast-mediated cellular crosstalk supports hepatocellular carcinoma progression. Hepatology 2021, 73, 1717–1735.
- Xiang, H.; Ramil, C.P.; Hai, J.; Zhang, C.; Wang, H.; Watkins, A.A.; Afshar, R.; Georgiev, P.; Sze, M.A.; Song, X.S.; et al. Cancer-associated fibroblasts promote immunosuppression by inducing ROS-generating monocytic MDSCs in lung squamous cell carcinoma. Cancer Immunol. Res. 2020, 8, 436–450.
- Feig, C.; Jones, J.O.; Kraman, M.; Wells, R.J.; Deonarine, A.; Chan, D.S.; Connell, C.M.; Roberts, E.W.; Zhao, Q.; Caballero, O.L.; et al. Targeting CXCL12 from FAP-expressing carcinoma-associated fibroblasts synergizes with anti-PD-L1 immunotherapy in pancreatic cancer. Proc. Natl. Acad. Sci. USA 2013, 110, 20212–20217.
- Kato, T.; Noma, K.; Ohara, T.; Kashima, H.; Katsura, Y.; Sato, H.; Komoto, S.; Katsube, R.; Ninomiya, T.; Tazawa, H.; et al. Cancer-associated fibroblasts affect intratumoral CD8+ and FoxP3+ T cells via IL6 in the tumor microenvironment. Clin. Cancer Res. 2018, 24, 4820–4833.
- Yu, M.; Guo, G.; Huang, L.; Deng, L.; Chang, C.S.; Achyut, B.R.; Canning, M.; Xu, N.; Arbab, A.S.; Bollag, R.J.; et al. CD73 on cancer-associated fibroblasts enhanced by the A2B-meidated feeforward circuit enforces an immune checkpoint. Nat. Commun. 2020, 11, 515.
- McCoy, M.G.; Nyanyo, D.; Hung, C.K.; Goerger, J.P.; Zipfel, W.R.; Williams, R.M.; Nishimura, N.; Fischbach, C. Endothelial cells promote 3D invasion of GBM by IL-8-dependent induction of cancer stem cell properties. Sci. Rep. 2019, 9, 9069.
- Hao, J.; Zhang, Y.; Yan, X.; Yan, F.; Sun, Y.; Zeng, J.; Waigel, S.; Yin, Y.; Fraig, M.M.; Egilmez, N.K. Circulating adipose fatty acid binding protein is a new link underlying obesity-associated breast/mammary tumor development. Cell Metab. 2018, 28, 689–705.e5.
- Kato, S.; Abarzua-Catalan, L.; Trigo, C.; Delpiano, A.; Sanhueza, C.; García, K.; Ibañez, C.; Hormazábal, K.; Diaz, D.; Brañes, J. Leptin stimulates migration and invasion and maintains cancer stem-like properties in ovarian cancer cells: An explanation for poor outcomes in obese women. Oncotarget 2015, 6, 21100.
- Takai, K.; Le, A.; Weaver, V.M.; Werb, Z. Targeting the cancer-associated fibroblasts as a treatment in triple-negative breast cancer. Oncotarget 2016, 7, 82889–82901.
- Giordano, C.; Barone, I.; Vircillo, V.; Panza, S.; Malivindi, R.; Gelsomino, L.; Pellegrino, M.; Rago, V.; Mauro, L.; Lanzino, M.; et al. Activated FXR Inhibits Leptin Signaling and Counteracts Tumor-promoting Activities of Cancer-Associated Fibroblasts in Breast Malignancy. Sci. Rep. 2016, 6, 21782.
- Polydorou, C.; Mpekris, F.; Papageorgis, P.; Voutouri, C.; Stylianopoulos, T. Pirfenidone normalizes the tumor microenvironment to improve chemotherapy. Oncotarget 2017, 8, 24506–24517.
- Long, K.B.; Gladney, W.L.; Tooker, G.M.; Graham, K.; Fraietta, J.A.; Beatty, G.L. IFNγ and CCL2 Cooperate to Redirect Tumor-Infiltrating Monocytes to Degrade Fibrosis and Enhance Chemotherapy Efficacy in Pancreatic Carcinoma. Cancer Discov. 2016, 6, 400–413.
- Wang, K.; Shan, S.; Yang, Z.; Gu, X.; Wang, Y.; Wang, C.; Ren, T. IL-33 blockade suppresses tumor growth of human lung cancer through direct and indirect pathways in a preclinical model. Oncotarget 2017, 8, 68571–68582.
- Xie, G.; Dong, H.; Liang, Y.; Ham, J.D.; Rizwan, R.; Chen, J. CAR-NK cells: A promising cellular immunotherapy for cancer. EBioMedicine 2020, 59.
- Peng, D.; Tanikawa, T.; Li, W.; Zhao, L.; Vatan, L.; Szeliga, W.; Wan, S.; Wei, S.; Wang, Y.; Liu, Y.; et al. Myeloid-Derived Suppressor Cells Endow Stem-like Qualities to Breast Cancer Cells through IL6/STAT3 and NO/NOTCH Cross-talk Signaling. Cancer Res. 2016, 76, 3156–3165.
- Yokoi, E.; Mabuchi, S.; Komura, N.; Shimura, K.; Kuroda, H.; Kozasa, K.; Takahashi, R.; Sasano, T.; Kawano, M.; Matsumoto, Y.; et al. The role of myeloid-derived suppressor cells in endometrial cancer displaying systemic inflammatory response: Clinical and preclinical investigations. Oncoimmunology 2019, 8, e1662708.
- Wculek, S.K.; Malanchi, I. Neutrophils support lung colonization of metastasis-initiating breast cancer cells. Nature 2015, 528, 413–417.
- Liu, S.; Zhang, C.; Wang, B.; Zhang, H.; Qin, G.; Li, C.; Cao, L.; Gao, Q.; Ping, Y.; Zhang, K.; et al. Regulatory T cells promote glioma cell stemness through TGF-beta-NF-kappaB-IL6-STAT3 signaling. Cancer Immunol. Immunother. 2021.
- Xu, Y.; Wan, Y.; Liu, J.; Wang, Y.; Li, S.; Xing, H.; Tang, K.; Tian, Z.; Rao, Q.; Wang, M. Regulatory T cells promote the stemness of acute myeloid leukemia cells through IL10 cytokine related signaling pathway. Blood 2017, 130, 2727.
- Facciabene, A.; Peng, X.; Hagemann, I.S.; Balint, K.; Barchetti, A.; Wang, L.-P.; Gimotty, P.A.; Gilks, C.B.; Lal, P.; Zhang, L. Tumour hypoxia promotes tolerance and angiogenesis via CCL28 and T reg cells. Nature 2011, 475, 226–230.
More
Information
Subjects:
Oncology
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
829
Revision:
1 time
(View History)
Update Date:
19 Jul 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





